Abstract
Objective:
This study aimed to compare the degradation behavior and protection mechanisms of biodegradable WE43 alloys coated with two different calcium phosphate (Ca–P) phases—octacalcium phosphate (OCP) and hydroxyapatite (HA)—to identify an effective surface modification strategy for biodegradable material applications.
Methods:
OCP and HA coatings were deposited on WE43 substrates and characterized using X-ray diffraction (XRD) and scanning electron microscopy (SEM). The degradation behavior was evaluated through immersion tests in Hanks’ balanced salt solution (HBSS) by monitoring surface morphology, magnesium ion release, and pH variation over time.
Results:
XRD confirmed the successful formation of OCP and HA phases, while SEM revealed distinct morphologies: porous, plate-like structures for OCP and dense, compact layers for HA. Immersion testing demonstrated that both coatings reduced magnesium ion release and stabilized the solution pH compared with uncoated WE43. The HA coating exhibited superior corrosion resistance, maintaining the lowest Mg2⁺ release and most stable pH, attributed to its high crystallinity, compact structure, and self-healing apatite reprecipitation during immersion.
Conclusion:
Both Ca–P coatings improved the corrosion resistance of WE43 alloy; however, the HA layer provided markedly enhanced and more stable protection. The HA-coated WE43 alloy shows great potential for biodegradable implant applications requiring controlled degradation and sustained biocompatibility.
Introduction
Magnesium (Mg) and its alloys have emerged as one of the most promising classes of materials for biodegradable metallic implants owing to their excellent biocompatibility, mechanical properties close to those of natural bone, and complete biodegradability within the physiological environment.1–3 Unlike traditional permanent metallic implants such as titanium or stainless steel, magnesium-based materials can gradually dissolve after fulfilling their structural function, eliminating the need for secondary surgery to remove the implant.4–6 This unique advantage has stimulated extensive research into Mg-based biomaterials for orthopedic and cardiovascular applications.6–8
Among the various magnesium alloys developed, WE43—comprising magnesium with additions of yttrium (Y) and neodymium (Nd) as rare earth (RE) elements—has gained particular attention for its superior mechanical strength, thermal stability, and corrosion resistance compared to other commercial Mg alloys.9–11 The presence of RE elements refines the microstructure and enhances the formation of thermally stable intermetallic compounds, improving overall performance.12,13 However, despite these advantages, WE43 still suffers from excessively rapid corrosion in physiological environments. The aggressive attack of chloride ions in body fluids leads to localized corrosion and hydrogen gas evolution, which can cause tissue inflammation and compromise the implant’s structural integrity before the surrounding tissue fully heals.14–17 Therefore, controlling the degradation rate of WE43 is a critical challenge that must be addressed for successful biomedical application.
Surface modification techniques have been widely explored as an effective means to improve the corrosion resistance and biocompatibility of Mg alloys. Methods such as micro-arc oxidation (MAO),18,19 sol–gel coatings, polymeric films, 20 and calcium phosphate (CaP) coatings21–24 have all been investigated. Among these, CaP-based coatings have attracted particular interest because they not only act as a corrosion barrier but also exhibit excellent osteoconductivity and bioactivity.21–24 Moreover, calcium phosphates are chemically similar to the inorganic component of bone and teeth, which enhances interfacial bonding and facilitates osseointegration during the healing process.25,26
Hydroxyapatite (HA, Ca10(PO4)6(OH)2) and octacalcium phosphate (OCP, Ca8H2(PO4)6·5H2O) are two major crystalline phases of CaP widely studied for biomedical coatings. HA is the most thermodynamically stable phase under physiological conditions, characterized by low solubility and strong bioactivity, making it ideal for long-term stability at the implant interface. 27 OCP, on the other hand, is metastable and exhibits higher solubility and faster resorption in vivo, which can be advantageous for promoting early bone remodeling and gradual integration with host tissue. 28 Moreover, OCP can serve as a precursor to HA, transforming under physiological conditions into a more stable phase. This dynamic behavior suggests that a well-controlled OCP coating could provide both initial bioactivity and long-term protection.
Previous research has demonstrated that the type, crystallinity, and microstructure of CaP coatings significantly influence their corrosion protection ability and biological response.29–31 Dense and uniform coatings generally offer better corrosion resistance, while porosity and phase composition affect ion exchange and bioactivity. Nevertheless, comparative studies that directly evaluate the degradation behavior of OCP- and HA-coated WE43 alloys under identical conditions are limited. Understanding the relationship between coating composition, structure, and corrosion performance is essential for optimizing surface treatment strategies for biodegradable Mg implants.
In this study, WE43 magnesium alloy was coated with hydroxyapatite (HA) and octacalcium phosphate (OCP) using a chemical conversion method, and their degradation behavior was systematically evaluated under identical conditions. While previous research has investigated surface coatings for Mg alloys, few studies have directly compared the corrosion behavior of HA- and OCP-coated WE43 alloys. Using X-ray diffraction (XRD) and scanning electron microscopy (SEM) for structural characterization, along with immersion tests and Potentiodynamic Polarization test in HBSS to assess corrosion resistance, this work elucidates how coating phase composition affects degradation mechanisms. By providing a direct comparison between uncoated, OCP-coated, and HA-coated WE43, this study offers novel insights into optimizing surface treatments for biodegradable Mg implants, highlighting both corrosion control and potential bioactivity enhancement.
Materials and methods
Sample preparation
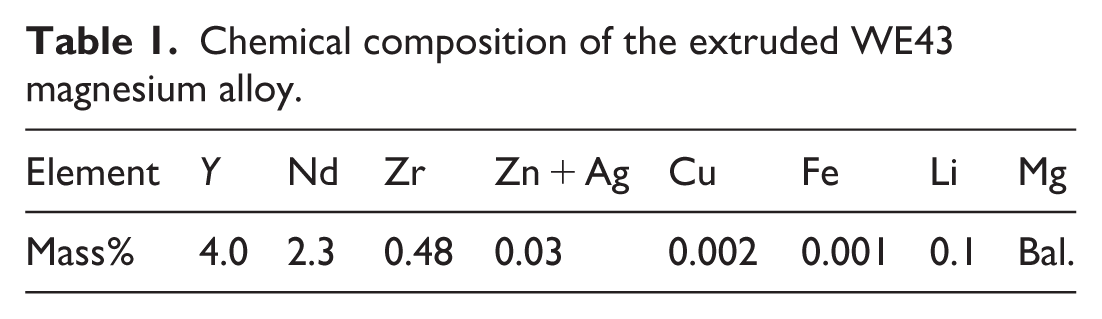
The substrate used in this study was an extruded WE43 magnesium alloy, whose nominal chemical composition is summarized in Table 1. The alloy rod (17 mm in diameter) was sectioned into disk-shaped specimens with a thickness of 2 mm. Each specimen was ground sequentially using silicon carbide (SiC) abrasive papers up to #2000 grit to achieve a uniform surface finish, followed by ultrasonic cleaning in ethanol and air drying before the coating process.
Chemical composition of the extruded WE43 magnesium alloy.
Coating formation
Ethylenediaminetetraacetic acid calcium disodium salt hydrate (Ca-EDTA, C10H12N2O8Na2Ca), potassium dihydrogen phosphate (KH2PO4), and sodium hydroxide (NaOH) were employed for the preparation of the coating solutions. Equal volumes of 0.5 mol·l⁻1 Ca-EDTA and 0.5 mol·l⁻1 KH2PO4 solutions were mixed thoroughly under continuous stirring. Subsequently, NaOH solution was gradually added to adjust the pH of the mixed solution to 5.5 and 7.5, corresponding to the preparation conditions for the OCP and HA coatings, respectively. The resulting coating solutions were then heated to 90°C, and the polished WE43 disk specimens were immersed in the respective treatment solutions at 90°C for 2 h to allow coating formation.
Immersion test
Immersion tests were conducted in accordance with ASTM G31-03 standard guidelines
32
to evaluate the
Salt concentration of the HBSS solution.

Hanks’ Balanced Salt Solution (HBSS) was prepared in-house following the standard Sigma-Aldrich H1387 formulation. Briefly, 9.7 g of Hanks’ balanced salts (H1387, Sigma-Aldrich) were dissolved in 1.4 l of nanopore water. The final solution contained (in mM): NaCl (137.9), KCl (5.33), CaCl2 (1.26), MgSO4 (0.81), MgCl2 (0.49), Na2HPO4 (0.34), KH2PO4 (0.44), D-glucose (5.55), and NaHCO3 (4.17).
To buffer the solution, 14.16 g of HEPES acid (H3375-5000G, Sigma-Aldrich, USA) and 16.65 g of HEPES sodium salt (H7006-500G, Sigma-Aldrich, USA) were added, along with 3.3 g of sodium bicarbonate (S8875-500G, Sigma-Aldrich). The pH was adjusted to 7.4 at 37°C using 1 M NaOH. The solution was stored at 4°C prior to use.
The specimens were placed in a humidified incubator maintained at 37°C and 5% CO2 throughout the immersion period. The ratio of the testing solution volume to the total exposed surface area of the specimen was controlled at 30 ml·cm⁻2 to ensure consistent degradation conditions.
Immersion durations of 1, 3, 7, 14, and 28 days were selected for evaluation. At each scheduled interval, a small aliquot (100 μl) of the immersion medium was sampled to determine the concentration of released Mg2⁺ ions. The quantification was performed using a colorimetric magnesium assay kit (Magnesium B-Test Wako, FUJIFILM Wako, Tokyo, Japan), which employs xylidyl blue-I as a chromogenic reagent.33,34 The absorbance of the Mg–xylidyl blue-I complex was measured at 520 nm using a microplate spectrophotometer (Multiskan GO, Thermo Scientific).
In addition, the pH of the immersion solution was recorded at each testing interval to monitor changes in the chemical environment associated with magnesium dissolution and coating degradation. After each immersion period, the specimens were retrieved from the testing medium, gently rinsed with deionized water, and dried for subsequent surface characterization.
Potentiodynamic polarization test
Polarization tests were conducted in HBSS. The sample surface was masked with epoxy resin, leaving an exposed area of 1 cm2 in contact with the solution at 37°C. The open-circuit potential (OCP) was monitored for 1800 s, after which potentiodynamic polarization measurements were performed from −0.5 V versus OCP at a scan rate of 1 mV·s⁻1. A saturated Ag/AgCl electrode and a platinum electrode served as the reference and counter electrodes, respectively. The test was conducted in triplicate for each type of sample.
Characterization
The phase composition and surface morphology of the coated specimens were characterized using X-ray diffraction (XRD) and field-emission scanning electron microscopy (FE-SEM). XRD analysis was performed on a Bruker D8 Advance diffractometer equipped with Cu Kα radiation (λ = 1.5406 Å), operated at 40 kV and 40 mA. The diffraction patterns were collected over an appropriate 2θ range to identify crystalline phases present on the coating surface. Surface and cross-sectional morphologies, as well as microstructural features of the coatings, were examined using a field-emission scanning electron microscope (JEOL JSM-7600F).
Result
X-ray diffraction (XRD) analysis
Figure 1 presents the X-ray diffraction (XRD) patterns of the uncoated WE43 alloy, OCP-coated WE43, and HA-coated WE43 samples. The diffraction peaks of the uncoated WE43 alloy are located at approximately 2θ = 32.2°, 34.4°, 36.6°, 47.8°, 57.4°, and 63.0°, corresponding to the (100), (002), (101), (102), (110), and (103) planes of the hexagonal close-packed magnesium phase (PDF#35-0821). These peaks confirm the crystalline structure of the alloy and the absence of secondary phases such as MgO.

XRD patterns of three different samples: uncoated WE43, OCP coated WE43, and HA coated WE43.
Following the OCP coating process, several new peaks appear at around 2θ = 4.9°, 16.7°, 25.9°, and 53.5°, which can be indexed to the (100), (210), and (300) planes of octacalcium phosphate (OCP, PDF#26-1056). The presence of both Mg and OCP peaks indicates that the coating layer is relatively thin, allowing partial X-ray penetration to the substrate. The OCP-related peaks are relatively broad and of low intensity, suggesting limited crystallinity and fine-grained characteristics typical of OCP coatings formed under moderate thermal conditions.
In contrast, the HA-coated WE43 sample exhibits several additional diffraction peaks located at 2θ = 25.9°, 28.0°, 49.3°, and 53.1°, corresponding to the (002), (211), (112), and (310) planes of hydroxyapatite (HA, PDF#09-0432). The HA reflections are sharper and more intense than those of the OCP-coated sample, indicating improved crystallinity and a well-developed apatite phase. No secondary phases such as tricalcium phosphate (TCP) or calcium oxide (CaO) are detected, demonstrating that the coating process yielded a phase-pure HA layer without decomposition or contamination.
Overall, the XRD analysis confirms the successful deposition of crystalline OCP and HA coatings on the WE43 alloy. The phase purity and increased crystallinity of the HA coating suggest enhanced stability and coating integrity with the substrate. These structural characteristics are expected to contribute to improved bioactivity and corrosion resistance of the coated WE43 alloy in physiological environments.
Surface morphology analysis (SEM)
Figure 2 shows the SEM micrographs and cross-sectional SEM of the uncoated WE43 alloy, OCP-coated WE43, and HA-coated WE43 samples at different magnifications. The uncoated WE43 surface appears relatively smooth, with visible polishing marks and a few fine surface defects, indicating the typical morphology of a mechanically polished magnesium alloy. No significant surface features or deposited phases are observed, confirming the absence of surface modification.

Surface and cross-sectional SEM images of: (a) uncoated sample; (b, d and f) OCP coated sample, (c, e and g) HA coated sample.
After OCP deposition, the surface morphology changes markedly. At low magnification, the OCP-coated surface appears uniformly covered with a continuous layer. Higher-magnification images reveal that the coating is composed of plate-like and flake-shaped crystals closely packed together, forming an interlaced structure characteristic of octacalcium phosphate (OCP). This morphology suggests a uniform nucleation and growth of OCP crystals across the substrate surface, producing a dense and adherent coating layer.
In contrast, the HA-coated WE43 sample exhibits a more compact and crystalline surface morphology. At low magnification, the surface appears homogeneous without visible cracks or delamination. The high-magnification micrograph shows that the coating consists of densely packed, rod-like or needle-shaped HA crystals uniformly distributed over the surface. These crystals are finer and more regularly arranged than those of the OCP coating, indicating that the HA layer possesses higher crystallinity and surface integrity.
Cross-sectional SEM observations further confirm the successful formation of the OCP and HA coatings on the WE43 substrate. As shown in Figure 2(f), the OCP-coated sample exhibits a continuous coating layer with a thickness of ~4–5 μm, tightly adhered to the substrate. The OCP layer appears relatively uniform, although slight variations in thickness and a somewhat porous internal structure can be observed, which are consistent with the plate-like crystal morphology seen on the surface. In comparison, the HA-coated sample (Figure 2(g)) presents a more compact and denser coating layer with a relatively uniform thickness of ~5 μm. The HA coating shows a clearer and more well-defined interface with the substrate, indicating improved coating integrity and cohesion. No obvious interfacial cracks or delamination are observed for either coating, suggesting good adhesion between the coatings and the WE43 alloy substrate.
Overall, the SEM observations confirm the successful formation of continuous and adherent calcium phosphate coatings on WE43 alloy. The distinct plate-like morphology of the OCP layer and the compact rod-like structure of the HA layer are consistent with their respective crystal structures, as supported by the XRD analysis. The dense and homogeneous HA coating is expected to provide improved corrosion resistance and enhanced bioactivity compared with the uncoated and OCP-coated samples.
Surface morphology after immersion test
Figure 3 shows the macroscopic, SEM images and cross-section SEM of the uncoated, OCP-coated, and HA-coated WE43 samples after immersion in HBSS solution for 28 days. The uncoated WE43 surface exhibits extensive corrosion and degradation after immersion, with dark regions and heavy deposits observed across the entire surface. The high-magnification SEM image reveals a rough and cracked morphology with large corrosion pits and flakes of corrosion products, indicating severe localized attack and loss of surface integrity. These features are characteristic of the rapid degradation behavior of magnesium alloys in chloride-containing environments.

Macro surfaces, SEM images, and cross-section SEM images of samples after immersion test of 28 days: (a and d) uncoated sample, (b, e and g) OCP coated sample, (c, f and h) HA coated sample.
In contrast, the OCP-coated WE43 sample shows noticeably reduced surface degradation. The surface remains relatively intact, though partially covered with corrosion products and minor deposits. The SEM micrograph displays a compact layer of corrosion products formed on top of the OCP coating, suggesting that the coating provided partial protection by delaying direct electrolyte penetration. Some microcracks and pores are visible, which may serve as localized sites for corrosion initiation during prolonged exposure.
The HA-coated WE43 sample demonstrates the most stable surface condition among the three. The macroscopic image shows minimal discoloration or deposit accumulation compared to the uncoated and OCP-coated samples. The SEM image reveals that the HA-coated surface remains largely covered by a dense layer of fine, globular apatite-like particles, indicative of secondary apatite formation during immersion. This suggests that the HA layer not only provides a physical barrier against corrosion but also promotes bioactive surface reactions leading to the deposition of bone-like apatite.
Cross-sectional SEM images further confirm the stability of the OCP and HA coatings after 28 days of immersion in HBSS. As shown in Figure 3(g), the OCP-coated WE43 sample still retains a distinguishable coating layer on the substrate surface. The OCP layer exhibits a non-uniform thickness with localized cracks and porous regions, indicating partial structural degradation while remaining largely adhered to the substrate. The coating–substrate interface is still visible, suggesting that the OCP layer was not completely dissolved during immersion, although its integrity was locally compromised.
In comparison, the HA-coated sample (Figure 3(h)) shows a more continuous and compact remaining coating layer with relatively uniform thickness. The HA coating maintains a well-defined and intact interface with the WE43 substrate, with fewer microcracks or discontinuities observed. This indicates that the HA coating possesses higher chemical stability and structural integrity during prolonged immersion, effectively preserving its barrier function and contributing to the improved corrosion resistance of the HA-coated WE43 alloy.
Overall, the post-immersion analysis confirms that both OCP and HA coatings effectively reduce the corrosion of WE43 in HBSS, with the HA coating offering superior protection and enhanced surface bioactivity. The compact and adherent morphology of the HA coating likely contributes to its improved performance compared with the more porous OCP layer.
Mg2⁺ ion release behavior
Figure 4 shows the cumulative Mg2⁺ ion release from uncoated, OCP-coated, and HA-coated WE43 samples during immersion in HBSS for up to 28 days. The uncoated WE43 sample exhibited the highest Mg2⁺ ion release throughout the entire period, increasing rapidly during the initial stage and continuing to rise steadily, reaching approximately 6.5 mg/cm2 after 28 days. This behavior indicates continuous and accelerated corrosion of the bare alloy in the physiological environment.

Accumulated amount of Mg2+ ion released into solutions during immersion.
In contrast, both coated samples released significantly lower amounts of Mg2⁺ ions, confirming the protective effect of the calcium phosphate coatings. The OCP-coated sample showed a moderate release rate, with the Mg2⁺ concentration rising sharply during the first 7 days and gradually stabilizing thereafter, reaching about 3.5 mg/cm2 at day 28. This trend suggests that the OCP coating provided partial protection but still allowed some ion diffusion through microdefects and pores.
The HA-coated WE43 sample demonstrated the lowest Mg2⁺ ion release among all groups. The release increased slightly during the first week and then stabilized at around 2.5 mg/cm2 after 28 days. This indicates that the HA layer effectively reduced the corrosion rate by forming a stable and compact barrier against electrolyte penetration. The early stabilization of Mg2⁺ release also implies enhanced coating integrity and corrosion resistance.
Overall, the Mg2⁺ ion release results are consistent with the surface observations and XRD analyses, confirming that the HA coating provides the most effective corrosion protection, followed by the OCP coating, while the uncoated WE43 undergoes rapid degradation in HBSS.
Variation in pH of the immersion solution
Figure 5 presents the change in pH of the HBSS during immersion of uncoated, OCP-coated, and HA-coated WE43 samples for up to 28 days. The initial pH of the HBSS was approximately 7.4, which increased progressively with immersion time for all samples due to the release of Mg2⁺ ions and the generation of hydroxide ions during magnesium corrosion.

The change of pH of the solutions during immersion.
The uncoated WE43 sample exhibited a rapid increase in pH during the first week, reaching around 9.0, and stabilized thereafter at approximately 9.8 after 28 days. This sharp increase indicates intensive corrosion and hydrogen evolution, which are typical of unprotected magnesium alloys in aqueous environments.
In contrast, both OCP- and HA-coated samples showed significantly lower pH elevations, confirming the corrosion-inhibiting effects of the calcium phosphate coatings. The OCP-coated sample demonstrated a moderate increase in pH, reaching ~9.0 after 28 days, while the HA-coated sample maintained the lowest pH level throughout the immersion period, stabilizing at approximately 8.5 after 14 days. The slower and lower pH rise observed for the coated samples indicates a reduced corrosion rate and improved chemical stability of the surface.
Among the coatings, the HA layer exhibited superior protective efficiency, as evidenced by the smallest deviation from physiological pH. This stability suggests that the HA coating effectively mitigates Mg dissolution and maintains a more biocompatible environment by suppressing alkalization of the surrounding medium.
Potentiodynamic polarization
Potentiodynamic polarization measurements were conducted to evaluate the electrochemical corrosion behavior of the uncoated, OCP-coated, and HA-coated WE43 samples. The corresponding polarization curves are presented in Figure 6, and the extracted corrosion parameters, including corrosion potential (Ecorr) and corrosion current density (Icorr), are summarized in Table 3.

Potentiodynamic polarization curves of the three samples in HBSS.
Corrosion potential (Ecorr) and corrosion current density (Icorr).

The uncoated WE43 exhibited the most negative Ecorr (−1.47 V) and the highest Icorr (5.01 × 10⁻6 A·cm⁻2), indicating high susceptibility to anodic dissolution and poor corrosion resistance in the testing solution. After coating with OCP, both Ecorr and Icorr shifted toward lower corrosion activity, with Ecorr increasing to −1.38 V and Icorr decreasing to 1.00 × 10⁻6 A·cm⁻ 2 . This reduction in corrosion current density suggests that the OCP coating acted as a partial physical barrier, slowing charge transfer at the alloy/electrolyte interface.
Among the three samples, the HA-coated WE43 exhibited the most positive Ecorr (−1.30 V) and the lowest Icorr (1.58 × 10⁻7 A·cm⁻2), indicating the greatest improvement in corrosion resistance. The marked decrease in Icorr compared with both the uncoated and OCP-coated samples is attributed to the denser and more uniform morphology of the HA coating, which provides a more effective barrier against electrolyte penetration.
Although the anodic branch of the HA-coated sample appears steeper than that of the OCP-coated sample, the corrosion behavior should be primarily interpreted based on Ecorr and Icorr values rather than the anodic slope alone. The steeper anodic response may be associated with localized activation following partial electrolyte penetration through the coating, while the overall lower corrosion current still reflects a significantly reduced corrosion rate. Therefore, the polarization results indicate that both coatings improve corrosion resistance, with HA providing the most effective protection.
These electrochemical findings are consistent with the immersion test results, which showed reduced degradation and improved surface stability for the coated samples. In particular, the HA-coated specimens exhibited the lowest overall degradation tendency, supporting the conclusion drawn from the lower Icorr values that the HA layer acts as a more protective and stable barrier than the OCP coating.
Discussion
Formation mechanism of OCP and HA coatings
The XRD and SEM results indicate that distinct calcium phosphate phases—octacalcium phosphate (OCP) and hydroxyapatite (HA)—were successfully formed on the WE43 surface. The formation of these phases can be explained by the supersaturation and subsequent crystallization processes in the phosphate solution, governed by local pH, temperature, and ionic concentration. Initially, magnesium dissolution at the WE43 surface generates Mg2⁺ ions and elevates the local pH due to hydrolysis reactions. 35 This localized alkalization promotes precipitation of calcium phosphate species through the following general reaction sequence 36 :

When the solution pH is moderately acidic to neutral (pH 5.5–6.5), OCP
In the present study, the OCP coating exhibited lower crystallinity and higher porosity, consistent with incomplete conversion to HA under mild conditions. In contrast, the HA coating showed sharper XRD peaks and a smooth, compact morphology, suggesting complete transformation and crystal maturation. Similar phase evolution has been reported by Iijima et al. 37 and Temizel et al., 38 who demonstrated that HA coatings grown at higher pH or longer deposition times exhibit improved crystallinity and adhesion to magnesium substrates. The initial Mg dissolution thus plays a dual role—providing localized alkalinity for Ca–P precipitation and simultaneously creating a diffusion gradient that affects the phase composition of the coating.
Protection mechanism of coating layers
Figure 7 presents the surface morphologies and corresponding EDS analyses of the uncoated, OCP-coated, and HA-coated WE43 samples after immersion in HBSS for 28 days.

SEM micrographs and EDS analysis on the surface of the immersed specimens after 28 days: (a) uncoated sample, (b) OCP coated sample, and (c) HA coated sample.
The EDS spectrum revealed a high oxygen content (63.6 at.%) together with magnesium (32.9 at.%), indicating that Mg(OH)2 was the predominant corrosion product formed on the surface of uncoated sample. The presence of visible cracks and a discontinuous corrosion film suggest weak surface protection and the formation of unstable layers that likely detached during immersion. When exposed to HBSS, the highly reactive magnesium matrix readily reacts with water to form Mg(OH)2 accompanied by hydrogen evolution. Although this hydroxide film provides temporary protection, it is easily destabilized by Cl⁻ ions in the solution, which convert Mg(OH)2 into soluble MgCl2. This reaction breaks down the surface film and promotes localized corrosion. The repeated cycle of film formation and dissolution leads to the development of a loose and porous corrosion layer, consistent with the cracked morphology observed in SEM images and the high Mg and O signals detected by EDS. As a result, the uncoated alloy undergoes rapid and uncontrolled degradation, exhibiting poor surface stability after prolonged immersion, in agreement with previous reports.21,22,39
In contrast, the EDS spectrum of the OCP-coated samples after immersion showed a significant increase in Ca (17.7 at.%) and P (10.9 at.%) and a sharp reduction in Mg (3.5 at.%), confirming that the OCP layer remained largely intact and acted as an effective protective barrier during immersion. The Ca/P ratio (~1.62) closely corresponds to the stoichiometric value of octacalcium phosphate, indicating that the coating largely preserved its phase composition and structural stability during the immersion period. 29 However, slight variations in the Ca/P ratio and local surface morphology suggest that partial transformation of OCP into a more stable, apatite-like phase may have occurred through ion exchange and reprecipitation processes in the HBSS environment. 28 Such transformation behavior has been widely reported for OCP coatings on magnesium alloys, where OCP gradually hydrolyzes to form hydroxyapatite or calcium-deficient apatite under physiological conditions.28–30
The EDS data of the HA-coated sample revealed Ca and P contents of 16.9 at.% and 11.4 at.%, respectively, with a Ca/P ratio of approximately 1.48, which is close to the theoretical stoichiometric value of hydroxyapatite (1.67). 27 The low Mg content (3.1 at.%) further indicates minimal substrate exposure, demonstrating that the HA coating provided strong corrosion protection and high chemical stability during immersion in HBSS. The nearly stoichiometric Ca/P ratio suggests that the coating retained its crystalline integrity and did not undergo significant dissolution or structural degradation throughout the 28-day period.
The dense and homogeneous morphology observed on the HA-coated surface supports this finding, indicating that the HA layer effectively prevented the diffusion of aggressive ions such as Cl⁻ toward the substrate. Hydroxyapatite is known for its excellent chemical stability and low solubility under near-neutral physiological conditions, which enables it to serve as a long-term corrosion barrier on magnesium substrates.22–24,40 The coating acts through both physical shielding and electrochemical stabilization, reducing direct contact between the electrolyte and the reactive Mg matrix and thus inhibiting hydrogen evolution and the formation of unstable Mg(OH)2 corrosion products. 40
Conclusion
This study demonstrated that calcium phosphate coatings significantly improve the corrosion resistance and surface stability of WE43 magnesium alloy. Both octacalcium phosphate (OCP) and hydroxyapatite (HA) coatings were successfully formed on the alloy surface, as confirmed by XRD and SEM analyses. The OCP coating exhibited a plate-like, porous morphology with lower crystallinity, whereas the HA coating formed a compact and highly crystalline layer.
Immersion tests revealed that both coatings effectively reduced magnesium ion release and moderated pH fluctuations in the physiological range; however, the HA-coated samples provided the most stable performance. The superior protective behavior of the HA layer was attributed to its dense structure, chemical stability, and self-healing capability through secondary apatite reprecipitation.
Overall, these findings confirm that HA-coated WE43 alloys exhibit superior corrosion resistance and chemical stability compared to both uncoated and OCP-coated counterparts. The developed coating strategy provides a promising approach to control degradation kinetics and enhance the bio functionality of magnesium-based biodegradable implants. Future work will focus on optimizing coating adhesion, evaluating in vitro cytocompatibility, and assessing in vivo degradation behavior to further validate the biomedical potential of HA-modified WE43.
Footnotes
Author contributions
LVH designed and conducted the experiments; DNN conducted the experiments; PMK analyzed and evaluated the data; NVN supervised, analyzed and evaluated the data; LVH and NVN prepared the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Guarantor
NVN.
