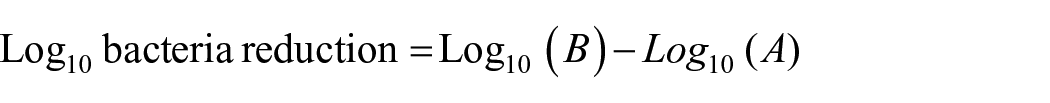Abstract
Introduction:
Intensive research is dedicated to the development of novel biomaterials and medical devices to be used as grafts in reconstructive surgery, with the purpose of enhancing their therapeutic effectiveness, safety, and durability. A variety of biomaterials, from autologous bone to polymethylmetacrylate, polyether ether ketone, titanium, and calcium-based ceramics are used in cranioplasty. Porous hydroxyapatite (PHA) is reported as a possible material for bone reconstruction, with good signs of biocompatibility, osteoconductive and osteointegrative properties. In the present paper we studied the possible antibacterial properties of PHA in a laboratory test in order to provide a possible overview of the occurrence of post-operative infections in PHA cranioplasty.
Method:
The test method has been designed to evaluate the potential antimicrobial activity of specimens under dynamic contact conditions to overcome difficulties in ensuring contact of inoculum to the specimen surface. The test was conducted using Staphylococcus aureus ATCC6538 as a bacterial strain.
Results:
Two experimental sets were performed to evaluate the antimicrobial properties of the specimens against two different Staphylococcus aureus concentrations. The first preliminary test (a) verified the antibacterial property at 0, 1, 2, and 4 h of contact time; the second confirmatory test (b) was repeated to verify the antibacterial property at 0, 4, 8 h. In the first experiment, after the first hour of contact, the bacterial inoculum was reduced by 7.96% compared to “inoculum only,” which increased to 26.11% at the second hour, and up to 52.33% after 4 h. In the second experiment, the confirmation test showed that bacterial growth reaches maximum inhibition after 4 h of contact. At 4 h, there was a higher bacterial reduction of 72.93%, which decreased at 8 h (36.45%).
Conclusion:
Analyzing the growth trend of viable microorganisms under Dynamic Contact Conditions it can be seen that PHA cranioplasty appears to inhibit exponential growth by inducing bacterial stasis in the early hours of contact, reaching a maximum reduction within 4 h, in this adopted experimental condition.
Introduction
Nowadays, intensive research is dedicated to the development of novel biomaterials and medical devices to be used as grafts in reconstructive surgery, to enhance their therapeutic effectiveness, safety, and durability. In this respect, graft versus host response represents one of the most important and serious concerns in clinical practice, particularly when manifesting in post-operative complications such as infections.1 –3
One of the main areas affected by such technological development is the cranial reconstruction. In the last years, a variety of biomaterials, from autologous bone to polymethylmethacrylate (PMMA), polyether ether ketone (PEEK), titanium, and calcium-based ceramics have been used in cranioplasty.
Despite their great advantages, none of the materials currently available for cranioplasty is free from complications. An exemplificative case is given by the autologous bone, still considered the “gold standard” material despite the high incidence of infection and bone resorption (29% to more than 50% in adults to 80% in pediatrics4 –8) but also by the other heterologous materials, which have reported clinical significant rate of postoperative complications, also related to the material properties, such as enhanced cold/heat conduction and osteopenia induced by the mismatch in stiffness at the bone/implant interface.4,5,7,9 –15
One of the most recently developed materials used in cranial reconstruction is porous hydroxyapatite (PHA), a ceramic material widely used in cranioplasty, that is reported to have good signs of biocompatibility, osteoconductive and osteointegrative properties.16 –18 These features are related to its composition closely mimicking human bone favoring cell adhesion, proliferation, and osteogenic differentiation. Studies suggest that PHA can establish a biochemical continuity with host bone tissue, inducing the formation of a protective film made of “slime mucopolysaccharide deposits” at the surface, which may provide resistance to bacterial proliferation. 19 In addition, recent studies report that calcium phosphates including PHA demonstrate inherent antibacterial properties and good eukaryotic vs. prokaryotic cell selectivity.20 –23
The present paper aims to provide an experimental overview of the occurrence of postoperative infections in PHA cranioplasty, which has been done through the results from a laboratory test performed to evaluate the antibacterial property of a PHA implant (CustomBone Service (CBS), Finceramica Faenza S.p.a., Italy).
Material and methods
To achieve the goal of the study, a laboratory test has been conducted according to the reference standard ASTM E2149-20, modified accordingly for the specific study, to evaluate the antibacterial properties. The details of how the tests have been conducted are included in the Supplemental materials.
The test method has been designed to evaluate the potential antimicrobial activity of specimens under dynamic contact conditions to overcome difficulties in ensuring contact of inoculum to the specimen surface. The test method ensures good contact between the bacteria and the substrate in the exam, by constant agitation of the test specimen in a challenge suspension during the test period. Regarding the clinical application, it was considered that the device is used in cranioplasty, with direct contact with the blood vessels from the tissues in the implant site.
The test was conducted using Staphylococcus aureus ATCC6538 as a bacterial strain since a recent meta-analysis performed by Chen et al. 24 investigated implant infections in neurosurgical procedures shows that Staphylococcus aureus is the main pathogen in neurosurgery.
A porous material can be considered a worse case than a non-porous material since a porous scaffold has a larger specific area that could encourage bacterial growth.
The samples tested were representative specimens of PHA cranioplasty prosthesis that is composed of porous stoichiometric HA (range of porosity 35%–65%). To be able to carry out a representative and reproducible in vitro test, it was decided to consider planar specimens of size 35 mm × 35 mm considering an average thickness of 6.5 mm of the implanted prostheses to better simulate the in vivo behavior.
For what concern the preparation of the PHA, it starts from a semi-finished workpiece obtained by manipulating hydroxyapatite with a porous matrix of a predefined size (patented technology). The PHA samples were prepared by imbibition: cellulose sponges with controlled pore morphology were treated in an HA slurry to obtain the bimodal porous structures. The semi-finished piece is then subjected to a lengthy drying and sintering process. The result is a porous block of hydroxyapatite. The porous hydroxyapatite block is then machined with CNC (Computerised Numerical Control) machines to obtain a device identical to the approved model. The use of stoichiometric HA as the raw material and the manufacturing process allows obtaining a finished PHA sample with a hydroxyapatite phase representing more than 99% with a crystallinity degree over 97% (result verified by XRD pattern analysis using Rietveld method according to the reference standard ISO 13779).To allow the experimental bacterial growth, Tryptic Soy Broth (TSB) has been used for culture growth and an inoculum with the bacterial strain has been prepared. The volume of inoculum has been reduced to the minimum volume needed for the test to ensure complete contact between the tested material and the bacteria and to allow at least 3 aliquots to be withdrawn from each container.
Two different concentrations of inoculum: 1–5 × 105 cfu/ml and 1–5 × 102 cfu/ml in TSB of the Staphylococcus aureus strains were exposed to the test item in the presence and in the absence of the test item (control) during a selected number of time points. At each time point, the number of viable bacteria for both suspensions by performing plating experiments on serial dilutions was determined. These concentrations have been chosen for specific reasons: 1–5 × 105 cfu/ml is the concentration indicated in the ASTM E2149 standard used as a test reference. 1–5 × 102 cfu/ml is a lower concentration in which bacterial growth should be promoted if compared to the ASTM concentration since more space and more nutrients are available for bacteria in 20 ml of growth medium. Since the purpose of the in vitro test was to evaluate the antimicrobial activity of PHA product in comparison to the inoculum only (control), by looking at the growth trend of viable bacteria, a lower, more growth-promoting concentration was chosen.
For each specimen and each inoculum only, one sterile 180 ml screw-cap Erlenmeyer flask, containing 20 ml of working dilution of bacterial inoculum was prepared. The series of flasks has been placed on the wrist-action shaker at 37 ± 1°C. Immediately at time 0 and at the appointed contact time enumeration by serial dilution of each sample in triplicate has been performed. Plates have been incubated at 37 ± 1°C for 24 h, bacterial colonies present on each plate have been counted and recorded.
Two different experimental sets of samples were performed, and the following contact times were evaluated for each Staphylococcus aureus concentration:
(1) Preliminary test: 0, 1, 2, and 4 h.
(2) Confirmatory test: 0, 4, and 8 h.
The time of intervals have been chosen since the bacterial growth curve, in an in vitro test, is characterized by four consecutive and distinct phases: lag phase, exponential phase, stationary phase and death phase. The exponential growth phase is the step where the bacterial population exponentially increases as long as there is sufficient space and nutrients to replicate. The antibacterial property of the tested item compared to the control (only inoculum) was evaluated in the time intervals (up to 8 h) that include the exponential phase, to better appreciate the difference between the two groups.
The percent or log bacterial reduction (Log10) is calculated by comparing the bacterial results from the tested samples to “inoculum only” after the specified contact times.
The percentage of reduction has been determined using the following formula:
The log bacteria reduction has been determined using the following formula:
where:
A = cfu/ml for the flask containing the treated substrate after the specified contact time.
B = cfu/ml for the “inoculum only” (control) flask after the specified contact time.
Statistical analysis
In the preliminary test, the growth trend between sample and inoculum was observed. Statistical analysis was not performed as the purpose of the test was to study the trend only. Following the preliminary test, the trend was further investigated with statistical analysis in the confirmatory test using one sample t-test.
Results
Two experimental sets were performed to evaluate the antimicrobial properties of the CustomBone specimens against two different Staphylococcus aureus concentrations. The first preliminary test (a) verified the antibacterial property at 0, 1, 2, and 4 h of contact time; the second confirmatory test (b) was repeated to verify the antibacterial property at 0, 4, 8 h (Tables 1–4).
Results of bacterial inoculum prepared at 1–5 × 105 cfu/ml in TSB.

Results of bacterial inoculum prepared at 1–5 × 102 cfu/ml in TSB.

Results obtained for inoculum at 1–5 × 105 cfu/ml in TSB.

Results obtained for inoculum at 1–5 × 102 cfu/ml in TSB.

(a) For the bacterial inoculum prepared at 1–5 × 105 cfu/ml in TSB, the results have been summarized in Table 1. After the first hour of contact, the bacterial inoculum has been reduced of 7.96% compared to “inoculum only,” which increased to 26.11% at the second hour, and up to 52.33% after 4 h.
For bacterial inoculum prepared at 1–5 × 102 cfu/ml in TSB, the results have been summarized in Table 2. There has been a significant difference also in these cases. At the first hour, there has been a decrease of 35.41%, which increase up to 44.78% at the second hour up to 58.65% after 4 h.
Analyzing the growth trend, shown in Figures 1 and 2, of viable microorganisms under Dynamic Contact Conditions, it can be stated that CustomBone appears to inhibit exponential growth by inducing bacterial stasis within 4 h of contact time, in the adopted experimental condition (Figure 3).
(b) A second session of the experiment was repeated to confirm the inhibition of bacterial growth. For the bacterial inoculum prepared at 1–5 × 105 cfu/ml in TSB, the results have been summarized in Table 3. The confirmation test showed that bacterial growth reaches maximum inhibition after 4 h of contact. At 4 h, there was a higher bacterial reduction of 72.93%, which decreased at 8 h (36.45%).
For the bacterial inoculum prepared at 1–5 × 102 cfu/ml in TSB, the results have been summarized in Table 4. There has been a significant difference also in these cases at 4 h of contact (70.59%), which decreases at 8 h of contact (43.24%).
Analyzing the growth trend, shown in Figures 4 and 5, of viable microorganisms under Dynamic Contact Conditions, it can be confirmed that PHA cranioplasty appears to inhibit exponential growth by inducing bacterial stasis within 4 h of contact time, in the adopted experimental condition (Figure 6).

Growth trend of viable S. aureus (1–5 × 105 cfu/ml) counted at 1, 2, 4 h (preliminary test).
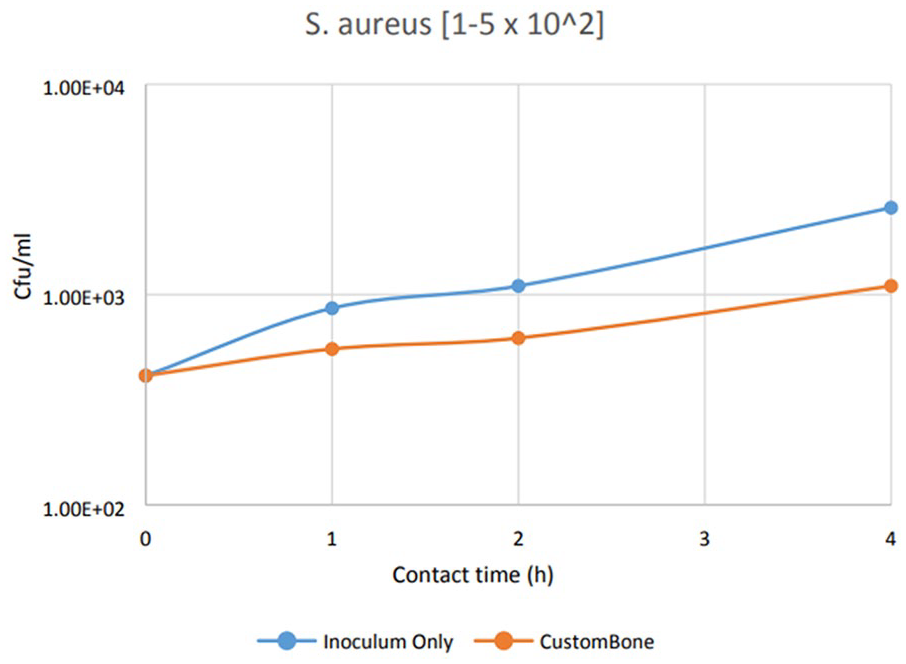
Growth trend of viable S. aureus (1–5 × 102 cfu/ml) counted at 1, 2, 4 h (preliminary test).

Scanning electron microscope (SEM) analysis at low magnification shows the macro structure of the material used as osteoconductive grafts. The morphology of sponge matrix (scaffold type A) is shown in panels a. Black spots represent interconnections between neighbour pores. SEM analysis at higher magnification of scaffold A is also shown (b). Bars: a = 1000 µm, b = 5 µm.
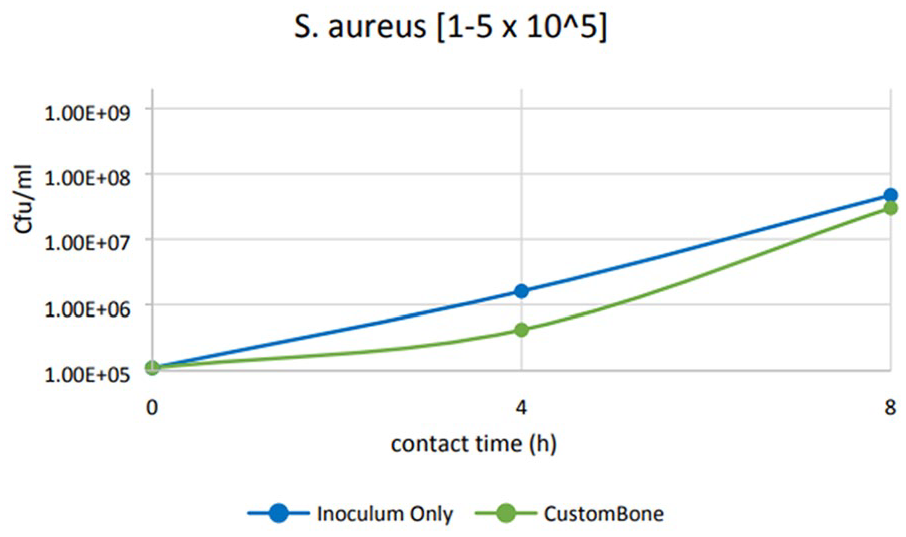
Growth trend of viable S. aureus (1–5 × 105 cfu/ml) counted at 4 and 8 h (confirmatory test).

Growth trend of viable S. aureus (1–5 × 102 cfu/ml) counted at 4 and 8 h (confirmatory test).

Diffraction patterns related to PHA. The XRD was obtained using Rietveld method according to ISO 13779. The hydroxyapatite phase represents more than 99%.
In the confirmatory test (Figures 4 and 5), the difference between the growth in the sample group and the inoculum only at 4 and 8 h in the two conditions (1–5 × 102 cfu/ml in TSB and 1–5 × 105 cfu/ml in TSB) was verified using one sample t-test (p < 0.05). All the comparisons resulted statistically different.
Discussion
The literature reported evidence that a certain rate of infections occurs within the first 60 days from implantation, irrespective of the investigated material. However, in the long-term period, infections in heterologous cranioplasties seem to be related to the different materials. 3
PHA cranioplasty behavior concerning post-operative infections may be different: when a scaffold is introduced into the human body, its chemical composition, surface texture and roughness, pore size and distribution, and also mechanical properties—such as stiffness—altogether play an important role in modulating the foreign body reaction, which represents the host’s inflammatory response to an exogenous material.1,2 This interaction usually occurs and resolves within the first 2–4 weeks following the device implantation, even though the foreign body reaction at the tissue/material interface is present for the in vivo lifetime of the medical device. In this preliminary time-lapse, it is expected that bone cells adhere to the implant, forming new bone tissue and establishing a tight interface with the implant, which is a key aspect for the scaffold stability and the subsequent stages of extensive colonization by the new bone. With biocompatible materials, early resolution of the acute and chronic inflammatory responses lasts no longer than 2 weeks. The persistence of the acute and/or inflammatory responses beyond 3 weeks usually indicates an adverse reaction against the foreign body, which can manifest in the development of an infection. These infections have been reported to occur in as many as 5%–10% of patients with implanted prosthetic devices and represent the major source of morbidity and mortality. 25
In light of these considerations, it can be devised that the physicochemical features of the implant play a pivotal role in determining the occurrence of infections, particularly in the late stage.
HA-based prostheses are made of porous, highly crystalline hydroxyapatite (HA), a biocompatible and osteoconductive calcium phosphate (Ca/P) material, with a chemical composition (range Ca/P = 1.65–1.82) and structure that resembles the mineral component (average Ca/P = 1.71) of human bones. This biomaterial is highly porous (range of porosity 35%–65%), with a trabecular structure and multi-scale pore size that are essential to ensure new bone penetration in the inner part of the scaffold and adequate exchange of nutrients and physiological liquids to provide new bone formation.
The association of a composition close to the mineral bone and the presence of wide-open and interconnected porosity are features that make porous HA very effective for extensive osteoconduction and osteointegration. This has great relevance for bone regeneration, particularly in cranioplasty procedures,26,27 to give stability to the bone-implant interface that facilitates new bone apposition and proliferation in large bone defects thus permitting the faster recovery of the original biomechanical performance.
Thanks to molecular recognition exerted by cells, PHA substrates permit direct cell adhesion, as compared to other materials which usually interact with cells either via (i) a layer of apatite covering the implant surface due to supersaturation of surrounding physiological fluids, rich in Ca and PO4 ions, as occurring with titanium or silica-based materials28,29 or (ii) through a fibrous tissue layer. The activation of biomimetic recognition mechanisms by cells allows foreign bodies to be accepted by our physiologic system with increased safety; this is confirmed by the high number of titanium, or polymeric devices successfully implanted so far. However, impaired reactions at the cell level can occur in a significant number of cases, more easily if the scaffold features are poorly affine with the host tissues so revision surgery after 10–20 years is a very frequent issue. Particularly, in the case of polymeric implants, a general concern is related to the production of free radicals as well as acidic and harmful by-products due to non-enzymatic dissolution, which may alter cell metabolism and vitality, and jeopardize tissue healing, particularly in the case of large bone defects.30,31 Impaired cell activity due to such phenomena is a possible reason for the existence of persistent inflammatory states and consequent possible alteration of the immune system, potentially leading to the proliferation of infective bacterial strains. 32
With the above considerations, it appears that bone-like composition and structure able to favor the natural physiological homeostasis and metabolic processes, can be relevant factors that not only promote extensive bone regeneration but at the same time can establish unfavorable conditions for adhesion and proliferation of bacteria and infective strains.
The recourse of revision surgery significantly increases patient morbidity and raises healthcare costs but, more importantly, can raise the risks of secondary infections. 33 In this respect, the use of porous materials with the ability to adsorb and retain drugs in their structure may offer significant advances, thanks to the possibility of performing antibiotic therapies directly in situ, possibly avoiding the removal of the original implant. These properties are functional to uptake a significant amount of antibiotic drugs that can be released over a prolonged time-lapse and with controlled kinetics.34,35 This is relevant for more effective drug delivery profiles that help to provide sustained antibiotic therapy and bypass the drawbacks and ineffectiveness related to systemic administration. 36
The present laboratory study confirms the capability of PHA implants to prevent bacterial infections. In fact, as demonstrated, bacterial growths are different between the sample and the inoculum alone (negative control). This evidence is strongly associated with PHA clinical evidence in which this material has a lower infection rate than other synthetic materials used in cranioplasty as reported in the literature. 37
As previously reported by Embery et al. 19 it could be assumed that the higher resistance to bacterial proliferation of PHA is associated with the presence of deposits of slime mucopolysaccharides that act as a protective film. This hypothesis is reinforced by experimental evidence showing that there is less bacterial growth in PHA samples than in the negative control (inoculum only). See Figure 7 that showing a schematic illustration of the antibacterial mechanism of samples against Staphylococcus aureus bacteria.

Schematic illustration of the antibacterial mechanism of samples against Staphylococcus aureus bacteria.
Inherent antibacterial properties in biomaterials such as hydroxyapatite open the possibility of reducing, or making more effective, the use of antibiotic drugs in bone surgery, thus aiding to contrast the ever-increasing bacterial resistance to antibiotics, which is today a primary cause of increased adverse outcomes and increasing cause of death in the post-operatory period. 38
Different antibacterial studies was conducted both with doped HA with bioactive ions and undoped HA.
Bakhsheshi-Rad et al. 39 demonstrated that zinc-doped hydroxyapatatite coating reduced the number of E. coli colonies to 92% examined according to the disc diffusion antibiotic sensitivity and colony forming units (CFU) conduted to verify the good antibacterial properties of the material. The antibacterial mechanism is attribuited not only to the HA but also to the presence of metal ions such Zn2+ that form strong bonds with thiole, amino and carboxyl groups of membrane proteins of microorganism, leading to structural alterations. The structural alteration of the microorganism membrane leads to increased permeability, causing the microorganism cells to be unable to effectively control proteins transport via the plasma membrane and, eventually, leading to cell death.
The antibacterial effect on E. coli of hydroxyapatite coated with Zn2+ ions was demostrated by Iqbal et al. 40 as well using the disk-diffusion technique and spred plate method concluding that doping of zinc into HA can help in resisting bacterial growth effectively.
Multi-doped hydroxyapatites was developed and studied for osteogenic and antibacterial effect by Ballardini et al. 20 and Sprio et al. 21 in comparison to undoped hydroxyapatite. The material was tested with Candida albicans, Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus. It has to be noted that undoped HA results confirmed the potential antibacterial effect of this material alone, reducing the bacterial growth at different time points, 6, 12, and 24 h, of all the microorganism tested. Furthermore, the results showed more than 80% bacterial reduction after 24 h in the doped HA, both in Ga-substituted and Zn-substituted apatites, compared with undoped HA. This confirms that the release of bioactive ions from the HA material has good antibacterial properties.
Anyway, the present study is consistent with the findings of Carlson et al., 41 which determined the antimicrobial effects of various biomaterials, including hydroxyapatite, by performing a count of viable bacteria after exposure to the biomaterial for up to 12 h. Hydroxyapatite significantly inhibited the growth of four tested bacteria. Staphylococcus epidermidis and Pseudomonas aeruginosa at 12 and 6 h respectively. In addition, a trend of growth suppression against Streptococcus and Staphylococcus aureus was also noted. In particular, the bacterial growth of Staphylococcus aureus decreased in the early periods, up to 4 h.
Further tests should be performed to explore the antimicrobial activity of PHA with additional bacterial strains, other than Staphylococcus aureus.
Despite the authors’ best efforts, the present study presents several limitations. The results of this typology of study method are highly dependent on the contact time chosen for the study. An antimicrobial surface that “passes” at 1 h of contact is almost certainly more effective than a surface that “passes” at 24 h of contact.
Conclusions
According to the results obtained, the test item does not cause any appreciable reduction of viable microorganisms. For what concerns the analysis of the growth of S. aureus in the biofilm, it appears that the porous hydroxyapatite appears to inhibit exponential growth by inducing bacterial stasis in the early hours of contact, reaching a maximum reduction within 4 h, in this adopted experimental condition.
The growth inhibition demonstrated in vitro during the first few hours of PHA cranioplasty contact with the Staphylococcus aureus microorganism may contribute to the inhibition of biofilm formation, thus reducing cases of infection in patients receiving the PHA product. Further tests are needed to investigate this aspect related to biofilm inhibition.
Supplemental Material
sj-pdf-1-jbf-10.1177_22808000241311389 – Supplemental material for Cranioplasty infection in porous hydroxyapatite: Potential antibacterial properties
Supplemental material, sj-pdf-1-jbf-10.1177_22808000241311389 for Cranioplasty infection in porous hydroxyapatite: Potential antibacterial properties by Ismail Zaed, Corrado Iaccarino, Francesca Faedo, Laura Grillini, Elisabetta Galassi, Alessandro Dotti, Angelo Nataloni, Francesca Carolina Mannella and Andrea Cardia in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.