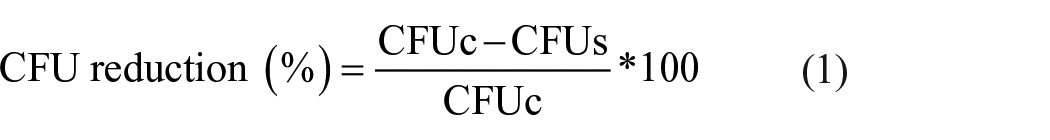Abstract
In the past few years, due to the Covid-19 pandemic, the interest towards textiles with antimicrobial functionalities faced a significant boost. This study proposes a rapid and convenient method, in terms of reactants and equipment, for fabricating antimicrobial coatings on textiles. Through the electroless silver plating reaction, silver coatings were successfully applied on cotton and polyester, rapidly and at room temperature. Functionalized samples were characterized by morphological (optical and scanning electron microscopies) and chemical tests (X-ray photoelectron spectroscopy, XPS) to investigate the nature of the silver coating. Although distinct nanoparticles did not form, XPS analysis detected the presence of silver, which resulted in an increased surface roughness and hydrophobicity of both cotton and polyester textiles. Ag-coated samples exhibited approximately 80% biocompatibility with murine L929 fibroblasts or human HaCaT cells, and strong antibacterial properties against Escherichia coli in direct contact tests. In antiviral experiments with SARS-CoV-2 virus, treated cotton showed a 100% viral reduction in 30 min, while polyester achieved 100% reduction in 1 h. With a human norovirus surrogate, the Feline Calicivirus, both treated textiles have a faster antiviral response, with more than 60% viral reduction after 5 min, while achieving a 100% reduction in 1 h. In conclusion, this study presents a fast, efficient, and low-cost solution for producing antimicrobial textiles with broad applications in medical and healthcare scenarios.
Introduction
The widespread use of textiles with antimicrobial properties underscores their significance in maintaining hygiene and minimizing infection transmission in different contexts, from everyday life to medical and healthcare, sports, commercial or industrial.1,2 Ongoing research initiatives are looking for ways to improve the efficacy, resistance and eco-compatibility of antimicrobial textiles, thereby reducing health and environmental problems. 3
Numerous natural and synthetic materials that possess intrinsic antimicrobial properties, such as polyphenols, chitosan, poly-ε-lysine, polyethyleneimine, quaternary ammonium compounds, propolis, or metal compounds and nanoparticles and surfactants, have been employed to defeat harmful pathogens. 4 Among them, metal and metal oxide nanoparticles (NPs), such as silver, copper, or zinc oxide NPs, have been extensively studied 5 due to their unique properties derived from the nanoscale, as introduced by Feynman in 1959. 6
Conventional finishing methods of textiles include dipping, impregnation, or padding, while more advanced methods include electrospinning, microencapsulation, spray deposition, or plasma and magnetron sputtering.1,5,7 For instance, Cui et al. 8 fabricated polyacrylonitrile, silica dioxide and the surfactant cetyltrimethylammonium bromide electrospun nanofibrous membranes killing and blocking the spread of bacteria in aerosols. Among conventional methods, pad-dry-cure is largely used to incorporate silver nanoparticles (AgNPs) into textiles and enhance their antibacterial properties. For instance, Lee et al. 9 applied ethanol-based nanosilver colloids on cotton and polyester fabrics, demonstrating the successful integration of AgNPs into textiles and remarkable antibacterial properties against Staphylococcus aureus and Klebsiella pneumoniae after 24 h. Wu et al. 10 proposed a mild route to prepare antibacterial cotton fabrics based on AgNPs binding on the fibers through chemical bonds by citric acid. This finishing process produced hybrid fabrics with excellent antimicrobial activities against Escherichia coli and Staphylococcus aureus, with 98.7% and 91.8% percentages of reduction, respectively.
The methods of incorporating NPs can be alternatively divided into ex situ or in situ. 5 While ex situ methods require the formation of NPs prior to their incorporation onto the target substrate, in situ approaches are faster and more efficient since the formation and incorporation of NPs occur directly and simultaneously onto the target substrates. 11 Vigneshwaran et al. 12 formed AgNPs on cotton by the autoclave-assisted chemical reduction of silver nitrate (AgNO3), that exhibited potent antibacterial effects against S. aureus and K. pneumoniae. More recently, an antimicrobial hydrophobic coating has been applied to cotton fabric by encapsulating AgNPs in a protective silicone layer. 13 Similarly, Ribeiro et al. 14 successfully functionalized polyester with AgNPs using chitosan or hexamethyldisiloxane as additional layers that ensured the stability and controlled release of AgNPs. In our previous study, 15 we introduced a novel one-step deposition method, combining the silver mirror reaction with sonication or agitation, to create antibacterial and antiviral Ag-coated polypropylene surgical masks. These biocompatible coatings demonstrated antibacterial properties and potent activity against SARS-CoV-2 virus. 15
This study aims to develop antimicrobial silver coatings on textiles, specifically cotton and polyester, potentially applicable in medical-health contexts, using an in situ functionalization method based on the electroless silver plating technique. This autocatalytic reaction is conventionally applied to conductive metal substrates 16 but can also be applied to non-conductive surfaces. 17 The classical plating procedure includes several stages, namely pre-treatment, sensitization, activation, plating, and post-treatment. 16 These steps are important for enhancing the adhesion and effectiveness of the plating on the target substrate. 18 The novelty of this research consists in the development of antibacterial and antiviral coatings on textiles through the electroless silver plating reaction, using a reduced number of steps and chemical reagents, exploiting the photocatalytic properties of AgNO3, at room temperature.
Methods
Chemical reactants
Silver nitrate crystals (AgNO3, MW 169.87 g mol−1), tin (II) chloride (SnCl2 MW 189.62 g mol−1), and hydrochloric acid were purchased from Merck KGaA (Darmstadt, Germany). All reagents were used without further purification. Deionized water (dH2O, Dia Class 120, Quality Invents s.r.l., Milan, Italy) was obtained via a reverse osmosis process. Mouse fibroblasts L929 (ICLC ATL95001) were obtained from Biobanking and Cell Factory Hospital San Martino (Genova, Italy). HaCaT keratinocytes were obtained from Istituto Zooprofilattico Sperimentale della Lombardia e dell’Emilia-Romagna “Bruno Ubertini”. E. coli strain was purchased from Thermo Fisher Scientific (Milan, Italy). White 100% polyester (Raso Saturno) and 100% cotton textiles were used without any initial pre-treatment.
Electroless silver plating on cotton and polyester textiles
Ag-coated polyester and Ag-coated cotton fabrics (5 cm × 5 cm) were obtained by submerging them separately in 6.25 mL of tin (II) chloride solution 10 mM with 12.5 µL of hydrochloric acid 0.1 M for 1 min. Then, after rinsing in deionized water for 30 s, the substrates were immersed in 6.25 mL of AgNO3 solution 6 mM for 30 s and rinsed again in water for 30 s. All the coating steps were carried out at room temperature, in daylight and static conditions, using glass beakers. Samples, dried overnight at room temperature, were prepared in triplicate.
Morphological analysis
Samples morphology was analyzed by stereo microscopy (Axio Zoom V16 equipped with a digital camera AxioCam 506 Mono, Carl Zeiss, Oberkochen, Germany) at different magnifications, and via scanning electron microscopy (SEM, FlexSEM 1000, Hitachi, Tokyo) by varying magnifications and voltages (5 or 10 kV). Prior to imaging, all samples were sputter-coated with gold.
XPS analysis
X-ray photoelectron spectroscopy (XPS) was carried out using a PHI Versaprobe 5000 spectrometer, equipped with an Al K-alpha monochromatic line (1486.6 eV), and by using a double charging neutralization procedure, made up by Ar+ and an electron beam, to compensate surface charging phenomena for non-conductive materials. Survey scans have been acquired before High Resolution (HR) spectra, to check for all the elements present on top of the materials. Both survey and HR measurements were collected using a 100 µm size diameter analysis spot. Deconvolution procedures on HR spectra have been carried out by using Casa XPS Software (version 2.3.18).
Wettability
The wettability of samples was assessed by static water contact angle (WCA) measurements. A 3 µL drop of dH2O was deposited on the surface of samples at room temperature, and the average WCA was obtained from digital images analyzed with ImageJ 1.52 software. At least 10 measurements per sample were taken.
Biological properties
In vitro biocompatibility assessment
The biocompatibility of Ag-coated samples towards murine L929 fibroblasts or human HaCaT cells was evaluated by the MTT assay after an indirect contact test following ISO 10993-5 protocols, 19 as previously reported. 15 Briefly, cells were exposed for 24 h to extracts obtained by incubating UV-sterile samples in the RPMI-based culture medium for 24 h. After the exposure, cell viability was calculated by comparing the absorbance of samples at 550 nm with reference values, that is, absorbance of cells exposed to the culture medium devoid of samples.
Antibacterial activity assessment
The antibacterial effectiveness of Ag-coated textiles was evaluated against E. coli (Gram-negative). 15 The bacterium was cultured in Mueller-Hinton broth at 37°C. Bacterial suspensions (75 μL) containing 50 colony forming units (CFU) were inoculated onto UV-sterilized round samples (ø 8 mm). Following several time points of incubation, 5, 15, 30, 60 and 120 min, broths were collected and plated on sterile Mueller-Hinton agar. After overnight incubation at 37°C, CFU numbers were counted. The antibacterial rate is expressed as percentage reduction of CFU number and was calculated according to the following equation (1):
where CFUc is the number of CFU counted for untreated samples and CFUs is the number of CFU counted for Ag-coated samples. The antibacterial activity of Ag-coated polyester and cotton was furtherly investigated by a qualitative analysis of E. coli growth in Mueller-Hinton broth using samples employed from direct contact assay. Briefly, after inoculum collection, samples of the 5 and 60 min time points were submerged in 1 mL of broth and incubated under agitation at 37°C for 24 h. Broth without any samples or inoculum was used as control, and E. coli growth was evaluated based on the turbidity of each suspension.
Antiviral properties assessment
The antiviral properties of samples (ø 14 mm), UV-sterilized for 1 h per side, were evaluated as previously reported.20,21 African green monkey kidney cells (Vero E6) and clinical isolate of SARS-CoV-2, kindly gifted from the San Raffaele Hospital (Milan, Italy). The virus expanded and titrated on Vero E6 cells had a titer of 4.6 × 105 TCID50/ml determined by the Reed and Muench formula and used at 0.1 multiplicity of infection (MOI).
Crandell-Reese Feline Kidney cells (CRFK, ATCC, CCL-94) and Feline Calicivirus F9 strain (FCV, ATCC, VR789) were purchased from ATCC and cultivated according to the manufacturer’s instructions. FCV was cultivated on CRFK cells and infection was conducted on CRFK cells at a 0.5 MOI for 72 h.
The antiviral properties of functionalized samples were assessed at 1, 5, 10, 30, and 60 min of contact. The results were expressed as average % inhibition of viral growth, normalizing the values on an infected but untreated control.
Functional tests of fabrics: Differential pressure
Differential pressure test, or breathability, was evaluated using Bulldog Plus-R (XEARPRO, Cogliate, Italy). The differential pressure, expressed in Pa cm−2, was calculated using the following formula (2):
where Xm1 is the pressure in Pa measured by the pressure gauge at the bottom side of the sample, Xm2 is the pressure in Pa measured by the pressure gauge at the top side of the sample, and 4.9 is the area in cm2 of the tested sample.
Statistical analysis
Data are presented as mean ± standard deviation (SD). One-way or two-way analysis of variance (ANOVA) was used for comparing data, a p-value less than 0.05 was considered statistically significant.
Results
Electroless silver plating on cotton and polyester textiles
Electroless silver plating on pristine cotton and polyester textiles was performed in less than 3 min at room temperature with very few steps and without any pre or post treatment. Indeed, textiles were first immersed in a SnCl2 solution for few seconds then washed with deionized water, and finally they were immersed in an AgNO3 solution and washed again. Although the electroless functionalization process applied to either material was the same, their different characteristics led to small variations in the resulting Ag coatings. From macroscopic observations, the electroless plating confers different colors to the treated fabrics. Ag-coated polyester has a pinkish color, while Ag-coated cotton turns brown (Figure 1).

Macroscopic appearance of uncoated (a) or Ag-coated (b) cotton, and uncoated (c) or Ag-coated (d) polyester textiles; scale bars 1 cm.
Morphological characterization
The stereomicroscopy analysis of uncoated or Ag-coated cotton and polyester samples shows that the silver coating on both textiles exhibits a consistent and even appearance (Figure 2). SEM imaging at various magnifications did not reveal the presence of silver in the form of nanoparticles. However, after the electroless silver plating, the surface roughness of the fibers appears increased compared to the control samples (Figure 3).

Morphological characterization by stereomicroscopy of uncoated (a) or Ag-coated (b) cotton, and uncoated (c) or Ag-coated (d) polyester textiles; scale bars 1 mm.

Morphological analysis by SEM imaging at 2000× magnification of uncoated cotton (a) and polyester (d), Sn pre-treated cotton (b) and polyester (e) and Ag-coated cotton (c) and polyester (f); scale bars 20 µm.
XPS analysis
XPS analysis has been conducted both on cotton and polyester Ag-coated samples. From survey spectra (see Figure 4), we can appreciate the presence of the main elements due to the substrates (C, O, N, and Si) and to the chemical decoration (Ag, Sn) and some remnants of precursors (Cl and N). The relative atomic concentrations (at. %) calculated for each sample, are reported in Figure 4. Ag-coated polyester sample shows almost double the concentration of Ag (2.0 at. %) and Sn (1.6 at. %), with respect to Ag-coated cotton (0.9 at. %). If we move to the high-resolution peaks, especially the one related to Ag3d and Sn3d doublets (see Figure 5(a) and (b)), we can establish the oxidation states for each species. Starting from Ag3d regions (Figure 5(a)), we can see that both samples show asymmetric peaks, which require at least two components for each peak of the doublet, to properly deconvolute the experimental signal. By using Gaussian-Lorentzian curves and Shirley background, we obtain a first component at 367.8 eV (±0.2 eV), due to Ag(I) chemical shift 22 and another smaller component at 368.8 eV (±0.2 eV), which is due to Ag(0). 23 The ratio between these two chemical states is, respectively, Ag(I)/Ag(0) = (80.6/19.4)% for Ag-coated cotton sample and (86.2/13.8)% for Ag-coated polyester. Regarding the Sn signals (see Figure 5(b)), we can clearly see a simple doublet due to only one component, which is ascribed to Sn(IV) oxidation state, 24 with Sn3d5/2 peaks located at 486.8 eV (±0.3 eV) for both samples. Thus, from the chemical point of view, no significant differences are evident in these two samples, which show the same oxidation states for both Ag and Sn, apart from the relative concentration of these elements, which is doubled, in the Ag-coated polyester sample.
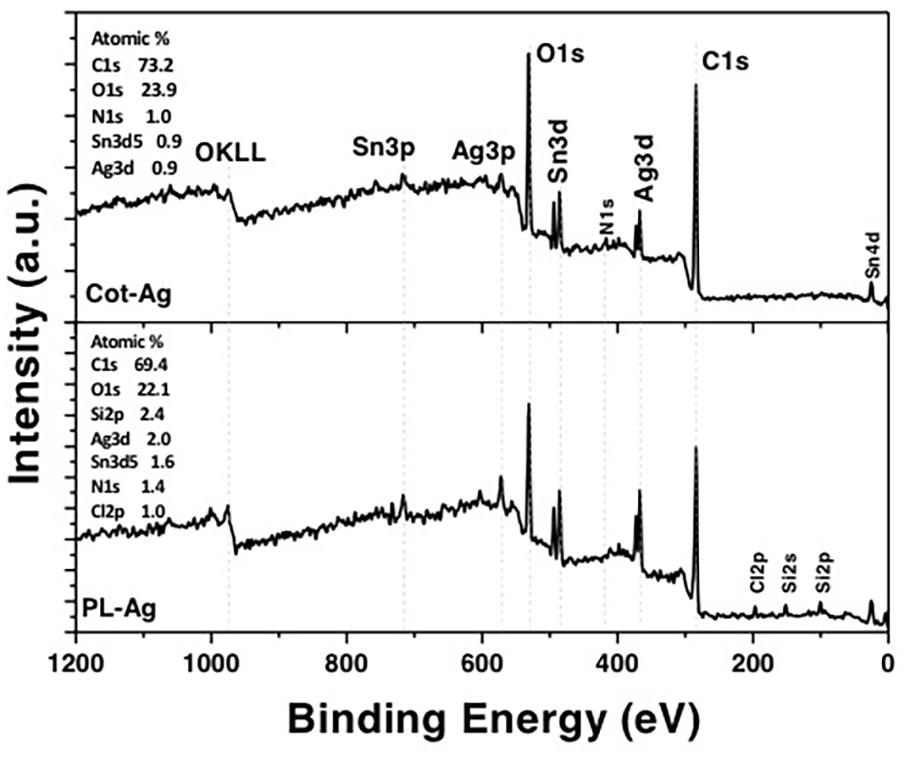
XPS survey scan for Ag-coated cotton (Cot-Ag) or polyester (PL-Ag) samples. Relative atomic concentrations values are reported in each graph.
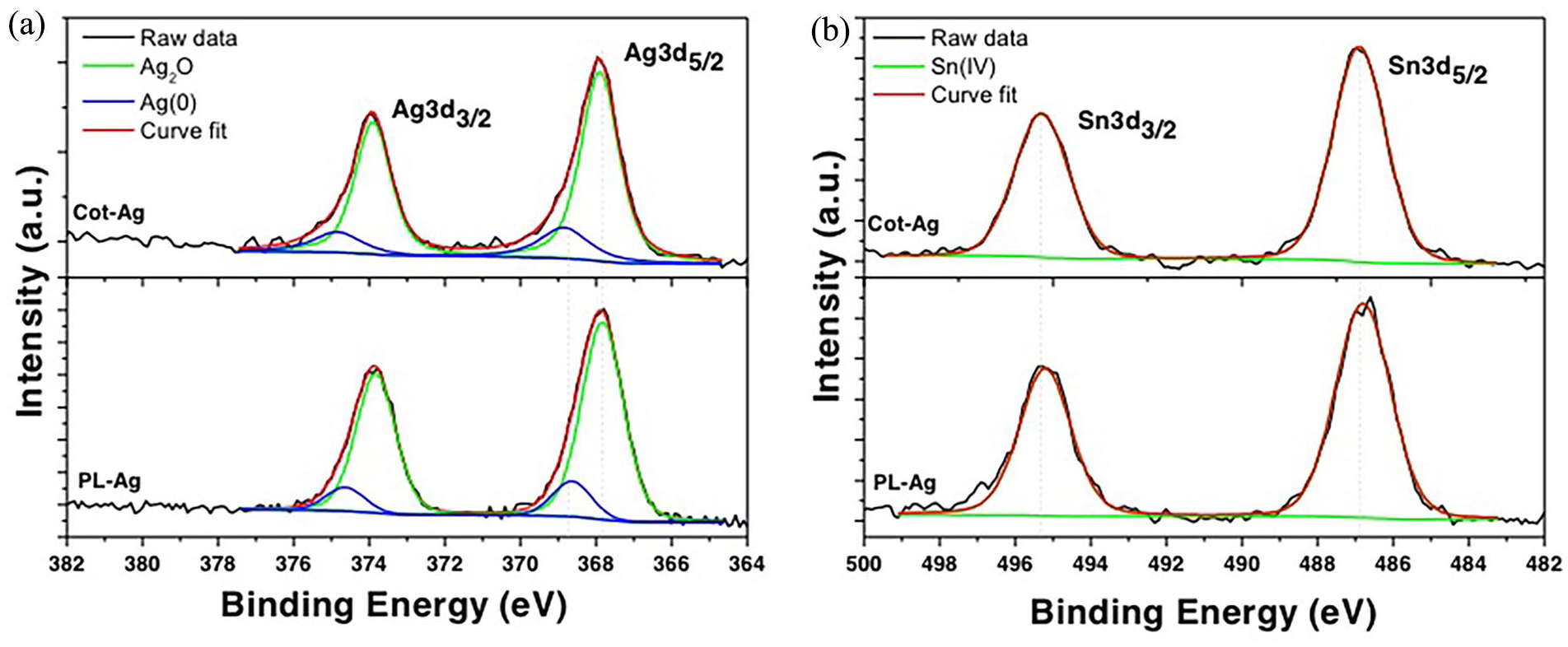
XPS HR spectra for Ag3d (a) and Sn3d (b) regions, for both Ag-coated cotton (Cot-Ag) or polyester (PL-Ag) samples. Deconvolution procedures have been reported in each graph, for clarity.
Wettability
Changes in wettability were investigated by static water contact angle measurements (Figure 6).
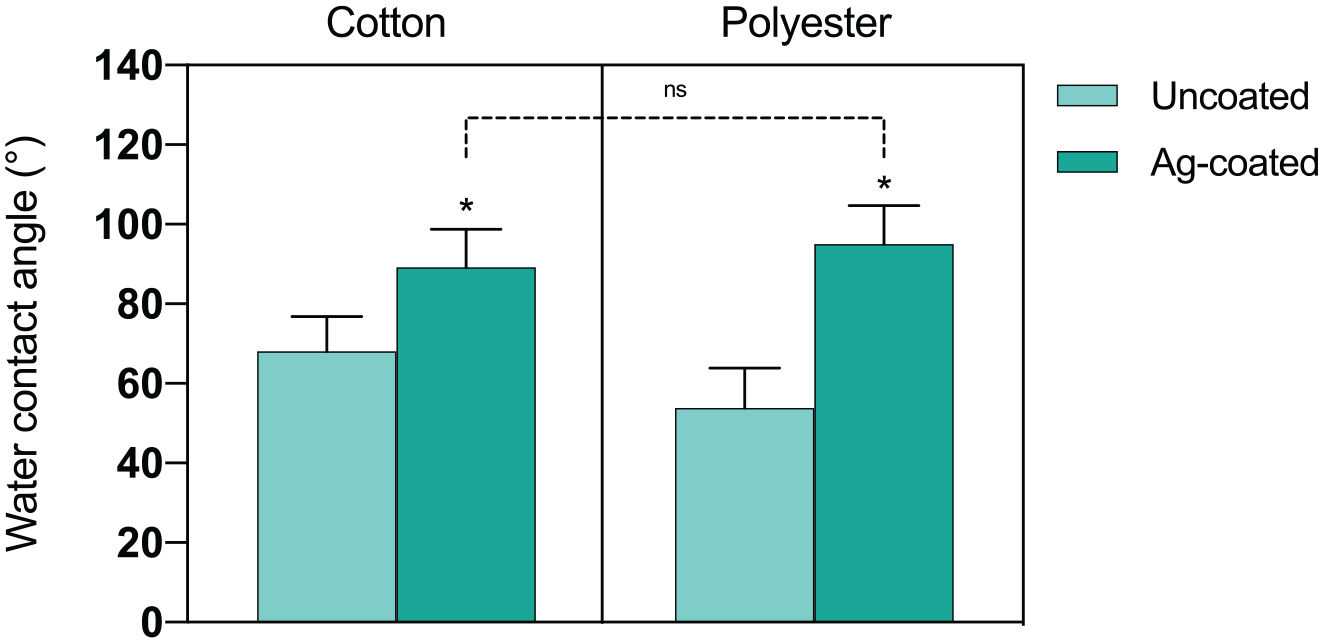
Water contact angle of cotton and polyester uncoated or Ag-coated samples. Histograms represents the mean ± SD (n > 10).
Both pristine polyester and cotton are hydrophilic, with WCA values of 54° and 68°, respectively. In both cases, the presence of the Ag coating significantly increased the WCA values, leading to more hydrophobic behavior. Despite the initial differences of uncoated samples, both Ag-coated fabrics present similar wettability, with an average WCA of 95° for polyester, and 89° for cotton, with no statistically significant differences.
Biological properties
Biocompatibility toward human keratinocytes
The biocompatibility of Ag-coated cotton or polyester was evaluated with L929 murine fibroblasts, or HaCaT human keratinocytes cells by an indirect contact test. Results expressed as a percentage of cell viability are plotted in Figure 7. All samples have cell viabilities above the 70% threshold value, so that are considered biocompatible for both cell types. Regarding polyester, the viability of Ag-coated fabrics did not significantly decrease compared to the uncoated fabric, with 84% ± 4% and 86% ± 6% cell viability for L929 and HaCaT cells, respectively. Regarding cotton, a similar behavior was observed, with cell viability for the Ag-coated fabric equal to 78% ± 4% and 81% ± 5% for L929 and HaCaT cells, respectively.
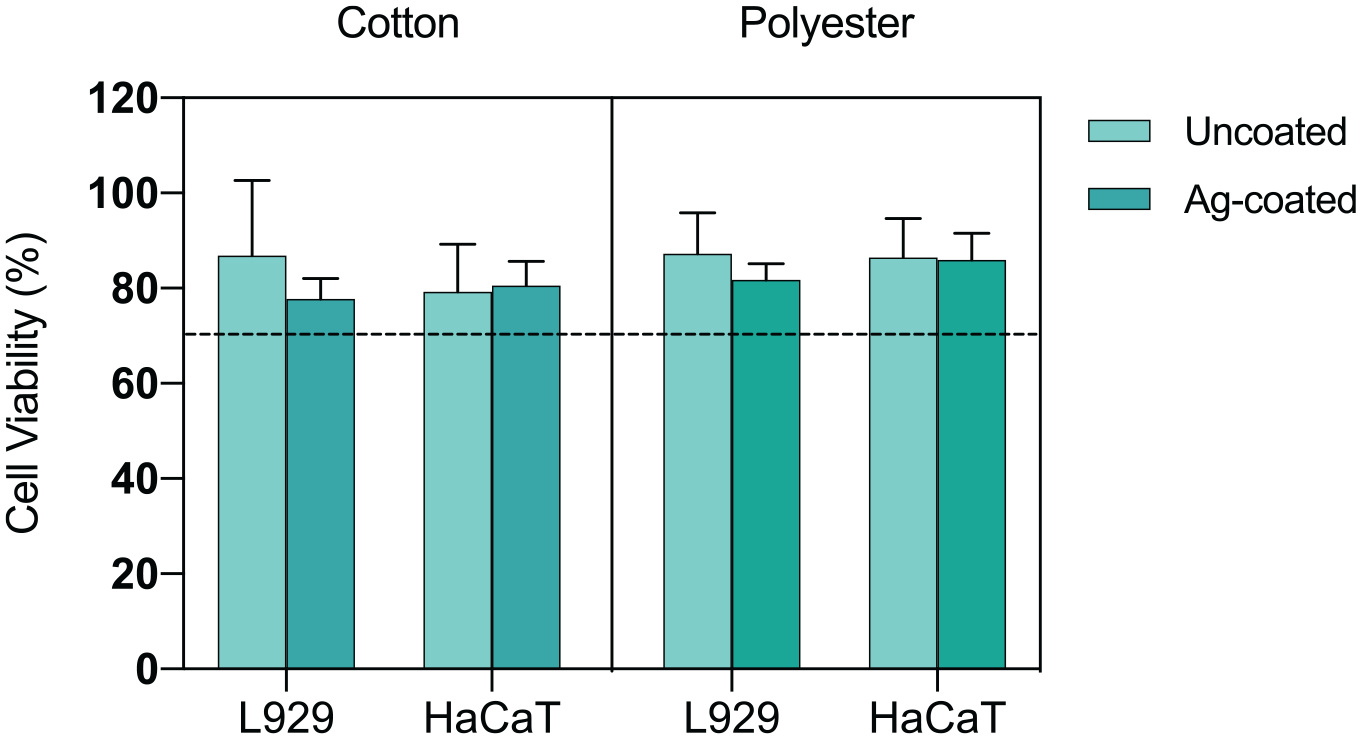
Biocompatibility of cotton and polyester uncoated or Ag-coated samples by indirect contact tests. Histograms represents the percentage of cell viability of L929 or HaCaT cells expressed as mean ± SD (n > 3). Dashed line at 70% is the common threshold for biocompatibility.
Antibacterial activity against E. coli
The antibacterial activity of Ag-coated samples was tested by a direct contact assay based on measuring the E. coli CFU number after its inoculation on the samples. The percentage reduction of the bacterial CFU number compared to the uncoated textile was considered as a measure of the antibacterial efficacy. As shown in Figure 8, silver coating on polyester possesses a significant antibacterial effect against E. coli. Specifically, Ag-coated polyester causes a good percentage reduction of the CFU number at all the tested time points, decreasing the CFU growth of 38% and 95% already at 5 and 30 min, respectively, and achieving the 100% reduction of CFU after 120 min of direct contact. Good results were also obtained testing Ag-coated cotton, achieving 47% and 78% of CFU reduction at 60 and 120 min of direct contact, respectively.
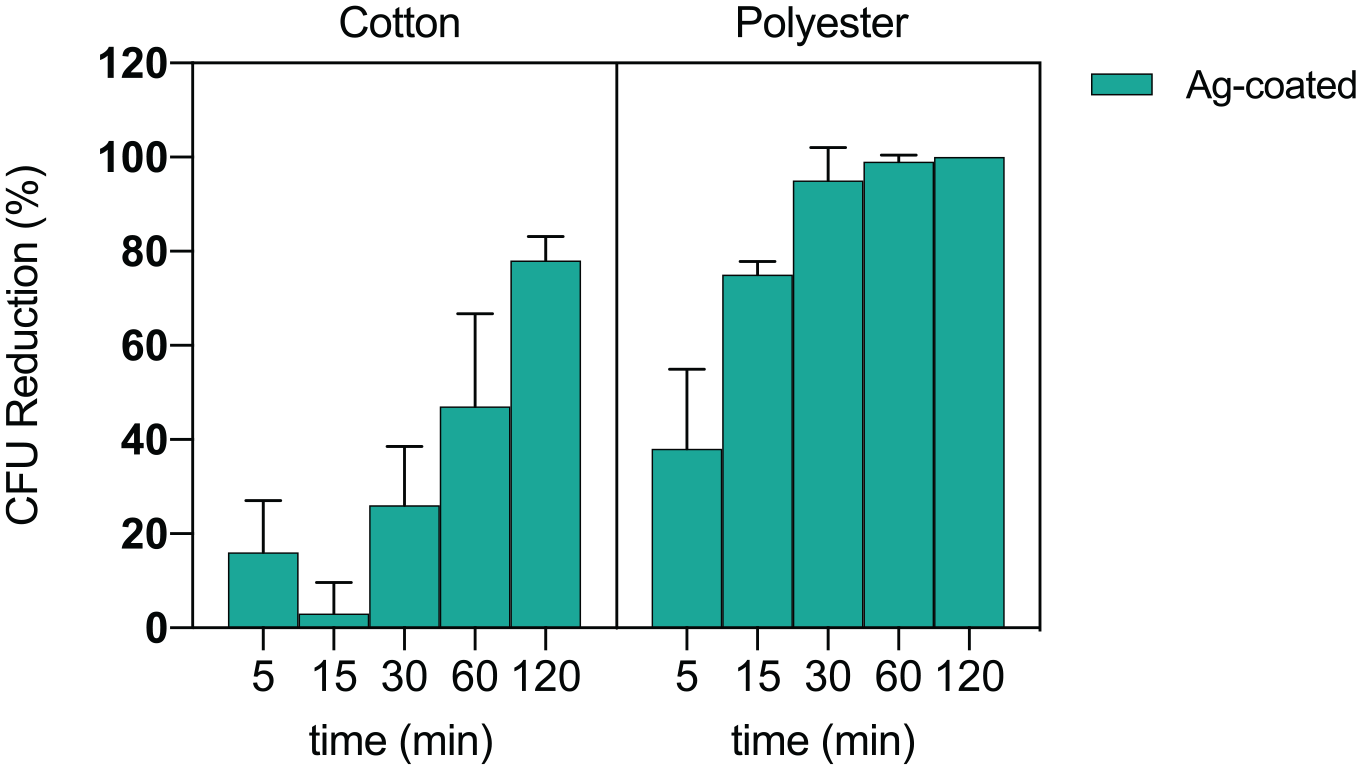
Antibacterial activity of Ag-coated cotton and polyester textiles versus E. coli, at different time points. Data are presented as percentage reduction of CFU number with respect to the uncoated controls. Results are expressed as mean ± SD calculated from at least three independent experiments.
The antibacterial properties of the tested samples were further confirmed by a qualitative analysis of E. coli growth in broth suspensions incubated for 24 h with the samples already used during the direct contact assay. Both suspensions containing uncoated textiles, previously tested with the inoculum for 5 and 60 min, were turbid, indicating the proliferation of the bacterial cells that remained trapped within the samples after their washing. On the contrary, the suspensions containing Ag-coated samples were clear, demonstrating the ability of the silver coating to prevent E. coli growth.
Antiviral properties against SARS-CoV-2 and Feline Calicivirus
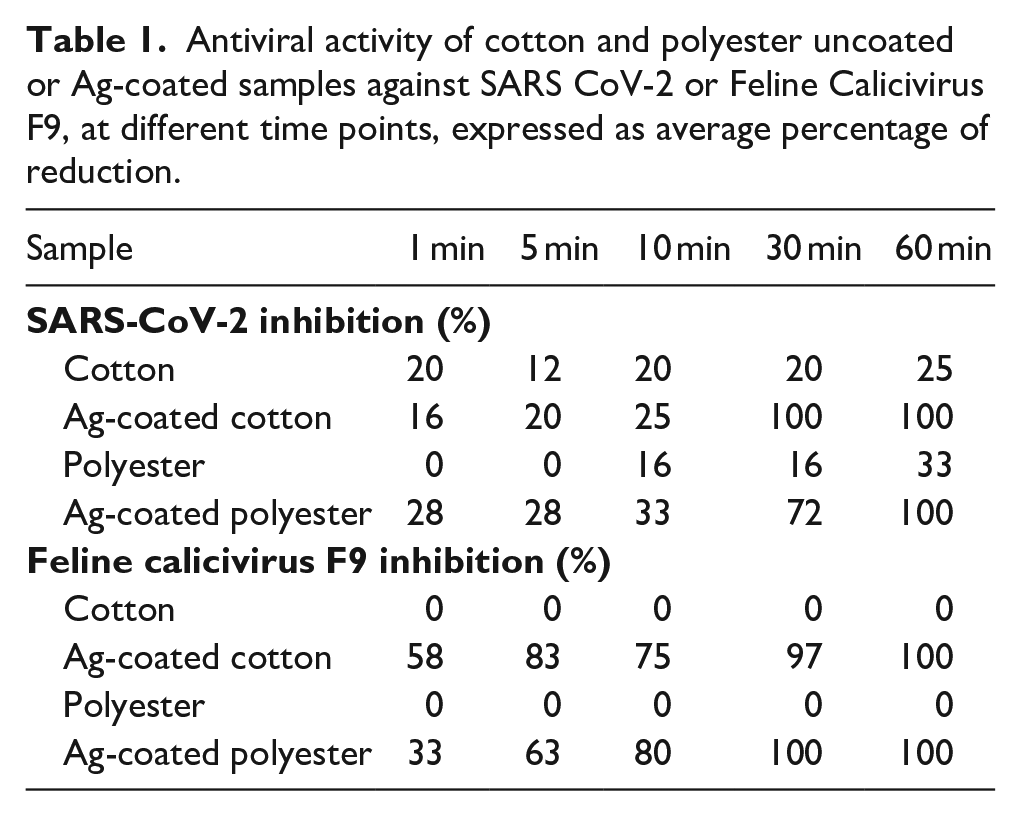
Antiviral properties of Ag-coated textiles were performed against SARS-CoV-2 or human norovirus surrogate FCV after 1, 5, 10, 30, and 60 min of contact. The results of virus inhibition are reported in Table 1. Regarding SARS-CoV-2, the Ag-coated cotton fabric showed better antiviral properties than the Ag-coated polyester fabric at earlier time points. In fact, at 30 min of contact, Ag-coated cotton had an antiviral capacity of 100% with respect to 72% of Ag-coated polyester. For longer contact times no differences were observed between the samples as they both reached 100% of viral inhibition at 60 min. Regarding FCV, Ag-coated cotton and polyester have a more pronounced and faster antiviral response, with 83% and 63% of viral inhibition, respectively, after only 5 min of contact, while a complete inhibition was reached after about 30 min.
Antiviral activity of cotton and polyester uncoated or Ag-coated samples against SARS CoV-2 or Feline Calicivirus F9, at different time points, expressed as average percentage of reduction.
Differential pressure assessment
Differential pressure evaluations were performed by measuring the differential pressure on samples. After the silver coating, only a slight decrease of ∆P is observed: untreated and treated cotton showed ∆P values of 15 and 12 Pa cm−2, respectively, while untreated and treated polyester showed ∆P values of 55 and 53 Pa cm−2, respectively.
Discussion
In this study, we introduced a simple in situ method for coating fabrics, specifically cotton and polyester, to provide them with antimicrobial properties. The Ag-coated textiles were obtained in a few minutes at room temperature through the electroless silver plating process, using SnCl2 as a sensitizing agent and AgNO3 as a photo-reducing agent.
In situ coating approaches, as reported by Perera et al., 25 are more favorable in terms of uniformity, antimicrobial properties and durability retaining textile functional properties closer to the original one compared to ex situ one. In our previous work, 15 we successfully coated polypropylene surgical masks through the silver mirror reaction. However, when the same coating method was applied to either cotton or polyester fabrics, it failed to produce biocompatible antibacterial textiles. Therefore, we sought an alternative method of functionalization, and investigated the electroless silver plating process. Usually, this method requires several steps to successfully functionalize non-conductive textiles,17,18 whereas in this work only tin sensitization is performed before AgNO3 photo-reduction by daylight irradiation. Tin sensitization is a well-known method to favor the direct and uniform deposition of metal layer on non-autocatalytic surfaces. The substrate immersion in SnCl2 solution with HCl and the subsequent washing step in water forms a gel-like layer with dispersed tin ions on the fibers surface. The immersion in AgNO3 creates silver nucleation sites on tin-sensitized fibers that become catalytic sites for the electroless silver plating reaction.26,27
Along with SnCl2 sensitization, silver photo-reduction is directly dependent on the reaction time and light irradiation conditions, such as daylight 28 or UV. 29 In this study, the in situ formation and adhesion of the silver coating were obtained by daylight irradiation. The formation of AgNPs is also directly influenced by the reaction time, with both the quantity and size of particles increasing proportionally with time.28–30 Herein, morphological evaluations by SEM analysis did not show significant differences between uncoated and Ag-coated textiles, even at high magnifications (Supplemental Figure S1). Furthermore, EDX analysis (not shown) failed at identifying the presence of elemental silver in both types of textiles. Probably the relatively short reaction time (only 30 s in AgNO3 solution) employed during the electroless silver plating has been insufficient to allow AgNPs formation. Similarly, in a previous work, AgNPs were detected only through transmission electron microscopy, whereas SEM analysis only revealed a change in surface roughness. 29 To prove this hypothesis, we increased the coating time of samples. Indeed, we demonstrated that, by extending the reaction time in AgNO3 to 2, 6 and 24 h, AgNPs formed both on cotton (Supplemental Figure S2) and polyester (Supplemental Figure S3).
Contrary to EDX, XPS analysis detected the presence of silver. The different amount of Ag species, 0.9 at. % and 2.0 at. %, respectively, for cotton and polyester samples could be possibly ascribed to a difference in the original surface roughness of the two fabrics, which has an influence and plays a significant role in the final use of the textile itself. At the same time, the periodic variations in the surface morphology of the fabric, due to the regular woven patterns of the yarns, cause a respective variation in the geometric roughness measurement. According to Sülar et al.’s 31 extensive work on cotton and polyester woven fabrics roughness and frictional properties, cotton textiles (either plain or twill weaves) show an average lower value of surface roughness compared to polyester ones. Thus, an average higher degree of roughness is expected to provide more anchoring points in which our Ag species can be accommodated. 32 Moreover, this may be linked also to the interaction of the substrate with water. Indeed, cotton is naturally hydrophilic thanks to the abundance of hydroxyl groups, whereas polyester is usually a hydrophobic material, although the polyester fabric used in this study is hydrophilic due to the weave structure and low density that allow fast water penetration. 33 The electroless deposition of silver on the textiles significantly changed the original wettability of samples; the more hydrophobic behavior observed for treated fabrics can be attributed to the presence of silver 13 and to the increased surface roughness of the fibers, as observed by SEM.29,33
Compared to other studies, these results confirmed that we were able to obtain Ag-coated textiles through the electroless deposition by employing fewer steps, reagents, and shorter reaction times. Indeed, Jiang et al. 18 employed four main steps to coat cotton and polyester textiles: (i) cleaning with a detergent, (ii) sensitization with SnCl2, (iii) plating process in a AgNO3 and glucose solution and (iv) washing with water. In another work, electromagnetic interference shielding and antibacterial cotton textiles were fabricated by Riaz et al. with a multiple step approach. 34 First cotton was dip-coated several times in a PEDOT:PSS solution along with polyethylene glycol and a silane crosslinking agent. Then, after PSS removal, sensitization and activation steps with SnCl2 and AgNO3 were applied, and finally copper and silver layers were applied by electroless deposition. Although good antibacterial efficacy against S. aureus was obtained, the reaction time for the electroless deposition processes was longer than 2 h. Similarly, Liu et al. 35 proposed a silver functionalized cotton material for smart wearable equipment using a combination of polymer brush grafting and electroless deposition, with an optimal electroless deposition time of 20 min.
The primary aim of our study was to produce antimicrobial and biocompatible coatings for healthcare settings, within a short reaction time and at room temperature, regardless of the formation of distinct AgNPs. However, many studies on antimicrobial textiles, or antimicrobial coatings in general, lack of biocompatibility evaluations.7,9,12,13,18,29,30 Due to the contact of many fabrics with human skin, the biocompatibility of the Ag nano-finished fabrics was studied either with L929, or HaCaT cells, which are the principal cell type of the epidermis (∼80%). Results from indirect contact tests demonstrated that both fabrics are highly biocompatible. Compared to the uncoated samples, the antimicrobial Ag coating on either fabric did not significantly decrease cell viabilities, which remained around 80% for both cell types.
As already reported, various textiles coated with silver possess high antibacterial properties, even though they require long time (usually 24 h) to exert their action.9,12,18,29,33
On the contrary, the electroless silver plating process used in this work allows to obtain an efficient antibacterial Ag coating even on such different textiles, as cotton and polyester, that both exerted their action in very short contact times (from 30 to 120 min). Nevertheless, the antibacterial efficiency of Ag-coated polyester samples is higher than the Ag-coated cotton ones. This is probably due to the double amount of Ag2O on the polyester surface with respect to the cotton one, as from XPS results. In fact, it was observed that the antibacterial activity increases with the Ag dosage deposited on fibers. 35 Ag-coated textiles showed great antiviral properties against SARS-CoV-2 and FCV. Both these viruses are transmitted mainly by airborne particles or respiratory droplets and are highly contagious. In particular, Ag-coated polyester achieved 100% SARS-CoV-2 inhibition after 60 min of contact, while the complete defeat of the virus with Ag-coated cotton was achieved even faster, after only 30 min of contact. With the human norovirus surrogate, FCV, both functionalized textiles have a faster and more effective virucidal response, halving the viral load after less than 5 min. On the contrary, other antimicrobial textiles functionalized with silver fail to reach a complete inhibition of the virus, even with longer contact times,11,15 while Kumar et al. 29 presented comparable results at 30 min of contact with SARS-CoV-2.
The synergic effect of Ag intrinsic antimicrobial properties and modified surface characteristics (greater roughness and hydrophobicity) are responsible for efficient textiles antimicrobial activity. In particular, the increased roughness results in an increased contact area with the antimicrobial agent while its hydrophobicity inhibits bacterial/virus adherence. 29
Finally, this functionalization method not only allows to confer bactericidal and virucidal activity to cotton and polyester, but also guarantees the preservation of textile functional properties, such as differential pressure.
Conclusions
The electroless silver plating is an autocatalytic process that can be employed to functionalize textile materials, providing antibacterial and antiviral activity. The protocol designed in this work offers a rapid, effective, and convenient process in terms of reactants and equipment to coat cotton and polyester textiles.
The silver coatings changed the surface roughness of fibers and increased the hydrophobic behavior of both the textiles, and they did not affect the differential pressure parameters. Ag-coated cotton and polyester also demonstrated optimal biological properties, such as high biocompatibility with murine L929 fibroblasts or human HaCaT cells, and fast antibacterial activity against E. coli from 5 min of contact. Notably, these coatings applied on cotton or polyester inhibited SARS-CoV-2 and FCV viruses, achieving 100% viral reduction in about 60 or 30 min of contact, respectively. The chemical binding, stability, storage conditions, and resistance to washing of the coating could be further investigated to confirm the durability of the antimicrobial properties for commercial long-term applications.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000241277383 – Supplemental material for Electroless silver plating on fabrics for antimicrobial coating: comparison between cotton and polyester
Supplemental material, sj-docx-1-jbf-10.1177_22808000241277383 for Electroless silver plating on fabrics for antimicrobial coating: comparison between cotton and polyester by Ivan Vito Ferrari, Micaela Castellino, Anissa Pisani, Giulia Giuntoli, Aida Cavallo, Tamer Al Kayal, Paola Mazzetti, Alfredo Rosellini, Maria Sidoti, Antonino Cataldo, Mauro Pistello, Giorgio Soldani and Paola Losi in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
The authors wish to thank Dr. Vassili Fotis for his private donation which allowed the purchasing of SEM microscope employed in this study.
Authors contribution
Conceptualization: IVF and PL; Methodology: IVF, MC, AP, GG, ACav TAK, PM, AR, MS, and ACat; Data curation: IVF, MC, AP, GG, ACav, and PM; Writing—original draft: IVF, MC, AP, GG, and ACav; Writing—review and editing: IVF, MC, AP, GG, ACav, MP, and PL; Supervision: PL; Funding acquisition: GS and PL. All authors have read and agreed to the published version of the manuscript.
Data availability
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financially supported by STOP-VIRUS project. This research project is funded by Tuscany Region through the “Programma Attuativo Regionale” financed by FSC and through MUR FAR funds.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.