Abstract
Alginate scaffold has been used widely for controlled release applications because of its ability to provide three-dimensional supports for formation of a gel matrix. Alginate gel scaffolds for drug delivery matrices were prepared using a fluidic device. N2 gas was used in the fluidic device to generate bubbles in the gel layer. The hydrogel matrices with induced voids were compared with hydrogel matrices without voids. This study attempted to identify the release mechanism of vitamin B12 from the two types of prepared scaffolds, and the data were fitted with different release kinetic models. The results revealed that the alginate scaffold exhibited a controlled release profile and that the corresponding release mechanism followed a first-order kinetic model. Hydrogel scaffolds fabricated with biocompatible polymers using fluidic methods could be promising for controlled drug delivery systems.
Introduction
Alginate scaffolds play a major role in the formation of three-dimensional (3D) matrices and have great potential for controlled release applications, making them the subject of extensive research. Use of polymer gel from natural and synthetic sources is now well accepted due to its biocompatibility and the fact that it is biodegradable. 1 The ability of gel layers to hold biological agents and allow their controlled release into a targeted tissue over a long period of time makes them even more promising for further investigation. The gel layer can also act as a scaffold after loading with matrix-forming cells where regeneration of tissue is required.
Recently, many researchers have reported controlled drug delivery using different types of scaffolds. In drug delivery applications, drug uptake is improved by the induction of additional porosity in the alginate gel layer. 2 Moreover, a uniform distribution of this porosity throughout the gel scaffold is essential for optimal function.
Several techniques, such as suspension freeze drying, fiber bonding, gas foaming, thermal phase separation, electro-spinning, and the use of supercritical CO2, etc, are used to introduce voids into the gel layer, but these techniques, in addition to being very expensive, all expose the gel to either thermal or chemical treatment. 3 Direct introduction of bubbles into a thin hydrogel layer by using a fluidic device does not rely on chemical reactions and gives better control to achieve a highly ordered pore structure. Such introduction of bubbles into the gel layer using a micro fluidic device is a simple and good technique to control both void size and the porosity of the scaffold. In this technique, bubbles form and then mono-disperse within the scaffold. The self-assembly of bubbles occurs very quickly, forming highly organized structures. The technique is also very economical as compared with other fabrication techniques.
The present study describes the fabrication, using a fluidic arrangement, of an alginate scaffold that can serve as drug delivery device, and the study of its solute release behavior and release kinetics. Vitamin B12 is used as a model drug for kinetic analysis in our study. The proposed method results in an alginic carrier with a uniformly distributed pore structure that influences the uptake and release of drug molecules. With the development of porous hydrogel, sodium alginate has caught the attention of researchers working on drug delivery systems. In recent years, sodium alginate has shown tremendous potential in the pharmaceutical field due to its biodegradability, biocompatibility, and non-toxic properties. 4 Moreover, simple modifications allow fabrication of multifunctional hydrogel structures. Thus, alginate gel is now widely considered for controlled drug delivery release studies.5–8
Pluronic F127 is a surfactant that is non-ionic in nature, 100% active, and relatively non-toxic. In general, pluronic F127 is composed of white, waxy, free-flowing granules that are practically odorless and tasteless. It has thermo-responsive properties in aqueous solution. The viscosity of pluronic solution increases with increasing temperature, which has the property of stabilizing the bubbles.
Fabrication of alginate scaffolds using microfluidic techniques has been reported.9–11 In a fluidic device, two glass capillaries are heated and pulled in such a way that one will be placed inside the other. The gas flow through the inner capillary is pulled by the flowing liquid and breaks to form bubbles. The resulting scaffold, as presented in this paper, is based on the generation of homogeneous bubbles by a co-flow device, where the liquid phase is formed by mixing aqueous polymeric solution with surfactant solution, with N2 gas being used as the gas phase. The alginate scaffold with embedded voids generated using the above technique forms a non-sticky gel film, where voids distributed homogeneously in the gel scaffold are expected to act as additional reservoirs that can take up a greater amount of drug molecules and hence increase the release rate. Uniformly distributed voids in the gel scaffold can also be useful in the field of tissue regeneration.
The present study reports alginate hydrogel carriers for controlled release of vitamin B12. The novelty of this research resides in the following two features: (1) easy and preparation of the gel layer under mild conditions without the use of toxic chemicals; and (2) the introduction of voids using fluidic arrangements that generate highly ordered mono-dispersed bubbles. After optimizing formulation of the alginate gel using the fluidic method, the morphology, mechanical strength, and release kinetics were investigated at different temperatures and with different gel systems.12–13 Release behavior from hydrogel scaffolds under shaking conditions was also investigated. Furthermore, the release profiles of vitamin B12 from the different scaffolds were fitted to different kinetic models to reveal release mechanisms.
The main aim of this work was to attempt the fabrication of alginate scaffold using a fluidic device. Vitamin B12 was selected as the model drug for the study of release kinetics. The drug release kinetic study investigated the effect on the kinetic rate of surface morphology, varying diameter, drug dissolution, drug loading concentration, etc. The water-soluble model drug inherently followed near first-order kinetics with an initial high release rate. The quantitative and qualitative analysis of the formulation may alter drug release performance. In controlled drug release formulations, analysis of drug dissolution data in vitro to anticipate in vivo performance is a rational approach. 14 To examine the kinetic release data, we applied the following models:
(a) Zero-order model
(b) First-order model
(c) Higuchi model
(d) Hixson-Crowell model
Materials and methods
Materials
Sodium alginate was purchased from Sigma-Aldrich (St. Louis, MO, USA). The viscosity of 2% solution at 25°C is 250 cP. Pluronic F 127 was purchased from Sigma-Aldrich. The critical micelle concentration of Pluronic F 127 is 4-11. Calcium chloride dihydrate (CaCl2·2H2O) pellets were purchased from Merck (New Delhi, India). Vitamin B12 was purchased from SRL (Mumbai, India).
Methods
An aqueous solution of alginate was prepared by dissolving 4% sodium alginate powder in distilled water. A mechanical stirrer was used at a stirring speed of 2000 rpm for 4 h to dissolve the sodium alginate completely. The pH of the alginate solution was measured and found to be 7.54. An aqueous solution of pluronic F127 surfactant was prepared separately in a beaker by adding 4% of pluronic F127 to distilled water. The mixture was kept on a magnetic stirrer for 1 h at a rotating speed of 300 rpm. The solution was then kept in a refrigerator for 24 h maintained at 4°C for complete dissolution. The pH of the pluronic solution was measured to be 6.56. The resultant solution of alginate and pluronic was prepared by adding alginate and pluronic solution in equal proportions, and mixing in a magnetic stirrer at a rotation speed of 100 rpm for 15 min. The resultant solution maintains a pH of 6.90. The viscosity of the resultant solution was measured at different shear rates at room temperature using parallel plate geometry of a rheometer from Anton Paar.
A co-flow device was fabricated using two glass capillaries pulled by heating. N2 gas was allowed to flow through the inner capillary and the polymer-surfactant solution was allowed to flow through the outer one. The flow rate of the liquid was controlled by a syringe pump (Figure 1) from Harvard Apparatus (Holliston, MA, USA) and was kept constant throughout the whole experiment. The flow rate of N2 gas was also maintained constant using a mass flow controller (Alicat Scientific, Tucson, AZ, USA) supplied from a gas cylinder. The flow rates of liquid and gas were maintained at 5.0 ml/min and 1.0 ml/min, respectively. The experimental procedure was described in detail in our previous article. 1 The aqueous film of alginate with entrapped bubbles was collected in a Petri dish for crosslinking with CaCl2. Magnified images of the alginate film were captured using a microscope with a camera mounted on it, and the whole system was attached to a computer for continuous monitoring. Davis software from LaVision was used to calculate the diameter of bubbles in the aqueous film. Next, the aqueous solution of CaCl2 was poured over the alginate film from one side of the Petri dish to form a gel structure within the liquid phase. Excess CaCl2 solution was removed from the gel structure by dipping it in the distilled water. A scaffold without bubbles was also prepared in a separate Petri dish to allow comparative study of the same composition. The scaffold was then dried in a vacuum oven at a temperature of 50°C; a constant vacuum pressure of 60 Torr was maintained throughout drying.15–16 The weight of the scaffold was monitored regularly using a digital weighing balance during the drying period until the weight of the scaffold reached a constant value. A scanning electron microscope (SEM) (JSM 5800; JEOL, Tokyo, Japan) was used to capture images of dried scaffold with and without voids. X-ray diffraction using an x-ray diffractometer (PANalytical, Eindhoven, The Netherlands) was also applied to characterize both types of scaffold (with and without voids). Fe-filtered Co K-alpha 0.178901 nm radiation at 30 mA and 40 kV was used, with a minimum step size of 2θ as 0.001 and a time per step of 19.685 s.
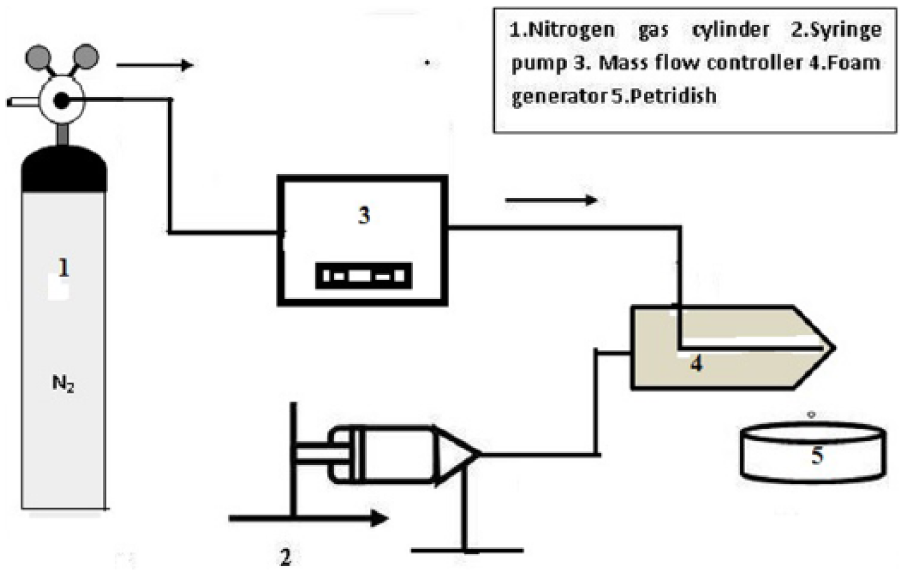
Schematic diagram of experimental set up.
Next, dried scaffolds with and without voids were dipped separately into a solution consisting of 0.02% vitamin B12 in 0.9% brine solution. At this stage, the objective was to allow the scaffold to absorb the maximum possible amount of vitamin B12 solution. The porosity of the different types of scaffolds were calculated by gravimetric and volumetric analysis and was presented in our previous article. 1 After uptake, the vitamin B12-loaded scaffold was taken out slowly from the vitamin B12 solution and immersed in a beaker containing 500 ml of 0.9% brine solution. The beaker was then kept on a vertex shaker with constant shaking at 90 rpm. Samples (5.0 ml) were withdrawn from the beaker at regular intervals during the shaking, and replaced with an equal amount of NaCl solution to maintain a constant volume in the beaker. The absorbance of the collected samples was measured using a UV visible spectrophotometer (Perkin Elmer Lambda 35) at a wavelength of 361 nm. Vitamin B12 concentrations were calculated by comparison with absorbance values of pre-calibrated data from known concentrations.
Kinetic model for vitamin B12 release studies
In the present study, a diffusion-based drug delivery system was fabricated and used for loading of vitamin B12 as discussed in our previous articles.17–18 In all drug release studies, kinetic models make an important contribution to identifying the mechanism of drug release. Many kinetic models have been adopted to discuss the exact mechanism of drug release from different types of supports. We used four different types of kinetic model for vitamin B12 release from different scaffolds. The kinetic models used in this study were: (1) a zero-order kinetic model (2) a first-order kinetic model, (3) the Higuchi model, and (4) the Hixcon-Crowell kinetic model.19–20
Zero-order model
The zero-order model describes a system where the rate of drug release does not depend on its concentration,

where
First-order model
The first-order model describes drug release from the scaffold where the rate of release depends on the concentration of the drug,

where
Higuchi model
Drug release from a scaffold was described by Higuchi in 1961. The Higuchi model describes the release of drug from the scaffold as the square root of a time-dependent process based on Fick’s law of diffusion. It is expressed as

where
The data obtained from the drug release from the scaffold is plotted as cumulative percentage of drug release vs the square root of time. The higher value of correlation coefficient describes the diffusion control mechanism of drug release from the scaffold.
Hixson-Crowell model
The Hixson-Crowell model describes the release of drugs from a scaffold when there is a change in surface area of the scaffold due to the presence of induced voids.
The relation between drug release and time can be expressed by the equation

where
The kinetic release data obtained from drug release from the scaffold were plotted as the cube root of the percentage of drug remaining in the scaffold vs time.
Results and discussion
The viscosity data of aqueous alginate solution with pluronic F127 at varying shear rate is depicted in Figure 2. The results of viscosity measurements show that, with increase in shear rate, the viscosity of the alginate-pluronic solution decreases. This result describes the shear thinning nature of alginate solution after mixing with pluronic F127.
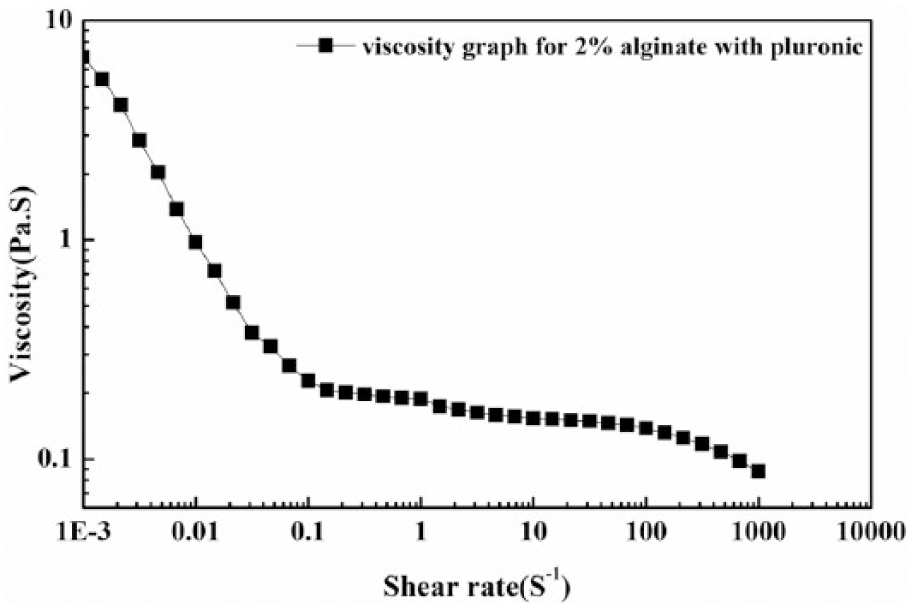
Viscosity as a function of shear rate.
A microscopic image of the alginate bubbles is depicted in Figure 3A. The co-flow device was used to generate bubbles using alginate-pluronic solution as the liquid phase and nitrogen as the gas phase. Image processing using Davis software and computer programs were used to calculate the diameter of the bubbles. The average bubble diameter was found to be 500 μm in the thin film of alginate-pluronic F127. A 4% CaCl2 solution was poured into the Petri dish so that the thin film of alginate became a gel. Ca+2 ions participated in the formation of a 3D network of calcium alginate gel by forming inter-chain ionic bonds. The Ca+2 ions exchanged rapidly with Na+ ions in the alginate to form a gel layer. The aqueous solution of alginate immediately converted into a white milky structure after the CaCl2 solution was added, and a free resting non-sticky gel scaffold with embedded voids developed in the Petri dish after a few minutes. The gradual penetration of surplus crosslinking solution into the air bubbles was also observed. A vacuum oven was used to dry the gel film to constant weight to ensure complete drying. Figure 3C presents a SEM image of dry film after complete drying without induced voids, while Figure 3D shows a SEM image of the gel film with induced voids. A reduction in void size was observed after complete drying. The weight and thickness of the film reduced four-fold after drying for all scaffolds, viz, scaffold A, B, C, and D (scaffolds A and C represent scaffolds without bubbles, and scaffolds C and D represent scaffolds with bubbles). Figure 3D clearly shows that the bubbles have been embedded in the alginate scaffold, and that void diameter is reduced from 500 μm to 200 μm due to complete drying. To characterize the alginate scaffold for crystallinity of molecules, XRD was performed (Figure 3B). The broad peak at 2θ value of 15.4° with
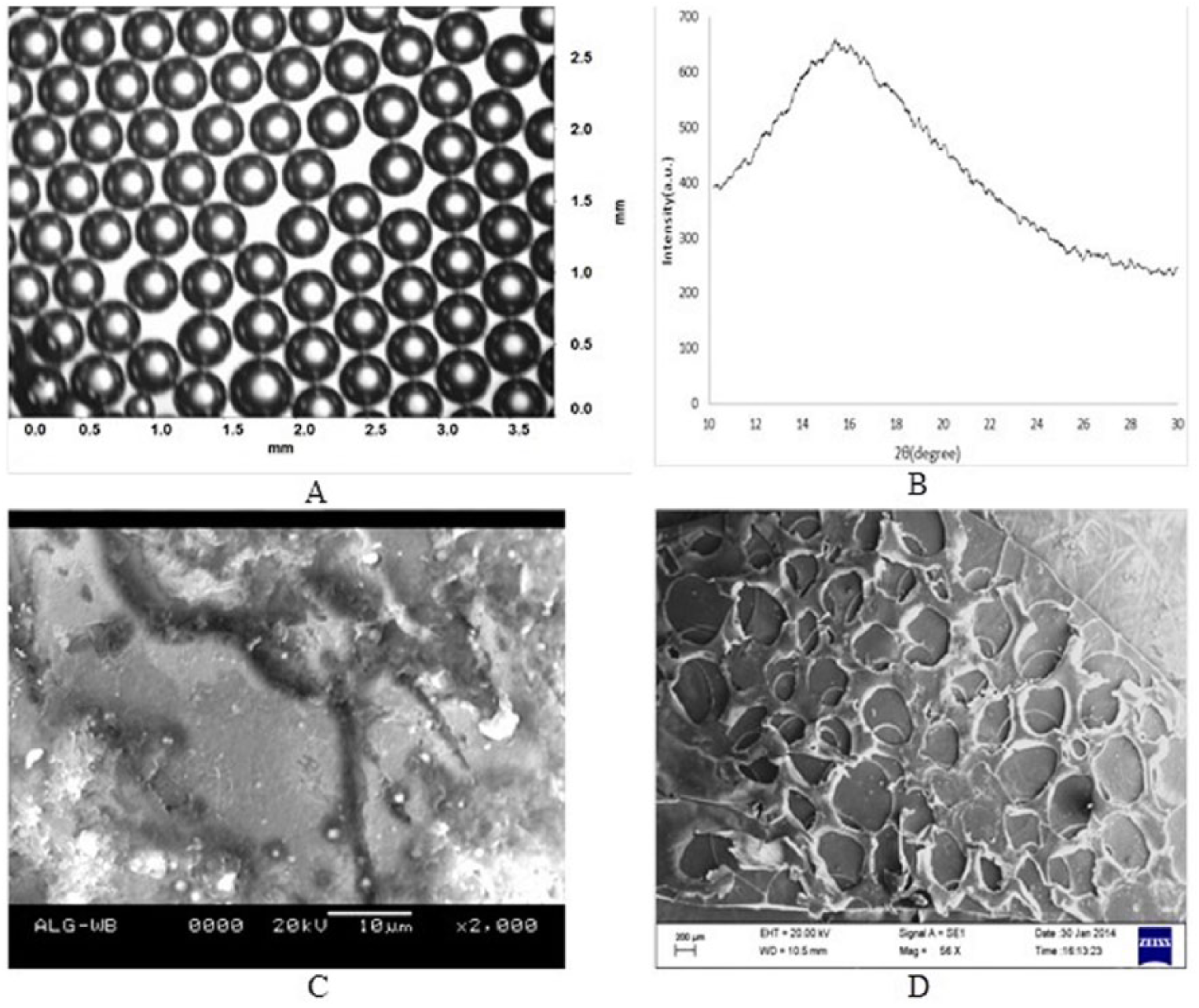
(A) Image of aqueous alginate solution under a light microscope. (B) XRD spectrum of alginate scaffold. (C) SEM image of alginate scaffold without bubbles. (D) SEM image of alginate scaffold with bubbles.
After the scaffold had absorbed vitamin B12, it was dipped into 0.9% (w/w) brine solution. For analysis of release mechanism of vitamin B12 from each scaffold, the beaker was kept on a vertex shaker with continuous shaking. Samples were collected at regular intervals for measurement of vitamin B12 concentration. A volume of brine solution equal to the volume of sample removed was poured into the beaker to make up the solution after every sample collection. A maximum of 1% SD in the calculated drug concentration was expected due to the addition of brine solution to the beaker after collection of each sample. The samples were collected for different scaffolds at regular time intervals to study the release kinetics. The release concentration of vitamin B12 from different scaffolds was estimated as

The amount of vitamin B12 released from the alginate scaffold was calculated from the amount of vitamin B12 remaining in the collected sample. The time data plot for the cumulative percentage of vitamin B12 released from the scaffold is depicted in Figure 4. The amount of vitamin B12 remaining after 3 h was significantly less for scaffolds with voids. The release rate of vitamin B12 was enhanced up to four-fold for scaffolds with induced voids compared with scaffolds without induced voids during shaking.
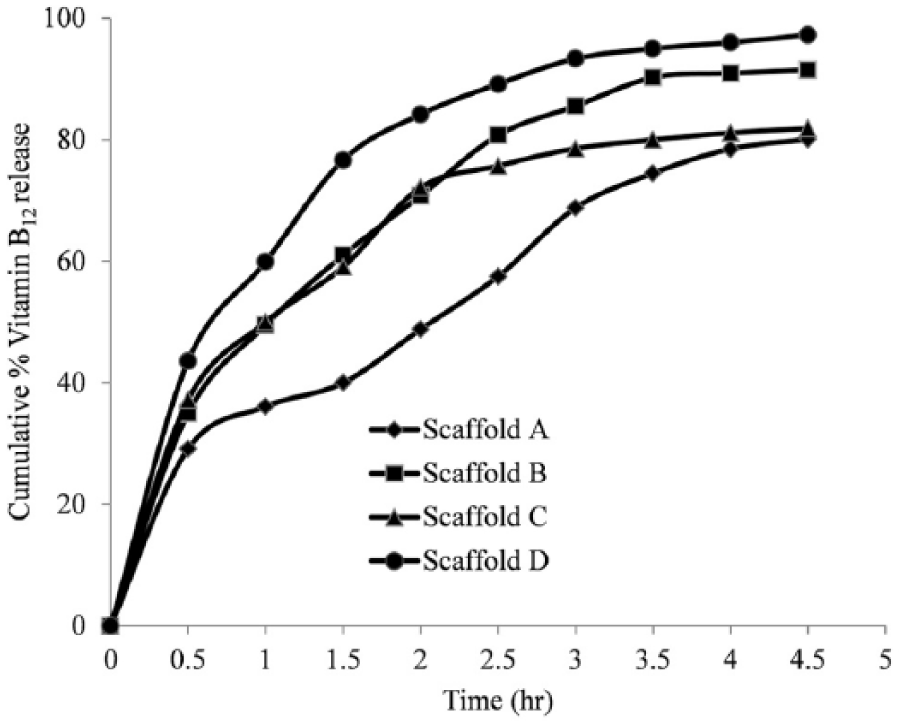
Time history data of vitamin B12 release from different scaffolds.
Vitamin B12 release mechanism
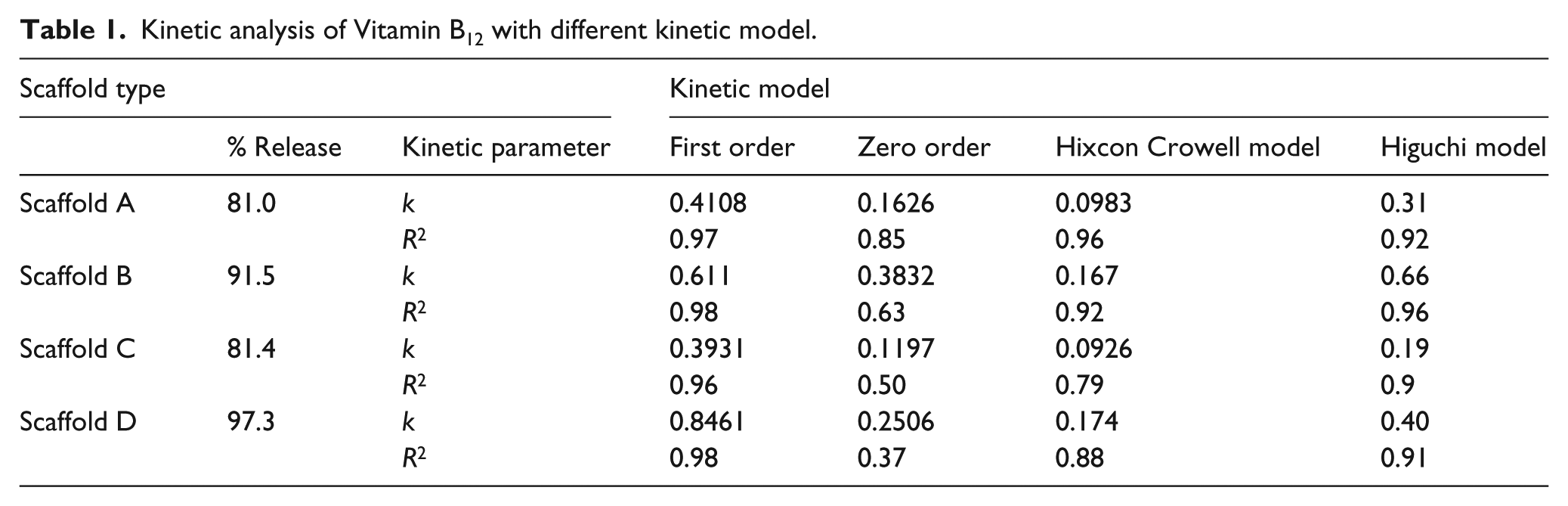
The time history data of vitamin B12 released from, and remaining in, the alginate scaffold during the dissolution process was evaluated using a UV-Vis spectrophotometer at λmax= 361 nm; data were fitted to the kinetic models. The value of regression coefficient (
From the experimental and model data, the most suitable fit can be suggested by values of
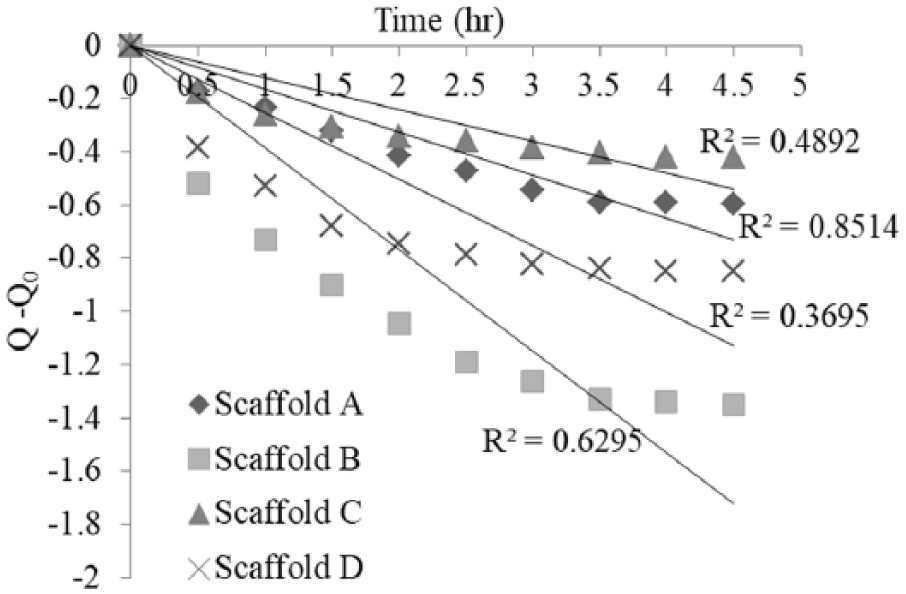
Zero-order kinetic fit for different scaffolds.
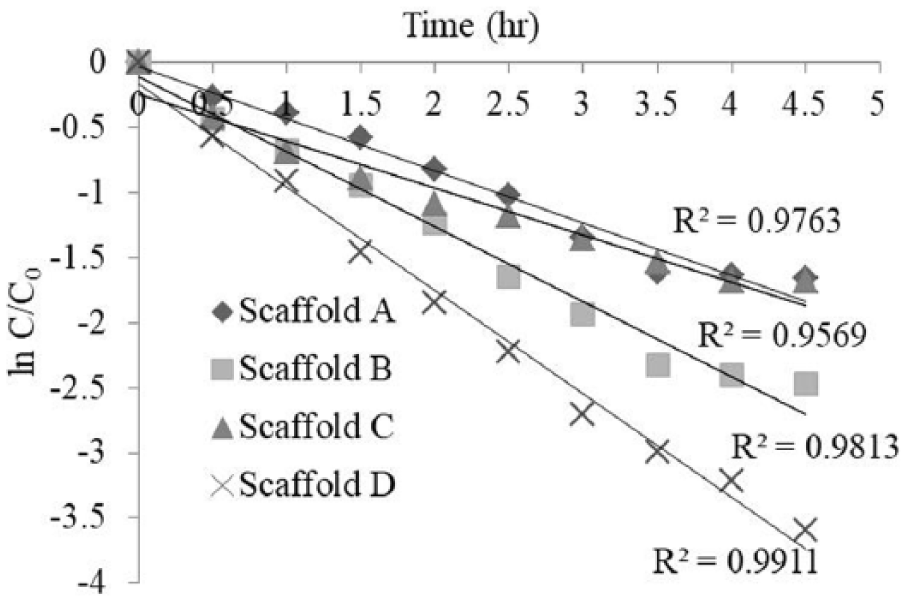
First-order kinetic fit for different scaffolds.
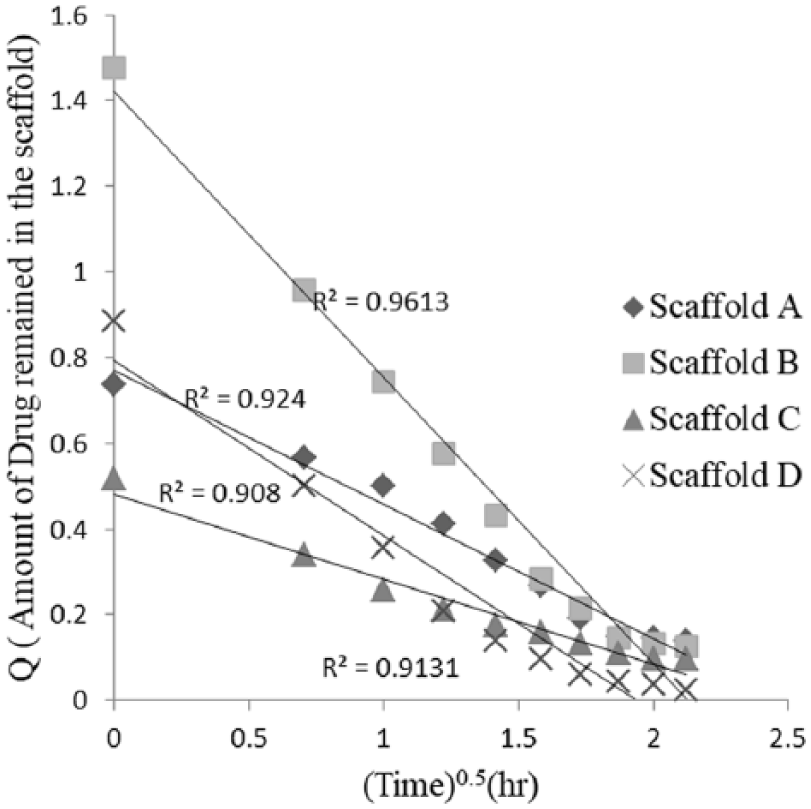
Higuchi kinetic model for different scaffolds.
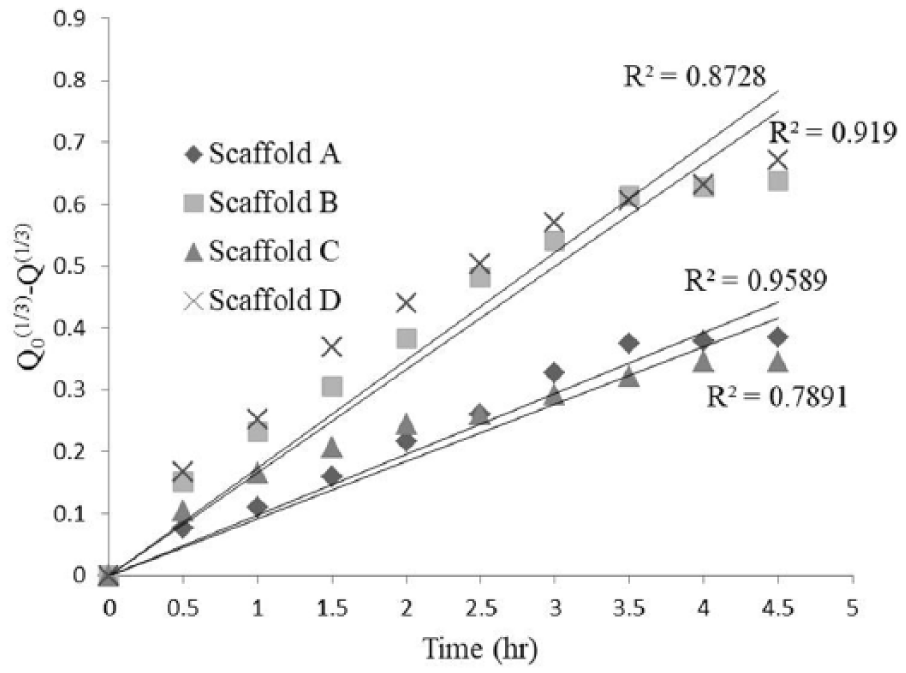
Hixcon Crowell kinetic model for different scaffolds.
Kinetic analysis of Vitamin B12 with different kinetic model.
Conclusions
A drug delivery system based on an alginate hydrogel scaffold was prepared successfully using a CaCl2 cross-linker. A fabricated micro-fluidic device was used to introduce voids in the gel scaffold. The 500 µm void size present in the swollen scaffold reduced to 200 µm after drying. The shrinkage in void size affected the absorption and release rate of vitamin B12. The scaffold swelled slowly after absorbing vitamin B12, and a high cumulative drug release was obtained within 5 h. Different scaffolds showed different release profiles for vitamin B12. Scaffold D, which contained induced voids, showed a higher percentage release of vitamin B12 and a higher reaction rate. Scaffolds B and D showed percentage release of >90%, whereas scaffolds A and C showed a percentage release of 80%. Mathematical kinetic models play a major role in interpreting the exact mechanism of drug release from scaffolds. The vitamin B12 release kinetics in this study were found to be best fitted with first-order kinetics for all scaffolds. The values of most regression coefficients were >0.96 (
Footnotes
Declaration of conflicting interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
