Abstract
Objective:
In this study, autologous bone grafts using bone-fixing nails made of magnesium-zinc-calcium ternary alloys were performed using rabbit skulls.
Material and methods:
Two types of nails for bone fixation were prepared: 2.5 mm width, 3 mm length and 2.5 mm width, 2 mm length. A disk-shaped bone with a diameter of 5 mm was resected from the parietal bone and fixed with a 3 mm long nail. As a control group, a 2 mm long nail was driven into the existing bone. The rabbits were sacrificed at 1, 4, 12, and 24 weeks after surgery. The resected samples were observed with micro X-ray CT, and embedded in methyl methacrylate to prepare non-decalcified specimens. The in vivo localization of elements was examined using energy-dispersive X-ray spectroscopy (EDS).
Results:
Micro X-ray CT images of samples showed volume reduction due to degradation in both the bone graft and control groups. No significant difference in the amount of degradation between the two groups was observed, however characteristic degradation processes were observed in each group. The samples stained with alizarin red S showed amorphous areas around the nails, which were considered as corrosion products and contacted directly with the newly formed bones. EDS analysis showed that corrosion products were mainly composed of magnesium and oxygen at an early stage, while calcium and phosphorus were detected on the surface layer during the long-term observation.
Conclusions:
The degradation speed of the magnesium alloy nails varied depending on the shapes of the nails and surrounding tissue conditions. A calcium phosphate layer was formed on the surface of magnesium alloy nails, suggesting that the degradation rate of the nail was slow.
Keywords
Introduction
Inert titanium (Ti)-based alloys are commonly used in the treatment of maxillofacial fractures and in grafting bones. However, such permanent fixation devices may require removal surgery, which increases patient burden and medical costs. 1
Resorbable bone fixation devices that mainly comprise polymers such as poly-L-lactic acid (PLLA), have been clinically used since the 1980s. 2 Materials with sufficient mechanical strength are often used for intraoral reconstruction, such as alveolar bone regeneration. 3 Calcium phosphate can strengthen polymeric bone fixation devices, 4 however, their mechanical strength is still lower than that of native bone, limiting their clinical applications. 5 These devices can also induce a persistent inflammatory cascade3,4,6 when absorbed by the body. 7
Although magnesium (Mg) alloys are degraded in vivo, they may be able to overcome the challenges posed by common bone fixation devices. Biodegradable Mg-based devices have shown promise in their clinical applications, such as in cardiovascular surgery (stents) and orthopedics (cruciate ligament reconstruction).8-11 A previous study indicated that Mg-based implants are osteoconductive in vivo. 12 Moreover, the tunability of Mg-based devices: is particularly promising as their mechanical properties and rate of biodegradation can be adjusted by modifying their alloy compositions and processing methods.13,14 This allows the application of Mg alloys in a wide range of clinical applications.
Mg-based bone fixation devices are rapidly degraded via hydrolysis in vivo. Alloying Mg with biocompatible elements and coating its surface can improve its mechanical and anti-corrosion properties. 15 However, the mechanism underlying the biodegradation of this material is unclear 16 ; further studies are needed to investigate the degradation of these devices over a long period of time.
Although Mg can be used to reconstruct jawbones during head and neck surgery,17,18 only few clinical applications of Mg-based devices have been reported. Herein this study, we used Mg alloy nails as a bone fixation device in autologous alveolar bone grafting and subsequently performed block bone grafting in rabbit skulls using pure Mg alloy nails. The color mapping images of the corrosion products formed on the surface of the degraded Mg-based devices in vivo helped in determining the process of their formation, thus enabling the formation of future unique designs to modulate degradation rates. 19
Materials and methods
Preparation of Mg alloy nails
Mg alloy devices made of Mg–zinc–calcium (Mg–Zn–Ca) ternary alloys with high mechanical strength were developed at the Graduate School of Engineering, Kobe University.
The cast alloy was fabricated via direct-chill casting of pure Mg (99.98% purity) with master binary alloys of Mg–6%Zn and Mg–6%Ca. After solution treatment at 773 K for 24 h, a cylindrical billet with a 90-mm diameter was machined. The chemical composition of the alloy was confirmed cdas Mg-0.21at% Zn-0.09at% Ca-0.004at% Al-0.001at% Fe-0.003at% Si via inductively coupled plasma (ICP).
The billet was first extruded at 723 K, with an extrusion ratio of 14:1. The extrusion bar was machined into a cylindrical billet with a 20-mm diameter. This billet was again extruded at 523 K, with an extrusion ratio of 25:1. Final extruded shape of the Mg–Zn–Ca alloy was a cylinder of 4-mm diameter. This cylinder was formed into arbitrary nail shapes via lathe machining (Figure 1(a) and (b)).

(a) A long nail. (b) A short nail. Scale bar = 1 mm. (c) The cross-sectional schematic drawing of animal experimental area. Right: A long nail used for fixing the grafted bone. Left: The control area where the short nail was implanted. PB: The parietal bone. GB: The grafted bone. (d) Surgical procedures. 1: The exposed surface of the skull. 2: After the graft was resected from the left parietal bone. 3: The grafted bone fixed by the long nail on the right parietal bone. 4: The defect of the left parietal bone was covered with an absorbable membrane. White circle dotted line: The short nail was fixed on the left existing bone as the control group. Scale bars = 5 mm.
Characterization of Mg alloy nails
The image quality (IQ) map and inverse pole figure (IPF) generated using electron backscatter diffraction (EBSD) were used to analyze the crystal grain size and orientation of the nail. Compression tests were performed to ensure that the nails were strong enough to penetrate bone without buckling or fracture. Nails with a diameter of 4 mm and a height of 8 mm were compressed along their axis at a strain rate of 10−3/s−1.
Animal experiments
Examination and approval for animal experiments
To compare the degradation of Mg nails in the autogenous bone graft and the existing bone (living bone) groups, rabbit skulls were used to simulate bone augmentation with autologous bone grafts. All experiments were approved by the Animal Experiment Technical Committee (2015DnA-065) and performed in accordance with Regulations for Animal Experiments and Related Activities at Tohoku University (Sendai, Japan).
Autologous bone graft models
Twelve male Japanese white rabbits (aged 14–16 weeks; weight range, 3.10–3.35 kg) were used in the experiments. The rabbits were anesthetized with a gas mixture of 8% isoflurane and 6 L oxygen, and 39 mg pentobarbital Na was delivered through the ear vein. Thereafter, the concentration of isoflurane was adjusted to 5%−6%, and the oxygen flow rate was adjusted as required by intraoperatively monitoring the animals’ peripheral temperatures of the rabbits during the surgery. Local anesthesia with 3.6 mL of 2% lidocaine containing 1:10,000 epinephrine was injected in the cranial periosteum.
All rabbits underwent the same surgical procedures. The head was incised to expose the parietal bone, and a circle-shaped free bone flap (autologous bone fragment) was resected by drilling the left parietal bone up to the dura mater using a 5-mm-diameter trephine bar. A nail hole of 1-mm diameter was drilled in the center of the resected autologous bone and the resected bone was fixed to the right part of the parietal bone with the Mg alloy nail with a length of 3 mm (bone graft group) (Figure 1(c)). Bone lacunae and blood flow were observed in the parietal bone. A PGLA membrane (GC membrane) was prepared (8 mm × 8 mm) and was used to cover the resected area of the left part of the parietal bone to minimize the blood flow effect from the defect. For the control group, a 2-mm Mg alloy nail was inserted into the left part of the parietal bone away from the bone defect (control) (Figure 1(d)). In this study, cortical bone perforation was not performed as significant bleeding was observed from the cortical bone surface.
The surgical site was thoroughly washed with saline, and the skin was closed and sutured.
Observation period
A total of 12 rabbits were randomized into four groups (n = 3 per group). According to preliminary experiments and previous reports,20–22 the skull experimental areas were resected and analyzed at 1, 4, 12, and 24 weeks postoperatively (three rabbits/week). The animals were sacrificed by injecting approximately 800 mg of pentobarbital into the ear vein. After thoracotomy, the descending aorta was ligated, and 4% paraformaldehyde was injected to fix the tissue.
Microcomputed topography imaging and morphometry
The resected areas were cut out as small as possible and immediately scanned by microcomputed topography (micro-CT) (Comscantecno, ScanXmate-E090). The scanning conditions were set at a current of 70 mA, a voltage of 60 kV, and a voxel size of 0.03 mm3. The volumes of the Mg alloy nails were measured through reconstructed 3D images using CT image analysis software (Ratok TRI/3D-BON), and the degradation rate per week during the observation period was calculated. Statistical analyses of the changes in the measured nail volumes were performed via two-way analysis of variance and the Tukey multiple comparison test (ATMS Stat Mate ver. 5.01).
Preparation of resin-embedded specimens and histopathological observation
After micro-CT scanning, all of the parietal bones were divided into two parts – right (the bone graft group) and left (control group) – and soaked in 100% alcohol for 48 h. The samples were then dehydrated, soaked in lemosol, and embedded in methyl methacrylate (MMA). Embedded samples were sliced into 500-µm thick sections (Leica SP-1800) with the long axis parallel to the nails and then adhered to 1.5 mm thick acrylic slides. All embedded samples were manually polished to a thickness of approximately 100 µm.
Polished samples were stained with alizarin red S and the morphology of corrosion products around the nails and Ca deposition was observed. Alizarin red S reagent (FUJIFILM Wako Pure Chemical Corp.) was mixed with 28% ammonium hydroxide at a pH of 6.36–6.40; this solution was added dropwise to the washed samples for 5 min. The stained samples were stored as digital images using a virtual slide system (Leica Scan Scope CS2).
Scanning electron microscopy/energy dispersive spectroscopy (SEM/EDS) analysis
The alizarin-stained layers of the resin-embedded specimens were removed by polishing. Next, a thin carbon film was deposited on each specimen surface via vacuum evaporation using an auto carbon coater (JEOL JEC-560). The specimen features were analyzed by scanning electron microscopy and energy dispersive spectroscopy (SEM/EDS) (JSM-6390LA; JEOL, TOKYO, JAPAN). EDS analysis was performed with an acceleration voltage of 15 kV, a working distance of 10 mm, and a spot size of 70. Element localization at the interfaces of the Mg alloy nails were mapped and observed.
Results
Intra- and postoperative follow-ups
The nails were not damaged during or after surgery. The rabbits did not experience any postoperative complications associated with the use of Mg alloy nails. No long-term local infection or inflammation was observed in the skulls after wound closure.
Characteristics of Mg–Zn–Ca ternary alloys
The IQ map showed some second-phase particles as the concentrations of Zn and Ca in the Mg ternary alloy were present in diluted concentrations. The IPF map indicated that the alloys had homogeneous structures of various orientations (Figure 2(a)). The average grain size was approximately 1.1 µm.

(a) IQ (image quality) map (left) and IPF (inverse pole figure) map (right) of a cross section in a nail specimen observed by SEM/EBSD. The arrow indicates the axial direction (A.D.) of the nail. (b) The Nominal stress-strain curve by compression tests. The specimens were Mg-0.2at.%Zn-0.1at.%Ca alloy having the diameter of 4 mm and height of 8 mm. The present alloy’s average grain size was measured to be 1.1 µm (Red line), and the control alloy’s was measured to be 6.7 µm (Blue line) by SEM/EBSD.
The yield stress and fracture strength of the experimental Mg ternary alloy were determined to be 311 and 625 MPa, respectively, under compression. As a control, another extruded Mg-0.2at.%Zn-0.1at.%Ca was compressed, whose average grain size was determined to be 6.7 µm via SEM/EBSD.
The nominal stress–strain relation of the Mg–Zn–Ca alloy was shown in the same figure to compare the yield strength in compression for a cylindrical specimen (Figure 2(b)).
Direct comparison of the compressive yield stress (σcys) revealed that the experimental Mg–Zn–Ca alloy was approximately 50% stronger than the control Mg–Zn–Ca alloy and comparable in strength to commercially available Grade II pure titanium (σys: ~275 MPa). Therefore, the former was strong enough to be used as a nail to guide bone regeneration. The superior strength of the Mg–Zn–Ca alloy was achieved by refining the grain size and alloying of trace Ca and Zn. We examined the mechanical properties in compression to focus on the device’s ability to withstand deformation when driven. The compressive yield stress of an extruded Mg alloy is typically lower than the tensile yield stress due to the crystallographic texture formed during extrusion. 23 The tensile yield stress of the present Mg–Zn–Ca alloy was also higher than the compressive yield stress due to the extrusion texture. This suggests that the alloy nail has a fixation force similar to that of pure titanium nails in terms of strength.
Morphological observation by micro-CT
Micro-CT images and histopathology results showed that the grafted bones were fixed well in all specimens without detachment of the Mg alloy nails. These nails showed corrosion products approximately 1 week postoperatively. Most of the nail bodies degraded completely after six postoperative months, though they almost completely degraded within 3 months (Figure 3).

The µ-CT images. (a) The three-dimensional (3D) image of bone graft at 1 week post-surgery. (b) The cross-sectional µ-µ-CT images of the bone graft group at 1, 4, 12, and 24 weeks post-surgery. (c) µ-CT images of the control group taken at 1, 4, 12, and 24 weeks post-surgery. Scale bar = 1 mm. (d) The images of extracted only Mg nails from the original image and reconstructed in 3D.
Changes in the volumes of Mg alloy nails
The changes in the volume of the degraded Mg alloy nails as well as the ratio of their volume to the total volume (residual volume ratio) are presented in Figure 4. The volumes of the nails in the two groups decreased rapidly from 1 to 4 weeks postoperatively. Their volumes did not change significantly between 4 and 12 weeks postoperatively but changed during 12–24 weeks after the procedure postoperatively (Figure 4(a)). The residual volume ratio was approximately 80% at postoperative 1 week, approximately 60% at weeks 4 and 12, and approximately 10% and 40% in the control and bone graft groups, respectively, at 24 weeks postoperatively (Figure 4(b)). The bone graft and control groups degraded similarly, according to their residual volume ratios. Both groups deteriorated by approximately 20%. The degradation rate decreased significantly from 1 to 4 weeks (p < 0.01), and the degradation rate during 4–24 weeks postoperatively was maintained within 5% (Figure 4(c)). There was no significant difference between the two groups.

(a) The changes in the nail volumes. (b) The changes in the ratio of the residual volumes of magnesium alloy nails to total volumes of the nails before disassembly (%). (c) The degradation rate per week (%).
Histopathology results
Control group
The nail bodies degraded uniformly at 1 week postoperatively. No bone resorption was observed in the surrounding bones (Figure 5(a)). Most nail bodies degraded at 4 weeks postoperatively, leaving only the nail heads undegraded (Figure 5(b)). The shapes of the nail heads did not change within the first 12 postoperative weeks (Figure 5(c)), but after 24 weeks the peripheral areas of the nail heads degraded with gas cavities (Figure 5(d)).

Non-decalcified specimens stained with alizarin red S. (a–d) Histopathological findings of the control group. (a) 1 week. (b) 4 weeks. (c) 12 weeks. (d) 24 weeks. (e and f) Histopathological findings in the bone graft group. (e) 1 week. (f) 4 weeks. (g) 12 weeks. (h) 24 weeks. GB: The grafted bone. EB: The existing bone. Scale bar = 1 mm.
Bone graft group
No obvious degradation was seen on the head surfaces of Mg alloy nails (Figure 5(e)). The grafted and existing bones partially united at 4 weeks postoperatively; however, localized gas intrusion into bone tissue was also observed (Figure 5(f)). At 12 weeks postoperatively, the nail body thinned and was absorbed by surrounding tissue due to deterioration. (Figure 5(g)). The entire nail bodies degraded by 24 weeks postoperatively. In addition, the bones around the remaining nails and the grafted bones were absorbed. The surfaces of the nail heads showed irregular degradation (Figure 5(h)).
Gas pockets were observed in both groups. Few inflammatory cells infiltrated the specimens, which agreed with the few postoperative complications experienced by the rabbits post-surgery but only few inflammatory cells were observed in the specimens.
Observation of nail surfaces
Figure 6 presents magnified images of the degraded areas of the nail surfaces. The degraded areas and the remaining nails were distinguishable in the specimens stained with alizarin red S. Amorphous hyaline appearances were seen in the degraded areas of the nail surfaces (Figure 6(a)); this was more noticeably in the control group. Spot-like degradation was observed in the corners of the nail heads and bodies, and the degraded areas appeared amorphous and hyaline-like (Figure 6(b)). The apexes of the nail bodies degraded with uniform thickness from the original outer shapes of the nails (Figure 6(c)). Some parts of the degraded areas were not stained and apparently distinguished from the existing nails (Figure 6(c)). Other apexes of the nails showed layers of corrosion products with irregular boundaries, which were in direct contact with the newly formed bones. Gas cavities around the corrosion product layers were also observed (Figure 6(d)).
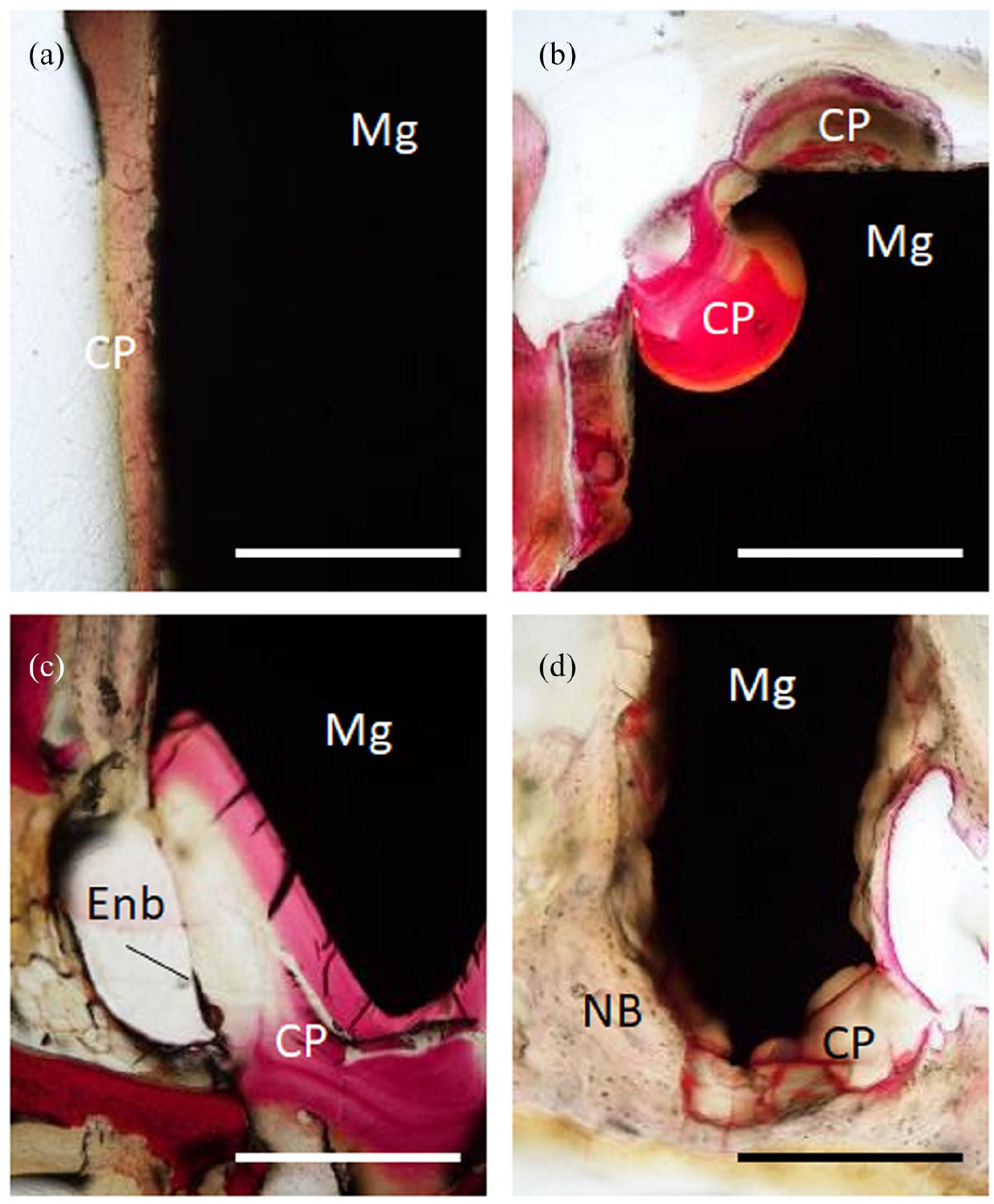
Histopathological findings of nail degradation in the bone graft group. Non-decalcified specimens stained with alizarin red S. (a) The thin layer of corrosion products formed on the nail surface. Scale bars = 500 µm. (b) The degradation of the nail head. Scale bars = 500 µm. (c) Unstained areas indicate corrosion products formed on the nail apex. Scale bars = 500 µm. Enb: The border between the existing nail and surrounding bone. (d) The lateral degradation of the nail body and formation of corrosion products. Scale bar = 1 mm. CP: Corrosion products. NB: The new bone.
Degradation patterns based on micro-CT and histopathological findings
Micro-CT and histopathology results revealed the degradation patterns of the nails in both groups (Figure 7). In the bone graft group, many of the nail bodies degraded laterally, becoming and simultaneously became thinner and shorter simultaneously. Moreover, the projecting areas of the nails tended to progressively degrade, and the nail body apexes were considered corrosive because they appeared transparent under micro-CT, showing vertical degradation. The nail heads remained for a longer period compared with the nail body, but eventually their also degraded. Similarly, the nail bodies surrounded by the existing bones in the control group rapidly degraded vertically, followed later by the nail heads.

The schematic diagram of nail degradation. (a) Lateral degradation. (b) Vertical degradation.
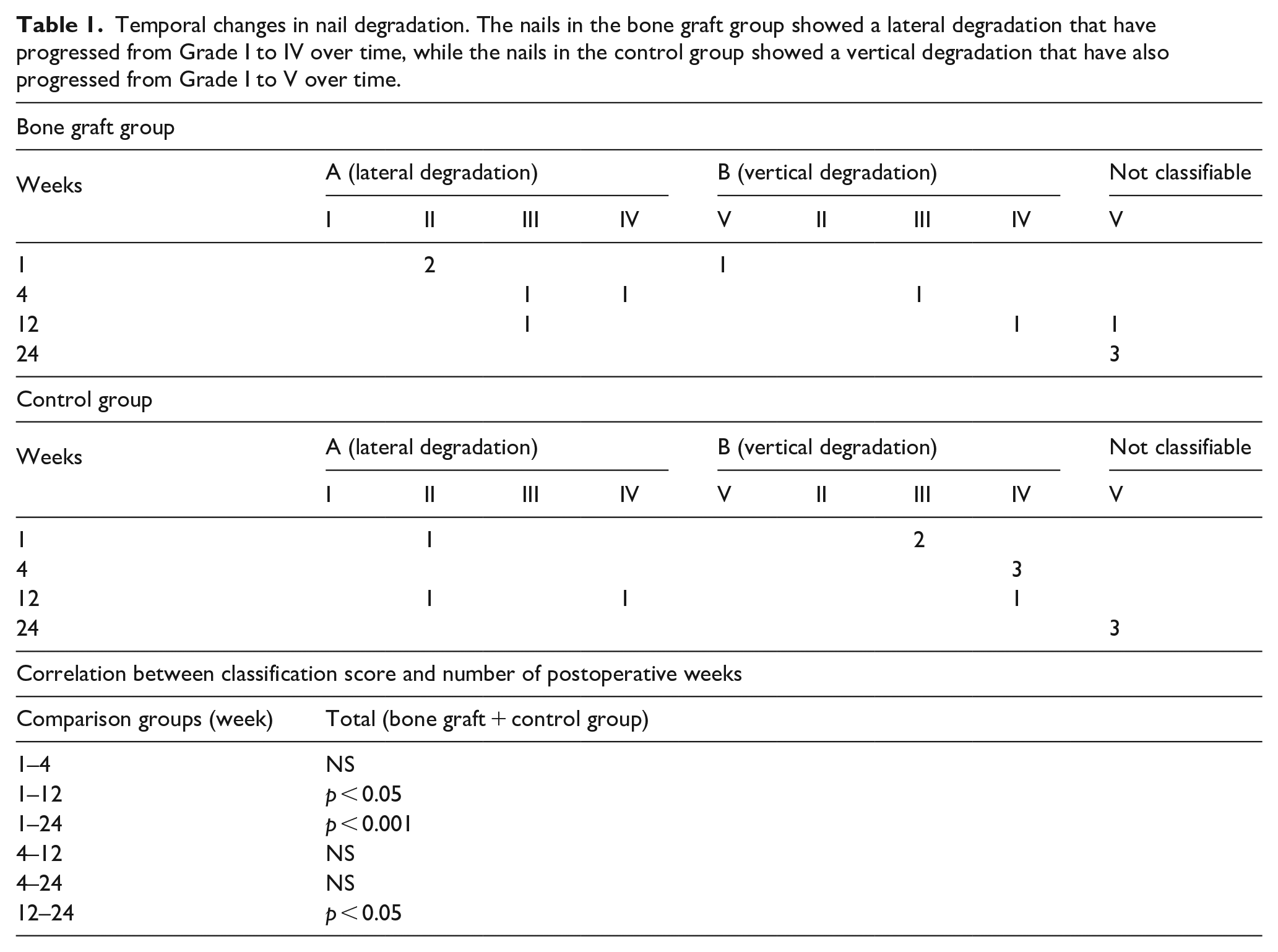
Table 1 shows the relationships between the number of cases, degradation types, and time. In the bone graft group, lateral degradation was seen in five cases, and vertical degradation was seen in three cases. In the control group, lateral degradation was seen in three cases, and vertical degradation was seen in six cases. The degree of degradation changed from I to V over time as shown in Figure 7 and Table 1. At 24 weeks postoperatively, all nail bodies completely degraded and were considered unclassifiable. Regardless of degradation pattern – vertical or lateral–the nail heads remained. The Newman–Keuls test was used to determine the correlation between the number of postoperative weeks and the classification score. Overall there was a significant difference between 1 and 12 weeks (p < 0.05), 1 and 24 weeks (p < 0.001), and 12 and 24 weeks (p < 0.05) postoperatively.
Temporal changes in nail degradation. The nails in the bone graft group showed a lateral degradation that have progressed from Grade I to IV over time, while the nails in the control group showed a vertical degradation that have also progressed from Grade I to V over time.
Results of EDS
The SEM and EDS mappings show the vertical degradation of the nails in the control group at 1 week postoperatively (Figure 8(a)). Mg and O were detected at the lateral sides of the nail head and body, whereas Ca and P were slightly detected at the surface layers of the nail.

(a) Elemental mapping images of the vertical degradation of the nails at 1 week post-surgery. The nail body (Nb), the nail head top (Nht), and the the nail head side (Nhs) degraded. Mg, O, Ca, and P were detected in corrosion around the nails. Cl: The corrosion product layer on the nail surface (Cl). (b) Elemental mapping images of the vertical degradation of the nails at 12 weeks post-surgery. The nail bodies and head degraded mostly degraded. Mg, O, P, and Ca were detected. Scale bar = 1 mm.
Figure 8(b) shows mapping images of same degradation pattern at 12 weeks postoperatively. Various concentrations of Mg, Ca, O, and P were detected on the surface layer of the deteriorated areas, whereas Ca and P were more prominent here than at 1 week postoperatively.
Figure 9(a) shows the lateral degradation of the nails in the bone graft group at 1 week postoperatively. Ca, O, and P were detected in the nail heads and body apexes and the parts of the nail bodies, specifically the area facing the gaps between the existing bones and grafted bones (the spot-like degraded area as shown by * in Figure 9(a)).

Elemental mapping images of the lateral degradation of the nails. (a–c) 1 week post-surgery. (d–f) 12 weeks post-surgery. (a, d) Overall view of the nail and localization of each element. The higher the amount of a specific elements, the higher its brightness. A color mapping image was created in the white square area of the EDS image to visualize the distribution of each element. The border of the grafted bone and existing bone. The nail body has deteriorated significantly (a*). GB: The grafted bone. EB: The existing bone. Nht: The nail head top. Nbt: The nail body tip. Scale bar = 1 mm. (b) the color mapping image at 1 week post-surgery. Mg (red), O (green), and C (blue). The Mg and O corrosion layer (Cor). The decrease of elements concentration in corrosion can be observed as a gradation (Δ). (c) Same image as (b) with a different coloring. Mg (green), Ca (red), and P (blue). There is very little uptake of Ca and P elements in the same area (Δ). (e) The color mapping image at 12 week post-surgery. Mg (red), O (green), and C (blue). Scale bar = 50 µm. (f) Same image as (e) with a different coloring. Mg (green), Ca (red), and P (blue). A Purple thin layer is formed on the surface of corrosion (Ca + P). Scale bar = 50 µm.
Superimposed images of Mg, O, and C by color mapping (Figure 9(b)) show 70-µm thick corrosion products layers, mainly including Mg and O colored in yellowish green. The element concentrations in the corrosion products layers were not uniform but relatively lower in outer layers. A blue color gradation in the outer layer of corrosion products means lower Mg and O but higher C. Superimposed images of color-mapped Mg, Ca, and P in the same area showed slight uptake of Ca and P, which was consistent with the region in which the concentrations of Mg and O were low (Figure 9(c)).
Figure 9(d) shows the lateral degradation of the nails in the bone graft group at 12 weeks postoperatively. The nail bodies became thinner, and the nail heads partially degraded.
Thin layers containing Ca and P were detected on the surfaces of the nail heads. Corrosion product layers of the nail bodies were thick and contained Mg, Ca, O, and P (Figure 9(d)). Carbon deposits were confirmed in the yellowish green regions of the corrosion products of the nail bodies, which mostly contained Mg and O (Figure 9(e)). The 10-µm purple-colored layers were confirmed as the exterior of the corrosion products and contained mostly Ca and P. (Figure 9(f)).
Discussion
Mechanical stability during bone healing determines the clinical outcomes.24,25 Mg-based bone fixation devices may be able to overcome the challenges posed by conventional Ti-based devices, such as insufficient mechanical strength and biocompatibility. Hase et al. 26 reported that adding aluminum or Zn to a Mg–Ca alloy, which is used as a ternary alloy, can improve the mechanical properties of the Mg alloy. Here, we created a Mg–Zn–Ca ternary alloy to study how it degrades and regenerates bone in rabbit skulls. Compression tests showed that the strength of this Mg alloy matches that of Grade 2 Ti alloy according to the ASTM standard. This Mg alloy nail easily penetrated the bone and fixed the grafted bone. The small Mg device used here degraded within 3–6 months, indicating that similar Mg-based alloys hold promise as new and strong biodegradable bone fixation devices.27–29
Patients with chronic renal failure should avoid excessive Mg intake. 30 However, this Mg alloy is small and degrades slowly, maintaining Mg homeostasis. 22 Histopathology showed the formation of a thick fibrous capsule and slight inflammatory cell infiltration around the Mg alloys, indicating that Mg alloys are likely safe in vivo. 31 No postoperative complications or weight loss was observed the current study. Future work should explore how the human body responds to locally applied Mg-based bone fixation devices.
Human bones heal in approximately 12–24 weeks, during which implant fixation is required. 32 Here, initial fixation seemed necessary for at least 4 weeks postoperatively because bone unions between the grafted bones and existing bones in histopathological findings were observed only after this duration. Although Mg alloys may degrade rapidly in the early postoperative period, a subsequent corrosive layer may inhibit later degradation. The grafted bone did not detach in any of the experimental animals. According to this result, the nail strength to bone fixation was thought to be 12 weeks postoperatively, and it degraded completely at 24 weeks.
The degradation behaviors of the Mg alloy nail bodies can be categorized into two types: lateral degradation, indicating that the bodies degraded from their sides, and vertical degradation, indicating that the bodies degraded from their apexes. The nail bodies completely degraded at approximately 12 weeks postoperatively, but the nail heads remained for a longer time. Sato et al. 31 previously showed that blood flow affects Mg alloy degradation, which can explain here whether the nail heads and bodies interfaced with tissue fluid and blood. Specifically, bone marrow blood flow was thought to degrade the nail bodies in the control group, whereas hemorrhagic blood and tissue fluid in the bone graft group flowed into the interfaces between the existing bones and grafted bones, which may have accelerated the degradation of the side of the nail body. Lateral degradation thinned the nail bodies and generated more gas, which may have damaged the nails.
Convex–concave shaped areas such as corners and apexes of the nails degraded rapidly. These shapes may be attributed to the difficulty of forming corrosion products. The tissues surrounding the nail should be considered when optimizing the function of Mg alloy nails.
This study identified the composition of corrosion products formed as the Mg alloy nails degraded in vivo. Our findings matched those of Rahim et al., 33 who reported that the corrosion products formed on surface-treated Mg devices accumulated the same elements. Our EDS analysis revealed that mixed layers of Mg and O initially formed on the surfaces of the Mg alloy nails, suggesting that corrosion products layers contained MgO or Mg (OH)2. However, the chemical instability of these early corrosion products cannot completely suppress the degradation of Mg-based devices. Eventually, small amounts of Ca and P were gradually observed in the outer layers of corrosion products, on which ion exchanges were presumed. Images at 12 weeks postoperatively suggested that stable Mg phosphate and Ca phosphate may have formed on the outer surface layers of corrosion products. Kraus et al. 34 reported that electrochemically depositing a Ca phosphate coating on the surface of Mg alloy improved bio-affinity, decreased lymphocyte infiltration, and accelerated new bone formations. Song et al. 35 also found that treating a Mg–Zn alloy’s surface with Ca phosphate suppressed its degradation rate and release of hydrogen gas. These may be attributed to corrosion layers containing phosphoric acid compounds on the nail’s surface. Calcium phosphate was released in corrosion product layers adjacent to gas cavities, which may have decreased the degradation rate of Mg-based devices with an irregular supply of blood flow. Controlling Mg’s degradation rate may help form a corrosion-resistant Ca phosphate layer.
We could not prove the superior osteoconductivity because of Mg alloy nails. However, the inflammatory response to hydrolysis of the nail was mild and gas release was also deemed to be temporary during bone remodeling.12,32 The results of this study indicate that understanding the metabolism of tissue fluids that affect gas generation and the effect of device geometry on degradation and gas generation must be studied. Castiglioni et al. 16 suggested that the Mg ions preserved bone homeostasis,29,36,37 but their role in bone metabolism was still unclear. Rahim et al. 33 also reported that an excessive increase in Mg concentration suppressed alkaline phosphatase activity of osteoblasts in vitro. Future studies should study additional factors linked to Mg and bone formation, such as local pH changes, blockage of growth factors due to blood flow disturbances, and baroreceptor stimulation.
Conclusion
This is one of few studies that apply an Mg-based biomedical device in vivo. Mg holds great promise in clinical applications such as dental and oral surgery. The Mg ternary alloy developed here was strong enough to fix bone in rabbit skulls. The chemical composition of its corrosive products revealed the formation of a layer of Ca phosphate that may have slowed biodegradation. Our morphological observations should help design Mg-based devices in future and control their degradation rates in vivo. These results indicated that a Mg alloy device can be clinically used as a biodegradable fixation device in the head and neck area.
Footnotes
Acknowledgements
The authors would like to thank Miho Oikawa from Division of Oral Pathology, Tohoku University Graduate School of Dentistry for the excellent technical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number JP20K18551.
