Abstract
Objectives:
This study aims to investigate the effect of coating time on the formation of hydroxyapatite (HA) coating layer on ZK60 substrate and understand the biodegradation behavior of the coated alloy for biodegradable implant applications.
Methods:
Biodegradable ZK60 alloy was coated by HA layer for different times of 0.5, 1, 2, and 4 h by chemical conversion method. After coating, all the coated specimens were used for immersion test in Hanks’ solution to understand the effect of coating time on the degradation behavior of the alloy. The degradation rate of the coated alloy was evaluated by Mg2+ ion quantification and pH change during immersion test. The microstructure of the coating layer was examined by scanning electron microscope (SEM) equipped with an energy-dispersive X-ray spectroscopy (EDS) before and after immersion to understand the degradation behavior of the coated alloy.
Results:
HA coating layers were formed successfully on surface of ZK60 specimens after 0.5, 1, 2, and 4 h with different microstructure. Optimal coating quality was observed at 1 or 2 h, characterized by well-formed and uniform HA layers. However, extending the coating duration to 4 h led to the formation of cracks within the HA layer, accompanied by Mg(OH)2. Specimens coated for 1 and 2 h exhibited the lowest degradation rates, while specimens coated for 0.5 and 4 h showed the highest degradation rates. Furthermore, analysis of degradation products revealed the predominance of calcium phosphates formed on the surface of specimens coated for 1 and 2 h. Conversely, specimens coated for 0.5 and 4 h exhibited Mg(OH)2 as the primary degradation product, suggesting a less effective corrosion barrier under these conditions.
Conclusion:
The HA layer formed after 2 h demonstrated as the most effective coating layer for enhancing the corrosion resistance of the ZK60 alloy for biomedical applications.
Introduction
Biodegradable magnesium (Mg)-based alloys are attractive materials for temporary load-bearing implantation because of their outstanding properties. Mg is an essential element in human body and influences significantly on bone metabolism. 1 Lightweight Mg alloys have a density of 1.74–2.0 g/cm3 that is similar to the density of human bone (1.8–2.1 g/cm3). 2 Moreover, the elastic modulus of Mg (40–50 GPa) is close to that of natural bone (3–20 GPa); therefore, minimizing the stress shielding effect. 2 Importantly, Mg alloys are able to dissolve in human body environment thanks to the chemically active property of Mg 3 ; therefore, the secondary surgery to remove the implant after bone healing is not needed. These advantages make Mg alloys become very potential candidates for biodegradable implant applications.4–6 However, the fast degradation rate resulting in rapid deterioration of the mechanical performance is still a major drawback of these alloys.7,8
In the past decades, many methods have been developed to control degradation rate of Mg alloys including surface treatment, adding alloying elements, or microstructure modification.4–8 However, the effectiveness of these methods is still limited. Therefore, the objective of this research is to develop a coating method to improve corrosion resistance of Mg alloy for biodegradable implant application.
In biomedical applications, even though with small amount, alloying elements play crucial roles on mechanical strength, corrosion behavior and biocompatibility of the alloys. In Mg-Zn-Zr alloys, both zinc (Zn) and zirconium (Zr) are considered to be bio-safe elements.9,10 Several studies proved that this system alloy exhibited good osteoconductivity and osteoinductivity.11,12 Furthermore, the addition of Zn and Zr into Mg matric enhances greatly mechanical properties of the alloys, which can support fractured bone during its healing process.13,14 Hence, Mg-Zn-Zr alloys are a potential candidate for biodegradable orthopedic implants and was chosen as substrate for this study.
One of the appropriate strategies to enhance not only corrosion resistance but also biocompatibility of the alloys is surface treatment with hydroxyapatite (HA, Ca10(PO4)6(OH)2) because HA accounts for the most major component of human bone, improves osseointegration of bone tissue and accelerates bone formation. 15 Among the surface treatments that introduce the HA coating on Mg alloys, the chemical conversion method is environmentally friendly, cost effective and applicable for all shape of the substrate. 11 Previous studies focused on the enhancement of corrosion resistance of pure Mg, Mg-Al-Zn and Mg-RE alloys after coated HA by this coating method.16–18 Up to date, the influence of coating duration on the formation and biodegradable performance of the HA coating formed on the Mg-Zn-Zr alloy has not been investigated yet.
Previous studies showed that coating time is an important parameter that may affect significantly the formation, mỉcrostructure, and the quality of HA coating layer.17,19 The short coating duration resulted in thin and inhomogeneous coating layer, while too long coating time led to formation of cracks or corrosion initiation to the substrate. Therefore, the objective of this study is to understand the effect of coating time on the formation of HA coating layer and degradation behavior of HA-coated Mg-Zn-Zr alloy that was coated for different durations: 0.5, 1, 2, and 4 h. By this optimization process, the best coating condition was achieved for biodegradable implant applications. In this study, Mg-6mass%Zn-0.5mass%Zr (ZK60) was selected as a substrate because this is a commercial alloy and is well-known for its high mechanical strength.
Experiment and method
Material preparation
Commercial ZK60 alloy used for this study was supplied by Osaka Fuji Industry, Japan with chemical composition described in Table 1. The ZK60 bars with diameter of 20 mm were machined into thin disc specimens with thickness of 2 mm. The specimens were then ground with SiC papers with the grit from #320 to #1200. All the specimens were then ultrasonically cleaned with ethanol and dried in air for the coating process.
Chemical compositions of ZK60 alloy (mass%).
Coating process
The coating process was started with coating solution preparation. Ethylenediaminetetraacetic acid calcium disodium salt hydrate (C10H12CaN2Na2O8, Ca-EDTA) 0.5 mol/L solution was mixed with potassium dihydrogen phosphate (KH2PO4) 0.5 mol/L solution by the volume ratio of 1:1. After that, pH of the mixed solution was adjusted to 7.5 by NaOH.
Three specimens were immersed in 100 mL coating solution for each coating condition. The specimens were coated at 90°C. The coating time was changed to 0.5, 1, 2, and 4 h. After the coating process, the specimens were retrieved from the coating solution, cleaned with distilled water, and dried for material characterization and corrosion test.
The coated specimens were then characterized by Bruker D8 Advance X-ray diffractometry (XRD) using CuKα radiation and field-emission scanning electron microscope (SEM, JEOL 7600F).
Immersion tests
Hanks’ solution was prepared for immersion test. The composition of the prepared Hanks’ solution is as follows: NaCl 8 g/L, KCl 0.4 g/L, Na2HPO4.H2O 0.06 g/L, KH2PO4 0.06 g/L, MgSO4.7H2O 0.2 g/L, NaHCO3 0.35 g/L, CaCl2 0.14 g/L. The pH of Hanks’ solution was adjusted to 7.4. The immersion test was carried out following ASTM G31-03 in an incubator with temperature of 37°C for 28 days.
After 1, 3, 5, 7, 14, and 28 days, the amount of Mg2+ ions in the solution was quantified by colorimetric method. 20 The pH of the solution was measured at these period as well. After 28 days, all the specimens were removed from the solutions, cleaned, and dried for further analysis.
The immersed specimens were analyzed by an optical microscope (OM), a scanning electron microscope (SEM) equipped with an energy-dispersive X-ray spectroscopy (EDS).
Results
Formation of the HA coating
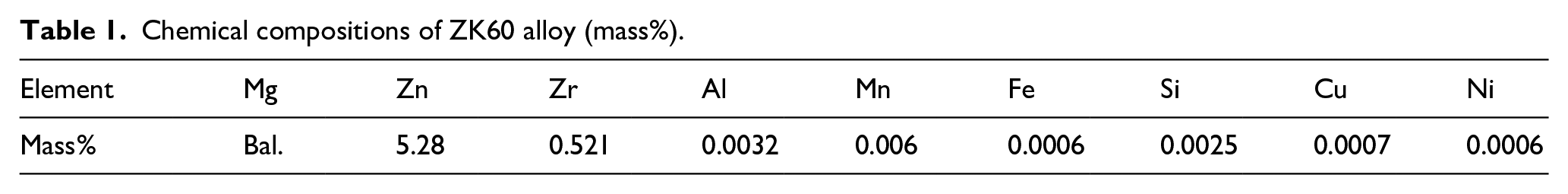
Figure 1 shows the XRD patterns of the alloy specimens after coating for 0.5, 1, 2, and 4 h, respectively. It could be seen that, the peak corresponding to HA was detected at approximately 26° in the XRD patterns of all specimens. This demonstrated that HA was formed on surface of all specimens. The highest intensity was detected by HA peak formed on the specimen coated for 2 h, while specimen coated for 0.5 h showed the lowest intensity. Peak corresponding to Mg(OH)2 was detected in the XRD pattern of the specimen coated for 4 h.
XRD patterns of the specimens after coating for 0.5, 1, 2, and 4 h.
SEM micrographs of the surface of the coated specimens in Figure 2 illustrates the growth of the HA coatings from 0.5 to 4 h. Up to 0.5 h, the HA grew in small agglomerates of rod-shaped crystals. The displacement of the agglomerates caused the nonuniformity of this coating. As the time increase, the radical HA rods grew up to fulfil the displacement, which increased the uniformity of the coatings after 1, 2, and 4 h, as shown in Figure 2(b) and (c). The coating after 2 h of treatment showed the highest uniformity. Additionally, cracks were observed only on the coating after 4 h.

SEM micrographs of the HA coated ZK60 specimens: (a) coated for 0.5 h, (b) coated for 1 h, (c) coated for 2 h, and (d) coated for 4 h.
Corrosion behavior
Figure 3 reveals surface morphology of the HA coated specimens after immersion test for 28 days. Specimens coated for 0.5 and 4 h were corroded severely. Corrosion products were formed and covered almost the entire surface of these specimens. On the other hand, specimens coated for 1 and 2 h showed much less corrosion. The surface of these specimens remained almost unchanged. There was only some small local corrosion appeared on the surface of these specimens.

Surface morphology of HA coated ZK60 specimens after immersion for 28 days in Hank’s solution: (a) coated for 0.5 h, (b) coated for 1 h, (c) coated for 2 h, and (d) coated for 4 h.
Figure 4 shows SEM micrographs and EDS analysis on the surface of the different coated specimens after 28 days of immersion. In general, the original surface of all the specimens was covered by corrosion products after the immersion tests. On the surface of the specimens coated after 0.5 and 4 h, high amounts of Mg and O were detected, while the amounts of P and Ca were very low, Figure 4(a) and (d). The ration of atomic percent of Mg and O suggested that corrosion products on these two specimens contained mainly Mg(OH)2 and trace of Ca-P compounds. On the other hand, Ca, P and O accounted for a high amount, whereas the amount of Mg was relatively low on the immersed specimens coated after 1 and 2 h, in Figure 4(b) and (c). It implies that on these two specimens, Mg(OH)2 corrosion product was sparely formed, but the formation of the Ca-P compounds was promoted. It is noted that the Ca-P compounds and/or inorganic salts of Mg/Ca are more stable and biocompatible than Mg(OH)2.15,17,21 Therefore, the specimens coated after 1 and 2 h are expected to have better biomedical performance during degradation process.

SEM micrographs and EDS analysis on the surface of the immersed specimens: (a) coated for 0.5 h, (b) coated for 1 h, (c) coated for 2 h, and (d) coated for 4 h.
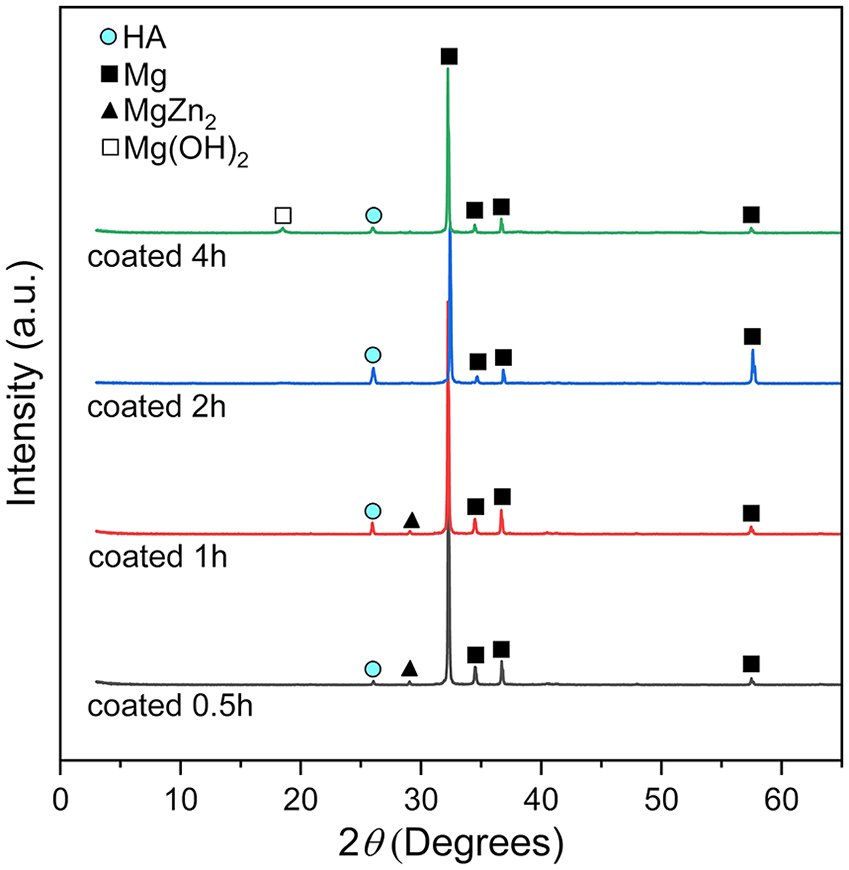
Figure 5 shows the accumulated amount of Mg2+ ions released into the immersion solution during immersion test because of the corrosion of the substrates. The highest amount of Mg2+ ions was measured in the solutions containing specimens coated for 0.5 and 4 h. This indicates that highest degradation rate occurred by these two specimens. Specimen coated for 2 h show the lowest amount of Mg2+ ion released into the solution, suggesting for its highest corrosion resistance. For all the specimens, corrosion occurred very fast at the first period of 7 days. Then, the corrosion was slower and got stable after 14 days.
Accumulated amount of Mg2+ ion released into solutions during immersion.
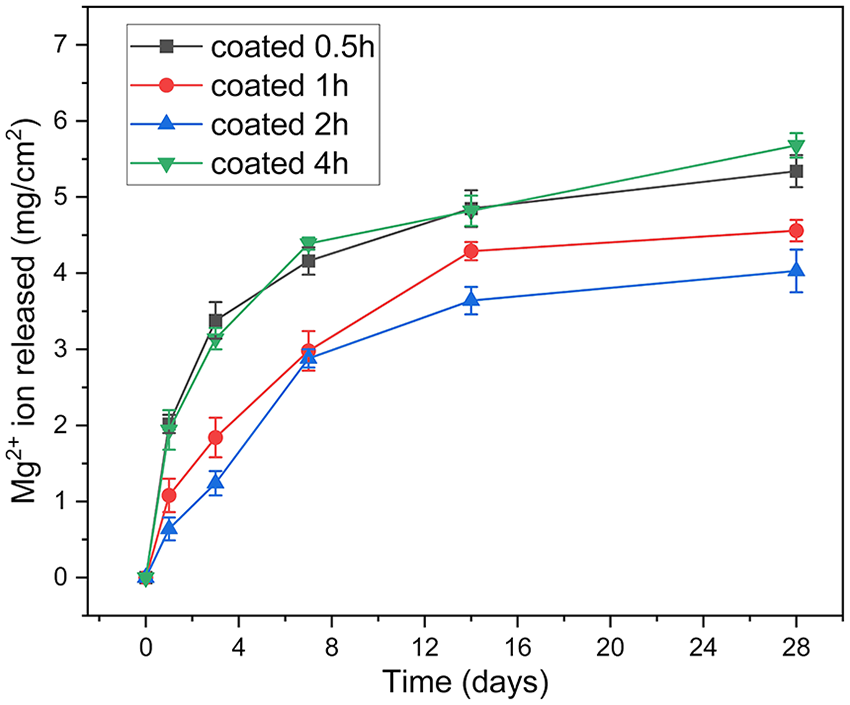
Figure 6 depicts the change of pH of the testing solutions during immersion period. In general, the tendency of pH change was similar to that of released Mg2+ ion amount. The solutions containing specimen coated for 4 h showed the highest pH at most of the testing periods. After 28 days of immersion, the pH value of this specimen reached about 10.5. The solution containing specimen coated for 0.5 h showed a slightly lower pH level, compared to specimen coated for 4 h. While the specimens coated for 1 and 2 h showed almost the same pH level during all testing periods. The pH values of these specimens reached to around 8.8 after 28 days. For all specimens, pH changes significantly during the first 14 days, after that pH stayed unchanged or slightly increased.
The change of pH of the solutions during immersion.
Discussion
Based on the XRD and SEM results of this study (Figures 1 and 2) and the growth mechanism of the HA coating formed in the chemical conversion coating method,17,22 the formation mechanism of the HA coating by time on the ZK60 alloy was illustrated as in Figure 7. The corrosion occurred immediately after the alloy was immersed into the coating treatment, which released abundant amount of Mg2+ and OH− associated with evolution of H2 gas, as in equations (1) and (2). The great release of OH− on the surface area of ZK60 initiated the formation of both HA nuclei and Mg(OH)2, in equations (3) and (4), respectively. The supplement of Ca2+ and PO43− was from the coating treatment. All the reactions were described in Figure 7(a). However, HA nuclei were accelerated more preferentially than Mg(OH)2 owning to the low solubility of HA. 22 The nucleation of HA then formed into the dense layer which has dome shape, as illustrated in Figure 7(b). Afterwards, this dense inner layer moderated the corrosion of the alloy, which conditioned the growth of the radically rod-shaped HA on the HA domes, as shown in Figure 7(c). The new formation of the rod-shaped HA was observed on the specimen coated after 0.5 h in Figure 2(a). As the coating time increased, both the dome-shaped layer and the rod-shaped layer grew. For the specimens coated after 1 and 2 h, the growth of HA increased the uniformity of the coating, in Figure 2(b) and (c). However, in addition to the growth of the HA layer, more Mg(OH)2 was formed to become an intermediate layer between the HA coating and the substrate, in Figure 7(d). That explains the detection of Mg(OH)2 in XRD patterns of the specimen coated for 4 h. In addition, the presence of a thick Mg(OH)2 layer was also a reason to cause cracks of the in the 4 h coating specimen as shown in Figure 2(d).


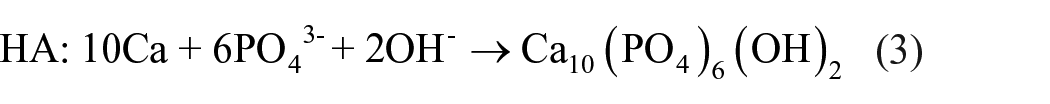

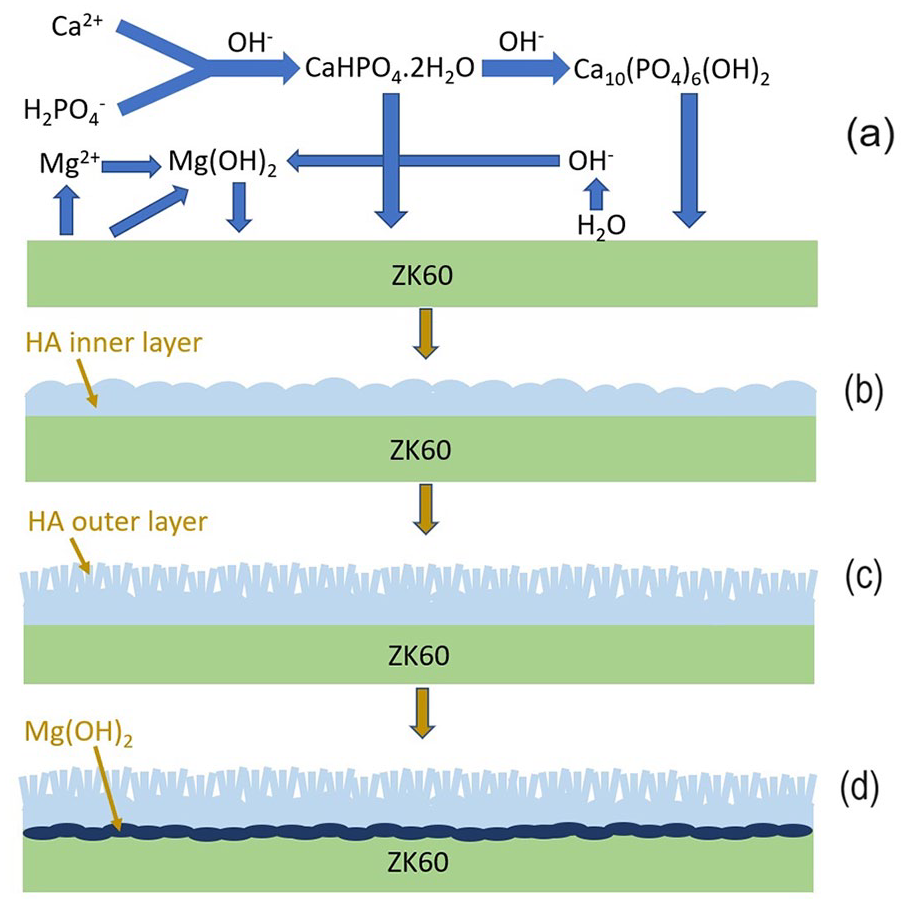
Schematic diagram of the formation mechanism of the HA coating layer by time.
As described above, H2 gas was evolved due to the corrosion reactions of the alloy during the formation of the coating. This evolution caused defects in the coating, by which corrosion could occurred as the immersion solution penetrated easier to attack the substrate. 21 The specimen coated for 0.5 h exhibited the least uniformity and thickness, which became a vulnerable coating. Therefore, it showed a very low corrosion resistance and resulted in the highest degradation rate. However, although owing higher uniformity and thickness, the specimen coated for 4 h showed a similar corrosion rate with that coated for 0.5 h, as in Figures 5 and 6. The low corrosion protection of this specimen is due to cracks and a thick layer of Mg(OH)2 existing in this coating (Figures 1 and 2(d)). After passing through the HA coating from the cracks, the immersion solution dissolved this intermediate Mg(OH)2 layer easily since Mg(OH)2 is sensitive in Hanks’ solution which contains a high concentration of Cl− ions. 23 Afterwards, the dissolution of Mg(OH)2 might cause detachment of the coating, leading to further corrosion of the substrate. This resulted in the generation of high amount of Mg2+ ions observed on the specimen coated for 4 h (Figure 5). Also, because of the effects of the treatment time, the specimen coated for 2 h exhibited higher uniformity and thickness than that coated for 1 h, implying a slightly better corrosion resistance (Figures 5 and 6).
Interestingly, after 28 days of immersion in Hanks’ solution, the degradation products were observed differently on the coated specimens. These degradation products could act as corrosion barriers to prevent further corrosion of the specimens to some extents. 24 It is observed that Mg(OH)2 was the main product adhered on the specimens coated for 0.5 and 4 h, whereas Ca-P compounds were mainly precipitated on the specimens coated for 1 and 2 h, Figure 4. Because Ca-P compounds are more chemically stable than Mg(OH)2, the specimens coated for 1 and 2 h would be well protected from corrosion than the other two specimens. This is one of the reasons explaining the higher corrosion protection of the specimens coated for 1 and 2 h. Ca-P compounds have been observed to serve as effective corrosion barriers for Mg alloys by forming protective layers on the alloy surface.25,26 These layers hinder the penetration of corrosive solutions, such as chloride ions or biological fluids, thereby retarding the initiation and propagation of corrosion. Moreover, the formation of a stable and adherent corrosion product layer, predominantly consisting of magnesium hydroxide further enhances the corrosion resistance of the alloy.27,28 Furthermore, previous studies proved that the HA coating was able to directly react with the simulated body fluid to form degradation products consisting of Ca-P compounds, which was similar to apatite in human bone.29–31 Ca-P compounds exhibit inherent biocompatibility since they are main component of bone tissue.32,33 Ca-P compounds have been shown to promote osteogenesis and osseointegration, essential processes for the successful integration of implants with host tissues.34–37 This enhancement of biocompatibility not only facilitates the acceptance of Mg-based implants by the host organism but also contributes to their functional integration and long-term stability. Therefore, not only having a positive effect on corrosion protection, Ca-P degradation product may enhance biocompatibility of the specimens.
In general, based on the results of this study, the specimen coated for 2 h is suggested to be a perspective material for orthopedic implants owing to its highest corrosion resistance and biocompatible degradation products.
Conclusion
In this study, HA coatings were formed successfully on the surface of ZK60 alloy substrate by the chemical conversion method after adjusting the coating time to 0.5, 1, 2, and 4 h. The effects of the coating time on the formation and corrosion of the coatings are concluded as follows:
- HA grew; as a result, the thickness and uniformity of the coating increased, as the treatment time prolonged. However, up to 4 h, cracks and Mg(OH)2 were observed in the coating, leading to a high corrosion rate.
- Due to the influence of thickness and uniformity, the 2 h coated specimen showed the lowest biodegradation rate, while the coatings for 0.5 and 4 h showed the similar lowest degradation rates when immersed into Hanks’ solution.
- Regarding to degradation products after immersion tests, biocompatible and stable Ca-P compounds were mainly detected on the specimens coated for 1 and 2 h. On the other hand, Mg(OH)2 was the main precipitates formed on the specimens coated for 0.5 and 4 h.
The results suggest that the HA 2 h coated ZK60 specimen is the most suitable for orthopedic application as a biodegradable material.
Footnotes
Contributorship
LVH and DNN designed and conducted the experiments. VND, PMK, LVT, and NVN analyzed and evaluated the data. LVH and NVN prepared the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
NVN.
