Abstract
Objectives:
To review the progress of Platelet Rich Fibrin (PRF) as a biomaterial in dentistry and to highlight its promising application as a safe and biocompatible autologous platelet concentrate. Publications were searched in GeenMedical, X-mol, GoogleScholar, and PubMed from October 2024 with no language restrictions. The literature was searched for relevant databases and journals on the use of PRFs in dentistry up to October 2024, and the inclusion criteria included randomized controlled trials, clinical trials, case series, and systematic reviews.
Conclusion:
PRF is a second-generation platelet concentrate that is sourced from oneself, has fewer adverse effects, and is simple and safe to prepare. These materials include growth factors and fibrin scaffolds, which are extensively utilized in regenerative medicine. By outlining PRF, we found that good results can be achieved when PRF is used to treat these conditions.
Clinical significance:
The application of PRF in dentistry is widespread, particularly in periodontal soft and hard tissue regeneration, oral lichen planus, and pulpal regeneration. This article reviews the background, classification, and preparation methods of PRFs, along with their dental applications. We anticipate further research on various PRF derivatives in the future, which will significantly improve the utilization of PRF in oral applications and offer fresh insights for diagnosing and treating oral diseases.
Introduction
Platelet concentrates, as a natural autologous source of human peripheral blood, have been widely used in various fields of medicine for more than 20 years. It mainly includes platelet-rich plasma (PRP), platelet-rich fibrin (PRF), and concentrated growth factor (CGF). The preparation parameters, applications, and forms of the resulting platelet derivatives are shown in Table 1. In 1954, Kingsley introduced PRP, which has been utilized in regenerative medicine for over 2 decades. It was initially developed with an anticoagulant to prevent rapid coagulation of blood before centrifugation. 1 Autotransplantation is considered the standard in regenerative medicine because of the absence of immune rejection. 2 PRP, a first-generation platelet concentrate, is enriched with platelets, leukocytes, and growth factors 3 PRP is activated by thrombin and release various growth factors through degranulation. Alpha particles, which are spherical or elliptical structures encapsulated by a unit membrane, deliver growth factors and cytokines that may enhance healing. They contain numerous bioactive mediators. 4 PRP is applied broadly for treating ulcers, burns, muscle repairs, bone disease, and post-operative tissue recovery. 5 The basic principle of PRP treatment is the application of concentrated platelets at the site of injury to enable tissue repair by releasing a variety of active factors, thereby initiating a hemostatic cascade reaction, the formation of new connective tissue and blood flow reconstruction. 6 Nevertheless, the activation of platelets and the formation of a fibrin clot in PRP require the inclusion of an anticoagulant, such as bovine thrombin. However, this addition can provoke adverse immune responses. 7 It has been noted that bovine thrombin contains anticoagulant factor V, which increases the risk of life-threatening coagulopathy, thereby poses a threat to life. 8 Moreover, anticoagulants can impede wound healing by inhibiting blood clot formation, a crucial step in the natural wound-healing process. 1
Classification, characterization, centrifugation parameters, and forms and applications of platelet derivatives.

For these reasons, Choukroun proposed PRF in 2001, a second-generation platelet concentrate that is safer and simpler to prepare without the need for anticoagulants. 9 Widely adopted in the 1990s, PRF is enriched with growth factors and cytokines essential for tissue regeneration and exhibits good biocompatibility and regenerative capabilities. 10 PRF offers the advantages of PRP but with a more prolonged and sustained release of growth factors. Eizaburo Kobayashi et al. 11 compared growth factor release among three platelet concentrates—PRP, PRF, and A-PRF—and reported that PRP facilitates rapid delivery by releasing high levels of growth factors at an early stage, whereas PRF and A-PRF demonstrate slower, sustained release. This is attributed to the higher leukocyte content in PRF, which supports the extended release of growth factors. 12 Unlike PRP, which requires an anticoagulant during preparation to ensure platelet activation and fibrin formation, PRF itself contains fibrin, which promotes wound healing and considerably reduces the biochemical handling of blood and its associated risks. 13 While PRP involves a two-step centrifugation protocol, Monika Prokurat and other scholars 14 have reported three methods for preparing PRP: (i) With respect to the differences present in each component of the blood, the blood is centrifuged, and after centrifugation is complete, the denser material is deposited at the bottom of the tube, while the less dense material is pushed up to the uppermost level. (ii) Two-step centrifugation results in a more concentrated PRP, in which whole blood is first centrifuged, the PRP is separated from the remaining layers, and only the most concentrated part of the PRP border is collected. (iii) Whole blood collection from the patient is collected in a test tube containing an anticoagulant substance; this method allows the PRP to be obtained directly from the patient’s blood without the need to separate other blood components. PRP is separated from other components of the blood by centrifugation or filtration whereas Choukroun’s method for PRF involves only one centrifugation and avoids the use of anticoagulants during preparation.
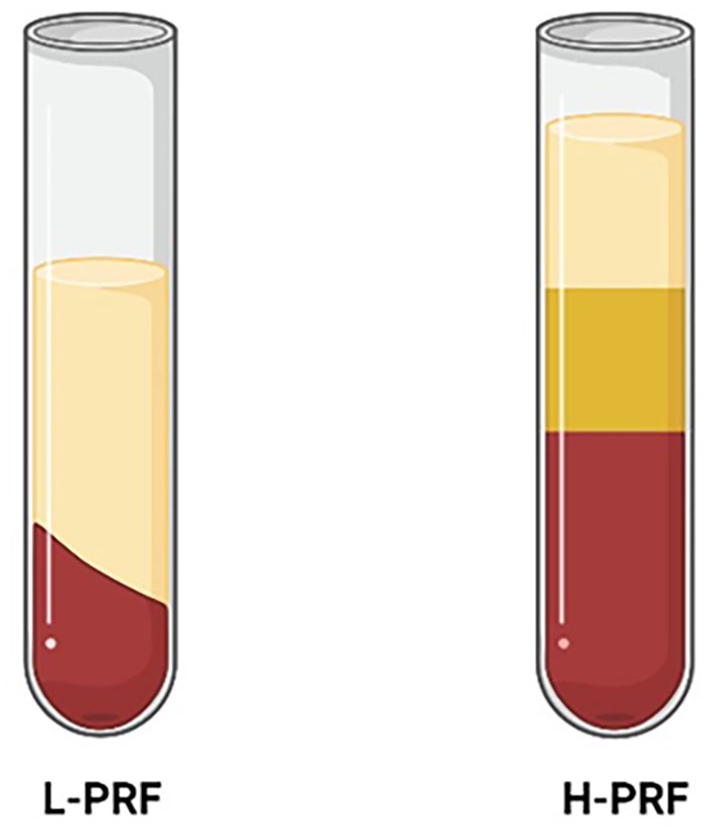
PRF has garnered increasing attention since its introduction in 2000 and has been utilized in regenerative medicine for over 20 years. It comprises platelets, white blood cells, fibrin, and growth factors. Angiogenesis stimulation and tissue healing promotion can be induced by it, and its preparation process does not necessitate the addition of anticoagulants, rendering it safe, simple, and convenient. 15 Currently, a variety of PRF derivatives have been investigated by scholars, including Leukocyte-Platelet-Rich Fibrin (L-PRF), Advanced Platelet-Rich Fibrin (A-PRF), Injectable Platelet-Rich Fibrin (I-PRF), Titanium Platelet-Rich Fibrin (T-PRF), Horizontal Platelet-Rich Fibrin (Horizontal-PRF), Concentrated Platelet-Rich Fibrin (C-PRF), and Platelet-Rich Fibrin Mixture (Alb-PRF). PRF derivatives are commonly used in regenerative medicine, especially in periodontal soft and hard tissue regeneration, alveolar ridge preservation and sinus floor lifting. 16 Blood was obtained by centrifugation of whole blood, and after centrifugation, the blood was divided into three layers: the PPP layer (supernatant), the PRF layer, and the red blood cell layer. 11 Under the influence of centrifugal force, erythrocytes with comparatively greater masses settle toward the bottom of the test tube, whereas leukocytes, platelets, and plasma, owing to their lower masses, are propelled upward. 3 PRF, which is characterized by biocompatibility and a wealth of growth factors, offers a fibrin matrix. Unlike PRP, the fibrin matrix of PRF acts as a three-dimensional scaffold for leukocytes and platelets, as well as their released products, enabling delayed release of their contents to facilitate improved wound healing. 17
Platelet-rich fibrin
Biological properties
PRF includes platelets, white blood cells, and a fibrin scaffold. Platelets, which are pivotal in PRF, undergo activation upon interaction with endothelial cells following tissue injury, prompting the release of growth factors (Figure 1). 2
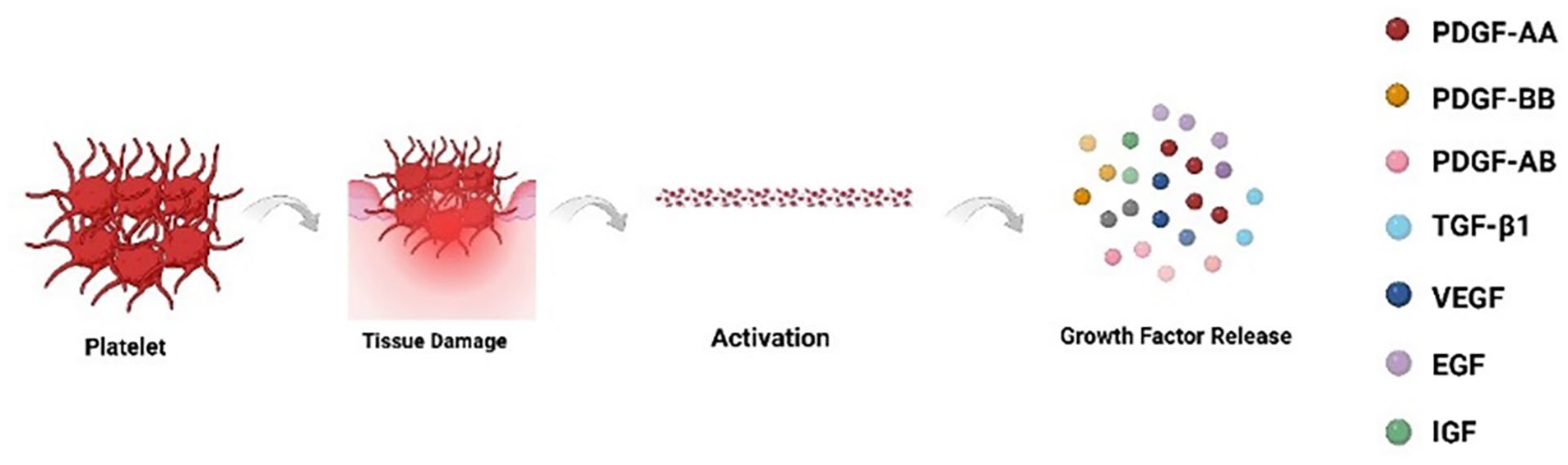
Platelets undergo activation upon interaction with endothelial cells following tissue injury, prompting the release of growth factors. 2
This triggers a sequence of healing responses. The normal process of wound healing includes hemostasis, inflammation, tissue formation, and tissue remodeling, and platelets can initiate the healing process by secreting various growth factors. 18 Two key factors in tissue regeneration are growth factors and fibronectin as a scaffold. 19 Growth factors, peptide molecules that occur naturally, function as powerful biological mediators, regulating wound healing processes and fostering tissue regeneration. 2 The regenerative potential of PRF relies heavily on its ability to release growth factors. In addition to activated platelets and leukocytes, fibrin scaffolds serve as repositories for growth factors, ensuring continuous and prolonged release. 20 The seven key growth factors in the blood are the platelet-derived growth factor (PDGF) family, which includes PDGF-AA, AB, BB, etc., transforming growth factor (TGF-β1), vascular endothelial growth factor (VEGF), insulin-like growth factor (IGF), epidermal growth factor (EGF), These growth factors are pivotal for tissue regeneration. The PDGF family includes AA, BB, and AB. This protein acts as a potent proangiogenic growth factor with diverse roles, including inducing neovascularization, assisting in bone healing, and encouraging stem cell division. Additionally, it stimulates cell proliferation and collagen synthesis in fibroblasts. It plays an important role in the wound healing process. 21 It is the first growth factor present in wounds and initiates connective tissue healing by promoting collagen and protein synthesis. 22 TGF-β1 23 plays a vital role in periodontal tissue regeneration and is recognized as the most potent cytokine in fibrosis. TGF-β1 is an important regulator of endothelial cell growth. 23 It can promote collagen synthesis and angiogenesis, prevent collagen breakdown, and promote tissue regeneration. 24 It is often referred to as a fibrosis drug. PRF serves as a rich source of TGF-β activity, with proteomic analyses, such as those conducted by Kargarpour, 23 identifying TGF-β as a primary growth factor responsible for gene activation in oral fibroblasts. Its effects include inducing the nuclear translocation of Smad-3, increasing Smad3 phosphorylation, and closely influencing tissue regeneration. VEGF 25 acts as a vasodilator, enhancing microvascular permeability. It has been shown to be the most potent growth factor for promoting tissue angiogenesis. VEGF also plays a significant role in tissue remodeling, with studies demonstrating increased new bone formation when VEGF is incorporated alone into various bone biomaterials. It increases collateral vessel formation, vasodilation, and neuroprotective effects. 26 In addition, it promotes endothelial cell proliferation, differentiation, and migration. 27 IGF is a major mediator of growth hormone, a growth factor involved in mediating cell proliferation, differentiation and apoptosis. 24 It can regulate the growth and function of skeletal muscle and has an anabolic effect on growth. 28 particularly when combined with other growth factors, may enhance bone wound healing. It increases bone turnover in individuals with low bone density, regulates proliferation and differentiation across most mesenchymal cell types, and offers cytoprotective effects. IGF can also synergize with other growth factors, such as PDGF, to promote periodontal bone regeneration. EGF 24 stimulates epidermal regeneration, accelerates wound healing by stimulating keratinocyte and dermal fibroblast proliferation, and amplifies the effects of other growth factors in the final stages of production. Growth factors in platelet derivatives and their functions in Table 2.21,22,24,28
Types of growth factors and their functions created by the author.

Leukocytes play a vital role in the wound healing process, particularly in antimicrobial activity and immune response modulation. Like platelets, they also release multiple growth factors to facilitate tissue healing and regeneration. 29 Fibrin scaffolds, three-dimensional mesh structures formed during centrifugation, are created gradually and naturally through auto-thrombin action. They can encapsulate growth factors and cytokines, enabling them to function effectively. 30
Evolution of the PRF
PRF originates from the patient’s own body and does not contain anticoagulants. However, it contains leukocytes and platelet fibrin, which contain growth factors and cytokines involved in key stages of the healing process, 31 making it autologous and thereby avoiding immune rejection. It maintains a stable biological composition, activity, and three-dimensional structure. In recent years, several derivatives have emerged and are categorized into seven types on the basis of their preparation methods: L-PRF, A-PRF, I-PRF, T-PRF, H-PRF, C-PRF, and Alb-PRF (Figure 2).

Classification of PRF derivatives. PRFs can be categorized according to equipment and preparation method.
Leukocyte-rich platelet fibronectin
The first proposed derivative of PRF was leukocyte-rich platelet fibronectin (L-PRF), which is a fibrin matrix that allows growth factors and cytokines to recombine, 32 which remains the most common and originally developed form of PRF derivative. It possesses immunological and antimicrobial properties that support regeneration and healing processes effectively. L-PRF has been successfully utilized as an adjunct to enhance tissue healing and plays a crucial role in the healing process overall. 33 Its applications span widely, including use in maxillary sinus floor lifting and extraction socket healing. It stimulates wound healing and regeneration, 16 and the 3D structure of the L-PRF membrane has very good elasticity, density, flexibility, and strength. These characteristics can promote not only wound healing and repair but also soft and hard tissue regeneration. 32
Advanced platelet-rich fibrin
As most leukocytes and platelets in L-PRF tend to settle at the bottom of the centrifuge tube, with some even penetrating the erythrocyte layer. 3 To ensure a more even cell distribution, the concept of low-speed centrifugation has been proposed, with the main objective of improving the distribution and quality of growth factors. 34 A-PRF is a modified form of L-PRF obtained by low-speed centrifugation. Reducing the centrifugation time results in a more homogeneous distribution of cells in addition to an increased release of growth factors. 35 Compared with L-PRF, A-PRF offers a greater concentration of growth factors, which is achieved by extending the centrifugation time and reducing the speed while building upon the principles of leukocyte-rich platelet fibrin (L-PRF). This method enhances the presence of neutrophils beyond the PRF clot. In a study by Kobayashi et al., 11 when L-PRF was compared with A-PRF, A-PRF exhibited greater growth factor release, likely because of its higher leukocyte content. A-PRF is extensively applied in regenerative dentistry. In research by Gupta et al., 36 A-PRF was used in alveolar sockets post-mandibular obstruction third molar extraction, showing enhanced efficacy and healing potential compared with those of control sites undergoing natural healing. Thus, A-PRF has the ability to increase the healing potential of both soft and bone tissues in extraction sockets. Compared with standard L-PRF, A-PRF has a greater concentration of growth factors, an increased number of cells, and a looser three-dimensional structure. 37
Injectable platelet-rich fibrin
I-PRF has become a versatile biomaterial, a non-clotting version of liquid PRF obtained by low-speed centrifugation. It is an injectable form that results in improved wound healing. 38 I-PRF is a fluid form of PRF that remains viable for 10–15 min. It can be utilized independently or in combination with various biomaterials. 39 Studies conducted by scholars such as Miron, 39 who compared PRP and I-PRF, have highlighted their distinct properties. While PRP triggers greater cell proliferation, I-PRF induces higher levels of cell migration and expression of growth factors. As growth factors play crucial roles in tissue regeneration, investigations by Karde, 40 who compared I-PRF with other platelet concentrates, have demonstrated its superior antimicrobial efficacy. Thus, I-PRF shows enhanced regenerative potential, increases the presence of growth factors within wounds, and facilitates wound healing. I-PRF contained the highest concentration of platelets and leukocytes of all the solid-based PRF matrices. Its growth factors promote tissue regeneration. 41
Titanium platelet-rich fibrin
To mitigate the adverse effects associated with the use of glass-coated plastic tubes, researchers have explored titanium-prepared platelet-rich fibrin. 42 This novel platelet concentrate is prepared by activating titanium tubes, resulting in a robust and dense fibrin network with prolonged absorption compared with L-PRF. Platelets may be more effectively activated via titanium tubing than via silica activation, which contributes to the unique feature of T-PRF, which enhances its biocompatibility. 43 T-PRF serves as a superior autologous healing material, requiring only the patient’s blood for preparation. 44 Studies conducted by Bhattacharya et al., 45 which utilized immunohistochemistry to evaluate and compare cells in PRF membranes prepared from titanium and silica glass tubes, revealed that T-PRF results in a denser fibrin network and cellular structure than L-PRF. These findings suggest that titanium-prepared PRF offers advantages over PRF prepared from silica-glass tubes, making it a preferred choice in periodontal regeneration. Esra Ercan and other researchers have demonstrated its importance as an autologous drug carrier with high loading and release of Doxy, making it a promising treatment for periodontitis and peri-implantitis. 42
Horizontal platelet rich fibrin
H-PRF is a new platelet concentrate prepared by horizontal centrifugation that results in a more homogeneous distribution of cells and an average clotting time of 71 min for pure liquid H-PRF. 46 H-PRF is superior to L-PRF in terms of better platelet and leukocyte concentrations, greater structural stability, more uniform cellular distribution, and the ability of H-PRF to prolong the release of various growth factors. 47 Badakhshan et al. 48 compared the antimicrobial properties and wound healing capabilities of H-PRF with those of two groups of commercially available collagen membranes. Research has revealed that H-PRF is the most effective long-term bacterial barrier and the most favorable epidermal growth factor. These findings suggest that in the future, the use of H-PRF membranes alongside GBR surgery could reduce postoperative infections and expedite wound healing processes. In addition, H-PRF can promote tissue regeneration and prevent degradation, and studies by scholars such as Zheng 49 have shown that H-PRF is a homogeneous, moldable gel. The H-PRF gel obtained by heating at 75°C for 10 min was characterized by a short solidification time and good rheological properties. H-PRF offers elevated levels of white blood cells and platelets, indicating advantages in tissue regeneration. Research by Zhang et al. 50 revealed that horizontal centrifugation can increase the number of immune cells in H-PRF by up to fourfold. Compared with L-PRF, H-PRF has superior antimicrobial properties and promotes bone regeneration, benefiting both hard and soft tissues.
Platelet-rich fibrin mixture
Recently, researchers have reported that ordinary PRF only lasts for 2–3 weeks of degradation in vivo, so they explored a PRF obtained by heating, called Alb-PRF, which can degrade in vivo for up to 4–6 months. 51 Alb-PRF as a stable, opaque plastic film utilized as a soft tissue barrier. 52 This biomaterial shows promise for application as a filler in oral surgery, implantology, and dermatology. Compared with other PRFs, the Alb-PRF membrane exhibits excellent biocompatibility and unique properties. It offers an extended maintenance time and an in vivo degradation period of 4–6 months. Additionally, its composition, which contains albumin, enables heat treatment to prolong the absorption properties of PRF. In a study led by Gheno et al., 53 three types of membranes—L-PRF, H-PRF, and Alb-PRF—were implanted into the backs of nude mice to observe wound healing rates and degradation characteristics. The results indicated that while the H-PRF and L-PRF membranes presented varying degrees of absorption after 21 days, Alb-PRF remained volumetrically stable throughout the observation period. These findings suggest that Alb-PRF holds significant potential as a biobarrier membrane in GBR surgery and as a biofiller material in the future.
C-PRF
C-PRF is known as concentrated platelet-rich fibrin, and a modified centrifugation protocol was used to obtain C-PRF with a liquid consistency and a high concentration of platelets and growth factors. It is rich in growth factors, and it has been shown that it has a unique advantage over C-PRF, namely, the ability to sustainably release growth factors, and that it works synergistically with bone allografts. 54 It is known as concentrated platelet-rich fibrin and is abundant in growth factors. In research conducted by Masako Fujioka-Kobayashi et al., two protocols, I-PRF and C-PRF, were implemented to compare the levels of seven key growth factors in the blood. The findings revealed a significant increase in growth factor levels in C-PRF throughout the study duration, particularly highlighting the release of PDGF-AA, TGF-β1, and EGF. The C-PRF protocol results in a threefold increase in growth factor release. 55
Preparation of PRF derivatives and factors affecting their preparation
Preparation of PRF derivatives
Leukocyte-rich platelet fibrin
L-PRF: L-PRF preparation involves the collection of 9–10 mL of blood in a blood tube. Initially, centrifugation was typically performed at 3000 rpm/10 min. A specially designed tube was used in the preparation process, and the L-PRF compression clot formed a fibrin matrix membrane. 56 However, in recent years, it has been commonly conducted at 2700 rpm for 12 min. Following centrifugation, the blood separates into three layers: the top layer comprises platelet-poor plasma (PPP), the middle layer consists of L-PRF, and the bottom layer contains erythrocytes. The Intra-Spin L-PRF (Intra-Lock Inc., USA) device is predominantly utilized for this preparation process (Figure 3).
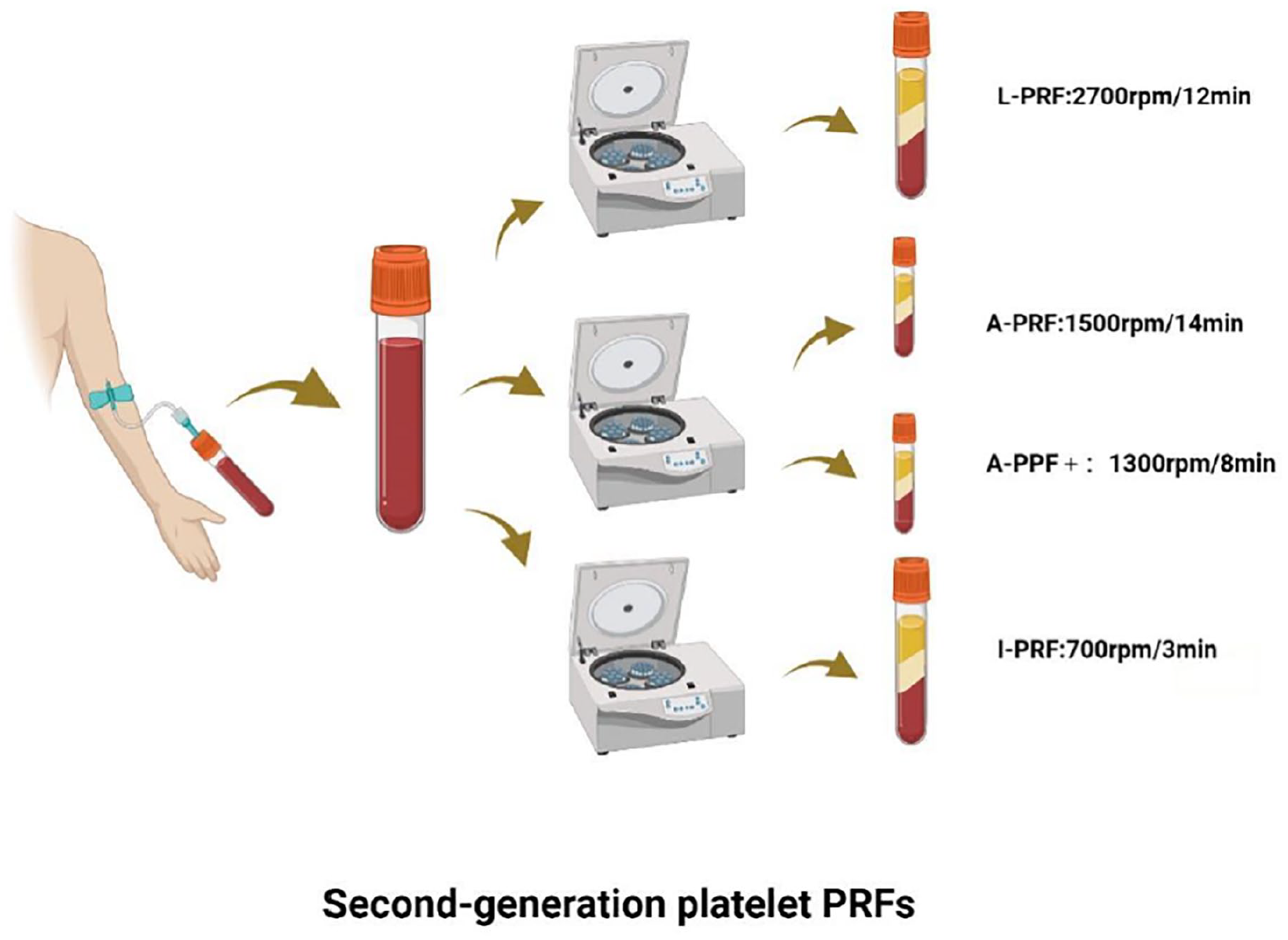
Preparation of second-generation PRF derivatives, including L-PRF, A-PRF, and I-PRF.
Advanced platelet-rich fibrin
A-PRF: A-PRF is a modification of L-PRF that was developed with an extended centrifugation time and a reduced centrifugation speed. To prepare A-PRF, 9–10 mL of blood is collected in a blood tube and then centrifuged at 1500 rpm for 14 min. Compared with L-PRF, A-PRF results in higher concentrations of growth factors, higher cell numbers, and denser fiber structures. The concentrations of growth factors, cells, and platelets were further increased in A-PRF+ with the preparation parameters of 1300 rpm/8 min. 37 Following centrifugation, the blood separates into three layers: the uppermost layer is the PPP layer, the middle layer is the A-PRF/A-PRF+ layer, and the bottom layer is the erythrocyte layer. Compared with L-PRF, A-PRF has greater regenerative potential and is commonly employed in periodontal regeneration and implant surgery. 57
Injectable platelet-rich fibrin
Prior to 2014, PRF was only available in solid form in the clinic, after which there was a push for the development of I-PRF, which sustains the release of a range of growth factors and induces the expression of type I collagen. 13 The lower centrifugal force and shorter centrifugation time of I-PRF are based on leukocyte-rich platelet fibrin (L-PRF). For the preparation protocol, 9–10 mL of blood was collected in a centrifuge tube and centrifuged at 700 rpm for 3 min. 41 The centrifuged blood was partitioned into three layers: the uppermost layer comprised the PPP layer, the middle layer constituted the I-PRF layer, and the bottom layer consisted of the erythrocyte layer. Compared with L-PRF, this I-PRF layer presented a greater number of cells and a greater concentration of growth factors.
Titanium platelet rich fibrin
T-PRF: A total of 10 mL of blood was withdrawn from the patient’s anterior elbow vein and transferred to a sterile medical-grade titanium tube. The preparation parameters included centrifugation at 3500 rpm for 15 min. The prepared product was divided into three layers: the uppermost layer was the PPP layer, the middle layer was the T-PRF clot, and the lowermost layer was the RBC layer. The T-PRF clot can be compressed to form a film. 58 In T-PRF, the presence of titanium dioxide (TiO2) on the inner surface of the blood collection tube facilitates the activation of platelets, leading to the formation of T-PRF. 45 This suggests that titanium is more hemocompatible than glass and produces more polymerized fibrin (Figure 4). 59

Preparation of titanium-rich platelet fibrin. It uses Grade IV titanium tubing instead of glass tubing.
Horizontal platelet rich fibrin
H-PRF: Horizontal platelet-rich fibrin is a new PRF derivative. The preparation was carried out by centrifugation at 700 ×g for 8 min using a horizontal centrifugation unit. 47 After centrifugation, the blood was partitioned into three layers: the uppermost layer consisting of the PPP layer, the middle layer comprising the H-PRF layer, and the bottom layer containing erythrocytes. H-PRF was created utilizing a horizontal centrifugation unit, ensuring an even distribution of cells throughout all layers (Figure 5).

Preparation of horizontal platelet-rich fibrin. This PRF derivative was prepared by horizontal centrifugation.
Platelet-rich fibrin mixture
Alb-PRF: To overcome the rapid degradability of platelet concentrates and to better maintain their stability, researchers have developed the Alb-PRF protocol. 60 The process involves initial centrifugation at 700 RCF for 8 min to obtain H-PRF, followed by separation of the centrifuged blood into three layers: the top layer as the PPP layer, the middle layer as the H-PRF layer, and the bottom layer as the erythrocyte layer. The uppermost PPP layer was heated at 75°C for 10 min in a heating device to yield denatured albumin (Alb). Upon cooling to room temperature, the remaining PRF is placed in a cooling device. The cooled Alb subsequently combines with the liquid PRF via a female-female connector, resulting in Alb-PRF platelet-rich fibrin mixtures. 61 Alb-PRF has a diverse range of applications and has shown promising efficacy in socket filling post-extraction as well as in soft tissue regeneration. The Alb-PRF mixture in a glass bowl naturally forms an Alb-PRF collagen film (Figure 6).
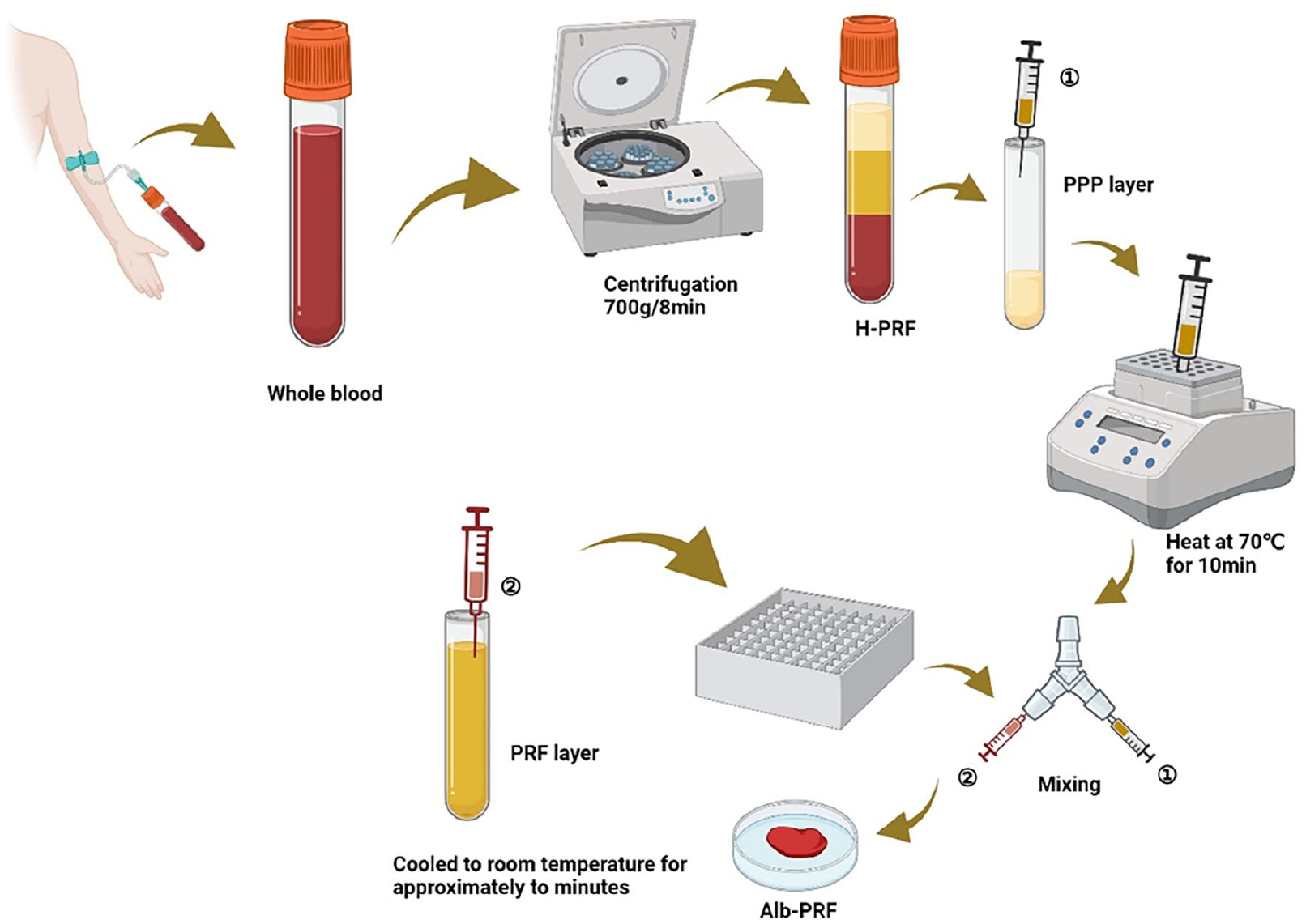
Preparation of Alb-PRF. It is a mixture.
Concentrated platelet-rich fibrin
C-PRF: Of blood from volunteers, 10 mL was collected and centrifuged at 2000 ×g for 8 min, after which 0.3–0.5 mL of hematocrit brown-yellow layer was collected as C-PRF. 62 The traditional I-PRF protocol boosts platelet yield by 2–3 times and leukocyte yield by 50%.Compared with the conventional I-PRF regimen, C-PRF is centrifuged at higher RCF values, especially in the hemoconcentrated brown-yellow layer, where higher concentrations of platelets and leukocytes are collected, resulting in about 10-fold higher concentrations of platelets and leukocytes and a 3-fold increase in growth factor release (Figure 7). 10
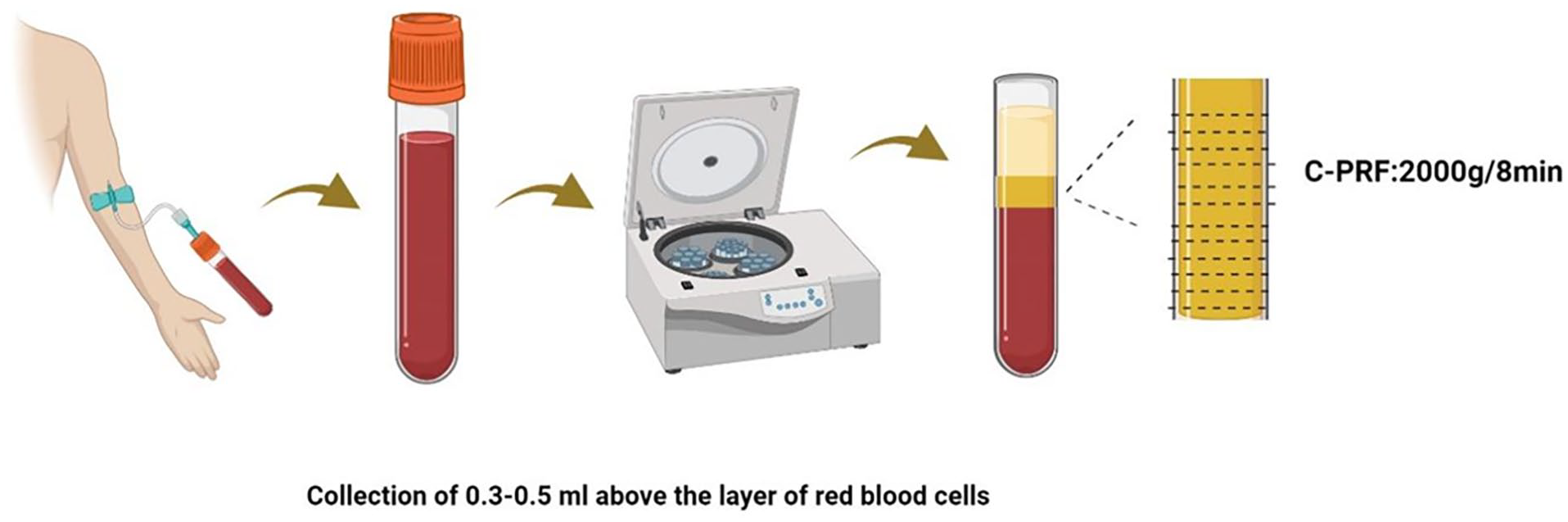
Preparation of C-PRF. Platelet-rich fibrin was concentrated, forming a gray layer measuring 0.3–0.5 mL above the erythrocyte layer.
Effect of preparation conditions on PRF derivatives
Effect of the centrifugal tube material on the PRFs
The nature of the PRF liquid or solid depends on the nature and chemistry of the tube used to draw the blood. The centrifuge tubes utilized for PRF preparation are categorized as either hydrophilic or hydrophobic. Hydrophilic materials, such as glass, silica-coated plastic, or titanium, facilitate the formation of solid clots. On the other hand, hydrophobic materials such as polyethylene terephthalate (PET) result in liquid PRF. 63 PRF derivatives are predominantly crafted using glass tubes, which effectively induce fibrin clot formation by triggering the coagulation cascade reaction upon blood contact with the glass surface. However, in recent times, researchers have begun investigating centrifuge tubes containing silicone, as major medical device manufacturers have ceased glass tube production, leading to a shortage. In addition to the risk of glass breakage in glass tubes, 56 Tsujino et al. 64 explored the preparation of PRF using centrifuge tubes containing silicone. SEM revealed that before centrifugation, there was overall uniform blurring of the wall of the tube, and some spots were dense. Following centrifugation, the wall of the tube became transparent, indicating the presence of silica particles in the prepared PRF product. However, silica poses health risks and is not advised for use. 65 Consequently, researchers have pursued alternative materials for centrifuge tubes. They reported that titanium-prepared platelet-rich fibrin (T-PRF), a novel platelet concentrate, yielded clots in titanium tubes similar to those observed in glass tubes. Compared with centrifuge tubes containing silicone, titanium tubes offer superior biocompatibility. 43 Titanium-containing tubes show better osseointegration, hemocompatibility, and platelet activation than tubes made of silica and histologically present a stronger fibrin network, which promotes tissue regeneration. 66
Impact of centrifugal devices on PRFs
Centrifugal devices are employed to centrifuge blood, with variations in rotor radii, angles, and RCF values across different devices. To investigate potential effects on the PRF product, Miron et al. 67 compared two PRFs—one at high speed and one at low speed—using three different centrifugation devices: the IntraSpin Device (IntraLock), the Duo Quattro (PRF process), and the Salvin (Salvin Dental). They discovered that the material of the centrifuge tubes had a significantly greater influence on the final size of the PRF clot than did the centrifuge device itself.
Effects of centrifugation mode on PRFs
Depending on the position of the rotor, the preparation of PRFs involves fixed-angle centrifugation and horizontal centrifugation. In fixed-angle centrifugation, the tube is inclined to the ground plane, whereas in horizontal centrifugation, it runs parallel to the ground. This distinction 25 : PRF prepared through fixed-angle centrifugation has an uneven cell distribution in layers, with the plasma and erythrocyte layers being tilted or “angularly” separated. Conversely, horizontal centrifugation results in more uniform PRF delamination. Compared with fixed-angle centrifugation units, horizontal centrifugation units require only 2/3 of the time needed for PRF preparation. 68 Within the plasma layer, PRF horizontal centrifugation devices accumulate up to four times as many leukocytes as fixed-angle centrifugation devices do. Currently, most PRFs are prepared via fixed-angle centrifugation, which is limited by cell migration to the bottom of the tube, resulting in uneven distribution. Conversely, horizontal centrifugation can produce fibrin membranes with more stable structures capable of the sustained production of growth factors and anti-inflammatory cytokines. 69 Studies have shown that horizontal centrifugation devices induce greater release of growth factors. Horizontal centrifugation results in a greater concentration of platelets and leukocytes, a greater ability to separate cells, and less shear force on cells (Figure 8). 70
Fixed-angle centrifugation versus horizontal centrifugation.
Influence of the centrifugal parameters on the PRFs
Centrifugation parameters, such as speed (rpm) and duration, vary across PRF preparation methods. The fundamental PRF, leukocyte platelet-rich fibrin (L-PRF), is produced through fixed-angle centrifugation for 12 min at 2700 rpm. Recently, low-speed centrifugation has led to the use of injectable platelet-rich fibrin (I-PRF), a liquid that can be used alone or with other biomaterials. Compared with L-PRF, I-PRF is obtained by centrifugation at 700 rpm for 3 min, with reduced force and a shorter time. Research by Miron et al. 67 indicated that I-PRF contains higher growth factor concentrations than L-PRF does. Furthermore, advanced platelet-rich fibrin (A-PRF) is achieved by reducing the speed and increasing the centrifugation time at 1500 rpm for 14 min, whereas A-PRF+ can be obtained by centrifugation at 1300 rpm for 8 min. 37 A-PRF offers more platelets and leukocytes, with a loosely structured fibrin matrix favoring growth factor release over L-PRF. Studies by Ramos et al. 71 revealed A-PRF’s superior chemical properties and degradation resistance compared with L-PRF, particularly in pain management. Owing to some limitations of glass tubes as well as silicone-containing tubes, scholars have explored titanium-containing centrifuge tubes. With respect to the preparation parameters of T-PRF, centrifugation at 3500 rpm for 15 min has been suggested. Some scholars reported that C-PRF was obtained by centrifugation at 2000 ×g for 8 min, and compared with I-PRF, C-PRF has a 10-fold higher concentration of white blood cells and platelets. H-PRF was prepared via centrifugation at 700 ×g for 8 min via a horizontal centrifuge, which is superior to the PRF derivatives prepared via fixed angle centrifugation. Alb-PRF was prepared on the basis of H-PRF by heating. Among the above PRF derivatives, L-PRF, A-PRF/A-PRF+, and T-PRF are in solid form (clots or membranes); I-PRF and C-PRF are in liquid form; and Alb-PRF is in gel form. 70
Effect of time on PRF
Several factors, including centrifugation speed, centrifugation method, and tube material, influence the quality and size of the PRF product. In recent studies, researchers have expanded their investigations and highlighted the importance of time 72 in the efficacy of PRF derivatives. Robin et al. 73 examined the impact of the time interval between blood collection and centrifugation on PRF derivatives. This experiment assumed that centrifugation immediately after blood collection was T0 and that centrifugation 1–6 min after blood collection was T1–T6; moreover, the authors concluded that the mass of PRF membranes usually decreases with increasing delay time. The largest PRF membranes were obtained when centrifugation occurred immediately after blood collection, whereas the smallest membranes resulted from a 6-min delay (T6) before centrifugation, indicating that delaying the process reduces the membrane size. Thus, to optimize the benefits of PRF, blood should be centrifuged promptly after collection. In a similar study, Miron et al. 74 reported that longer delays before centrifugation led to smaller PRF membranes. A slight reduction was noted at 90 s post blood draw, whereas a significant reduction occurred at 120 s, suggesting an optimal window of about 60–90 s between blood draw and centrifugation initiation for standard-sized PRF membrane production. Additionally, Castro et al. 75 investigated the timing before and after blood centrifugation after collection, revealing that shorter intervals between centrifugation and blood collection yielded larger PRF and derivative membranes, whereas longer intervals resulted in smaller membranes. Centrifuging blood within 60 s of collection yielded the best results. In summary, immediate centrifugation after blood collection yields larger PRF membranes suitable for clinical applications.
PRF in dentistry
PRF, derived from autologous blood through centrifugation, is biocompatible. It has been applied in disease diagnosis and treatment, either alone or in conjunction with other materials, and is administered via injection or placement on skin and mucous membranes, among other methods, to release diverse growth factors. Its utilization in treating conditions such as pulpal tissue regeneration, 76 implant alveolar ridge site preservation, 77 and periodontal soft tissue regeneration 78 has been extensively researched. For PRF applications in the dental field, see Figure 9. Details about the specific role of PRFs in the oral cavity, their groupings, and their morphology at the time of action are shown in Table 3.
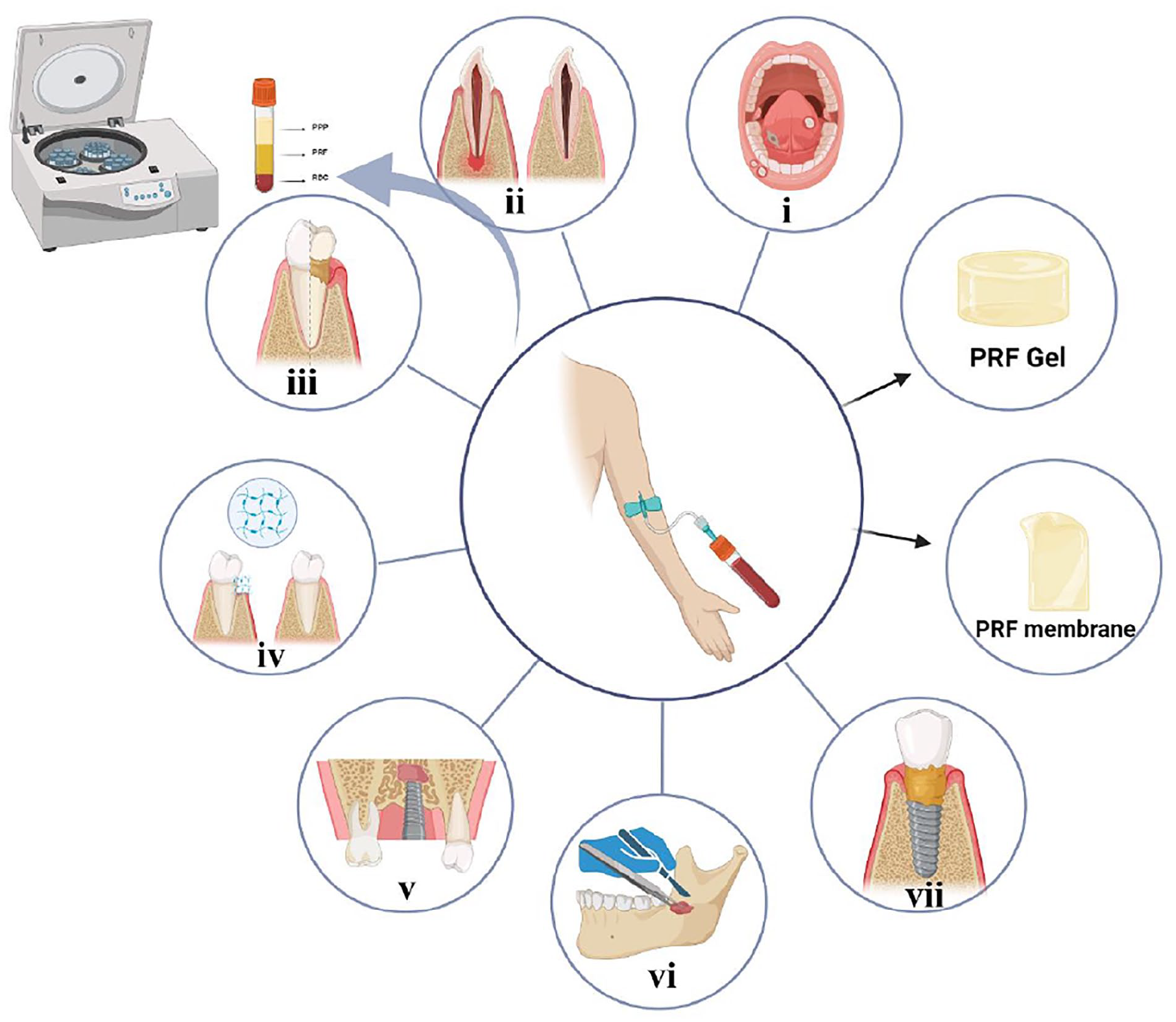
PRF in dentistry. PRF is used in various areas of dentistry in the form of films and gels. i: oral lichen planus, oral ulcer; ii: tooth pulp regeneration; iii: periodontal soft tissue regeneration; iv: periodontal bone regeneration; v: maxillary sinus lift; vi: alveolar ridge preservation; vii: peri-implantitis.
PRF in periodontics
PRF in periodontal soft tissue regeneration
Periodontitis is a chronic inflammatory disease that affects the hard and soft tissues of teeth and, if left untreated, can eventually lead to tooth loss. 79 Periodontal tissue comprises the gingiva, periodontal membrane, alveolar bone, and dental bone, 80 which securely affixes the tooth to the alveolar bone, resists occlusal forces, and provides a seal between the oral mucosa and the hard dental structures. The regeneration of periodontal tissue involves the renewal or rebuilding of damaged regions to restore the morphology and functionality of these structures. Insufficient periodontal support can lead to gum recession, tooth mobility, a reduced aesthetic appearance, and other issues. Gingival recession (GR) refers to the retraction of the gingival margin away from the enamel bone junction toward the root apex, including recession of the gingival papillae and exposure of the tooth root to the oral environment. 81 The primary objective of periodontal therapy is to regenerate lost tissues, establish new connective tissue attachments, and, crucially, form new alveolar bone.
Current techniques for addressing gingival recession and tissue restoration include surgical methods and bioactive materials. Surgical methods include guided tissue regeneration (GTR), 82 open flap debridement (OFD), and subepithelial connective tissue grafts combined with a coronally advanced flap (SCTG + CAF), 83 among others. Bioactive materials include enamel matrix derivatives (EMD), 52 platelet-rich fibrin (PRF), and demineralized freeze-dried bone allografts (DFDBA).
The GTR procedure is used to rebuild the periodontal tissue around the teeth by separating the gingival epithelium from the alveolar bone using a closed barrier membrane. The application of the membrane to the operative area prevents connective and epithelial tissue from migrating through it, allowing the roots to regenerate and divide to produce new periodontal support tissue. 84 Despite the apparent success of the GTR procedure, some studies have shown a risk of root resorption. 84 SCTG + CAF is regarded as the optimum approach for treating gingival recession; surgical approaches can yield a degree of periodontal soft tissue augmentation. However, the limitation of the surgical approach is the high level of trauma. The process might induce fear in patients, and autografted soft tissues may be utilized at a second surgical site, often accompanied by postoperative symptoms such as pain, bleeding, psychological stress, and masticatory disturbances. 83 Consequently, recent research has indicated that soft tissue injections can increase gum recession and facilitate tissue regeneration. Thoma et al. 85 demonstrated that soft tissue injections can ameliorate gingival recession with increased patient acceptance, reduced bleeding, and pain, eliminating the need for grafts and reducing the risk of necrosis. Owing to their rich content of growth factors that enhance tissue regeneration, plasma products have recently gained widespread clinical use, and Ozsagir et al. 86 utilized microneedle injections combined with I-PRF beneath the membranogingival junction, which improved gingival thickness and the gingival biotype. Miron et al. 87 compared PRF with other treatment options for root coverage in Miller class I or II gingival recession and reported that CAF combined with PRF resulted in better root coverage. Compared with the combination of CAF alone, the combination of PRF and CAF significantly increased root coverage and clinical attachment levels.
In summary, the use of PRF in periodontal tissue regeneration is both safe and effective. The fibrin matrix in PRF, which is rich in growth factors, may serve as a novel bioscaffold carrier for future periodontal regeneration engineering alongside various regenerative techniques.
PRF in the treatment of periodontal bone defects
Periodontal disease is a chronic, multifactorial inflammatory disorder characterized by the destruction of tooth-supporting tissues, often resulting in intraosseous defects. It is considered a risk factor for various chronic systemic diseases, such as coronary heart disease and diabetes. 88 The primary manifestations include loss of clinical attachment, formation of periodontal pockets, and bleeding upon probing, leading to the degradation of supporting structures such as the periodontium, alveolar bone, and dentin. 89 The regeneration of periodontal tissues involves a balance between anabolic and catabolic processes, 90 with bone regeneration dependent on both bone formation and resorption. 91 The aim of periodontal regeneration is to rebuild damaged periodontal tissues and form new bone, including the integration of the periodontium into the alveolar bone, which begins with the deposition of osteoids. 84
Common treatments for periodontal disease include surgical techniques and the use of bioactive materials, including open flap debridement (OFD). OFDs are particularly effective for treating periodontal intraosseous defects. Abdulrahman et al. 91 compared the outcomes of PRF combined with OFDs to those of OFDs alone in treating intraosseous defects in patients with stage III periodontitis. Nine months post-operation, both treatments significantly improved clinical attachment loss and probing depth, yet the addition of PRF yielded superior results.
Despite their effectiveness, surgical methods can be invasive. Even more advanced is the use of biomaterials for controlled delivery, which function as three-dimensional scaffolds, thus creating a supportive environment for regeneration, and biomaterials can come into direct contact with living tissue without adverse reactions. 84
In recent decades, the use of biomaterials such as enamel matrix derivatives (EMDs), platelet concentrates (APCs), and amniotic membranes (AMs) has been extensively explored and applied in clinical settings for periodontal regeneration. However, materials of heterogeneous origin may elicit specific immune responses in tissues.
Zhou et al. 92 evaluated the efficacy of four biomaterials—platelet-rich plasma (PRP), platelet-rich fibrin (PRF), enamel matrix derivative (EMD), and amniotic membrane (AM)—in conjunction with demineralized freeze-dried allograft bone grafting (DFDBA) for treating periodontal intraosseous defects. The applications of PRF and PRP were effective in reducing probing depth and clinical attachment loss. Notably, PRF alone was effective in reducing gum recession. Fábio França Vieira e Silva and other scholars summarized the effectiveness of PRF compared with other regenerative treatments to treat periodontal bone defects, including PRF + OFD and PRF + metformin, and reported that compared with other treatment modalities, PRF significantly improved the regeneration of periodontal bone defects. 93 These findings suggest that PRF may be an optimal adjunct to enhance periodontal regeneration in patients with intraosseous defects.
Application of PRF in oral and maxillofacial surgery
PRF application for filling extraction sockets
Missing teeth can have a considerable impact on a patient’s chewing ability, pronunciation, and aesthetics. Most traumatized and fractured teeth cannot be saved and need to be extracted. 94 Numerous studies have shown that bone loss occurs after tooth extraction. 95 Rapid resorption of the alveolar bone occurs 3–6 months after extraction, and changes in the shape of the alveolar bone may affect the outcome of the implant restoration.
Alveolar ridge preservation 96 is a procedure aimed at halting or reducing alveolar bone resorption subsequent to tooth extraction. The objective is to preserve the morphology of the alveolar ridge through the utilization of various materials, such as bone grafts, soft tissue grafts, guided bone regeneration (GBR), and biologically active materials, with the goal of minimizing the reduction in both the coronal alveolar bone height and width. Alveolar preservation, a preventive measure, involves inserting bone substitute materials (BSMs) into the extraction socket to maintain alveolar bone integrity. 97 Common complications after tooth extraction include pain, swelling, clenching, and infection.
A variety of biomaterials have been developed to facilitate the healing of alveolar sockets. These include plasma products, PRP, PRF, and CGF, which are designed to enhance wound healing and biocompatibility. Among these, PRF is frequently chosen for its cost-effectiveness, ease of production, and availability. Gupta et al. 36 assessed the impact of advanced A-PRF versus natural healing on extraction sockets, evaluating pain, swelling, and soft tissue healing. The findings revealed significant improvements in pain reduction and soft tissue healing at A-PRF-treated sites compared with those at control sites on the third and seventh days post-extraction, which was attributed to the anti-inflammatory properties of A-PRF, which release growth factors that enhance wound repair.
Recent studies have noted that L-PRF, H-PRF, and A-PRF membranes undergo spontaneous fibrinolysis within 3 weeks post-subcutaneous implantation in immunocompromised subjects, 53 making them unsuitable for use that requires long-term volume stability. 98 To address this, researchers developed a novel PRF variant by heating the liquid platelet-poor plasma (PPP) layer, extending the reabsorption properties of the albumin gel from 2 weeks to more than 4 months. 61 Compared with traditional PRFs, Alb-PRF, a new form of platelet-rich fibrin, has enhanced biocompatibility and stability. Yang et al. 52 evaluated clinical and biochemical outcomes after mandibular third molar extraction via Alb-PRF on one side and L-PRF on the other, measuring pain, tooth closure, infection, and swelling. The results showed that while both PRF effectively reduced surgical pain, Alb-PRF significantly lessened edema after 7 days, indicating superior performance in post-extraction alveolar treatment.
In conclusion, the application of PRF has proven effective in managing extraction sockets, with heated albumin PRF (Alb-PRF) showing greater efficacy than conventional unheated PRF. This suggests substantial potential for PRF and its derivatives, such as Alb-PRF, in future applications within extraction sockets. Khan et al. 95 To evaluate the effectiveness of PRF versus DFDBA for alveolar ridge preservation in humans at different times, DFDBA and PRF placed in extraction sockets were used as an experimental group, whereas the control group was used for natural healing. Radiographic evaluation was used after extraction, 3 months after extraction, and 6 months after extraction, and the results revealed less bone resorption in the experimental group, confirming that DFDBA combined with PRF has some advantages over natural healing for alveolar preservation. Park et al. 99 divided the samples into four groups, blank, Bio-oss alone, L-PRF, and L-PRF mixed with Bio-oss, by comparing L-PRF combined with or without bone grafting materials for alveolar ridge preservation. The results in the group with L-PRF were superior to those in the group without L-PRF, demonstrating that L-PRF-filled defects produced a greater degree of parallel collagen fiber alignment. Its ability to promote alveolar socket regeneration alone is comparable to that of particulate xenografts, suggesting that it has a promising future application in alveolar socket preservation.
Application of PRF in oral implantology
PRF applied in maxillary sinus lift
The maxillary sinus is a closed cavity bordered internally by the nasal cavity, externally by the maxillary tubercle, superiorly by the orbit, and inferiorly by the alveolar bone. It is present at birth and completes its growth by the age of 18 years. 100 After extraction in the posterior maxilla, the vertical height of the alveolar bone decreases and can prevent implant placement, making maxillary sinus lift very important when implants are needed. It has been shown that a sinus floor lift using bone grafts can increase vertical bone height. 101
A prevalent surgical solution is a maxillary sinus lift, which increases bone volume and height. The blood clot formed in the sinus during this procedure facilitates bone regeneration. For posterior maxillary dental implant surgery, a minimum of 10 mm of vertical height from the crown to the sinus floor is needed. If this height is insufficient, sinus floor elevation can be performed to achieve the desired height. 102 Various techniques for lifting the maxillary sinus include sinus floor surgery, pharmacologically induced bone formation, and the use of biosynthetic materials. 103 However, the disadvantage of bone substitutes is that they do not have osteogenic or osteoinductive ability. 104
Maxillary sinus floor elevation surgery (MSFES) involves the implantation of different bone graft materials, such as bone substitutes or autologous bone, into the sinus cavity. While autogenous bone is considered the most effective for promoting new bone formation, allogeneic bone grafts are more commonly used. Simvastatin is frequently employed to elevate the sinus floor, along with biosynthetic materials such as tricalcium phosphate (TCP) and PRF. 105 Canellas et al. 106 noted that integrating bone marrow concentrate (BMAC), leukocyte- and platelet-rich fibronectin (L-PRF), recombinant human morphogenetic protein-2 (rhBMP-2), or enamel matrix derivatives into Bio-Oss® xenograft material (Geistlich Pharma) (Emdogain®, Straumann) enhances new bone formation in maxillary sinus floor lift procedures, with BMAC resulting in a significant increase in new bone formation. In recent years, researchers have suggested that growth factors released from platelet concentrate help promote bone remodeling and cell proliferation and reduce the amount of autologous bone required for regenerative surgery. 102 Platelet-rich fibrin (PRF) is a second-generation platelet concentrate enriched with growth factors. It is recognized for its significant advantage over other graft materials, as platelets play a crucial role in soft tissue and bone development and repair. PRF is considered an effective alternative to bone substitutes because of its antimicrobial ability and ability to release growth factors. The assessment conducted by Sebastian Dominiak et al. 107 examined whether PRF, as a grafting material in maxillary sinus lift surgery, represents a dependable alternative to xenografts. After a 3-year follow-up period, the utilization of PRF alone alongside simultaneous implantation of sinus lift demonstrated effectiveness in soft tissue augmentation. Hence, it is inferred that PRF holds promise as a reliable alternative to various biomaterials in forthcoming applications.
PRF in the treatment of peri-implantitis
Peri-implant disease is a collective term for “inflammatory reaction of the tissues surrounding the implant.” 108 Peri-implantitis is a pathological condition that occurs in peri-implant tissues and is characterized by inflammation of the peri-implant connective tissue and progressive loss of supporting bone. X-rays are required to determine whether there is progressive bone loss. Peri-implant mucositis is characterized by bleeding, redness, pus spillage upon gentle probing, and no marginal bone loss on radiographs. It has been suggested that peri-implant mucositis is a precursor and risk factor for peri-implantitis, but not all peri-implant mucositis becomes peri-implantitis. 109 The difference between the two is the presence of progressive or persistent peri-implant bone loss. 110 While researchers have provided various definitions for peri-implantitis, they have consistently utilized bone loss as a key criterion.
Commonly, treatments for peri-implantitis include surgery 111 and antibiotic-assisted therapy. 112 The goal of surgical intervention is to reduce or eliminate infection associated with peri-implantitis via techniques such as open flap debridement to preserve the soft tissue surrounding the implant. However, this approach may lead to increased recession of peri-implant soft tissue over a period of 1–5 years. 111 The antibiotics typically employed are metronidazole and amoxicillin, but their effects are not long-lasting. 112 Recent research has indicated that bioactive materials effectively regulate inflammation and accelerate healing. The optimal treatment for peri-implantitis appears to be soft tissue grafting. 113 PRF, an optimized blood clot with enhanced fibrin structure and mechanical properties, contains growth factors that promote cell proliferation. Burak Hamzacebi et al. 114 investigated the effectiveness of PRF combined with conventional flap surgery compared with flap surgery alone in treating peri-implantitis. Both methods were effective postoperatively; however, the PRF group showed significant improvements in probing depth reduction and increased clinical attachment, outperforming the control group in all measures. PRF membranes facilitate epithelialization of the peri-implant surface, leading to rapid wound closure and healing, potentially reducing pain and post-operative swelling, with significant future potential in the clinical management of peri-implantitis. 115
Impact of PRF on regenerative aspects of dental pulp
The dental pulp, also known as the pulp-dentin complex, consists of dentin and pulp that function as structural and functional units. The pulp supports the dentin, and the dentin provides a protective barrier for the pulp. 116 Maintaining the health and vitality of the pulp is essential for long-term tooth preservation. 117 When the pulp is compromised or inflamed, the pulp needs to be treated. Endodontic treatment includes various procedures, such as live pulp therapy, root canal therapy, root canal retreatment, surgical endodontic treatment, and regenerative endodontic procedures. 118 The most common treatment is a root canal.
Root canal therapy 117 involves the use of sterile fillings to seal the root canal space, removing inflamed or necrotic pulp tissue and replacing it with synthetic biomaterials. This is the most common treatment for pulp inflammation, although it results in the loss of immune defense, vascularization, and regenerative capabilities of the tooth. 119 It has been argued that root canal therapy is essentially an invasive tooth deactivation procedure, the success of which is assessed only by the maintenance of an asymptomatic tooth in the alveolar bone. 120 To overcome these limitations, the concept of regenerating the pulp has been explored, particularly through pulpal blood flow reconstruction, 121 which involves the hematological reconstruction of immature permanent teeth with infected necrotic pulp and apical abscesses. This approach encourages root development and hard tissue deposition, strengthening the dentin wall. Regenerative pulp is a biologically driven strategy to restore the integrity of mature permanent teeth by physiologically replacing damaged tooth structure while allowing the roots of young permanent teeth to continue to develop. Revascularization has now become the most common treatment modality in an attempt to revitalize necrotic young permanent teeth and mature permanent teeth.
In young permanent teeth with underdeveloped root tips, traditional root canal treatment poses challenges because of the thin dentin wall and open root tip. Researchers suggest the use of platelet-rich fibrin (PRF) for endodontic hemoreduction. PRF, a second-generation platelet concentrate, shows promise in regenerative medicine and can be used alone or in combination with bone graft materials to increase bone growth and vascularization. 122 Joshi et al. 123 applied PRF with MTA as an apical barrier in young permanent teeth and observed the clinical outcomes. PRF has been shown to accelerate wound healing and act as an internal matrix for MTA coagulation. When used as an apical barrier in the non-surgical treatment of pulpless young permanent teeth, PRF, in combination with MTA, promotes bone filling. These findings highlight the potential future application of PRF in pulp regeneration. Kavitha et al. 124 evaluated the effectiveness of PRF and concentrated growth factor (CGF) as scaffolds for regenerative root canal treatment. Increases in root length, decreases in apical foramen size, and reductions in periapical lesion volume were observed through preoperative and 18-month post-operative CBCT scans. Closure of the apical foramina and a reduction in lesion volume were noted in all patients, demonstrating the efficacy of platelet concentrates as scaffolds for the regeneration of the pulp-dentin complex. Pulp regeneration relies on the ability to predict pulp tissue formation, which can be determined only through regular sensitivity assessments. A related study by Mittal et al. 125 evaluated the likelihood of restoring endodontic sensitivity in four groups (periapical hemorrhage, PRF, collagen, and hydroxyapatite) using endodontic testing and periapical X-rays. Pulp sensitivity was observed in all the groups, suggesting pulp tissue formation, with the highest sensitivity noted in the PRF group, followed by the collagen, hydroxyapatite, and periapical hemorrhage groups. These findings suggest that neurovascular bundles mature and form normally over time. Hazim Mohamed Rizk and other scholars concluded from a randomized controlled trial that hemodilution reconstruction with PRF is a very promising method. They applied blood clot (BC) and platelet-rich fibrin (PRF) scaffolds for the restoration of apically open, bilateral, necrotic maxillary permanent mesial incisors in young permanent maxillary teeth, which resulted in a significant increase in root length, a significant improvement in periapical density, and a significant reduction in apical diameter in the PRF group. 126
In conclusion, PRF may play a positive role in regenerative endodontic treatment and may open new avenues in the future for the use of PRF for endodontic regeneration. 127
Impact of PRF on oral mucosal disease
The oral mucosa, comprising the moist lining of the oral cavity, is composed of epithelial and connective tissue, exhibiting structural and functional similarities to the skin; it serves as the body’s primary protective barrier. 128 Oral mucosal disease, also referred to as oral soft tissue disease, 129 includes a range of conditions affecting oral mucosal tissues, including oral ulcers, oral lichen planus, polymorphic exudative erythema, drug-induced stomatitis, and oral leukoplakia. 130 Diseases of the oral mucosa are associated not only with localized injuries but also with systemic diseases. For example, anxiety, depression, and immunological factors can trigger oral mucosal disease. 131
PRF in the treatment of mouth ulcers
Mouth ulcers represent a prevalent category of oral mucosal disease, with recurrent aphthous ulcers, also termed recurrent aphthous stomatitis, and recurrent mouth ulcers being the most prevalent type. 132 Recurrent aflatoxic ulcers are of unknown etiology and account for about 20% of the global population. 133 It has been suggested that it is an excessive inflammatory response caused by innate immune destruction. 134 These ulcers exhibit noticeable individual variations, characterized by features such as a yellow, red, concave, and painful appearance, with the ulcer surface typically covered by a yellow pseudomembrane encircled by a red halo band, displaying a concave center and causing significant discomfort. While typical recurrent aphthous ulcers tend to heal spontaneously within 1–2 weeks, prolonged persistence can adversely impact patients’ quality of life. Its pathogenesis is characterized by ulcerative lesions on the surface of the oral mucosa and proliferative lesions of the oral mucosal epithelium. Treatment strategies for oral ulcers aim to alleviate pain, promote healing, and prevent recurrence. 135 Commonly used treatments include corticosteroids, topical antibiotics, anti-inflammatory analgesics, and immunomodulators. However, all of these methods are only temporary and do not address the underlying cause. 136 Historically, systemic and topical glucocorticoids, such as 0.1% trimethoprim acetate, have been favored for the treatment of recurrent oral ulcers. 135 However, repetitive tretinoin injections have been linked to local tissue atrophy, hardening, reduced resistance, and susceptibility to fungal infections. 137 Further investigation revealed the effectiveness of 5% Ameclox in treating mouth ulcers, making it the sole FDA-approved prescription for recurrent mouth ulcers. 135 Its administration typically involves ointments or oral pastes. Nevertheless, owing to the unclear etiology of mouth ulcers, no definitive cure exists, and medication primarily focuses on pain reduction and relief. Platelet concentrates play an important role in mediating the tissue healing process, as they release growth factors and cytokines. 138 Platelet-rich fibrin (PRF) has gained traction in dentistry, with studies such as that by Ahmed et al. 139 assessing its efficacy in pain management and mucosal repair through a randomized controlled trial. By comparing the effectiveness of clobetasol propionate (control group) and PRF (experimental group) in oral ulcer treatment, it was determined that both significantly reduced pain (p ⩽ 0.05). Notably, the PRF group achieved complete pain relief by day 7, resulting in superior clinical outcomes and accelerated healing compared with those of the corticosteroid group. These findings underscore PRF as a safe and promising alternative for oral ulcer treatment, offering novel avenues for diagnosing and managing oral mucosal ulcers.
PRF in the treatment of oral lichen planus
Oral lichen planus (OLP) presents as a chronic inflammatory autoimmune condition. 140 Its exact cause remains elusive, and it affects various areas, such as the skin, scalp, nails, and mucous membranes; however, symptoms are more pronounced in the mouth and persist longer. 141 Its occurrence is associated with several risk factors, such as medications, infections, smoking, mechanical trauma, and radiation. 142 According to recent studies, the global prevalence of oral lichen planus (OLP) is 1.01%, and it is 3–4 times more common in women than in men. 143 The diagnosis of OLP relies on clinical and histological observations, with six recognized clinical types: reticular, vesicular, atrophic, plaque, papular, and blistering, all of which are driven by immune antigen-mediated T cells. 144 Individuals with OLP commonly report pain and a burning sensation in the oral mucosa, impairing functions such as eating, speaking, and swallowing.
Traditional treatments for lichen planus typically involve medication, including corticosteroids, 100 hydroxychloro-quine, 145 folic acid, vitamin B 144 , and Topical Calcineurin Inhibitors.” 146 Among these, corticosteroids 147 are the most commonly prescribed, including trimethoprim, clobetasol propionate, betamethasone sodium phosphate, dexamethasone, fludioxonyl propionate, and budesonide. 147 A study by scholars such as Nosratzehi 144 demonstrated that corticosteroids possess anti-T-cell-mediated and anti-inflammatory properties, thereby suppressing cell-mediated immunity and expediting healing in OLP patients. Nonetheless, their usage may lead to adverse effects such as pain, bleeding, ulcers, and secondary infections. 148
In recent years, researchers have shown that treating lichen planus with platelet-rich fibrin is effective. Bennardo et al. 149 conducted a randomized controlled trial to evaluate the efficacy of comparing tretinoin (TA) and PRF in the treatment of patients with oral lichen planus. In a study using a same-mouth controlled approach, PRF was injected into one side of the buccal mucosa, while TA was injected into the other side of OLP patients once weekly for 4 weeks. The results showed that PRF was as effective as TA in reducing the extent of OLP lesions and symptoms. These findings suggest that PRF could become a promising therapeutic option for OLP treatment in the future. Gupta et al. 148 conducted a similar study comparing the effectiveness of I-PRF and other interventions for OLP treatment. I-PRF, a type of L-PRF obtained by adjusting the centrifugation speed and time, has better antimicrobial properties and regenerative capacity than L-PRF. In this study, I-PRF was assessed for pain reduction, lesion surface area, and patient satisfaction. The findings indicated that both the I-PRF group and the control group experienced reductions in pain and lesion size. However, patients treated with I-PRF showed improvements in lesion extension and reported higher satisfaction levels. These findings suggest that I-PRF not only reduces pain and improves lesion size but also enhances patient satisfaction. In summary, PRF holds promise as a potential treatment for OLP. A meta-analysis of the effectiveness of PRP and PRF in patients with lichen planus by scholars such as Alshemmari et al. 150 revealed no significant differences between PRF and PRP in terms of lesion size, pain scores, clinical severity scores, or changes in adverse events compared with corticosteroids; both have some efficacy. PRP, compared with PRF, and in accordance with the order of treatments, showed that PRF was the best intervention. These findings suggest that PRF has great potential as a biomaterial and is expected to be an alternative to corticosteroids in the future as a new approach for the treatment of OLP, providing new clinical ideas.
PRF treatment for pediatric dentistry
PRF is widely used not only in adults but also in children with oral diseases, and this study reviews its use in children with jaw defects and pediatric alveolar clefts and as a direct pulp-capping agent for milk teeth.
PRF applied to children with alveolar clefts
Cleft lip and cleft palate are the most common congenital craniofacial anomalies, with a statistically significant prevalence of 1.4 per 1000 infants worldwide. Within Asia, the disease affects 1 in 500 infants. 151 Incomplete fusion of facial contours is the underlying cause of cleft lip and palate. Three out of four patients with cleft lip or cleft palate have alveolar clefts, 152 which develop as a result of surgery, trauma, infection, or congenital malformations. 153 The presence of an alveolar ridge gap disrupts the proper alignment and eruption of permanent teeth and interferes with speaking, eating and swallowing. 154 Its treatment is usually restored at the age of mixed dentition (6–11 years of age) by means of a second-stage alveolar bone graft to promote normal tooth eruption. 152 However, despite initial cleft lip and palate repair in these children at birth, complete restoration of the alveolar ridge, nasal floor, intraoral soft tissues, and dentition requires extensive skeletal and soft tissue reconstruction. Eid et al. 155 conducted a randomized controlled trial in which 36 children with alveolar clefts were randomized into 3 groups. When PRF with resorbable collagen membranes was applied separately to alveolar bone grafts, with an additional control group, significant osteogenesis was observed at 9 months post-surgery. Compared with the control group, the use of PRF membranes and collagen membranes as intermediate layers between the nasal layer and the autologous chin bone graft significantly improved bone formation and bone density in the alveolar eminence. On the other hand, PRF contains a variety of growth factors that promote wound repair. In the future, PRF is expected to be an alternative to collagen membranes as an adjunct for alveolar clefts.
PRF acts as a direct pulp-capping agent in milk teeth
Caries is a multifactorial disease that affects about 60%–90% of the global population, especially children, and it is one of the most prevalent health problems during childhood. 156 Its pathology is characterized by demineralization of inorganic material and decomposition of organic material. The incidence of caries is extremely high and can have a significant negative impact on health and quality of life. 157 Caries develop rapidly in children, and pulp infection may occur when caries involves and penetrates the dentin. If deep caries in milk teeth are not treated in time, tooth tissue loss, pulpal lesions, and periapical infections may occur, eventually leading to the loss of milk teeth. 158 Live pulpotomy, indirect pulp capping, direct pulp capping (DPC), etc., are options for conservative endodontic treatment. The aim is to maintain pulp vitality and protect dental health by preserving some or all of the pulp. DPC is less invasive than root canal treatment modalities. 159 Gomez-Sosa et al. 159 reported that the application of mineral trioxide aggregates (MTA), which are similar to biological dentin, has a higher success rate than the use of calcium hydroxide in teeth where the pulp has been exposed due to caries. It should be used as a substitute for calcium hydroxide. In recent years, owing to the development of plasma products, PRF has been widely used by scholars in pediatric dentistry, and Tiwari et al. 160 conducted a randomized controlled trial. To evaluate and compare the efficacy of platelet-rich fibrin (PRF) and mineral trioxide aggregates (MTA) as direct pulp capping (DPC) agents in milky molars, as determined by their clinical and imaging success rates, the experimental group covered the remaining pulp tissue with the PRF preparation, and the control group placed the MTA and placed a zinc oxide eugenol (ZOE) base and a glass ionomer (GIC) on top of the PRF and MTA, respectively. Clinical and imaging evaluations were performed at 1-, 3-, 6-, 9- and 12-month postoperative intervals. The overall success rate was 82.6% in the PRF group compared with 61.9% in the MTA group at 12 months. PRF proved to be more effective than MTA. Platelet-rich fibrin is expected to be used as an ideal alternative material for DPC restorations in deciduous teeth.
PRF applied to bone defects in children
Skeletal insufficiency in the jaws occurs spontaneously as a result of physician-induced factors caused by diseases such as cysts, tumors, and infections or surgical resection of prior periosteal nerve root disease in the bone. 161 Bone tissue defects affect maxillofacial functions such as neural activity, mastication, speech, and hearing. 162 Its treatment is a great challenge because, in cases of bone discontinuity, bone tissue does not regenerate spontaneously without surgical intervention, despite prolonged healing time. 163 Bone defects are usually treated with autografts and allografts. However, autografts involve the transfer of healthy tissue from the same individual to the injury site. Although this treatment approach has achieved remarkable therapeutic results in recent years, it has significant limitations, such as the scarcity of donor sites and the presence of a second operative zone. 164 In recent years, researchers have reported that tissue-engineered scaffolds can replace defective bone tissue, allowing bone to reform on its surface. The incorporation of bioactive molecules into scaffolds is by far the most widely used method for endowing scaffolds with osteogenic capacity. Materials used to restore bone tissue should have good biocompatibility as well as osteoinductive properties. PRF contains a large number of growth factors and has been extensively studied for its ability to promote regeneration. Koksi Sangma Shadap et al. 161 studied the role of platelet-rich plasma (PRP) and platelet-rich fibrin (PRF) and synthesized nanocrystalline hydroxyapatite (HA) and β-tricalcium phosphate (β-TCP) by comparing their effects on the regeneration of regenerated bone defect regeneration efficacy. In this study, 20 children with bone defects were selected and randomized into two groups of 10 each. The first group was filled with PRP + nanocrystalline HA with β-TCP, and the second group was filled with PRF + nanocrystalline HA with β-TCP. Bone density and bone regeneration were analyzed via X-ray, and the density of the bone defects was significantly greater in the PRF group than in the PRP group. PRF significantly improved and accelerated bone regeneration.
Summary and outlook
PRF, derived from autologous blood, comprises a natural fibrin matrix, platelets, white blood cells, and growth factors. Its safety, straightforward preparation, and excellent biocompatibility render it highly promising for applications in regenerative medicine. 165 PRF has been extensively used in various fields, such as oral and maxillofacial surgery, periodontal soft and hard tissue regeneration, alveolar ridge site preservation, and pulp tissue regeneration. 166 Several types of PRF derivatives have been developed, including L-PRF, A-PRF, I-PRF, H-PRF, T-PRF, Alb-PRF, and C-PRF. This paper categorizes PRF derivatives and outlines their biological properties, preparation methods, and applications to serve as a reference for the development and clinical application of additional PRF derivatives. Despite the advantages of existing PRF derivatives, such as easy preparation, accessibility, abundant growth factor content, and good biocompatibility, variations in the centrifugal force and significant differences in the centrifugation time can notably impact the platelet content. Additionally, few high-quality clinical trials have investigated the efficacy of PRF therapy and determined the optimal treatment approach. 167 In addition, PRF membranes have relatively poor mechanical properties and lack rigidity, limiting their use.
In summary, PRF stands out as a safe and biocompatible autologous platelet concentrate with significant advantages and promising prospects in the realm of regenerative medicine. 168 We anticipate further research on various PRF derivatives in the future, which will significantly improve the utilization of PRF in oral applications and offer fresh insights for diagnosing and treating oral diseases.
Footnotes
Acknowledgements
This study received no grants from funding agencies in the public, commercial or not-for-profit sector.
CRediT authorship contribution statement
Ping Song: Writing – original draft, Visualization, Methodology, Investigation, Data curation. Dawei He: review, editing. Song Ren: Visualization,Investigation. Lin Fan: Data curation, Investigation. Jiang Sun: Writing – review editing, Visualization, Supervision, Methodology.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

