Abstract
Background
Controlled and local drug delivery systems of anti-inflammatory agents are drawing increasing attention thanks to their possible pharmaceutical and biomedical applications. These systems have extended therapeutic effects and reduced side effects.
Methods
A single-step sol-gel process was used to prepare organic-inorganic hybrid materials based on silica (SiO2) and poly-ε-caprolactone, containing ketoprofen for controlled drug delivery applications. Fourier transform infrared spectroscopy analysis proved formation of H-bonds among the carbonyl groups of the polymer chains and Si-OH group of the inorganic matrix. X-ray diffraction analysis highlighted the amorphous nature of the synthesized materials. Scanning electron microscopy and atomic force microscope topography showed their homogeneous morphology and nanostructure nature.
Results
The bioactivity of the synthesized hybrid materials was shown by the formation of a layer of hydroxyapatite on the surface of samples soaked in a simulated body fluid (SBF).
Conclusion
Release kinetics in SBF were subsequently investigated by means of UV-VIS spectroscopy. A large amount of drug release occurred during the first few hours, then a slower drug release supplied a maintenance dose until the end of the experiment.
Introduction
Ceramics with medical applications are an interesting research and development field for obtaining biomaterials useful for implant production (1-4). With biomaterials, and more specifically with bioceramics, many parts of the human body can be replaced or repaired (1, 5). Regardless of the ceramic type used and the manner of implantation, the introduction of an implant in a living body always causes inflammation phenomena and frequently infection processes. These problems can be overcome by using local drug delivery systems to confine pharmaceuticals such as antibiotics, anti-inflammatory, anti-carcinogens, etc. (6-8). The possibility of introducing certain drugs into the ceramic matrices employed for bone and teeth repair is, undoubtedly, an added value to be taken into account.
The use of the conventional high-temperature procedures to conform glasses and ceramics into the desired form is very well known. The degradation temperature of a pharmaceutical compound is normally around 100°C, which is very low compared with the high temperature needed for the pieces to be compacted, which is around 1,000°C. Consequently, this is the main problem in including pharmaceuticals in these conventional glass and ceramic implants. So the scientific community is currently investigating new procedures for incorporating drugs into implantable biomaterials.
The sol-gel process has proven to be versatile and has been widely used in the preparation of organic/inorganic hybrid materials (9-11), nonlinear optical materials (12) and mesomoporous materials (13). The sol-gel chemistry is based on the hydrolysis and polycondensation of metal alkoxides M(OR)x, where M=Si, Sn, Ti, Al, Mo, V, W, Ce and so forth. The following sequence of reactivity is usually found Si(OR)4 << Sn(OR)4=Ti(OR)4 < Zr(OR)4=Ce(OR)4 (11). Because silicon alkoxides are not very reactive, the sol-gel process of the silicon alkoxides is slow and easy to control when producing a transparent gel. There is considerable interest in organic-inorganic hybrid/composite materials prepared via the sol-gel process. A variety of organic polymers have been introduced into inorganic networks to obtain the hybrid or composite materials with or without covalent bonds between the polymer and inorganic components, respectively. Sol-gel reactions are known to be affected by many synthetic parameters such as the structure and concentration of the reactants, solvents and catalysts, as well as the reaction temperature and rate of removal of products and solvents (14, 15). Furthermore, the presence of organic components modifies the morphology and physical properties of the sol-gel products. For example, a base-catalyzed sol-gel reaction usually results in translucent or opaque products with visible organic-inorganic phase separation. Under acid catalysis and carefully controlled reaction conditions, transparent and monolithic hybrid/composite materials can be obtained.
Recently, a family of monolithic and transparent hybrid materials have been synthesized via the acid or photo-acid-catalyzed sol-gel reactions of inorganic precursors such as tetraethyl orthosilicate (TEOS) with polymer precursors that contain reactive alkoxysilyl groups (16-19). A key issue that remains unresolved in these organic-modified materials is the degree of mixing of the organic-inorganic components – i.e., the phase homogeneity. The high optical transparency to visible light indicates that the organic-inorganic phase separation, if any, is on a scale of ≤400 nm. Many conventional methods for analyzing composite materials have not proven to be effective. For example, the changes and disappearance of well-defined glass transition of the polymer component as measured by differential scanning calorimeter (DSC) or dynamic mechanical analysis (DMS) suggest the diminution of phase separation but offer little quantitative information (20, 21). Transmission electron microscopy (TEM) often fails to provide useful morphological data because of weak contrast (20). Recently, there have been several reports on the use of atomic force microscopy (AFM) in the analysis of sol-gel materials (20, 22).
In the last 10 years, the study of organic-inorganic nanocomposite networks and gel has become an expanding field of investigation (23, 24). At first glance, these materials are considered as biphasic materials, where the organic and inorganic phases are mixed at the nanometer to submicometer scales. Nevertheless, it is obvious that the properties of these materials are not just the sum of the individual contributions from both phases; the role of the inner interfaces could be predominant. The nature of the interface has recently been used to divide these materials into 2 distinct classes (24). In class I, organic and inorganic compounds are embedded and only the weak bonds (hydrogen, van der Waals or ionic bonds) give cohesion to the whole structure. In class II materials, the phases are linked together through strong chemical bonds (covalent or ionic-covalent bonds). Class I and class II hybrids were prepared by sol-gel technique (25).
It is known that SiO2 glass is bioactive (26) – i.e., it is able to bond to living bone. As reported in the literature (27, 28), the essential condition for glasses and glass-ceramics to bond to living bone is the formation of a bone-like apatite layer on the surfaces. In vitro studies have been performed (27, 28) by soaking the glasses in a simulated body fluid (SBF) to study hydroxyapatite formation on the surface.
Poly-ε-caprolactone (PCL) is a biodegradable, synthetic, aliphatic polyester that has attracted wide interest for its possible applications as a biomaterial. PCL-based 3-dimensional scaffolds for orthopedic surgery (29) and film substrates for tissue engineering (30) have been proposed. Micelles of polycaprolactone-b-poly(ethylene oxide) copolymer have been used as drug delivery vehicles (31). Organic-inorganic nanocomposites have been prepared with montmorillonite (32), silicates (33), SiO2 (34) and TiO2 (35). We recently prepared, by the sol-gel method, several PCL-based hybrids for drug delivery by using different oxides, including CaO and/or SiO2 (36-40), TiO2 (41, 42) and ZrO2 (43-45). In all cases, our Fourier transform infrared (FT-IR) measurements gave circumstantial evidence of the formation of class I hybrids, characterized by the presence of hydrogen bonds among the carbonyl groups of the polymer and the OH- groups of the inorganic phase.
In this study, SiO2+PCL (PCL 0, 6, 12 and 50 wt%) materials are used as support matrices for controlled drug release. Silica gel, originally developed for engineering applications, is also currently being studied as a polymer for the entrapment and sustained release of drugs (46). The sol-gel method was applied to encapsulate ketoprofen (5, 10, 15 wt%) as a model drug. The drug-loaded amorphous bioactive materials were studied in terms of their drug release kinetics.
Materials and Methods
Sol-gel synthesis
The hybrid organic-inorganic materials (PCL 0, 6, 12 and 50 wt%) were prepared by means of the sol-gel process from an analytical reagent grade of TEOS in an ethanol, PCL (molecular weight=65,000 Da), water and chloroform (CHCl3) mixture. Water diluted with ethanol was added to the solution under vigorous stirring.
Figure 1 shows the flow chart of hybrid (SiO2+%PCL + % ketoprofen) synthesis by the sol-gel method. SiO2+PCL (PCL 0, 6, 12 and 50 wt%), each mixed with ketoprofen (5, 10 and 15 wt%) (Sigma-Aldrich) were also prepared by using analytical reagent grade (Sigma-Aldrich) as precursor material (see Fig. 1). After the addition of each reactant, the solution was stirred; the resulting sols were uniform and homogeneous. The gelification time was controlled by changing the concentration of PCL, as shown in Table I.
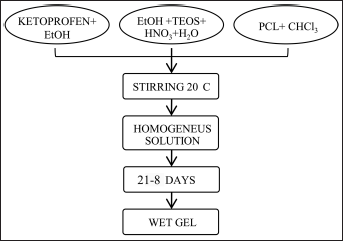
Flow chart of SiO2 + poly-ε-caprolactone (PCL) gel synthesis. TEOS = tetraethyl orthosilicate.
VARIATION IN GELIFICATION TIME, CONTROLLED BY CHANGING CONCENTRATION OF PCL
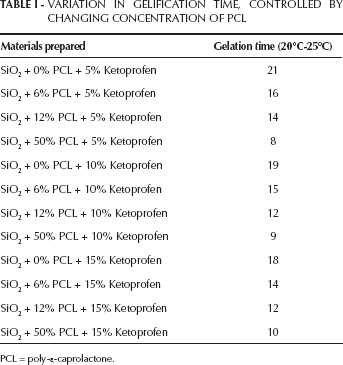
PCL = poly-ε-caprolactone.
After gelification, the gels were air dried at 50°C for 24 hours to remove the residual solvent; this treatment does not modify the stability of ketoprofen, and a glassy piece was obtained (Fig. 2). Discs with a diameter of 13 mm and a thickness of 2 mm were obtained by pressing a fine (<125 μm) gel powder into a cylindrical holder.

SiO2+PCL gel after drying.
Chromatographic experiments were carried out on a Shimadzu HPLC system, equipped with Class-VP 5.0 software, an UV spectrophotometric detector SPD-10AV-vp and 2 pumps LC-10ADvp, with low-pressure gradient systems. Samples of solutions were injected by a syringe via a Rheodyne loop injector. The loop volume was 20 μL. The analytical column was a Phenomenex C18 (150×4.60 mm; 5 μm). The flow rate of the mobile phase A (water) was set at 0.8 ml/min and that of the mobile phase B (methanol) was set at 0.2 ml/min. The total run time was 10 minutes.
HPLC grade methanol was obtained from Sigma-Aldrich. HPLC grade water was prepared using a Millipore (0.22-μm) system.
A standard solution of ketoprofen 3 mM in SBF was prepared, and the samples were taken at the end of the release from the materials.
The characteristics of the SiO2 gel, PCL and SiO2+PCL hybrid materials were ascertained by X-ray diffraction (XRD) analysis using a Philips diffractometer. Powder samples were scanned from 2Θ=5° to 60° using CuKα radiation.
The presence of hydrogen bonds between organic-inorganic components of the hybrid materials was ascertained by FT-IR analysis. FT-IR transmittance spectra were recorded in the 400-4,000 cm−1 region using a Pristige 21 Shimatzu system, equipped with a DTGS KBr (deuterated triglycine sulphate with potassium bromide windows) detector, with a resolution of 2 cm−1 (45 scans). KBr pelletized disks containing 2 mg of sample and 200 mg KBr were made. The FT-IR spectra were elaborated by IR solution software.
The microstructure of the synthesized gels were studied by scanning electron microscopy (SEM) Cambridge model S-240 on samples previously coated with a thin Au film, and by Digital Instruments Multimode atomic force microscopy (AFM) in contact mode in air.
Study in vitro bioactivity
To study their bioactivity, samples of the hybrid materials studied were soaked in an SBF with ion concentrations, as reported elsewhere (47), nearly equal to those of human blood plasma. During soaking, the temperature was kept fixed at 37°C. The ability to form an apatite layer was studied by submitting reacted samples to FT-IR, SEM and energy-dispersive spectroscopy (EDS) analysis. Taking into account that the ratio of the exposed surface to the volume of solution influences the reaction (28, 48, 49), a constant ratio of 50 mm2·mL−1 of solution was maintained as described by Hench et al (48). An electron microscope (Cambridge Stereo scan 240) equipped with an EDS LINK AN 10000 was used to verify the morphology of the coated sample and to perform a qualitative elemental analysis.
Study of in vitro release
For the study of release, the discs of the materials investigated were soaked in 15 mL of SBF, and continuously stirred at 37°C. The SBF was previously filtered with a Millipore (0.22-μm) system to avoid bacterial contamination. Release measurements were carried out by means of UV-VIS spectroscopy with a Shimadzu UV mini-1240. Absorbance values were taken at a wavelength of λ=259.5 nm, corresponding to the absorbance maximum value.
The calibration curve was determined by taking absorbance versus concentration between 0 and 3 mM as parameters. For this interval, the calibration curve fits the Lambert-Beer law (50):
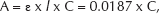
where A is the absorbance, ε is the molar absorptivity of the materials (for ketoprofen, ε=0.0187 L μmol−1 cm−1 (37)), l is the pathlength (l=1 cm−1) and C is the concentration (μM).
Results and Discussion
Gelification is the result of hydrolysis and condensation reactions according to the following reactions:
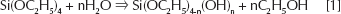
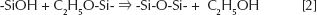
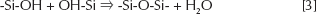
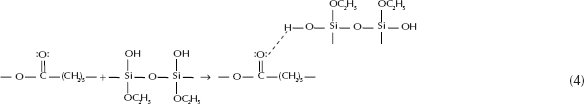
The reaction mechanism is not known in great detail; however, it is generally accepted that they proceed through a second-order nucleophilic substitution (51).
Reaction 4 shows the formation of a hydrogen bond between the carbossilic group of organic polymer and the hydroxyl group of inorganic matrix.
Sol-gel characterization
The existence of hydrogen bonds was proven by FT-IR measurements. Figure 3 shows the infrared spectrum of the SiO2 gel (Fig. 3, line a), the SiO2+PCL (6, 12 and 50 %wt) gels (Fig. 3b-d) and PCL (Fig. 3e). In Figure 3, line a, the bands at 3,400 and 1,600 cm−1 are attributed to water (28, 52). The bands at 1,080 and 470 cm−1 are due to the stretching and bending modes of SiO4 tetrahedra (53). In Figure 3, lines b-e, the bands at 2,928 and 2,840 cm−1 are attributed to the symmetric stretching of -CH2- of PCL. The band at 1,730 cm−1 is due to the characteristic carbonylic group shifted to low wave numbers. The broad band at 3,200 cm−1 is the characteristic O-H group of hydrogen bonds. The nature and the microstructure of the SiO2+PCL hybrid materials were studied by XRD, SEM and AFM.
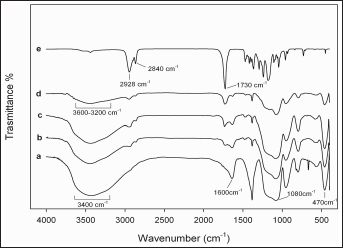
Fourier transform infrared (FT-IR) analysis of SiO2 gel (line a), SiO2 + PCL 6 wt% (line b), SiO2+PCL 12 wt% (line c), SiO2+PCL 50 wt% gels (line d) and PCL (line e).
The diffractograms in Figure 4a show that SiO2 gel exhibits broad humps characteristic of amorphous materials, while sharp peaks can be detected on the diffractogram of PCL and ketoprofen, which are typical of crystalline materials (Fig. 4b, c). On the other hand, the XRD spectrum of hybrid SiO2+PCL exhibits broad humps characteristic of amorphous materials (Fig. 4d), as well that of SiO2 gel.
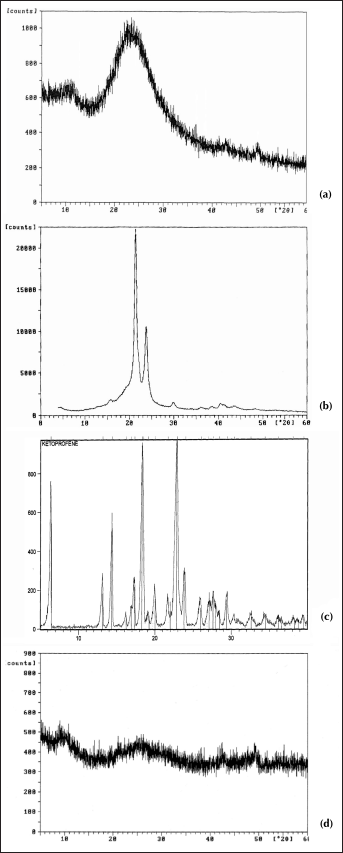
XRD diffractograms of SiO2 gel (
SEM micrographs showed that no appreciable difference between the morphology of the 4 amorphous materials could be observed. The samples appear as shown in Figure 5.

SEM micrograph of SiO2 + PCL gel.
The degree of mixing of the organic-inorganic components – i.e., the phase homogeneity, was ascertained applying AFM in the analysis of the sol-gel hybrid material.
AFM contact mode images can be measured in the height mode or in the force mode. Force images (z range in nN) have the advantage that they appear sharper and richer in contrast and that the contours of the nanostructure elements are clearer. In contrast, height images (z range in nm) show a more exact reproduction of the height itself (54). In this work, the height mode was adopted to evaluate the degree of homogeneity of the hybrid materials. The AFM topographic images of SiO2 and SiO2+PCL (0, 6, 12 and 50 wt%) gel samples are shown in Figures 6 and 7. As can be observed, the average domain size was less than 130 nm. This result confirmed that the SiO2+PCL gels synthesized could be considered organic-inorganic hybrid materials as suggested by literature data (55).
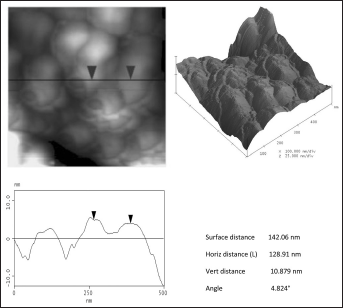
AFM image showing the microstructure of SiO2 gel.
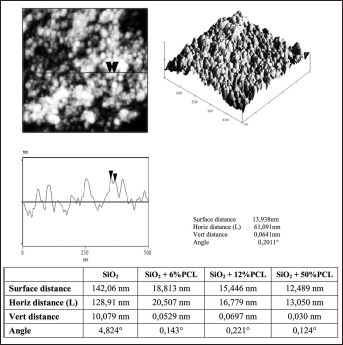
AFM image showing the microstructure of SiO2 + PCL gels.
Bioactivity test
The hybrid materials were soaked in SBF, as described by Kokubo (27), for in vitro bioactivity tests. The FT-IR spectra after different exposures to SBF up to 7, 14 and 21 days are shown in Figure 8b-d. Evidence of formation of a hydroxyapatite layer is given by the appearance of the 1,116 and 1,035 cm−1 bands, usually assigned to P-O stretching (53), and of the 580 cm−1 band usually assigned to the P-O bending mode (53). The splitting, already after a 7-day soaking, of the 580 cm−1 band into 2 others at 610 and at 570 cm−1 can be attributed to formation of crystalline hydroxyapatite (53). Finally the band at 800 cm−1 can be assigned to the Si-O-Si band vibration between 2 adjacent tetrahedra characteristic of silica gel (53). This supports the hypothesis that a surface layer of silica gel had formed as expected in the mechanism proposed in the literature for hydroxyapatite deposition (28, 56). Moreover an evaluation of the morphology of the apatite deposition and a qualitative elemental analysis were also carried out by electron microscopy observations on pelletized discs previously coated with a thin Au film.
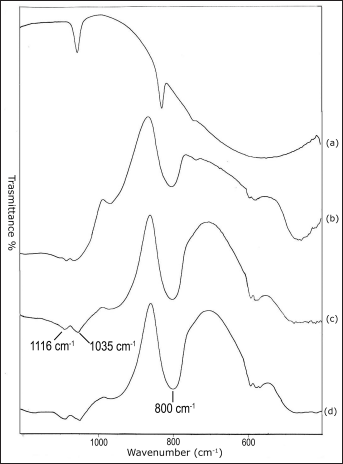
FT-IR spectra of SiO2 + PCL gel samples after different times of exposure to SBF: line a: not exposed; line b: 7 days exposed; line c: 14 days exposed; and line d: 21 days exposed.
The EDS reported in Table II confirms that the surface layer observed in the SEM micrographs (Fig. 9) consists of calcium phosphate, and can be noted that the amount of hydroxyapatite is directly proportional to PCL content.
EDS ANALYSES OF HYBRID MATERIALS 21 DAYS AFTER IMMERSION IN SBF

EDS = energy-dispersive spectroscopy; PCL = poly-ε-caprolactone; SBF = simuated body fluid.

Scanning electron microscopy micrograph of SiO2 + PCL gel after exposure to SBF for 21 days.
Release kinetics
Kinetic measurements of release from the materials studied were carried out in 15 mL of SBF incubated at 37±0.1°C and under continuous magnetic stirring at 150 rpm. Sink conditions were maintained throughout all studies. The discs used were obtained with particles size between 63 and 125 μm compressed at 3 tons, and aliquots of 600 μL were withdrawn at 1-hour intervals and replaced with an equal volume of release medium preequilibrated to temperature. The release was assayed by measuring the photometrical absorbance at 259.5 nm. To establish the relationship between the UV absorbance at λ=259.5 nm and the concentration of the solution, a calibration curve (R2=0.9907) was drawn for a standard solution with 4 levels of concentration: 0.0 mM, 1.0 mM, 2.0 mM and 3.0 mM (Fig. 10). All of the standard solutions were prepared in SBF.
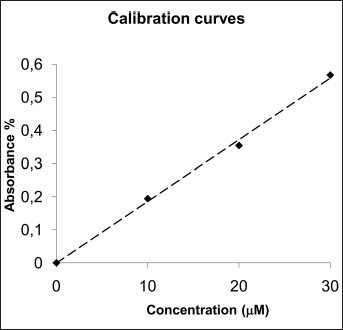
Calibration curve (259.5 nm) based on the concentration of ketoprofen (R2=0.9973).
Figures 11a, 12a and 13a, and 11b, 12b and 13b show the drug release rates expressed as a percentage of the drug delivered, related to the drug-loading value, or as a function of time, respectively. It was observed that from the SiO2+PCL (0, 6, 12 and 50 wt%) + ketoprofen 5 wt% gels, about 60 wt% of the drug was released in a relatively rapid manner during the initial 2 hours and that seemed to be completed within 7 hours without any evident difference in the time of release. For the SiO2+PCL (0, 6, 12 and 50 wt%) + ketoprofen 10 and 15 wt% gels, about 60 wt% of the drug was released during the initial 1 and 0.5 hours, respectively, and that was complete in about 3 and 4 hours, respectively.
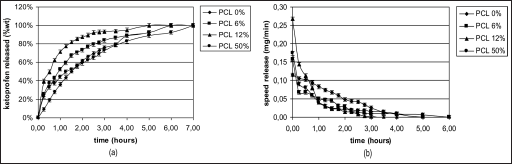
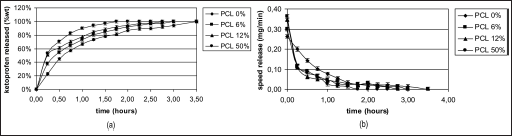
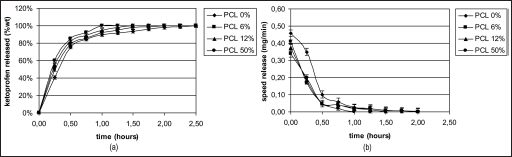
The differences observed in the release behavior between SiO2+PCL (0, 6, 12 and 50 wt%) + ketoprofen might be due to the different networks of the 4 gels that are determined by the different percentage content of PCL.
The 2-stage release observed in all cases suggests that the initial stage of release occurs mainly by dissolution and diffusion of the drug entrapped close to or at the surface of the samples. The second and slower release stage involves the diffusion of the drug entrapped within the inner part of the clusters.
An interesting observation is the general presence of a early lag period, which indicates the need for solvent penetration into the structure.
Figures 11b, 12b and 13b show this particular kinetic describing the changes of the release speed during the 2 stages.
Conclusions
The SiO2+PCL (0, 6, 12 and 50 %wt) materials, prepared via the sol-gel process, were found to be organic-inorganic hybrid materials.
The polymer (PCL) was incorporated into the network by hydrogen bonds between the carbonylic groups of organic polymer and the hydroxyl groups of inorganic matrix. The formation of hydrogen bonds was ascertained by FT-IR. Moreover the AFM and SEM analyses confirmed that the SiO2+PCL was a homogenous organic-inorganic hybrid material, because the average domains were less than 400 nm in size (55).
The release kinetics demonstrate that the materials investigated supplied high doses of the anti-inflammatory during the first hours when soaked in SBF, and then a slower drug release supplied a maintenance dose until the end of the experiment.
Finally, the formation of a layer of hydroxyapatite on the surface when the samples of the SiO2+PCL were soaked in SBF, showed by SEM micrograph and related EDS that the SiO2+PCL can be considered bioactive materials. The presence of the anti-inflammatory drug does not inhibit this bioactive behavior.
Further investigations are required for evaluating the mechanical properties of the synthesized hybrid material.
