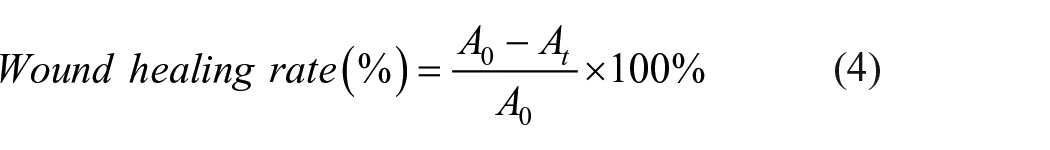Abstract
Chitosan and its derivatives show potent biocompatibility, biodegradability, antimicrobial activity, hemostatic effects, and wound healing properties. Their application in wound dressings has garnered substantial research interest. In this work, we prepared a drug-loaded hydrogel by mixing N-glycosylated chitosan with polyvinyl alcohol (PVA), followed by loading of ofloxacin. A 2:1 volume ratio of chitosan to PVA was found to be optimal based on swelling and water evaporation rates. The slow-drug-release performance of the blended hydrogel was best when the ofloxacin loading was 5.0%. The ofloxacin-loaded hydrogel shows excellent antimicrobial properties in vitro and wound healing ability in an in vivo rabbit full-thickness excision wound model. The chitosan/PVA blended hydrogel has great potential for use in wound dressings and sustained drug release.
Introduction
A wound dressing is a material used to cover the surface of a wound or to wrap the wound. In 1962, Winter proposed the theory of “moist healing,” which stated that a humid environment around the wound can promote wound cell migration. 1 This theory laid an important foundation for the development of modern wound dressings. An ideal wound dressing should maintain an adequate water vapor transmission rate, prevent microbial invasion, and have suitable mechanical strength. It should also be nontoxic, nonirritating, and easy to remove without trauma. 2 Furthermore, the materials should absorb exudates from wounds, and promote granulation and growth of epithelial tissue.
Chitosan is the most abundant natural biopolymer, second only to cellulose. It is mainly derived from fungi, shrimp shells, and crustacean exoskeleton deacetylation chitin. 3 Chitosan hydrogels have good mechanical properties and water retention capacity. They can be shaped according to the size of the wound and cover wounds well. Hydrogels can be prepared by a variety of methods, which are mainly divided into three categories: chemical, physical, and radiation-dependent crosslinking. 4 The chemical crosslinking approach mainly uses chemical crosslinking agents, such as glutaraldehyde and genipin, to generate covalent bonds between polymer molecular chains, thus forming a three-dimensional network structure. Physical crosslinking mainly involves crosslinking a polymer through intermolecular hydrophobic interactions, intertwined molecular chains, hydrogen bonds, and ionic bonds. Physically-crosslinked hydrogels are safer than chemically-crosslinked gels because they are nontoxic; they may also be biodegradable. 5 Radiation crosslinking mainly relies on an electron beam, γ-rays, ultraviolet light, or microwave radiation energy to promote the formation of netlike linkages inside a hydrogel. 6
The safety of wound dressing materials is always the first priority. Tsao et al. 7 tested the cytotoxicity of chitosan hydrogels with 3T3 fibroblasts. The chitosan hydrogels were noncytotoxic and promoted cell adhesion and proliferation. Yang et al. 8 prepared a new type of crosslinked gel using water-soluble chitosan, polyvinyl alcohol (PVA), and glycerol. The dressings were noncytotoxic toward fibroblasts and effective in promoting wound healing in a model of whole-layer injury. Moreover, chitosan hydrogels have antimicrobial properties 9 and can carry a drug, such as an antimicrobial agent, to enhance their bacteriostatic effect.
In this study, we blended N-glycosylated chitosan with PVA to form hydrogels using a freezing–thawing cycle method. Based on the swelling ratio and water evaporation rate as evaluation indexes, we chose the optimal proportion of N-glycosylated chitosan to PVA to prepare the hydrogel. The blended hydrogel was then coated with ofloxacin, an antimicrobial drug. Thereafter, we explored the properties of the ofloxacin-loaded hydrogel in sustained drug release, antimicrobial activity, and wound healing. In addition, we analyzed the hydrogel using Fourier-transform infrared spectroscopy (FTIR) and scanning electron microscopy (SEM) to identify its structure and explore its surface morphology.
Experiment
Materials
N-glycosylated chitosan (degree of deacetylation 67%) was obtained from the Laboratory of Pharmacy, Zhejiang University of Technology. Polyvinyl alcohol (PVA-1788) was purchased from Sinopharm Group Chemical Reagent Co., Ltd. Ofloxacin was purchased from Shanghai Biological Chemical Co., Ltd., China. Tryptic soy agar and YM agar were bought from BD Biosciences Co., Ltd., USA.
Preparation of N-glycosylated chitosan/PVA hydrogels
A 10% PVA solution was prepared by dissolving PVA powder in distilled water with gentle magnetic stirring for 3 h in a water bath at 95°C. N-glycosylated chitosan was dissolved in 0.1 M hydrochloric acid solution. After cooling, the PVA solution was added dropwise into the N-glycosylated chitosan solution to volume ratios 1:3, 1:2, 1:1, 2:1, and 3:1 chitosan solution to 10% PVA solution. The mixtures were blended at room temperature and stirred at 300 rpm for 30 min, then kept still overnight until all the bubbles had collapsed. Afterward, a freeze–thaw physical crosslinking method 10 (the sample is frozen at −20°C for 24 h, and thawed at room temperature for 24 h after taking out) was adopted to produce the composite hydrogel.
Swelling
We used the method described in Rao 11 to measure swelling of the hydrogels. First, a 1 cm × 1 cm hydrogel sample was weighed accurately (to 0.0001 g); the weight was recorded as the initial mass (m0). Then the sample was immersed in phosphate-buffered solution (PBS) at pH 7.4 for 6, 12, 18, 24, or 30 h at room temperature. At each time point, a sample was taken out and dried using filter paper. The weight of the sample (m1) was recorded to 0.0001 g. Equation (1) was used to calculate the swelling ratio:
Water evaporation rate
The water evaporation rate from hydrogels was determined using the methods reported in Fan et al. 12 and Cao. 13 W1 is the sample weight after reaching equilibrium swelling condition. After that, the hydrogels were placed in an incubator at 50°C and 50% humidity for 6, 12, 18, 24, and 30 h. The weight at each timepoint was recorded as W2. When the weight of the hydrogel reached constancy, it was considered dry; that weight was recorded as W3. Each weight was measured to 0.0001 g. The water evaporation rate of the hydrogel was calculated from equation (2):
Preparation of compound hydrogels carrying ofloxacin
In the previous experiments, we determined the optimal volume ratio of N-glycosylated chitosan to 10% PVA. A blended solution was prepared in this proportion. Then, an appropriate amount of ofloxacin was added. The mixture was stirred magnetically and then left until all the bubbles had collapsed. Subsequently, freeze–thaw physical crosslinking was used to obtain ofloxacin-loaded hydrogels with drug concentrations 2.5%, 5.0%, 10.0% (weight of drug to weight of hydrogel).
Characterization of the hydrogels by FTIR spectroscopy
FTIR analysis of hydrogels was carried using an FTIR spectrophotometer (Nicolet 170SX, Thermo Fisher Scientific Inc., USA) in the range of 400–4000 cm−1. Dry sample (1 mg) was mixed with 100 mg dry potassium bromide, then the mixture was pressed into a disk to record spectra.
In vitro drug release studies
We prepared ofloxacin standard solutions (1.0–10.0 μg mL−1) by dissolving appropriate amounts of ofloxacin in PBS (pH 7.4). According to the Chinese Pharmacopoeia 2010, 14 we measured the absorbance (A) of ofloxacin at 295 nm, then a standard curve was created with A as the ordinate and concentration (C) as the abscissa.
A sustained release study of ofloxacin was carried out in vitro according to the rotating basket method described in Pharmacopoeia 2010. Hydrogel samples carrying 2.5%, 5.0%, and 10.0% ofloxacin were immersed in 500 mL phosphate buffer (pH 7.4) at 37°C ± 0.5°C. The stirring speed was set to 100 rpm. At 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 20, 30, 40, and 50 h, 1 mL of sample was taken, filtered through a 0.45-μm microporous membrane, and measured at 295 nm. When a sample was taken, an equal volume of phosphate buffer was immediately added to the reaction vessel.
In vitro antimicrobial activity
We took the agar plate diffusion method in the Pharmaceutical Microbiological Examination Manual 15 as a guideline. A circular hydrogel with diameter 1 cm was prepared by tape casting and cutting. Two kinds plates were prepared – one was for bacteria, made using tryptic soy agar (BD 236950); the other was for fungi, made using YM agar (BD 271210). A circular film was placed on each of the plates before uniformly coating with microbial solution. The plates were incubated at 37°C for 24 h and the diameter of the microbial inhibition zone was measured. The following test strains were used: Escherichia coli ATCC 25922 (a Gram-negative bacterium), Staphylococcus aureus ATCC 25923 (a Gram-positive bacterium), and Candida albicans ATCC 10231 (a fungus). The diameter of the inhibition zone was calculated:
where H was the diameter of the antimicrobial ring (in mm), and h was the diameter of the film sample (mm).
In vivo wound healing
The full-thickness excision wound model 16 was constructed to observe wound healing. Six rabbits (three male and three female, 3-months-old, weighing 2.5–2.75 kg, obtained from the Experimental Animal Center of Zhejiang Chinese Medical University) were chosen randomly. After hair removal and anesthesia, four 1.5 cm × 1.5 cm full-thickness wounds were created on the dorsum of each rabbit. These wounds were randomly allocated to two groups: the control group and the experimental group. The control group was given a hydrogel dressing without addition of chitosan or ofloxacin. The experimental group was given an ofloxacin-loaded chitosan/PVA hydrogel dressing. The wound size was determined by IPP 6.0 software based on pictures. The rate of wound healing was calculated using equation (4):
where A0 is the area of the initial wound, and At is the open area of the wound at the time of biopsy (on days 3, 7, 10, or 15 in this study).
All animal procedures were in accordance with the guidelines for animal care of the Animal Ethical Committee of Zhejiang University of Technology.
SEM
Hydrogel samples were lyophilized before being shaped using a cold knife (−150°C). Then the samples were observed and photographed using a Hitachi S-4700 scanning electron microscope (Japan). For morphological characterization, hydrogels swelled to an equilibrium state in water were freeze-dried at −52°C for 24 h (Christ, Germany, Alpha 1–2). A cold knife was used to cut the freeze-dried samples, obtaining transverse sections. Samples were then examined using a JEOL JSM-5400 scanning electron microscope (JEOL, Tokyo, Japan).
Statistical analysis
One-way analysis of variance was used to evaluate the statistical significance of the wound healing results. The significance level was 0.05, and differences among the groups were evaluated for significance by comparing the probability (p) of the null hypothesis with this level.
Results and discussion
Swelling
The swelling percentage of hydrogels made using different N-glycosylated chitosan/PVA ratios is shown in Figure 1. An increase of swelling was observed with the increase of the proportion of N-glycosylated chitosan in the hydrogel. Among the five groups, the swelling percentage was highest when the CS/PVA volume ratio in the hydrogel was 3:1, followed by 2:1.

Swelling of chitosan (CS)/polyvinyl alcohol (PVA) hydrogels with different volume ratios of CS to PVA.
Swelling is an important indicator of the hydrogel dressing properties. 17 Dressings with good hygroscopicity can effectively absorb exudate from a wound, avoiding the infiltration of exudate into healthy tissue and protecting the surrounding tissue. Meanwhile, wet dressings can effectively clean the wound and ease pain during healing.
Water evaporation rate
In the present study, a common characteristic of the hydrogels, irrespective of the chitosan/PVA volume ratio, was that the majority of water evaporation occurred in the first 12 h, by which time the evaporation rate was about 60% (Figure 2). Equilibrium was reached by ~24 h. The water vapor evaporation rate of each group was <90% after 24 h, which indicated that the hydrogels could lock trace water in their structure and provide a favorable wetting environment for wound healing. When the CS/PVA volume ratio was 3:1, the water evaporation rate at 24 h was the highest; the water evaporation rate of the 2:1 v/v group was less by 5.60% than that of the 3:1 group.

Water evaporation rate of CS/PVA hydrogels with different volume ratios of CS to PVA.
A humid environment is conducive to wound healing. The lower the water evaporation rate of a hydrogel dressing, the better it is able to maintain a humid environment. Hydrogel dressings have a lower rate of water evaporation than other dressings. Clinically, this can reduce the replacement frequency of wound dressings and accelerate wound healing.
It was found that the hydrogels with CS/PVA volume ratio 2:1 had both a high swelling percentage and a lower water evaporation rate than gels with a 3:1 volume ratio. Thus, hydrogels with CS/PVA volume ratio 2:1 were chosen for further performance studies.
FTIR analysis
FTIR spectra of N-glycosylated chitosan (a) and the complex hydrogel (with CS/PVA volume ratio 2:1) (b) are presented in Figure 3. –OH peaks appear at 3049.2 and 3142.3 cm−1, indicating that after the addition of PVA, hydrogen bonds formed between the molecules. Peaks at 1420.5 and 1324.2 cm−1 in spectrum (a) are deformation peaks of chitosan CH2 and C–H bending vibration. After PVA was added, the peak at 1420.5 cm−1 in spectrum (a) moved to 1405.4 cm−1 in spectrum (b). There is a d-NH2 vibration peak at 1539.9–1638.7 cm−1, which is obviously weakened in spectrum (b).

Fourier-transform infrared (FTIR) spectra of: (a) N-glycosylated chitosan and (b) complex hydrogel (CS/PVA = 2:1 v/v).
When the hydrogel was loaded with 5.0% ofloxacin (Figure 4), the peak positions were significantly different from those for the non-drug-loaded hydrogel. After drug-loading, peaks characteristic of ofloxacin were observed at 1442.9, 1621.3, 1710.2, 2789.3, and 3310.7 cm−1 18. The peak at 1442.9 cm−1 represented the methyl group of the benzoxazine ring; that at 1710.2 cm−1 was attributed to C=O stretching of a –COOH group; the peak at 1621.3 cm−1 was ascribed to the quinolone amide group; and the absorption bands at 2789.3 and 3310.7 cm−1 were from stretching of methyl and hydroxyl groups, respectively.

FTIR spectra of 5.0% ofloxacin-loaded complex hydrogel.
In vitro drug releasing by the hydrogel dressings
For the ofloxacin standard curve in the range 1.0–10.0 μg mL−1, the linear regression equation was A = 0.0821C + 0.0264 (R2 = 0.9993; A is absorbance at 295 nm, C is concentration) (Figure 5).

Ofloxacin standard curve.
A hydrogel with a CS/PVA volume ratio of 2:1 was used for drug loading. Hydrogel samples with ofloxacin loadings of 2.5%, 5.0%, and 10.0% were prepared. The sustained release of ofloxacin in phosphate buffer (pH 7.4) at 37°C was investigated (Figure 6).

In vitro sustained release of ofloxacin from chitosan/PVA hydrogels.
For each sample, the release rate of ofloxacin was faster in the early stages of release. However it decreases over time, eventually reaching an equilibrium. After 20 h, the ofloxacin release rate of the hydrogels was 88.12%, 92.09%, and 67.65% for the 2.5%-, 5.0%-, and 10.0%-loaded gels respectively; after 50 h, the values were 89.76%, 99.44%, and 91.64% respectively. Thus the 5.0% ofloxacin loaded hydrogel achieved complete release of the drug by 50 h, but the 2.5% and 10.0% ofloxacin loaded hydrogels still had a small amount of drug embedded in the hydrogel structure at this time. Presumably, that in the 2.5% group was difficult to release because it was locked in the hydrogel structure, while in 10.0% group, ofloxacin release had not yet reached equilibrium. Indeed, in the 10.0% loading group, the ofloxacin release reached 98.63% at 62 h, then did not increase further. Since the 5.0%-loaded hydrogel reached complete drug release within 50 h, while the 10.0%-loaded hydrogel did not achieve complete drug release until 62 h, the 5.0%-loaded hydrogel was chosen as it was more suitable for the treatment of wounds.
In vitro antimicrobial activity
The results are shown in Table 1. The 5.0% ofloxacin-loaded hydrogels had excellent antimicrobial properties against the three tested microbial strains. The best inhibition effect was against E. coli.
Inhibition zones of each sample against studied strains.

E.coli: Escherichia coli; S.aureus.: Staphylococcus aureus; C.albicans.: Candida albicans.
In vivo wound healing test
The 5.0% ofloxacin loaded hydrogel with a CS/PVA volume ratio 2:1 was used in this test. All animals were in good condition after the operation and their activity was normal. All the wounds showed bleeding, exudation, and surrounding tissue edema. The healing time of the experimental group and the control group was (13.63 ± 0.53) and (16.25 ± 0.62) days, respectively. The wounds in the experimental group began to scab on the third day after operation. In general, the wounds were wet and clean. On the seventh day after operation, the wound areas were decreased in both the experimental and control groups, the wounds were moist and clean, and granulation was apparent. The surface of the newly formed epithelium was thickened. On the 10th day after operation, the wound areas in the experimental and control groups were significantly reduced compared with day 7, but the stratum corneum of the former group was thinner than that of the latter group. On the 15th day after operation, the wounds in the experimental group had essentially healed, while a small wound was still apparent in the control group (Figure 7). The wound healing rates are shown in Table 2.

Macroscopic observations of rabbit skin wound healing.
Comparison of wound healing rates between the control and experimental groups.

p < 0.05 compared with control group.
SEM
Hydrogels were expanded to equilibrium in water and lyophilized at −52°C for 24 h (Christ, Germany, Alpha 1-2). Cut the freeze-dried sample with a cold knife to obtain a cross section. Then, the samples were examined with JEOL JSM-5400 scanning electron microscope (JEOL, Tokyo, Japan). As shown in Figure 8, the internal structures of all the hydrogels were noncontinuous pores; that is, the prepared hydrogels had a good three-dimensional network structure. It can be seen from Figure 8(a) that the surface pore distribution of CS/PVA 2:1 v/v hydrogel material was not uniform and the surface roughness varied. The morphology of CS/PVA composite hydrogel loaded with 5.0% ofloxacin is shown in Figure 8(b). The addition of ofloxacin did not change the three-dimensional network structure of the composite hydrogel. However, the number of pores increased, and the size of the pores decreased. This may be due to dissolution of ofloxacin in the pores of the hydrogel. Strong hydrogen bonds were formed between ofloxacin and the hydrogel, which made the crosslinked structure of the composite hydrogel more compact.

Scanning electron microscopy of: (a) CS/PVA (2:1 v/v) hydrogel and (b) 5.0% ofloxacin-loaded CS/PVA hydrogel after freeze-drying.
Conclusions
Composite hydrogels were prepared by using CS and PVA as raw materials. When the volume ratio of CS to PVA was 2:1, the composite hydrogel had a good swelling ratio and water evaporation rate. Hydrogel with CS/PVA volume ratio 2:1 loaded with 5.0% ofloxacin achieved complete release of the drug within 50 h and was suitable for the treatment of trauma and burns. The ofloxacin-loaded hydrogel had good antimicrobial properties that could aid wound healing. Meanwhile the hydrogel created a moist environment for wounds, which also promoted wound healing. The blended hydrogel has potential as a dressing for treatment of trauma and burns, and for application in drug release.
Footnotes
Acknowledgements
Data availability
The raw/processed data required to reproduce these findings cannot be shared at this time due to technical or time limitations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank Zhejiang Foundation Public Welfare Research Project (authorization no. LGF19B060006) for financial support.