Abstract
The aim was to prepare experimental adhesive (EA) with ZrO2 nanoparticles (ZEA) and without it; and its interaction with dentin conditioned with photodynamic therapy (PDT), etch and rinse, and Er, Cr: YSGG laser (ECL). Methods consisted of the scanning electron microscope (SEM), energy dispersive X-ray (EDX), shear bond strength (SBS) failure mode, and rheological property analysis. The methods were employed to assess bond integrity; the shape of ZrO2 NPs and interfacial dentin and adhesive interaction. Ninety premolar teeth were collected and pre-bonding, conditioning of dentin was performed utilizing phosphoric acid (H3PO4), photodynamic therapy (PDT), and ER-CR-YSGG laser (ECL). The teeth were set to form bonded specimens using two adhesives EA and ZEA. The microscopic imagery of ZrO2 revealed irregular-shaped, non-edged particles. EDX analysis revealed Zr to be 75.7% whereas, O2 by weight was 25.3%. The highest SBS was demonstrated in dentin conditioned with etch and rinse and ZEA (19.05 ± 0.13 MPa). Whereas, the lowest SBS was observed in dentin surface treated with PDT and EA (12.25 ± 0.14 MPa). Most of the reported failure modes were adhesive. Rheological analysis revealed that with increasing frequencies the viscosities of adhesives were reduced. The presence of 10% ZrO by weight in adhesive improves SBS of restoration and this improvement in bond values is statistically significant compared to EA. Bond values may be influenced by surface treatment of dentin.
Introduction
Composites are considered as one of the most widely used restorative materials due to their exceptional esthetics and better mechanical properties. 1 Adhesion to the dentin is more challenging in contrast to enamel due to less mineralization and higher water content which makes the procedure technique sensitive. 2 The adhesive-hybrid interface plays a major role in composite restoration, retention, and bond strength. 3 Quality of the bond between different adhesives and dentin depends on resin monomers ability to penetrate in inter-collagen fibril spaces and resin tags formation. 4
The literature revealed that loss of bond strength in composite restoration with time is due to nano-leakage at the resin–dentine interface resulting in recurrent caries.5,6 Therefore, during the past few years efforts were made to modify the mechanical properties of dental adhesives which helps in strengthening the bond between dentin and adhesive. 7 Different fillers were added to the resin bonding system to reinforce the adhesive resin and to overcome the adhesion failure overtime. 5 Previous studies by Fanning, 8 and Miyazaki et al., 9 revealed that additive fillers in cooperation with the dentin bonding system reduce water sorption and solubility. It also enhances the mechanical properties of the adhesive thus improving the shear bond strength (SBS) with the dentin. 10
With the ongoing advancement in the field of dentistry, nanotechnology which deals with nano-size particles has gained attention due to its excellent biological properties. 11 Its application is introduced almost in all fields that is, restorative, prosthetics, endodontics, implantology, oral cancers, and periodontology. 12 Nanoparticles (NPs) possess excellent antibacterial, antiviral, and antifungal properties. They also improve the mechanical and physical properties of the adhesive as well as improve the bond between dentin and restorative material. 13 Nowadays, amongst the various type of inorganic fillers zirconium dioxide, ZrO2 has caught the attention of dental researchers. Zirconia being a chemical oxide is bio-inert material and water-insoluble. 14 ZrO2 NPs possess exceptional mechanical, physical, and photocatalytic properties that make them suitable additives for different bonding systems. 15 Furthermore, they are extremely biocompatible and esthetically pleasing due to their opaque and whitish appearance. 16 They also yield better compatibility with the resin monomers and possess high corrosion resistance. 17 However, their effect on the SBS of adhesive resin has not been determined yet.
According to available indexed literature, it was found that data related to the effect of different NPs as an additive in dental adhesives is limited. Similarly, the role of ZrO2 NPs on the mechanical properties, their characterization, and interactions with dentin bonding adhesives has not been determined yet. Therefore, the current study aimed to prepare adhesive with ZrO2 NPs and its interaction with dentin conditioned with photodynamic therapy (PDT); etch and rinse and Er, Cr: YSGG laser (ECL) using scanning electron microscope (SEM), energy dispersive X-ray (EDX), shear bond strength (SBS), and failure mode. It is hypothesized that the addition of ZrO2 NPs displayed better SBS to adhesive resin when compared to the adhesive without ZrO2 filler.
Material and methods
Synthesis of adhesives
ZrO2 NPs were purchased commercially (Sigma Aldrich, Cassopolis, MI). The preparation technique of adhesive was adopted from already reported work by Ye et al. 18 The preparation of adhesive consisted of 25% triethylene glycol dimethacrylate (TEGDMA), 50% bisphenol A glycol dimethacrylate (BISGMA), and 25% 2-hydroxyethyl methacrylate (HEMA) by weight, fused in 30% ethyl alcohol acting as a solvent. As an electron initiator diphenyliodonium hexafluorophosphate (1.0% n/n) was used along with 0.5% (n/n) ethyl 4-dimethylamino benzoate and 0.5% camphor-quinone photo-initiators. A magnetic stirrer with a condenser (Magnetic stirring hotplates MR, Hei-End, Buch Holm, Denmark) and three-necked flasks were used for mixture preparation to avoid photopolymerization. The adhesive was stored in a black container shielded with foil. The adhesive prepared was considered as control adhesive (EA).
To improve the adhesion of ZrO2 NPs to a matrix, commercially obtained ZrO2 NPs were salinized using 5% silane (Methyltrimethoxysilane, 97%, ACROS Organics, Fisher Scientific, New York, USA) added to 95% (acetone). To prepare ZrO2 experimental adhesive (ZEA) 10 wt % salinized ZrO2 NPs were added in EA. The weight of ZrO2 NP was measured in milligram (mg) and resin volume in milliliter (ml).
For homogeneity of ZrO2 NP in EA, dispersion of ZrO2 NPs was completed using sonication in a centrifuge for 10 min. Both the adhesive that is, EA and ZEA were kept at 37°C to permit evaporation of the solvent. The final adhesives were maintained at a temperature of 4°C and were used within a month due to short shelf life.
Characterization of nanoparticles
SEM (Axia ChemiSEM, Thermofisher Scientific, OR, USA) was employed for the investigation of ZrO2 nanoparticles. For fixation, aluminum stubs were used on which filler NPs were positioned. The ZrO2 NPs were sputter-coated with gold (Luxor, benchtop sputter coater Sheffield). At different magnification, micrographs were taken by a single investigator operated with an accelerating voltage of 20 kV. To investigate different elements EDX spectroscopy was also performed.
Tooth preparation, surface treatment, and bonding procedure
Disinfection of 90 premolar teeth was performed with chloramine trihydrate solution (Merck, Mumbai, India) for a duration of 48 h. Using polyvinyl pipes (3 mm) filled with heat cure acrylic resin all teeth samples were mounted up to a cement-enamel junction and stored in distilled water until further use. For SEM imaging three pairs of specimens were used. Whereas, SBS testing was performed on 10 specimens from each group following failure mode evaluation. Sample calculation was based on previous studies,23–29 a power analysis using G*Power 3.1 (Heinrich Heine University, Düsseldorf, Germany) indicated that a minimum sample size of nine for each group ensured a power of 80% to detect significant differences at a 0.05 significance level. Thus, 15 samples were used for each group. By removing occlusal enamel, dentin was exposed using a low-speed diamond wheel saw (Model 660, South Bay Technology) just 1 mm below dentinal enamel junction (DEJ). Based on the surface treatment of dentin structure specimens were divided into three groups.
Group 1: Etch and rinse: 10-s dentin etch with 35% phosphoric acid gel followed by a wash with distilled water and drying with cotton pellets.
Group 2: Photodynamic therapy (PDT): Methylene blue photosensitizer (MBP) at 50 mg/l was applied on dentin structure for 60 s activated by diode laser (SIRO-Laser Advance, Sirona) having a wavelength of 638 nm, 30 Hz frequency, and 2 W power for 30 s washed and air-dried.
Group 3: Er, Cr: YSGG (ECL): Dentinal surface of the specimen was photosensitized using ECL (Biolase-Waterlase I-Plus) in a non-contact circular motion at a distance of 2 mm under 60% water and 40% air using MZ8 tip in circular non-contact position at power 4.5 W and frequency 30 Hz. a non-contact circular motion for 60 s.
After surface treatment of dentin using different conditioning regimes 10 samples from each group were applied with formulated adhesive EA and ZEA. The application was made by a micro brush for 10-s smearing and air thinning of adhesive for 6 s for better penetration. A blue light was used for photopolymerization for 20 s. Using a resin condenser and a jig the adhesive surface was bonded with composite resin in incremental thickness of 1.5 mm and cured for 20 s from all sides. All specimens were placed in artificial saliva for 48 h until further use.
Artificial aging, shear bond strength testing, and interfacial failure mode investigation
Between the temperature of 5°C and 55°C all specimens underwent artificial aging in a thermocycler (Thermal cycler, BioRad, UK) for 30 s, 100,000 cycles replicating oral conditions. Following thermocycling of specimens, all samples were positioned in a universal testing machine for SBS, applying force parallel to the bonded surface at a crosshead speed of 0.5 mm/min. The force required to debond the filling material was measured in megapascal (MPa). The debonded surfaces were seen under a stereomicroscope at 60× magnification for failure modes and were categorized into adhesive, cohesive, and admixed failure types.
Rheological property analysis
The rheological properties of adhesives were measured using a rheometer (DMA 850, Waters, TA Instruments, UK) in a rotatory manner in a frequency sweep of 8 mm predetermined plate with an opening of 0.25, over a collection of angular frequencies between 01 and 100 rads at 25°C.
Statistical analysis
Using SPSS-20 (IBM SPSS Statistics) the SBS values were analyzed statistically. The mean values and standard deviation of SBS were estimated and investigated. Two-way analysis of variance ANOVA and post hoc multiple comparison test was used, p-values less than 0.05 were considered statistically significant.
Results
SEM and EDX of synthesized adhesive
The microscopic imagery of ZrO2 revealed irregular-shaped, non-edged particles in secluded and agglomerated form. It is seen that small particles have conjugated to develop into large irregular nanoparticles depicting cloud appearance (Figure 1). EDX analysis revealed essential ions (Zr and O2) in ZEA adhesive. In ZEA the percentage of Zr was noted to be 75.7% whereas, O2 by weight was 25.3% (Figure 2). In two groups that is, dentin surface treated with PDT and ECL resulted in no hybrid layer formation (Figures 3 and 4). This may be attributed to damage to the tubular structure. Also, in Figure 3 superficial layer of dentin fused and collapsed due to laser heat treatment. Dentinal structure treated with etch and rinse demonstrates hybrid layer with penetration of ZEA into the dentinal structure, demonstrating a good bond between dentin and adhesive along with high resin tag formation (Figure 5).

SEM image of ZrO2 nanoparticle. Amorphous Irregular shaped, non-edged particles in secluded and agglomerated form.

The EDX analysis revealed Zr and O2 particles in ZEA adhesive. Zr particles by weight 75.7% and O2 particles by weight 24.3%.
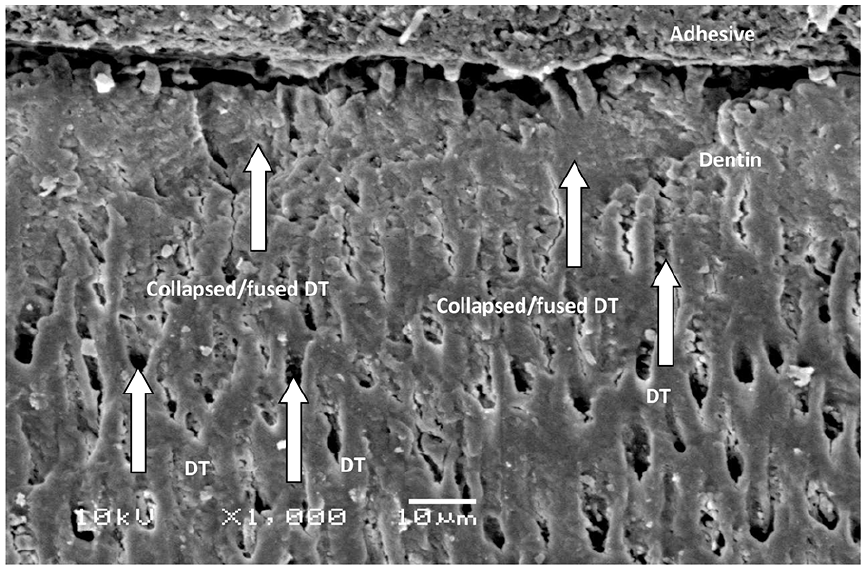
Collapsed/fused dentinal tubules in the superficial layer of dentin due to laser heat treatment along with the absence of resin tag and hybrid layer.

Absence of hybrid layer. Low and coarse resin tag (RT) formation along with poor penetration of adhesive. Absence of dentinal tubules.

Dentin treated with Etch and rinse showing standard hybrid layer (HL) along with good quality dentinal tubule and the high number of resin tag formation.
SBS testing and failure mode
The highest SBS was demonstrated in dentin conditioned with etch and rinse and ZEA (19.05 ± 0.13 MPa). Whereas, the lowest SBS was observed in dentin surface treated with PDT and EA (12.25 ± 0.14 MPa).
Intergroup comparison revealed that dentin structure conditioned with etch and rinse and adhesive ZEA and EA exhibited comparable bond integrity (19.05 ± 0.13 MPa) (18.12 ± 0.01 MPa) (p > 0.05). Moreover, dentin pre-treated with PDT and ECL and adhesive applied ZEA and EA demonstrated a significant difference in SBS values (PDT (14.62 ± 0.11 MPa) (12.25 ± 0.14 MPa)) and (ECL (16.11 ± 0.03 MPa) (14.15 ± 0.31 MPa)) respectively (p < 0.05) (Table 1).
Means and SD for bond strength values among study groups using ANOVA and Tukey multiple comparisons test.

EA: experimental adhesive; ECL: Er,Cr:YSGG; PDT: photodynamic therapy; ZEA: zirconium oxide experimental adhesive.
Different superscript characters denote statistically significant difference!.
Showing significant difference among study group (ANOVA) (Tukey multiple comparison test).
Dentin conditioned with etch and rinse and adhesive ZEA showed significantly better bond strength to dentin treated with PDT and ECL (p < 0.05). Likewise, dentin surface treated with ECL and adhesive EA demonstrated lower SBS to dentin conditioned with etch and rinse regime and higher SBS to PDT treated specimens (p < 0.05) (Table 1).
In the present study, no definitive patterns of modes of failure were observed. Most of the reported failure modes were adhesive (Table 2). Failure in the adhesion process is determined as adhesive failure in which fracture patterns are not found in the resin and dentin.
Interfacial failure types among investigated groups.

EA: experimental adhesive; ECL: Er,Cr:YSGG; PDT: photodynamic therapy; ZEA: zirconium oxide experimental adhesive.
Rheological properties analysis
In Figure 6 rheological properties of two adhesive ZEA and EA are depicted by complex viscosities along with the angular frequency at 0.001–1000 rads/s. It was estimated that with increasing frequencies the viscosities of adhesives were reduced. Non-Newtonian behavior was witnessed by both the adhesive ZEA and EA that is, reduction viscosity with increase shear rate. Low viscosity values were shown in ZrO2 5% by weight in group ZEA adhesive, compared to EA adhesive in the 0% group. It can be estimated that 5% of ZnO2 particles in ZEA improved fluidity of resin filler mixture in ZEA however at low frequencies no Newtonian plateau was observed in the same group. In the EA group, 0% ZnO2 nanoparticles displayed high viscosity than 5% ZnO2. This viscosity was not sustained at high frequencies as an intersection was forecasted between the viscosities of two adhesives (Figure 6).

Rheological properties of two adhesive ZEA and EA.
Discussion
The current study was postulated on the hypothesis that the addition of 5% ZrO2 NPs displayed better SBS to adhesive resin when compared to adhesive without ZrO2 filler after dentin surface treated with PDT, etch and rinse, and ECL. Interestingly, to our surprise addition of 5% ZrO2 particles when used in adhesive established SBS values better than EA. Therefore, the hypothesis of the present study was accepted.
For the clinical success of restoration dental adhesive plays a vital role. Weak adhesive bond results in microleakage, secondary caries, dentin sensitivity henceforth, decreasing the longevity of restoration with a poor prognosis. 19 The nanoparticles in general have been found to improve the mechanical properties of composite with great success. In recent years ZrO2 NPs have gained great attention due to their excellent biocompatibility properties and acceptable esthetics. Also, ZrO2 NPs have been found to improve abrasion, tensile strength, corrosion, and toughness of denture materials. 20 Considering these advantageous properties of ZrO2 these nano ZrO2 particles were incorporated into adhesive to improve SBS, dentin interaction, and durability. 21
ZrO2 NPs acquired and used in the present study were amorphous irregular-shaped, non-edged particles in secluded and agglomerated form (Figure 1). This type of structure makes ZrO2 NPs flow faster and smoother hence reducing friction and contact between the dentin surface. 22 The EDX mapping of ZrO2 NPs demonstrated two main ingredients Zirconium (Zr) and oxide (O2). The use of Zr in different dental applications and tissue engineering is not new. Dental applications in which ZrO2 NPs are incorporated may range from dental implants, ceramics, bone tissue scaffolds, radiopacifying agents, and different denture base materials. Zr as an element improves the mechanical strength when added in different dental materials improving its versatility and applicability.19,20
Dentin surface conditioned with etch and rinse displayed the highest bond integrity irrespective of the type of adhesive. Etch and rinse method of dentin surface treatment is still considered to be the gold standard. 23 Phosphoric acid on dentin structure results in resin tag formation by demineralizing hydroxyapatite crystals, funneling and revealing collagen fibrils, opening the dentinal tubules making the surface receptive to bonding (Figure 5).24,25 Similarly, dentin surface etched with ECL displayed bond strength lesser than etch and rinse but better than PDT. ECL photosensitization on dentin structure results in evaporation of water from the organic and intertubular structure along with thermomechanical ablation resulting in a dentinal structure which is scaly, flaky with fused and collapsed dentinal tubules (Figure 3).24,26,27 Recent work by Aranha et al. 28 demonstrated that laser irradiation of dentinal surface resulted in the formation of micro retentive resin tags. However, the present study exhibited that ECL resulted in no resin tags and hybrid layer formation (Figure 3).
Similarly, PDT using MBP as a dentin conditioner demonstrated the lowest SBS scores. MBP is cationic naturally, and its interaction with Calcium (Ca++) and Phosphate (P) ions in hydroxy appetite crystals of dentin results in precipitation, hence creating a barrier between adhesive and dentinal structure. Moreover, no resin tags on the dentinal structure may attribute to a low concentration of MBP (50 mg/l) used for surface treatment. Poor penetration of adhesive in dentinal tubules may be due to the hydrophilic nature of MBP diminishing SBS scores (Figure 4).25,29,30
Two adhesives were used that is, EA and ZEA after conditioning of dentin. 10% ZrO2 NP were incorporated in dental adhesive as a recent study by Lohbauer et al. 31 indicated that NPs used >10% by weight deteriorates bond integrity. The author speculates that the positive influence of inorganic fillers (ZrO2) in the adhesive is dependent on the type of surface treatment of dentin. The merger of ZrO2 NPs improved bond values compared to EA which improved bond strength. The positive influence of ZrO2 NPs was observed in all groups when dentin was conditioned with etch and rinse, ECL, and PDT where the dentin surface became receptive for bonding to ZEA compared to EA (Figures 3 and 5). Dentin conditioned with etch and rinse itself as a bonding regime has a significant role in improving bond values. However, dentin surface treated with ECL and PDT is not efficient therefore incorporation of ZrO2 NP in ZEA might have influenced surge in bond values compared to EA. The receptive nature of dentin resulted in close adherence of ZrO2 NPs particles with collagen dentin fibrils improving bond values. These findings were in line with already work reported by Aljamhan et al. 32 and Zhou et al. 33
The outcomes of the present study are inspiring but should be understood with caution. The degree of conversion (DC) of adhesive needs to be evaluated as high or low DC values warrant the success of an adhesive. Furthermore, to study inorganic fillers (ZrO2 NPs) more profoundly, micro-Raman spectroscopy of ZrO2 NPs before and after incorporation in adhesive needs further investigation. In vitro nature can be considered as one of the possible limitations as well. The in vivo environment is dynamic and challenging and offers multiple challenges like saliva, pH, temperature which may influence the prognosis of restoration. Fourier-transform infrared spectroscopy (FTIR) along with the different weights of ZrO2 NPs in adhesive needs to be evaluated and assessed in future and upcoming investigations.
Conclusion
The presence of 10% ZrO2 particles in adhesive improves SBS of restoration and this improvement in bond values is statistically significant compared to EA. For ZEA and EA dentin surface treated with etch and rinse is recommended. Dentin conditioning methods with PDT and ECL along with the weight of ZrO2 NPs in adhesive may influence bond values.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to the deanship of Scientific Research at King Saud University for funding the work through research group number (RGP-1438-028)
