Abstract
The biodegradable metals have great potential for the biomedical applications, which could be gradually degraded, absorbed, or excreted in the human body, avoiding the removal though secondary surgery. Zinc-based alloys are novel series of degradable metals for medical applications, and they are gaining lots of attention in the research field of absorbable metals. Zinc-silver (Zn-Ag) alloys show superior mechanical strength, good biodegradability, biocompatibility, and antibacterial properties, which render them to be potential candidates for biomedical applications. In this paper, we reviewed the development of Zn-Ag alloys in terms of mechanical properties, degradabilities, biocompatibilities, antibacterial properties, and potential applications in dentistry.
Introduction
Bioabsorbable metals are regarded as revolutionary biomedical materials, which are gained lots of interest in medical device applications, such as fracture fixation devices, cardiovascular stents, guided bone regeneration (GBR) materials, etc. 1 Currently, non-degradable metals such as titanium mesh for GBR need a second surgery to remove after its function is fulfilled. 2 Recent researches on biodegradable metals provide the possibility to avoid the second surgery and thus can greatly improve patient compliance and reduce the medical costs.3,4 Biodegradable metals have attracted extensive attention in the medical field due to their excellent mechanical properties and biodegradability. There are mainly three kinds of biodegradable metals: magnesium (Mg), iron (Fe), and zinc (Zn) based alloys. The Mg-based alloys are widely studied, however, their applications were limited by the rapid corrosion rate in physiological environments5–7 and the by-products, that is, hydrogen, produced in the degradation process of Mg alloy, which hinders them to match the growing demand of bone and not conducive to the healing of tissue.8,9 In contrast, the corrosion rate of Fe-based alloys is relatively slow compared to the clinical requirements, which unnecessarily prolongs the tissue exposure time to the metals.10,11 The degradation rate of Zn is between Mg and Fe, and the degradation products can be completely absorbed without hydrogen production. 12 As the second most abundant intracellular cation and the fourth most common cation in the body, Zn is an essential substance for hundreds of enzyme reactions which are related to growth, development, immune response, diseases, and even cancer. 13 For instance, Zn is necessary for osteogenesis and mineralization as it can inhibit bone absorption induced by osteoclast and promote osteoblast proliferation. 14 Given its important role in physiological and biological systems, Zn is known as “calcium in the 21st century.” 15 The lack of Zn will affect the growth and development, while the intake of Zn at ten times the daily dose rarely causes an adverse effect. 16 Therefore, there is a large window for Zn-based alloys in clinical application.
Although Zn-based alloys have promising degradation performance for biomedical applications, their mechanical properties are relatively limited. The tensile strength of as-cast pure Zn is 18 MPa, and the fracture elongation is only 0.32%. 17 Fortunately, the mechanical strength of the alloys can be significantly improved by adding Mg, copper (Cu), silver (Ag), calcium (Ca), strontium (Sr), aluminum (Al), and lithium (Li). 18 Among them, Zn-Ag based alloys showed not only enhanced mechanical strength, but also antibacterial properties, 19 which can prevent complications like infection that is a major challenge for the application of degradable metals in vivo. Therefore, Zn-Ag alloy is promising as a new generation of degradable metals with antibacterial properties and may be suitable for biomedical applications. To this end, the research advances of the Zn-Ag-based alloys as a new generation of biodegradable metals are reviewed in terms of their mechanical property, biodegradability, biocompatibility, and antibacterial property.
Mechanical properties
The mechanical properties of pure Zinc are relatively limited. The tensile strength of as-cast pure Zn is 18 MPa, and the fracture elongation is only 0.32%. 17 The melting point of Zinc is relatively low, that is, 420°C. 20 The low melting point of Zn facilitates the fabrication of Zn based alloys. Zn-Ag based alloys can improve its mechanical properties.
Effect of Ag content
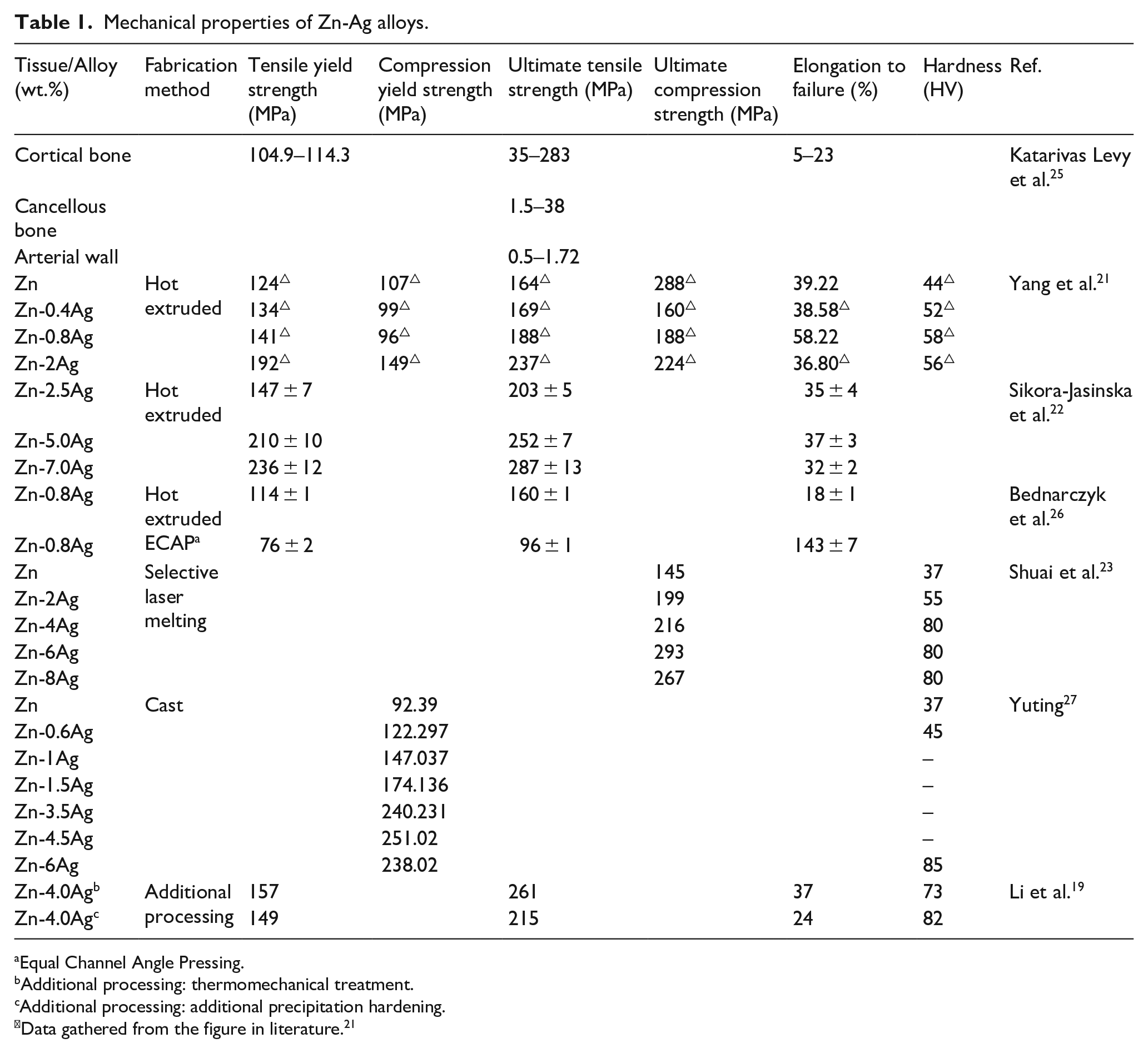
As an important component of Zn-Ag alloy, Ag has a great influence on the mechanical properties of Zn-Ag alloy. Table 1 summarizes the mechanical properties of different Zn-Ag alloys. Currently, the preparation methods of Zn-Ag based alloys can be divided into conventional methods and advanced processing techniques. The former mainly include casting and hot extrusion while the advanced processing techniques consist of powder metallurgy, additive manufacturing, severe plastic deformation and electroforming techniques, etc.
18
Regardless of the fabrication methods, the mechanical properties such as yield strength and tensile strength seem to reach a peak value at certain content. For instance, the addition of Ag can significantly improve the mechanical properties of the hot extruded Zn-Ag-based alloys.21,22 When the Ag content reached 7.0 wt.%, the yield strength and tensile strength reached the maximum values, that is, 236 and 287 MPa, respectively.
22
A similar pattern can be found with Zn-Ag alloys by selective laser melting technology. Zn-xAg (
Mechanical properties of Zn-Ag alloys.
Equal Channel Angle Pressing.
Additional processing: thermomechanical treatment.
Additional processing: additional precipitation hardening.
Data gathered from the figure in literature. 21
The fracture elongation (Ef) is a paramount parameter to evaluate the plasticity of materials to show the potential for metal processing. The elongation of pure Zn is closely related to its preparation method. For example, the Ef of the as-cast pure Zn is 0.32% while that of hot extruded pure Zn is 60% at 250°C. 28 For the hot-extruded Zn-Ag alloy, the maximum fracture elongation can be 58.22% ± 7.18% with the lower content of Ag (0.8%) at 260°C. 21 With the increase of Ag content (2.0%–7.0%), the fracture elongation has no further change, which is basically stable between 32% and 36%. 22
The influence of other elements
To further improve the mechanical properties of Zn-Ag based alloys, other metal elements can be incorporated. The available compositions of those alloys have been summarized in Figure 1.

The most commonly added elements are Li, aurum (Au), vanadium (V), zirconium (Zr), and manganum (Mn).29,30 The Zn-4Ag-xMn (
The orthopedic fixation devices and cardiovascular stents are the most potential applications for biodegradable metals. As for cardiovascular stents, the yield strength, tensile strength, and elongation of the materials need to be greater than 200 MPa, 300 MPa, and 15%, respectively.12,33,34 Regarding the orthopedic fixation devices, the yield strength should be more than 230 MPa. Based on the aforementioned data, Zn-Ag alloys can meet the requirements of those devices in terms of yield strength and fracture elongation, with the exception that their tensile strength is relatively limited. The addition of Mn optimizes the tensile strength of Zn-Ag-based alloys and Zn-4.0Ag-0.6Mn alloy is quite promising for these applications. 31 Since the data are limited by only a few studies focused on Zn-Ag-based alloys, more researches should be conducted with different proportions or multiple components to meet the clinical needs for biomedical applications.
Degradability
Degradation rate
Corrosion rates of biodegradable metals can be evaluated by immersion (corrosion rate of immersion, CRI) and electrochemical (corrosion rate of electrochemistry, CRE) experiments. Table 2 summarizes the degradation properties of Zn-Ag alloys in the available studies.
Degradation rate of Zn-Ag alloys.

Stimulated body fluid.
DMEM/F12: Dulbecco’s modified Eagle’s medium/nutrient mixture F-12.
Conversion from µm/year in the original text. 35
Generally, Zn-Ag alloy showed both higher CRI and CRE than pure Zn.22,23,27 According to the electrochemical theory, a larger corrosion potential (Ecorr) and a lower corrosion current densities (Icorr) indicates a smaller corrosion rate. 36 Compared with pure Zn, Zn-Ag alloys showed a smaller Ecorr and a larger Icorr, which indicated that the Zn-Ag alloys had a relatively greater corrosion rate than pure Zn. Besides, the increase of Ag content resulted in a higher degradation rate.22,27 This phenomenon may be due to the micro-galvanic corrosion between the second phase AgZn3 and the matrix.19,23,37 Since different biomedical applications require different degradation rates of the materials, Zn-Ag alloys have the potential to provide different degradation rates by altering the alloy composition.
Besides, the type of extraction of media can affect the corrosion rate of Zn-Ag based alloys. The extraction media containing buffering agents and glucose similar to those of human plasma is critical for the prediction of in vivo corrosion rate. 38 More details will be discussed in section 4.1.
Degradation products
According to ASTM standard F2129, in vitro corrosion test is commonly carried out with electrolytes containing simulated human plasma chemical composition. 39 The degradation mechanism of Zn in the electrolyte follows the reactions given in equations (1)–(7).33,40,41

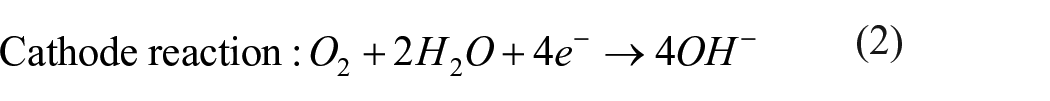




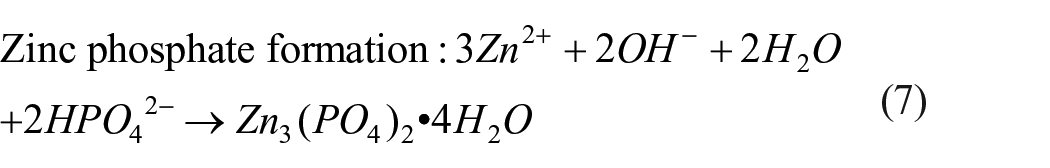
According to the micro-galvanic corrosion theory, the Zn in the alloys corrodes preferentially when both Zn and ZnAg3 is in contact with each other in the presence of an electrolyte. Indeed, the degradation products of Zn-Ag alloys generally have high Zn content with the Ag content below the detection threshold (<50 μg/L).19,22,23,32,35,42,43 For instance, the mass fraction of Zn in the degradation products of Zn-xAg (
The degradation process of Zn-Ag alloys is demonstrated in Figure 2 . It is worthy to note that no gas is generated in the degradation process of Zn-Ag alloys, which may have an obvious advantage over Mg-based alloys. The main precipitates produced in the degradation process are ZnO and Zn(OH)2.22,42 The other degradation products vary depending on different immersion solutions. The high concentration of Cl¬ in the physiological electrolytes can break the equilibrium between the formation and the dissolution of corrosion products, and consequently convert the insoluble ZnO and Zn(OH)2 into soluble chloride salts (equations (5) and (6)). 41 Besides, the HPO42− in the Dulbecco’s phosphate-buffered saline (DPBS) can react with released Zn2+ to form insoluble zinc phosphate (equations (7)). 33 It is noteworthy that the selective dissolution of zinc will result in an enrichment of AgZn3 phase at the alloy surface and eventually the AgZn3 particles may be released (Figure 3), 37 consequently leading to an increase in the concentration of silver ions in the solution. To the best of our knowledge, the degradation mechanism and biocompatibility of AgZn3 is not fully elucidated yet. Further studies are needed to clarify the degradability, cytotoxicity, and biocompatibility of AgZn3.

Schematic diagram of Zn-Ag alloy degradation: (a) ZnO/Zn(OH)2 precipitation are formed at the initial stage of degradation, (b) chloride ion degrades ZnO/Zn(OH)2 to produce soluble zinc chloride salts, and (c) zinc phosphate and calcium phosphate precipitates are formed when phosphate groups are encountered.
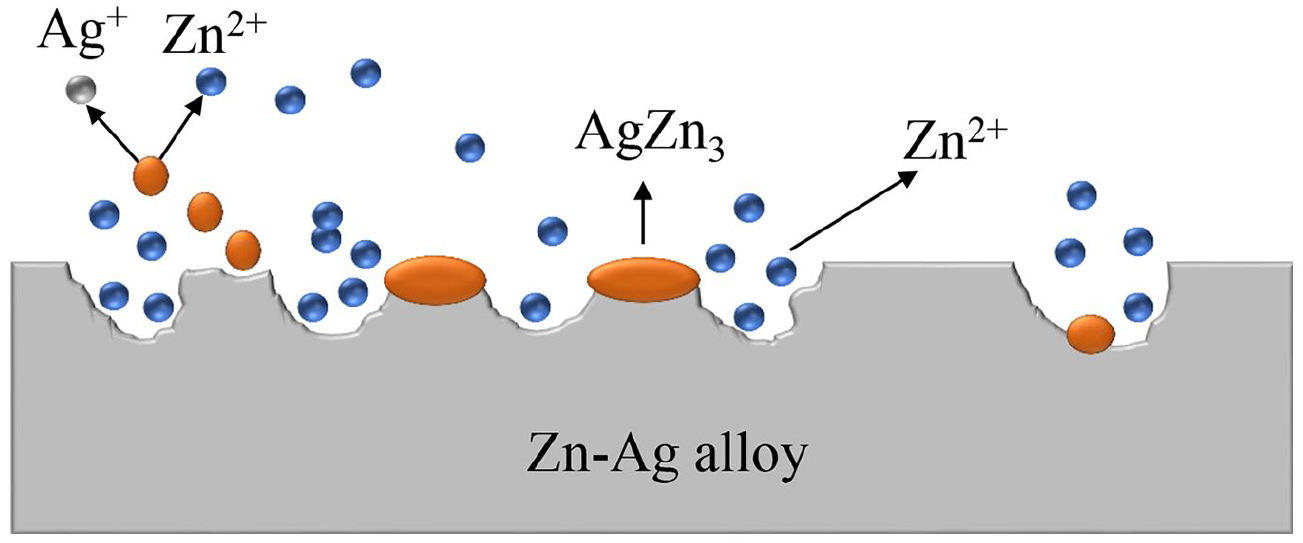
The corrosion mechanism of Zn-4Ag with selective dissolution of the zinc phase leaving the AgZn3 phase uncorroded.
Biocompatibility
In vitro studies
According to the international standard ISO 10993-5, extract test, direct contact test, and indirect contact test are the most commonly applied methods for in vitro cytotoxicity evaluation. As biodegradable metals, the biocompatibility of Zn-Ag alloys is closely related to their degradation products and surface properties.
Regarding biodegradable metals, an extraction test is the most commonly adopted method to evaluate their cytotoxicity. Mouse fibroblast line (L929) and human osteosarcoma cell line (Saos-2) were cultured in the extraction test of Zn-4Ag alloy at different concentrations (100%, 33.3%, 16.7%, 10%) for 24 h. The undiluted extracts showed obvious cytotoxicity, while the dilution of 10% and 16.7% of the original extract showed no cytotoxicity. 19 Similar results were also reported by other scholars.32,44,45 Therefore, the cytotoxic effect of Zn-Ag-based alloys can be significantly reduced with dilution.
Zn2+ is the major degradation product of Zn-Ag alloy. Ma et al. 46 cultured human aortic smooth muscle cells (HAOSMCs) in different concentrations of ZnCl2 solution for 24 h, and found that Zn2+ promoted the adhesion and proliferation of HAOSMCs with the concentrations lower than 80 μM while it has an inhibitory effect at the concentration of 100–120 μM. A similar biphasic effect was revealed on cellular osteogenic differentiation. 35 2% pure Zn and Zn-4Ag extract promoted osteogenic differentiation of TAg cells, while 50% extract inhibited osteogenic differentiation. 35
As the other major component of Zn-Ag-based alloys, Ag has been rarely studied on its cytotoxicity, which may be related to the fact that the release of silver in Zn-Ag alloy is mostly trivial and is below the detection threshold (<50 µg/L).32,35 The LC50 of silver was reported to be 11.0 µM for tumor cells (MG-63) and 9.0 µM for macrophages (RAW 264.7). 47 The discrepancy between the actual released silver concentration and LC50 may indicate that the silver has little effect on biocompatibility in Zn-Ag alloys, just as reported previously that metallic and alloyed silver is neither genotoxic nor cytotoxic. 48 Nevertheless, the deposition of a great amounts of insoluble silver precipitates in the human body can lead to argyria or argyrosis.49,50 It is thus suggested that the evaluation of the Zn-Ag based alloys in vivo before biomedical applications.
Nevertheless, the current extraction test of the biodegradable metals lacks standardization. Firstly, the cytotoxicity of Zn-Ag alloy is related to the type of immersion solution in vitro experiment.19,51 Törne et al. 52 compared the electrochemical behavior of pure Zn in PBS, ringer’s solution, simulated plasma, and whole blood, and found that the degradation rate of pure Zn in whole blood and simulated plasma was higher. This difference could be contributed to the rapid blood protein adsorption onto the surface of Zn, which not only hindered the formation of a protective film layer on the surface but also greatly increased the corrosion current. 53 Also, Li et al. 51 found that the concentration of Zn2+ released in the extract containing FBS for 24 h was higher than that without FBS, and consequently, the extract containing FBS showed obvious cytotoxicity to L929 and Saos-2 cells while the extract without FBS had no cytotoxicity. Secondly, the rapid accumulation of the degradation products of Zn-Ag-based alloys in vitro conditions is difficult to represent the real in vivo environment with circulating body fluids. The evaluation standard of ISO 10993-5 or ISO 10993-12 was even questioned not suitable for evaluating degradable metals as it was found that the extract of Mg alloys should be diluted between 6 and 10 times to be comparable with that in vivo conditions. 44 In addition, Fischer et al. 45 uncovered that the influence of osmotic pressure should be eliminated by applying 10 times more extraction medium than recommended by the ISO standards in vitro before conducting the extraction test. Since the cytotoxicity of degradable metals is closely related to the kinds of extraction solutions and their dilution times, more standardized methods should be developed considering various factors such as the type of immersion solution and the extract dilution to evaluate the cytotoxicity of degradable metals including Zn-Ag-based alloys.
Only a few studies are available about the cytotoxicity of Zn-Ag alloys with direct contact test. HAOSMCs were directly seeded on the alloys for 24 h, the proliferation and metabolic activity of HAOSMC only decreased slightly to moderately with no more cell death observed compared with the control group. 43 More in-depth studies are needed to elucidate the cytotoxicity of Zn-Ag-based alloys on different cells.
In vivo studies
Only a few in vivo experiments were carried out to evaluate Zn-Ag-based alloys. Zn-3Ag stents were placed in the swine iliac artery for 6 months, and the stents were endothelialized and covered with new intima without thrombus formation, thus indicating that Zn-Ag alloy has a good prospect as a cardiovascular stent. 43 Besides, Yang et al. 21 implanted Zn-2Ag alloys in rat femurs and found new bone formation around the alloy 8 weeks later with less than 10% degradation rate of the Zn-2Ag alloys. As the degradation rate is relatively slow, this degradation pattern may provide the mechanical properties as fracture fixation devices, which commonly require to maintain mechanical strength for 3–6 months. 54 Nevertheless, it is worth noting that local corrosion was not evenly distributed on the surface of the alloy with a damage depth of up to 50 μm. 21 Thus the evaluation of mechanical properties is recommended during all application periods to avoid complications.
Antibacterial properties
Antibacterial properties are of great importance for biomedical applications. Zn can inhibit the growth of bacteria and the formation of biofilm, 55 and Ag can effectively kill various microorganisms, such as bacteria, yeast, viruses, and fungi. 56 Regarding the antibacterial properties of silver, it can interact with three main components of the bacterial cells to produce the bactericidal effect: the peptidoglycan cell wall and the plasma membrane, bacterial (cytoplasmic) DNA, and bacterial proteins (Figure 4). 57 As expected, Zn-Ag-based alloys showed a broad-spectrum antimicrobial activity against both Gram-positive bacteria such as Staphylococcus aureus (S. aureus) 30 and Streptococcus goeth19,32 and Gram-negative bacteria such as Escherichia coli (E. coli).27,30,42 The antibacterial efficacy of the Zn-Ag alloys was positively related to Ag content, with Zn, Zn-1Ag, Zn-1.5Ag, Zn-3.5Ag, Zn-4.5Ag, and Zn-6Ag showing 20%, 40%, 65%, 99.78%, 99.99%, and 100% killing rate of E. coli, respectively. 27 Similarly, the antibacterial property of the Zn-Mg-Ag alloys enhanced with the increase of silver content. 58 The greater the amount of active silver ions are released, the more effective the antibacterial properties would be. 59 Therefore, the increase of silver content in Zn-Ag alloy may potentially increase the release of silver, which in turn improve the antibacterial properties, possibly just as reported previously in Mg-Ag alloys. 60 In addition, the shape of Zn-Ag alloys seemed to have an impact on the antibacterial property, as the antibacterial property of porous Zn-Ag alloy was better than that of bulk one. 42 This was also due to the porous alloys provide a larger surface area/volume ratio and thus increase the contact surfaces with bacteria. In rat models, pure Zn, Zn-Al alloy, Zn-Sr alloy, Zn-Mg alloy, Zn-Ag alloy, Zn-Ca alloy, and Zn-Cu alloy were implanted into the bone marrow cavity of the femurs and challenged with S. aureus and E. coli inoculation, the results revealed that all implanted materials showed antibacterial effect and Zn-Ag alloy had the optimal antibacterial performance. 61 Therefore, the antibacterial properties of Zn-Ag-based alloys are probably from both Zn and Ag elements. 32
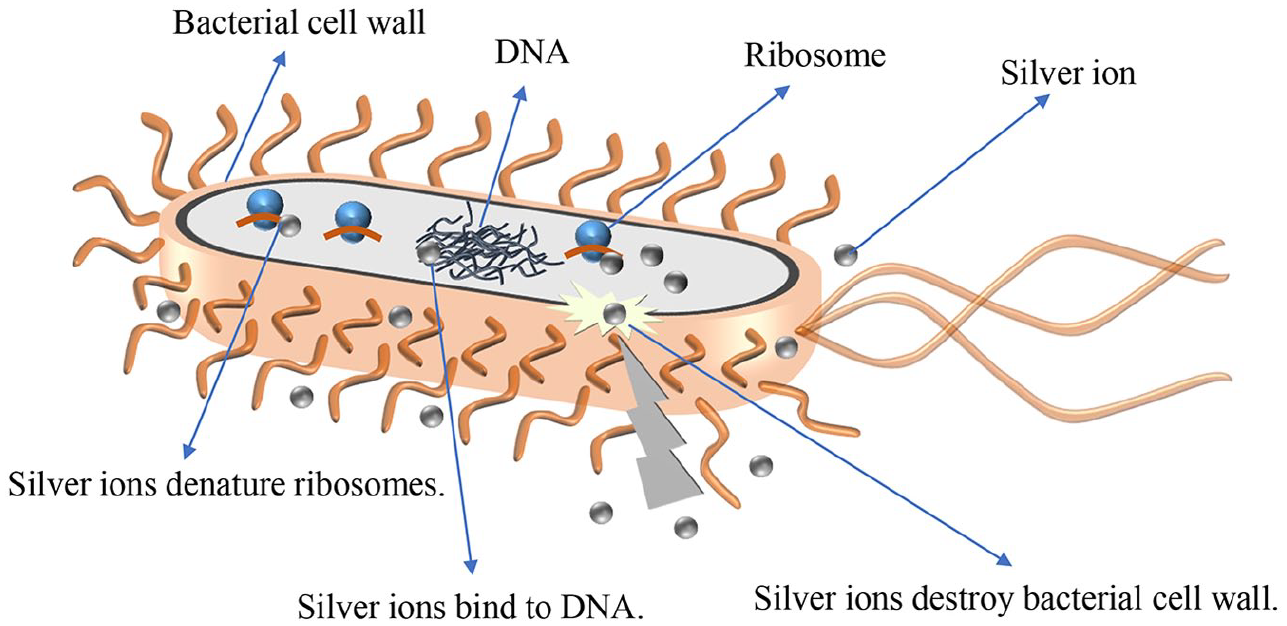
Mechanisms of the antibacterial activity of silver ions.
Medical applications
Zn-Ag based alloys, as relatively novel biodegradable metals, are still in their infancy for biomedical applications (Figure 5). Owing to their promising biodegradability and mechanical properties, Zn-Ag alloys are considered to be potential biodegradable materials for stenting applications. The tensile strength of hot extrusion Zn-4.0Ag-0.6Mn alloy reached to 302 MPa, meeting the requirements of vascular stent. 31 Currently, the only biodegradable alloy stent that is going through clinical trials is an Mg alloy stent. 62 Since the properties of Zn-Ag alloy are better than Mg alloy in many aspects, the former may be a promising candidate for a vascular stent. More in vivo studies and clinical trials are needed to explore the application of Zn-Ag alloy as cardiovascular stents. Secondly, the mechanical properties of the Zn-Ag alloy can meet the requirements of bone fixation. When Zn-Ag alloys were inserted into the right femur of rats, new bone tissues formed around the implants. 21 Zn-based alloys also showed excellent mechanical properties to support mandibular bone fracture healing, so they might be promising to serve as craniomaxillofacial osteosynthesis implants as well.63,64 Thirdly, Zn-Ag alloy can be used as biodegradable scaffolds for tissue engineering applications. Metal scaffolds play an important role in the treatment, repair, and regeneration of damaged tissues of trauma, and periodontal diseases. Zn-Ag alloys may have some advantages over traditional Titanium scaffolds by avoiding secondary surgery, reducing the risk of infection, and promoting bone regeneration. 65 Take the barrier membrane used in GBR as an example. Barrier membranes can be divided into biodegradable and non-biodegradable membranes. The absorbable membrane represented by aliphatic polyesters lacks rigidity and stability, and the non-absorbable membrane represented by titanium alloy requires a second operation. 66 The introduction of Zn-Ag-based alloys can potentially solve this dilemma by maintaining the space while gradually degrading. This property makes it potentially applicable in bone augmentation. Moreover, the porous Zn-Ag alloy scaffolds have already been prepared by air pressure infiltration and both their mechanical properties and antibacterial ability are higher than those of porous Zn scaffolds with similar porosity. 42 As porous scaffolds have been studied as candidates for orthopedic applications, 67 including bone augmentations, Zn-Ag alloy is promising to be another candidate. Fourthly, Zn-Ag alloy can be developed into a new type of degradable medical suture. The main component of the degradable suture is polymer, which is difficult to meet the dual requirements of good biodegradability and high mechanical strength. 40 Venezuela et al. 68 demonstrated that the tensile strength and elongation of suture materials should be more than 270 MPa and 10%, among which Zn-7Ag could meet this requirement. Biodegradable pure Zn wires with diameters of 3.0 and 0.3 mm were prepared. The ф0.3 mm pure Zn wire exhibited favorable cytocompatibility and demonstrated partly antibacterial effect on S. aureus, which could be a potential suture material. 69 Besides, Zn-Ag alloy suture is also within sight for clinical use. Moreover, Zn-Ag alloy is expected to be promising as an auxiliary device for wound closure such as zinc absorbable staples, plugs, micro clips, and rivets. 68 Though Zn-Ag alloys have great potential for medical applications, further researches are needed for the clinical transformation.

Zn-Ag alloys can be used in human body as: (a) degradable metal scaffolds for alveolar bone regeneration, (b) cardiovascular stents, (c) bone fracture fixators, and (d) suture materials.
Conclusions
Zn-Ag-based alloys possess attractive properties for biomedical applications including adequate mechanical strength, adjustable corrosion rate, low cytotoxicity, and antibacterial properties. These properties endow them with broad application prospects as biodegradable materials. Currently, the researches on Zn-Ag-based alloys are relatively limited, thus more studies are needed to explore the suitable compositions for biomedical applications. In this review, the advances in the development of Zn-Ag-based alloys are elaborated in detail, which hopefully can provide some insight for the following researches, and may be beneficial for researchers interested in transforming Zn-Ag-based alloys from bench to clinic as a new generation of biomedical degradable materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (No. 81901009, 81670993, and 81873716), Shandong Provincial Postdoctoral Innovation Project (202001009), the Construction Engineering Special Fund of “Taishan Scholars” of Shandong Province (No.ts20190975), Jinan Medicine and Health Science Technology Project (No. 201907090), China Postdoctoral Science Foundation (No. 2019M652409 and No. 2019TQ0187), and the Fundamental Research Funds of Shandong University. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors declare that no financial or other potential competing interests exist with regard to this study.
