Abstract
A major burden of the healthcare system resides in providing proper medical treatment for all types of chronic wounds, which are usually treated with dressings to induce a faster regeneration. Hence, to reduce healing time and improve the patient’s quality of life, it is extremely important to select the most appropriate constituent material for a specific wound dressing. A wide range of wound dressings exist but their mechanisms of action are poorly explored, especially concerning the immunomodulatory effects that occur from the interactions between immune cells and the biomaterial. Tissue-resident and monocyte-derived recruited macrophages are key regulators of wound repair. These phagocytic immune cells exert specific functions during the different stages of wound healing. The recognition of the substantial role of macrophages in the outcome of the wound healing process requires specific understanding of the immunomodulatory effects of commercially available or newly developed wound dressings. For a precise intervention, it is necessary to obtain more knowledge on macrophage polarization in different phases of wound healing in the presence of the dressings. The main purpose of this review is to collect clinical cases in which macrophage immunomodulation was taken into consideration as an indicator of the performances of novel or mainstream wound dressing materials, including those provided with antimicrobial properties.
Introduction
Wound healing is a complex process in which different cell types (e.g. macrophages) are involved, progression is strictly regulated, and different cellular activities overlap. 1 If an atypical progression takes place, a chronic wound may result, impacting the patient’s quality of life and the economic cost to the healthcare system 2.
Recent research has been focused on understanding the factors that influence the healing progress and new discoveries elucidating mechanisms of physiologic wound repair have been reported. The development of advanced wound dressing biomaterials, able to play an active role in wound repair, is also a key issue, which is attracting increasing attention.
Therefore, the main objective of this manuscript is to review established and recent concepts about wound repair, with a focus on chronic wounds, and to underline the importance of macrophages in this context, as well as the effect of different types of wound dressing biomaterials on macrophage function.
Chronic wounds
Chronic wounds: A never-ending nightmare
Non-healing wounds, also known as chronic wounds, of diverse etiologies, are a significant health problem worldwide, being a major cause of morbidity and mortality. Owing to the extent of its strong social impact, wounds have been described as a “silent epidemic.” 2 Concomitant with the increase of the obese population, increased diabetic patients, and prolongation of life expectancy, the number of chronic wound cases is ascending alarmingly. Accurate epidemiological analysis concerning chronic wounds is scarce; consequently, the determination of the precise cost of wound management is affected. Nevertheless, there is a body of evidence that the incidence of chronic wounds is increasing, becoming an economic burden in developed countries.2–4
A wound is characterized by the disturbance of the tissue integrity and structural organization of body cells, with or without destroying them. 5 If a wound fails to initiate its physiologic regeneration after 2–3 weeks and does not regain its structural and functional integrity (remaining open for 3 months), it is classified as a non-healing (chronic) wound.6–8 Examples of chronic wounds are diabetic foot ulcers (Figure 1(a)), venous leg ulcers (Figure 1(b)), and pressure ulcers (Figure 1(c)). 9
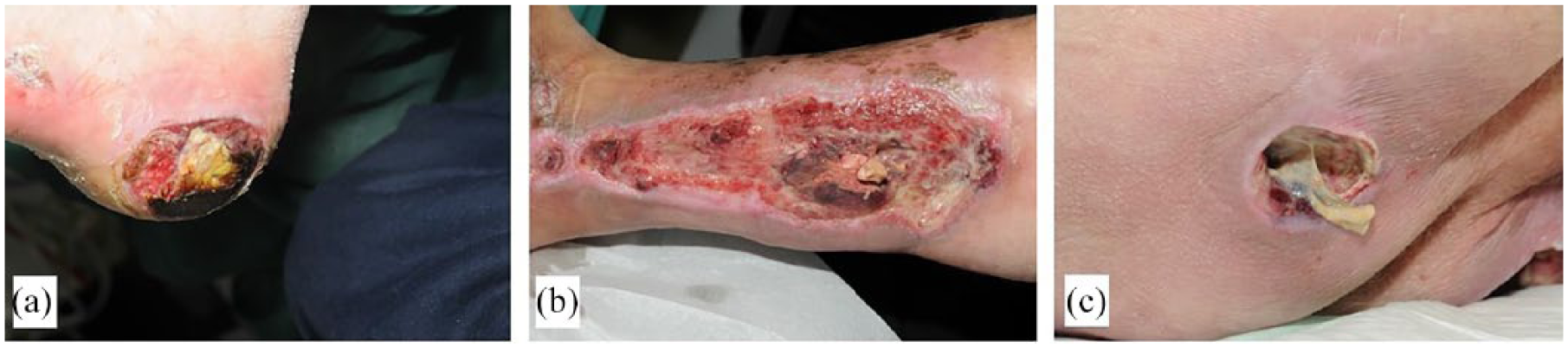
Morphology of chronic wounds : (a) colonized diabetic foot ulcer at the heel; (b) infected venous leg ulcer; (c) colonized pressure ulcer of the right ischium.
Disruption of healing process by bacterial infection and persistent inflammation
Chronic wounds are associated with an increased risk of infection, since long-term cutaneous wounds are more susceptible, owing to the loss of the protective barrier of intact skin. In fact, one of the main causes of impaired wound healing is the presence of high bacterial contamination. 10 The progression from contamination through colonization to infection on open wounds depends on the bacterial multiplication rate, crosstalk between different microorganisms, and the initiation of the innate immune response, such as the disturbance of a successful recognition and ingestion of invading microorganisms, owing to the failure of the opsonization effect. This effect involves marking pathogens with antibodies and factors of the complement system, in order to attract phagocytes (e.g. macrophages) to the site of contamination and induce the elimination of the pathogens.11,12 Figure 2 describes the most common effects induced by the progression of bacterial growth in wounds. Starting from a contaminated state, in which no major negative signals are observed, the system evolves to a severe state (established infected wound), in which pathogens proliferate and an inflammatory response is clearly activated.
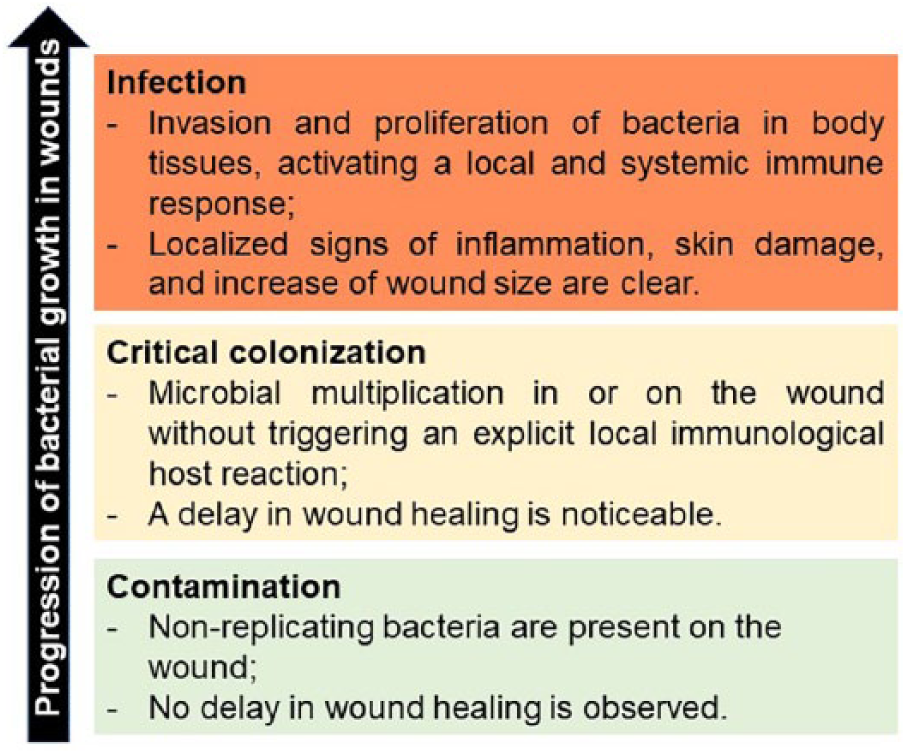
Description of cutaneous wounds by the establishment and growth of bacteria. 13
In the past years, there has been increasing evidence that bacteria exist in chronic wounds as organized communities, in which they are able to cooperate and communicate. These colonies are commonly defined as biofilms.10, 14,15 Biofilms are usually polymicrobial in nature and are surrounded by a self-produced extracellular polysaccharide matrix, which contains extracellular enzymes, plasmids, and signal molecules for chemical communication (quorum sensing). 16 In wound biofilms, blood components may also be enclosed in the matrix. 17 Moreover, bacteria show a higher tolerance to antibiotics and have a barrier protection against the host immune system.11,18
For successful removal of contaminating bacteria, inflammation is a crucial part of the normal wound healing process. However, when the microbial clearance is incomplete, the progression of bacterial growth on wounds reaches a critical colonization state and the immunologic inflammatory process will be prolonged. 1
The combination of the presence of bacteria (especially as a biofilm) and persistent inflammatory reaction involves the overexpression of pro-inflammatory cytokines (e.g. interleukin-1 (IL-1), tumor necrosis factor-α (TNF-α), chemokines (C-X-C motif ligand 9 (CXCL9), CXCL10, and CXCL11) and other signal molecules (e.g. signal transducer and activator of transcription 1 and nitric oxide synthase 2). This framework is the basis for the continuous non-healing status of chronic wounds.1,11
Natural or synthetic antimicrobial wound dressing biomaterials can play an important role in preventing or decreasing the high bacterial contamination and provide a chance for the continuation of normal wound healing. 21
Wound healing is a highly complex multicellular and regulated metabolic process. The normal wound healing stages, in timeline order, comprise four overlapping phases: hemostasis, inflammation, proliferation, and remodeling.22,23 In chronic wounds (Figure 3), bacterial critical colonization or infection is present and the inflammatory phase is persistent for a long period of time (from at least 4 weeks to several months). 6 During this period, several irregularities are present and, finally, no proper shift to the proliferation and remodeling phases occurs. 24
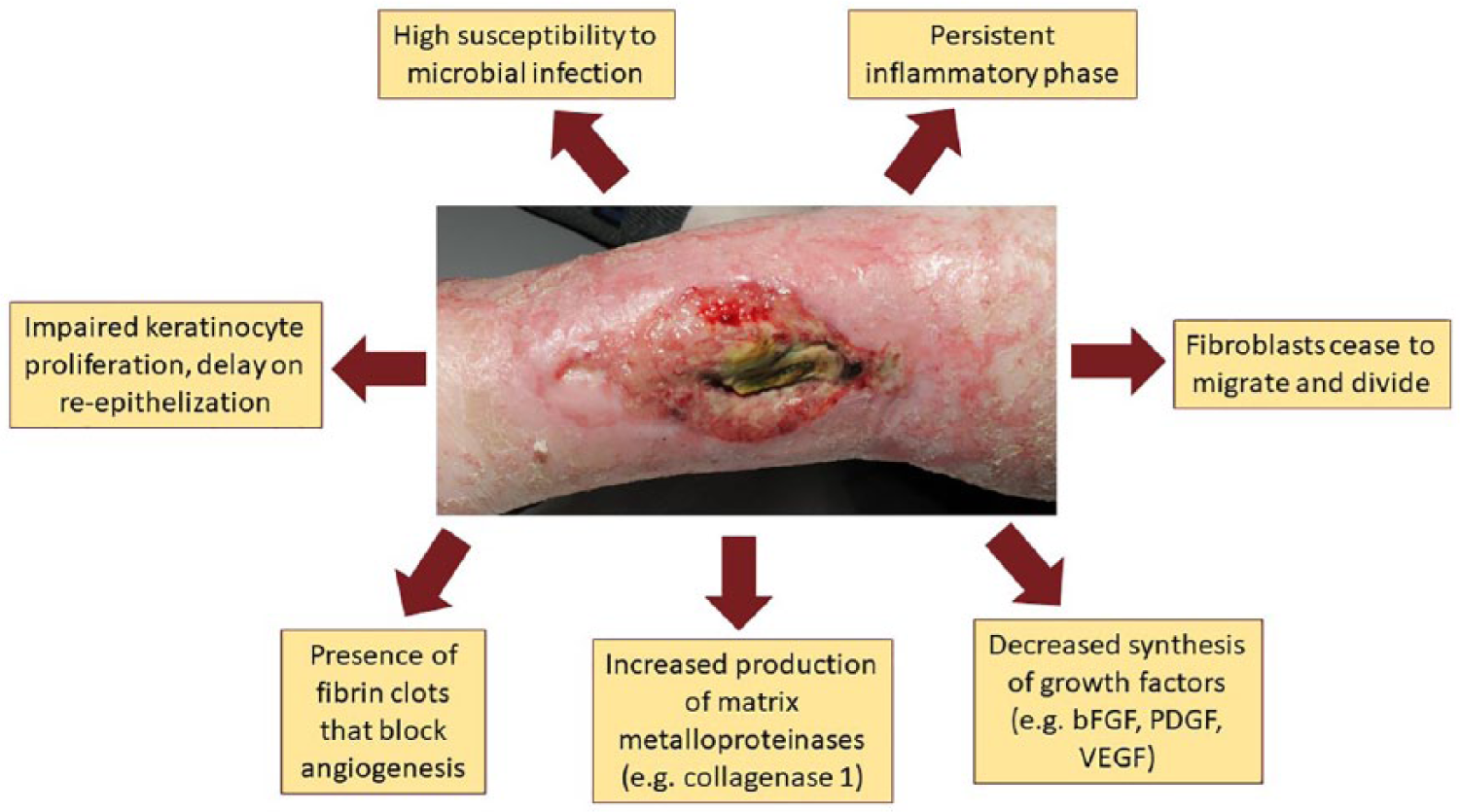
Abnormalities in the chronic wound environment.
Macrophages in wound repair and regeneration
Recruited monocyte-derived macrophages and tissue-resident macrophages are essential key players in response to a tissue injury. After an injury, circulating neutrophils are the first to reach the site of damage. This is followed by the infiltration of monocytes, which differentiate into macrophages, joining tissue-resident macrophages with the mission of restoring integrity. 25 Macrophages are mononuclear leukocytes, part of the innate immune system, and are required for successful skin regeneration. 26 In fact, macrophages are involved in all phases of wound healing. Until recently, this immune cell population was considered a “background actor”, but during the last decade scientists’ attention exponentially increased, leading them to unveil macrophage immunomodulation, which is relevant in many diseases. 27 Wound healing research has revealed that plasticity, flexibility, and heterogeneity of macrophages have a crucial participation in obtaining successful cutaneous regeneration. Das et al. have compiled several studies that clearly prove the participative role of wound macrophages in scavenging, phagocytosis, efferocytosis, antigen presentation, promotion of repair, extracellular signaling, and angiogenesis. 28 Special attention has been given to the capacity of macrophages to remove debris from dead and dying cells, as a resolution of inflammation and a shift toward regeneration. 29
Using a very simplistic nomenclature, macrophages are able to polarize into two categories, depending on the environmental stimuli: classically activated M1 pro-inflammatory macrophages, which are involved in combating infections, or alternatively activated M2 tissue-healing macrophages, which are connected with tissue remodeling28,30,31 This polarization occurs as a continuous spectrum, in which macrophages shift and gain different metabolic and phenotypic characteristics (Figure 4).

Macrophage polarization occurring in a spectrum between M1 and M2 macrophages.
During the inflammatory phase of cutaneous healing, M1 macrophages are predominant. These have an extremely important role in the phagocytosis of dead or dying cells and their components, besides orchestrating more macrophages immune cells at the site of injury. 32 M1-like macrophages are known for their high production of nitric oxide, an antibacterial oxidative metabolite, and for being efficient antigen-presenting cells. Their stimulation is induced, for example, by lipopolysaccharides of the outer membrane of Gram-negative bacteria or interferon-γ (IFN-γ), and consequently its metabolism is activated toward producing high levels of pro-inflammatory cytokines, such as IL-1β, IL-6, and TNF-α. 33 Additionally, this population is characterized by the release of reactive oxygen species, mainly derived from hydrogen peroxide (H2O2) and superoxide anions (O2−). 34 Reactive oxygen species have been described as chemo-attractants of monocytes to the wound location. 35
Conversely, M2-like macrophages appear in greater quantities during the proliferation and remodeling phases of wound healing, during which they express high levels of anti-inflammatory cytokines, such as IL-4 and IL-10, and the enzyme arginase-1, which is believed to be a main contributor to wound healing. 26 The classification of M2-like macrophages is still controversial and a subject of persistent study; hence, many types of subdivision are found in the literature, especially depending on specific diseases. It is not clear whether these intermediate phenotypes can be divided in such a strict categorized manner, owing to their plasticity through a continuous spectrum. However, it seems currently well accepted that M2-like macrophages are divided into four different subtypes (M2a, M2b, M2c, and M2d) in what concerning the wound healing process (Table 1).32,36,37
Most common characteristics of M2-like macrophages subtypes.23,32,38–41
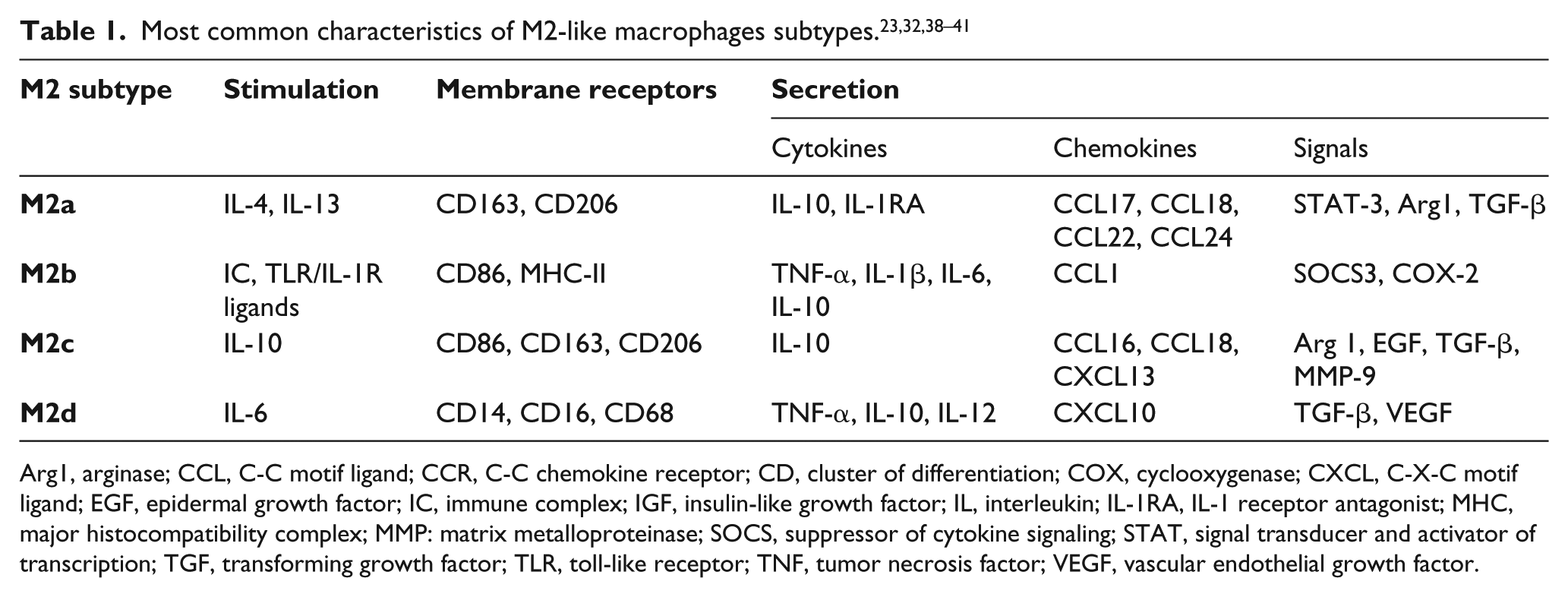
Arg1, arginase; CCL, C-C motif ligand; CCR, C-C chemokine receptor; CD, cluster of differentiation; COX, cyclooxygenase; CXCL, C-X-C motif ligand; EGF, epidermal growth factor; IC, immune complex; IGF, insulin-like growth factor; IL, interleukin; IL-1RA, IL-1 receptor antagonist; MHC, major histocompatibility complex; MMP: matrix metalloproteinase; SOCS, suppressor of cytokine signaling; STAT, signal transducer and activator of transcription; TGF, transforming growth factor; TLR, toll-like receptor; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor.
Briefly, M2a macrophages release platelet-derived growth factors and stimulate fibroblasts to proliferate and secrete extracellular matrix, an essential factor for angiogenesis in wound repair. 33 M2b macrophages have been shown to have an important function in decreasing the inflammatory process by synthesizing high levels of the anti-inflammatory cytokine IL-10, despite the fact that these cells still produce pro-inflammatory cytokines (e.g. TNF-α).23,26 M2c macrophages are intervenient in vascular and matrix renovation, owing to the high production of extracellular matrix. Lastly, M2d macrophages are pro-angiogenic and produce high levels of vascular endothelial growth factor. 32
Macrophages: Dysfunction in chronic wounds
Non-healing wounds can be exacerbated because of improperly acting macrophages. In normal wound healing, macrophages induce an initial pro-inflammatory response and shift to a pro-repairing action. This inflammatory-to-regenerative shift is completely dependent on their capacity to perform phagocytosis of dead neutrophils and remaining debris. 42 In healthy processes, M1 macrophages are partially converted to M2b macrophages to overcome inflammation; however, M1 macrophages can accumulate in chronic wounds, owing to environmental stimuli that do not allow the transition. 32 As a consequence, a highly inflammatory environment is maintained, and wound closure is delayed, as proved by the increased levels of IL-1 and TNF- α (pro-inflammatory cytokines produced by M1-like macrophages) in chronic wound fluids. 43 Moreover, the fluid also contains higher levels of matrix metalloproteinases than those usually observed during the proliferation and remodeling phases, leading to protein and extracellular matrix degradation, and, as a consequence, impaired tissue regeneration. 44 Dysregulation of M2-like macrophages is also a big concern. Alteration of their typical phenotype and metabolic characteristics dictates the level of collagen deposition and scar formation. 32 As an example, an overexpression of type-III collagen has been found in hypertrophic scars. 45
Wound dressings
The correct selection of wound dressing biomaterials is vital for a faster wound recovery of patients with non-healing cutaneous injuries. Hence, studies must provide information about biocompatibility, possible antibacterial efficacy, and material properties. 46
Such products as bandages and gauzes, of natural or synthetic origin, are commonly used to cover an acute wound to prevent contamination. 47 This type of dressing lacks many features for proper wound healing, such as maintaining wound hydration and preventing wound contamination. Modern wound dressings, based on polymers, can be made bioactive by the incorporation of antibiotics (e.g. gentamicin), metals (e.g. silver), or growth factors (e.g. fibroblast growth factor 1). 48
Unsurprisingly, the diverse surface morphologies and incorporated active components will induce an effect on the phenotype and metabolism of macrophages. Hence, it is extremely important to study the immunomodulatory effects of wound dressing biomaterials.
Wound healing biomaterials to the rescue: Are biomaterial–macrophage interactions friends or foes?
Countless wound dressings are commercially available, and a number of new dressings are under development, but their mechanisms of action are poorly understood. Therefore, there is a great interest in exploring immune cell–biomaterial interactions. Owing to the importance of the effector functions of macrophages, it has become essential to obtain evidence about the polarization of macrophages and immunomodulation in the presence of different biomaterials. 49 As previously discussed, macrophages are dysregulated in non-healing wounds. Furthermore, a correlation between prolonged M1 cell populations and delayed wound repair has been demonstrated. 50 The importance of a wound dressing biomaterial that is able to stimulate the shift from M1 to M2 tissue-healing macrophages, or at least to avoid M1 pro-inflammatory phenotype perpetuation, is thus clearly evident. In the rest of this paper, relevant work describing the effect of non-commercially and commercially available wound dressing biomaterials on the immunomodulation of macrophages (Table 2) will be analyzed. To test their response to wound dressings, various techniques can be applied to macrophages after exposition to the biomaterial influence, such as gene up- or downregulation evaluation by polymerase chain reaction, determination of expressed membrane markers through flow cytometry, secretion of cytokines and other signal molecules by enzyme-linked immunosorbent assay, and evaluation of posttranslational proteins by proteomics.
Selected studies that unravel alterations of macrophage phenotype or metabolism in the presence of wound dressing biomaterials.
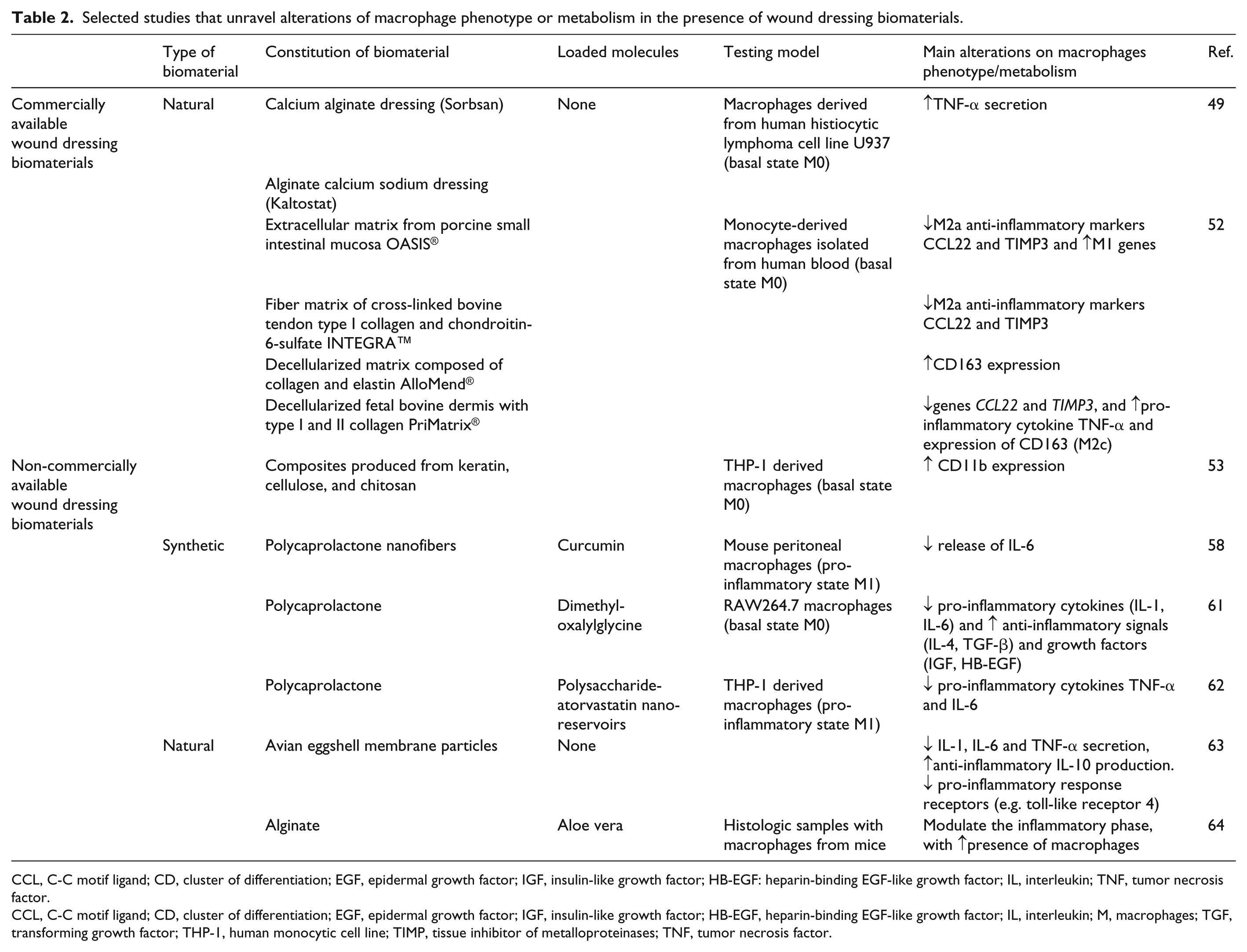
CCL, C-C motif ligand; CD, cluster of differentiation; EGF, epidermal growth factor; IGF, insulin-like growth factor; HB-EGF: heparin-binding EGF-like growth factor; IL, interleukin; TNF, tumor necrosis factor.
CCL, C-C motif ligand; CD, cluster of differentiation; EGF, epidermal growth factor; IGF, insulin-like growth factor; HB-EGF, heparin-binding EGF-like growth factor; IL, interleukin; M, macrophages; TGF, transforming growth factor; THP-1, human monocytic cell line; TIMP, tissue inhibitor of metalloproteinases; TNF, tumor necrosis factor.
Commercially available wound dressings
Alginate dressings are widely used in the treatment of exuding wounds and may enhance the healing process. Thomas and co-workers evaluated the capacity of interaction between alginates present in commercially available absorbent wound dressings with macrophages, 49 and observed that macrophages derived from the human histiocytic lymphoma cell line U937 in the presence of a calcium alginate dressing (Sorbsan) at 1 mg/mL stimulated the production of 302 ± 19 pg/mL of TNF-α and that the alginate calcium sodium dressing Kaltostat induced the production of 839 ± 36 pg/mL of TNF-α. Two other alginate dressings were also tested without significant differences. Thomas and co-workers concluded that alginate dressings have the potential to improve wound healing, owing to the ability to activate wound macrophages in producing TNF-α, a pro-inflammatory signal. 49 This cytokine has been found to be secreted by M2b and M2d macrophages but, contradictorily, it has been proved that prolonged expression of TNF-α contributes to a delayed wound healing, for example, in diabetic wounds. 51
In a study conducted by Witherel and collaborators, 52 the responses of monocyte-derived macrophages isolated from blood (one donor) to four currently applied wound dressing biomaterials were analyzed and compared. The four wound dressing biomaterials were:
(a)
(b)
(c)
(d)
The expression of M2a anti-inflammatory markers CCL22 and TIMP3 in the presence of OASIS® and INTEGRA™ was downregulated; this suggests a possible inhibition of extracellular matrix secretion and fibrosis, which are key phenomena for wound closure. OASIS® was also responsible for the largest increase of expression of M1 genes; hence, Witherel and collaborators 52 considered this product significantly pro-inflammatory. Although INTEGRA™ is constituted of collagen I and the anti-inflammatory chondroitin-6-sulfate, an opposite effect was observed and the reason might be the presence of glutaraldehyde in this dressing. OASIS® and INTEGRA™ seem to be a poor option for chronic wound cases.
In the case of PriMatrix®, a downregulation of the genes CCL22 and TIMP3 was also observed. Moreover, this wound dressing caused an overexpression of the pro-inflammatory cytokine TNF-α and increased the expression of CD163, associated with M2c, which is a less fibrotic phenotype. 52 For last, AlloMend® only induced an effect of the upregulation of CD163, and it was considered the dressing with the lowest influence on macrophage response. Overall, this work shows that the dressings have a direct effect on macrophage immunomodulation, and give us a very important take home message, which is “the same wound dressing can have a dual effect promoting the polarization to M1- and M2-like macrophages”.
Non-commercially available materials for wound dressings
Composites produced from keratin, cellulose, and chitosan have shown bactericidal and anti-inflammatory properties. 53 For instance, chitosan is well known for its highly antimicrobial activity against fungi and bacteria. 54 As a result of these properties, chitosan is used in the production of commercial wound dressing products, such as Tegasorb® and KytoCel®, alone or in combination with synthetic polymers, such as poly(ε-caprolactone) and polyethylene oxide.55,56 Anti-inflammatory properties were detected on THP-1 derived macrophages in the presence of composites produced from keratin, cellulose, and chitosan 53 Herein, the authors of these studies noticed that the composites induced an increase in the expression of CD11b on macrophages. As a support, it has been proven that the depletion of CD11b+ macrophages in a bacterial pathogen-induced inflammation model delayed inflammation resolution and antigen clearance, indicating its importance for proper wound healing. 57
In a study of Merrell and co-workers, curcumin-loaded poly(ε-caprolactone) (PCL) nanofibers were evaluated for wound healing applications.
58
PCL is a synthetic polymer utilized in wound dressing biomaterials
59
; it possesses several advantages over other synthetic polymers, including stability, low cost, mechanical strength, and biocompatibility.
60
In combination with the anti-oxidant curcumin, these novel nanofibers reduced the anti-inflammatory reaction, proven by the low release of IL-6 observed in mouse monocyte-derived macrophages, which were previously polarized to the M1 state by lipopolysaccharides.
Likewise, in a study in which the hydroxylase inhibitor dimethyloxalylglycine was embedded in PCL, a similar anti-inflammatory modulation of macrophages was demonstrated. 61 On RAW264.7 macrophages, Zhang et al. 61 observed a decreased production of the pro-inflammatory cytokines (IL-1, IL-6) and an enhancement of anti-inflammatory signals (IL-4, TGF-β) and growth factors (insulin-like growth factor, heparin-binding epidermal-growth-factor-like growth factor).
Schwinté and colleagues produced electrospun PCL containing polysaccharide-atorvastatin nano-reservoirs by using complexes with poly-aminocyclodextrin. 62 These membranes were tested on THP-1 stimulated by lipopolysaccharides. A pronounced decrease in the production of pro-inflammatory cytokines TNF-α and IL-6 (reductions of 60 and 80%, respectively) was verified for these M1-like macrophages. This strategy is very interesting to overcome the exacerbated inflammatory phase that occurs in non-healing injuries.
Natural biomaterials, such as avian eggshell membrane have been widely used on injured skin, especially in Asian countries. In a recent study, processed avian eggshell membrane particles have been found to contain collagen, hyaluronic acid, sulfated glycosaminoglycans, and
Other examples of non-commercial materials are the novel alginate dressings with aloe vera, which are able to modulate the inflammatory phase, promote angiogenesis, and stimulate type I collagen production. 64
Biomaterial–macrophage interactions: Main drawbacks
When a biomaterial is in contact within the human body, a series of events occur. These events include65,66:
Immediate protein adsorption on the biomaterial;
Cell infiltration (i.e. neutrophils, monocytes, and platelets) and adhesion to the biomaterial surface;
Production of chemokines and cytokines by adhered cells;
Macrophage fusion forming multinucleated foreign body giant cells;
Interaction between macrophages or foreign body giant cells and inflammatory or wound healing cells in general.
The formation of foreign body giant cells originates from fused variants of M1 macrophages in an attempted phagocytic response to eradicate a large foreign body.66–68 As a result of this effort, foreign body giant cells secrete reactive oxygen species and matrix metalloproteinases, provoking a failure to deposit extracellular matrix and the consequent local persistence of damage-associated molecular patterns and continuous activation of macrophages.69,70 Since regeneration is not achieved, tissue granulation occurs, finally forming a fibrous capsule.71,72 This is a very important effect in the cutaneous healing area, especially if a wound dressing biomaterial must be left in contact for a prolonged period.
This foreign body response also alters the material surface, leading to biomaterial degradation, inducing a continuous inflammatory response and consequent failure of injury repair, which finally has an undesirable impact on biocompatibility. 71 Hence, the formation of foreign body giant cells is the “dark side” of macrophages in interaction with biomaterials.
Final remarks
Biomaterial–macrophage interactions are a “hot topic” nowadays. A beneficial macrophage response in contact with a biomaterial is therefore crucial for a successful overcome of the perpetual inflammatory phase that occurs in chronic wounds. Thus, biomaterials that induce the “M1–M2 shift” are ideal candidates for promoting wound closure and avoiding susceptibility to contamination from the early stages. Not only a huge effort has been given to develop newly synthesized biomaterials with anti-inflammatory properties, but also a deeper understanding of the selective impact on wound macrophage polarization of currently applied biomaterials is being explored. However, the field of macrophage immunomodulation and biomaterial–macrophage interaction is still “immature”. The progress that has been made in understanding macrophage polarization under physiological versus pathological wound conditions has given a boost to research in biomaterial science. The focus on immunomodulatory effects may lead to the establishment of a novel generation of immunomodulating biomaterials, able to influence the behavior of the wound healing key players, such as different macrophage phenotypes.
Footnotes
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the “Drug-Free Antibacterial Hybrid Biopolymers for Medical Applications” HyMedPoly project and the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie Action MSCA-ITN-2014-EID: Marie Skłodowska-Curie Innovative Training Networks (grant number 643050).
