Abstract
A hydroxyapatite (HAp) coating on 2304 duplex stainless steel (DSS) through an electrophoretic deposition process has been investigated in this work. The deposition voltage was 30 V with a time of 2 min. Field emission scanning electron microscopy and energy dispersive X-ray spectroscopy analyses were used for the microstructural and chemical examination of coatings, respectively. The Ca to P ratio for the nano HAp coating on 2304 DSS has been determined as equal to 1.642. It was observed from X-ray diffraction patterns that HAp nanoparticles were successfully combined into the substrate. The corrosion behavior of all samples was tested in simulated body fluid using a potentiodynamic polarization study. A homogeneous structure with a thin crack-free layer was obtained. Moreover, the porosity of this coating was very low leading to a high corrosion resistance, thus promoting good biocompatibility.
Introduction
Duplex stainless steel (DSS) has a double phase structure of ferrite (α) and austenite (γ), and presents a superior combination of mechanical and corrosion properties.1–3 Generally, it contains equal parts of ferrite (body-centered cubic) and austenite (face-centered cubic) and is free from tertiary phases, such as carbides (Cr23C6), chromium nitrides (CrN, Cr2N), and intermetallics (σ, χ, R), which are mandatory to achieve the best mechanical properties and corrosion resistance of DSS. 4 The success of DSS has encouraged the manufacturers to form other categories of duplex including smaller or larger quantities of alloying elements. Presently, there are some categories of DSS that are grouped according to their values of pitting resistance equivalent number, such as standard duplex, lean duplex, hyper duplex, and super duplex.5,6 With regard to biomedical applications, studies7,8 have shown that the corrosion behavior of DSS is better than that of 316L stainless steel (SS) under potentiodynamic conditions. The implant’s life in the human body increases when the implant has a high corrosion resistance. However, investigation has also shown that in human bodies corrosion occurs slowly because of an electrochemical reaction once a metal has been implanted. 9 Over 90% of implants that are made from SS fail due to pitting and crevice corrosion. This limited corrosion behavior and metallic ion release from implants require enhancements in corrosion resistance by modifying the surface of SS. 10 Hydroxyapatite (HAp), Ca10(PO4)6(OH)2, is one of the materials that is grouped as bioactive, meaning it improves osseointegration and the growth of the bone. 11 HAp is known to be highly biocompatible, osteoconductive, bioactive, non-toxic, non-immunogenic, and non-inflammatory. 12 The HAp coatings prevent the corrosion in body fluid on surrounding tissues. 13 Several coating processes have been investigated using HAp coatings on the substrates of metals, such as sol–gel, 14 laser deposition,15,16 plasma spraying,17,18 electrophoretic process,19–23 electrolytic plasma oxidation,24,25 and so on. Among these coating methods, electrophoretic deposition (EPD) is particularly attractive for the deposition of HAp and other biomaterials on metallic surfaces. 26 EPD is a colloidal suspension method including two steps: electrophoresis and deposition. 27 EPD has various advantages, such as using economical equipment, short deposition time, room temperature processing, ability of coating on complex shapes, a fast processing rate, ability to control the thickness, and deposition of coatings with interconnected porosity for improving bone regrowth.28,29
DSS SAF 2304 has better mechanical properties, a high corrosion resistance, and contains a smaller amount of nickel compared with austenitic SS (316L), which gives it a medical advantage by avoiding the risk of nickel ions that cause allergies and infections in the human body when released. 30 In addition, the cost of 2304 DSS is lower when compared with many other SS alloys. Therefore, 2304 DSS is considered an alternative material for austenitic and ferritic SSs. Until now, investigations about the influence of HAp deposition on the corrosion behavior of 2304 DSS have not been carried out. Previously, several researchers31–33 studied HAp deposition on 316L SS by EPD and have shown it improves corrosion resistance. In this research, the EPD of HAp deposition on 2304 DSS was investigated along with its microstructure; surface morphology and corrosion behavior in simulated body fluid (SBF) were also investigated.
Materials and methods
Materials used
HAp nanoparticles (99% purity, CAS No. 1306-06-5, Hualanchem, China) with 20 nm particle size were utilized in the current work. The reason behind choosing this particle size specifically is that it possesses nanostructured materials with an extremely fine grain size and exhibits enhanced properties when compared with their coarse-grained counterparts. The smaller particle size usually results in a higher surface area and the amount of porosity is lower which may improve its mechanical properties and fracture toughness. This may be important to optimize the adsorption of specific proteins, which can improve osteoblast cell adhesion, enhance osteoblast proliferation and differentiation, and optimize biological functionality. On the other hand, the coarse particle size causes a rougher agglomeration and a more heterogeneous appearance with discontinuous grain growth than the fine one.34,35 Ethanol absolute (99.9% purity, Scharlau, Spain) was used for the suspension preparation. Iodine (99.9% purity, AnalaR, England) was also used as a dispersant and to enhance the surface charge (this resulted in positively charged particles). 36 The 2304 DSS plate (OutoKumpu, Sweden) was utilized as a material of substrate and was used as the counter electrode in the EPD cell.
Preparation of substrate
A wire cutting machine was used to cut the 2304 DSS plate into dimensions of 25 × 15 × 2 mm. The chemical composition of the 2304 DSS substrate is shown in Table 1. In order to improve the bonding and adhesion between the deposition and the substrate, prior to EPD, the substrates were ground using SiC emery papers of 80, 120, 320, 600, and 1200 µm grits. Sandblasting was done on the 2304 DSS surface by static pressure of 5 kPa with 60 µm alumina particles for 25 s. The distance from the nozzle orifice to the surface of substrate was 30 cm while the angle between the abrasive jet direction and the substrate surface was 90°. Next, the sandblasted 2304 DSS specimens were washed with de-ionized water, then cleaned ultrasonically in ethanol for 10 min. The cleaned specimens were dried for 15 min at room temperature and stored in vacuum desiccators to prevent oxidation.
Composition of DSS SAF 2304.

Preparation of suspension
The suspension component of HAp nanoparticles was prepared from 0.5 g/L of chitosan that had been dissolved in acetic acid (1 vol.%) according to research. 32 Next, ethanol (94 vol.%) and water (5 vol.%) were added to the suspension. The prepared suspension was stirred for 15 min using a magnetic stirrer at room temperature, then 10 g/L total concentration of nano HAp powder with a particle size of 20 nm was added to the suspension. The mixture of ethanol–water gives better colloidal stability for inorganic particles. 37 The prepared suspensions were stirred for 6 h. Afterwards, the suspension was dispersed ultrasonically for 30 min by using an ultrasonic homogenizer for dispersing (MTI Corporation) in order to break weak agglomerates and to produce homogeneous dispersion for the particles in the suspension. Next, the mixture was magnetically stirred for 10 min to obtain a homogeneous suspension before deposition.
EPD
The EPD system consisted of a 150-ml glass beaker and was equipped with a direct current (DC) power supply where the voltage can be changed as required to achieve the best deposition. Both 2304 DSS plates were placed in the EPD system as the cathode and anode with a 1 cm separation distance between them. Both 2304 DSS electrodes were submerged in the suspension inside a 200-ml glass beaker and placed parallel to each other within a separating distance of 10 mm. In order to obtain the best electrophoretic parameters, deposition was performed at 30 V using a regulated DC power supply for a deposition time of 2 min at room temperature. The surface area (1.5 cm2) of the substrate was coated by an EPD technique from a supersaturated solution of nano HAp particles, where the other side and edges were disguised with adhesive non-conducting tape. After coating was complete, the samples were dried for 24 h at 25 °C. Eventually, the coated samples were weighed using a calibrated microbalance (ABS 220-4, KERN) with an accuracy of ± 0.0001%.
Characterizations
Light microscopy
The microstructure of 2304 DSS before deposition was carried out using light microscopy according to ASTM E3-01. The preparation method includes grinding with SiC papers (120, 320, 600, 800, 1000, 1500, 2000 µm grit) with a speed of 200 rpm, then polishing with a diamond paste of 1 µm to achieve a smooth mirror finish. The solution for etching was prepared according to ASTM A923-01 by adding 40 g of NaOH to 100 ml of distilled water and applying a voltage of 2 V for 10 s with a Pt cathode.
Field emission scanning electron microscopy with energy dispersive X-ray spectroscopy
The surface morphology and cross-section of the depositions on the 2304 DSS substrate were studied using the field emission scanning electron microscope (FESEM) SUPRA 55VP (Zeiss). X-ray spectroscopy (EDS) attached to the FESEM was used to investigate the elemental composition.
X-ray diffraction
X-ray diffraction (XRD) was employed to define the presence of phases in the 2304 DSS, HAp powder, and HAp deposited on 2304 DSS by using LabX 6000 (Shimadzu, Japan). CuKα radiation (λ = 0.154056 nm) with nickel-filtered material was used and the scanning speed was 6 deg/min; the resultant peaks were compared with the standards peaks for each material by using the Joint Committee on Powder Diffraction Standards (JCPDS) cards.
Electrochemical corrosion tests
The electrochemical estimation of the corrosion behavior for the 2304 DSS before and after deposition was carried out in SBF, with composition of 0.0860 g/L NaCl, 0.0033 g/L CaCl2.2H20, and 0.0030 g/L KCl, at 37 ± 1 °C, to imitate the conditions of the human body. The electrochemical cell used for this purpose was fitted with a Pt electrode as an auxiliary electrode, a saturated calomel reference electrode, and 2304 DSS as a working electrode. The electrochemical corrosion tests were realized in a potentiostat/galvanostat (Wenking LB 81M, Germany) according to ASTM G5-94.
Results and discussion
Metallographic analysis
Figure 1 illustrates the microstructure of the 2304 DSS where the ferrite phase appears darker than the austenite phase. Using the ImageJ 1.46r program to analyze the microstructure, a fraction area of approximately 50.2% ferrite and 49.8% austenite was shown. A scanning electron microscopy investigation was performed to analyze the microstructure and morphology of the deposition surface. Figure 2(a) shows the morphological structures of coatings at a deposition time of 2 min at EPD potential of 30 V. It was noticed that the sample with a 2-min deposition time had a highly homogeneous and less porous structure than other coated samples. Figure 2(b) shows the high magnification of the HAp deposition for a 2-min deposition time, indicating a smooth, dense (with more closely packed microstructure) surface, with good homogeneity and less porous structure, where a crack-free and uniform coating was achieved. This less porous structure has a significant role in the action of the bioactive HAp depositions due to its improvement in biocompatibility and ability for bone growth. However, the high porosity of the implants has a negative effect on the mechanical properties, particularly the shear and bonding strength, leading to easier fractures.17,22,38 Figure 2(c) illustrates the cross-sectional morphology of HAp deposition at 30 V. It can be seen that the coated layer has a regular thin film structure of 15.3 μm in thickness with high compaction, homogeneity, and complete adhesion between the coating and the underlying substrate.

Microstructure of 2304 duplex stainless steel.

Scanning electron microscopy micrographs of hydroxyapatite coatings on 2304 duplex stainless steel at electrophoretic deposition time of 2 min: (a) low magnification, (b) high magnification, (c) transverse section.
Based on these results, the EPD parameters at 30 V for 2 min determined the best HAp deposition in this study.
Elemental composition of the coatings
In order to recognize the composition of the coatings at the surface of the 2304 DSS, EDS analysis characterized the chemical elemental composition of the 2304 DSS surface of uncoated samples (Figure 3). It was observed that Fe, Cr, Ni, and Mo main elements with a high degree of homogeneity for chemical constituent distribution were present. Figure 4 explains the analysis for chemical elemental composition of DSS 2304 after EPD. The presence of main elements Ca and P in the depositions on 2304 DSS were also observed. This proves that the elemental components of suspension in the EPD method were incorporated into the deposition. The existence of these strong peaks affirms the formation of successful nano HAp deposition on the 2304 DSS substrate.

X-ray spectroscopy analysis for chemical elemental composition of 2304 duplex stainless steel.

X-ray spectroscopy analysis for chemical elemental composition of 2304 duplex stainless steel after EPD.
In surgical applications, it is highly important that the elemental composition for the bioactive layer on the surface of the metallic implant is compatible with the composition of bone source. The Ca/P ratio is a very important feature for determining the characteristics of the calcium phosphate coatings that can be utilized in surgical applications. One may notice that there is a variation between the ratio of the theoretical HAp (1.67) and different bone types. 39 Table 2 shows the atomic percentage of Ca and P and the Ca/P ratio of the coatings. This EPD time is a very important parameter in determining the amount of Ca and P in bioactive layers on the surface of the 2304 DSS. The Ca/P ratio was determined to be 1.642, proving that HAp is deposited on the 2304 DSS substrate in the condition of 30 V for 2 min. The value of the Ca/P ratio near to those found in the human skeleton 40 indicates that these elements were distributed homogeneously throughout the deposition and this would also give the impression that these depositions are the best candidate for orthopedic applications.
Ca/P ratio of hydroxyapatite coating on 2304 duplex stainless steel.

Phases identification
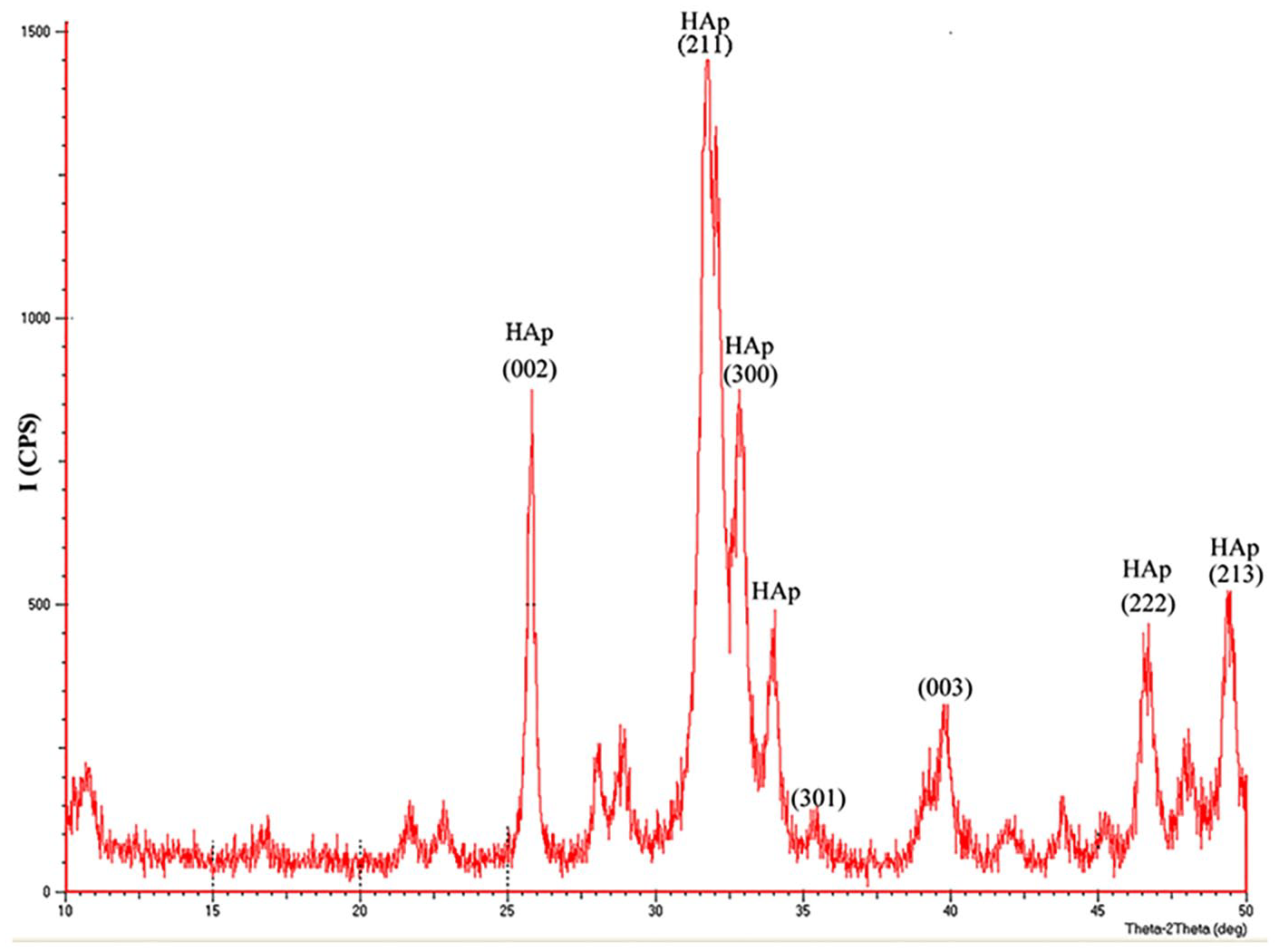
Figure 5 shows the phase identification for the substrate of the 2304 DSS before coating. The diffraction pattern of the ferrite phase (α) matches with the JCPDS Card No. 06-0694 peaks and the austenite phase (γ) matches with the JCPDS Card No. 33-0397 peaks and are conformable to the ferrite (α) and austenite (γ) phases in Figure 1. The results of XRD are in agreement with Fargas 41 and Tschiptschin. 42 The phase identification of HAp powder shown in Figure 6 were identical with the JCPDS File No. 9-432 peaks. The sharp and high peaks of HAp indicated high crystallite of HAp and no calcium oxide presence in the XRD pattern of HAp can be seen. Figure 7 shows the XRD pattern for the HAp deposited on the 2304 DSS sample at 30 V for 2 min deposition time. The nanosized materials used provide a higher surface area for the particles, which leads to a higher ion exchange with the surrounding medium and finally results in an accelerated HAp formation. It can be observed from this figure that the nanoparticles of HAp were successfully combined together into the deposition layer on the 2304 DSS. When comparing the uncoated 2304 DSS substrate with the coated one, according to Fargas 41 and Tschiptschin, 42 it can be seen that the strongest peak lines in the 2304 DSS phases become smaller after coating due to the HAp-deposited layer covering the 2304 DSS surface and consequently reducing the 2304 DSS peaks. The other peaks contain the DSS phases; the diffraction pattern of the ferrite phase (α) matches with the JCPDS peaks and the austenite phase (γ) matches with the JCPDS peaks in addition to the phases of HAp. The existence of these peaks is due to the entrance of X-ray into the substrate. No other additional phases were observed in the XRD analysis and this means a higher degree of crystallinity (no change in the crystal structure) and no decomposition of phases, by using the best applied deposition parameters used in the EPD process.
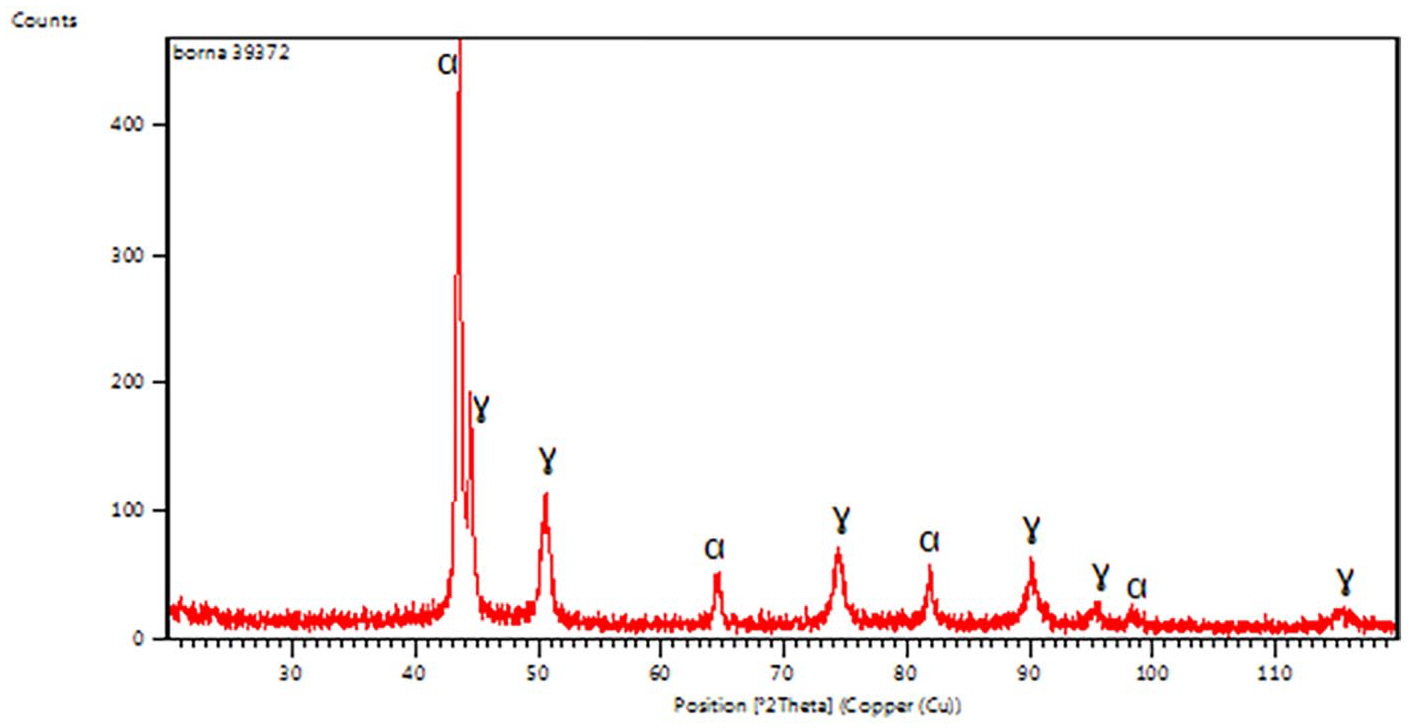
X-ray diffraction pattern of 2304 duplex stainless steel.
X-ray diffraction pattern of nano hydroxyapatite powder.

X-ray diffraction pattern of 2304 duplex stainless steel after hydroxyapatite electrophoretic deposition.
Corrosion resistance
The corrosion behavior for uncoated and coated 2304 DSS using a 2-min deposition time at 30-V applied voltage in the EPD process was obtained by a potentiodynamic test, as explained in Figure 8. The samples were submerged in SBF for 60 min and the open circuit potential was recorded after this period where a steady state potential was attained. It is well known that corrosion current density is a helpful parameter to represent the corrosion rate of metallic implants at the same conditions. The SBF environment containing chloride ions is capable of penetrating the deposition, then attacking the surface of the 2304 DSS. Corrosion current densities and corrosion potentials were derived from the potentiodynamic curves by employing Tafel extrapolation and are shown in Table 3.

Potentiodynamic polarization curves of 2304 duplex stainless steel and the 2304 duplex stainless steel deposited at 30 V for 2 min by electrophoretic deposition in simulated body fluid solution at 37 °C.
Electrochemical parameters of uncoated and hydroxyapatite-coated 2304 duplex stainless steel at 30 V for 2 min.

βa: anodic tafel slopes; βc: cathodic tafel slopes; CR: corrosion rate; DSS: duplex stainless steel; Ecorr: corrosion potential; Icorr: corrosion current density.
The uncoated substrate has a higher corrosion current density (1.090 µA/cm2) and thus the lowest corrosion resistance, due to metal ion dissolution which may occur on the surface of the 2304 DSS.
The coated sample at 30 V for 2 min has a lower corrosion current density (0.605 μA/cm2), which is attributed to the fine, crack-free and dense structure of the obtained coating as well as the solid adhesion between the coating and the 2304 DSS substrate, effectively able to inhibit the reaching of corrosive fluid to the substrate. The HAp-coated 2304 DSS has a higher corrosion resistance than the coated 316L austenitic SS when submerged in Ringer’s physiological solution, which has a lower corrosion current density than coated 316L SS as concluded by Farrokhi-Rad. 43 The results propose that the HAp coating successfully provides a protective film on the 2304 DSS surface and prevents corrosion. These results show that HAp-coated 2304 DSS has a better biocompatibility than HAp-coated 316L SS and, as such, can allow for a higher implant life. In addition, this excellent corrosion resistance makes 2304 DSS a highly suitable solution for medical purposes when it is used as a substrate coated with an HAp layer.
Conclusions
From the current study, the following can be concluded:
1 The morphology of coating was homogeneous and crack-free when coated with the best condition at 30 V for 2 min.
2 The Ca/P ratio of the coating on 2304 DSS was detected to be 1.642, a value similar to that found in the human skeleton, demonstrating that these coatings would be a robust contender for orthopedic applications.
3 The HAp coating on the 2304 DSS at 30 V for 2 min has the lowest corrosion rate value (0.277 mpy), i.e. high corrosion resistance in SBF, and thus it is more corrosion resistant than other uncoated samples.
Research Data
Proofreading_Certificate – Biomineralization of 2304 duplex stainless steel with surface modification by electrophoretic deposition
Proofreading_Certificate for Biomineralization of 2304 duplex stainless steel with surface modification by electrophoretic deposition by Ali Sabea Hammood in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
The author thanks Assistant Professor Morteza Farrokhi-Rad, Department of Materials Engineering, Faculty of Engineering, Azarbaijan Shahid Madani University, Iran for amendments to this paper.
Declaration of conflicting interests
The author has no conflict of interest to declare.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
