Abstract
The objective of this study was to determine the antimicrobial potential of AH plus supplemented with bismuth lipophilic nanoparticles (BisBAL NPs) on the growth of Enterococcus faecalis isolated from patients with endodontic infections. BisBAL NPs, synthesized with the colloidal method, were characterized, in its pure form or AH Plus-absorbed, by energy-dispersive X-ray spectroscopy and scanning electron microscopy (EDS-SEM). Antimicrobial activity was evaluated with disc diffusion assays, and antibiofilm activity with fluorescence microscopy. BisBAL NP-supplemented AH Plus had a 4.9 times higher antimicrobial activity than AH Plus alone (p = 0.0001). In contrast to AH Plus alone, AH Plus supplemented with BisBAL NP inhibited E. faecalis biofilm formation. The sealing properties of AH plus were not modified by the incorporation of BisBAL NPs, which was demonstrated by a 12-day split-chamber leakage assay with daily inoculation, which was used to evaluate the possible filtration of E. faecalis. Finally, BisBAL NP-supplemented AH plus-BisBAL NPs was not cytotoxic for cultured human gingival fibroblasts. Their viability was 83.7% to 89.9% after a 24-h exposure to AH Plus containing 50 and 10 µM BisBAL NP, respectively. In conclusion, BisBAL NP-supplemented AH Plus constitutes an innovative nanomaterial to prevent re-infection in endodontic patients without cytotoxic effects.
Keywords
Introduction
Microorganisms that colonize root canals and pulp chambers must be eradicated for successful clinical attention. 1 Common causes of endodontic failure include bacterial persistence, incorrect filling of the canal, and an improper sealing of the canal. 1 An optimal sealer should have a strong antimicrobial potential to avoid reinfection of root canals and the pulp chamber. AH Plus root canal sealer is a thermoplastic cement, applied in a paste-paste system for fast and clean mixing. 2 AH Plus has many excellent properties, like a suitable fluency, low contraction and solubility, but it lacks strong antimicrobial activity. In vitro disc diffusion assays revealed that AH Plus hardly affected Enterococcus faecalis growth. 3 However, the mix of AH Plus containing 2% insoluble antibacterial nanoparticles completely inhibited bacterial growth inhibition. 4 Furthermore, E. faecalis could be eradicated when AH Plus was supplemented with 3% of benzalkonium chloride. 5 Likewise, antimicrobial properties improved when AH Plus was supplemented with substances like amoxicillin or triple antibiotic paste, 6 cetylpyridinium chloride, 7 the herbal extract of Salvadora persica, 8 and nano-mangesium hydroxide 9 as compared to AH Plus alone. Unfortunately, the gain in antimicrobial activity is set back by diminished the sealing capacity of AH Plus, so that the net risk of infection may even be worse than AH Plus alone. Therefore, it would be interesting to improve the antimicrobial activity of AH Plus without loss of its excellent sealing characteristics.
Nanomedicine is a new discipline that offers the opportunity to develop “smart drugs” with high selectivity, controlled delivery, and few non-severe adverse side effects. 10 The high and increasing prevalence of multidrug-resistant bacteria is a huge challenge for clinical practice and current antibiotics are not effective to eradicate bacteria growing into biofilms. 11 Multidrug-resistant E. faecalis have been isolated from root canal biofilms from patients with endodontic infections and from the urinary tract of infected patients.12,13 It has been shown that silver and zinc oxide nanoparticles are as effective as conventional endodontics irrigants in detaching E. faecalis biofilm from root canals. 14 We have previously reported the efficacy of bismuth lipophilic nanoparticles (BisBAL NPs) against several oral pathogens, including E. faecalis . 15 We have also demonstrated that the cement mineral trioxide aggregate (MTA) supplemented with BisBAL NPs acquired antimicrobial and antibiofilm activities against E. faecalis . 16 As a continuation of this work, here we report the antimicrobial potential of AH Plus BisBAL NPs against E. faecalis, both a commercial strain and clinical isolates from patients with endodontic infection.
Material and methods
Study design and clinical sampling
This experimental case-control study on extracted endodontic samples was approved by the institutional bioethical committee (ID: IB-049/2021-05-CCR). Volunteers were recruited among patients who had been referred for endodontic retreatment to the Postgraduate Endodontic Clinic of the Faculty of Dentistry of the Autonomous University of Nuevo León (U.A.N.L.) between February 2019 and September 2020.
After a signed informed consent, 50 endodontic samples were recollected from patients with endodontic treatment failure (clinical diagnosis of previously treated). Before extraction, the samples were X-ray analyzed to determine the possible presence of calcification and resorption. Next, the dental piece was isolated with two rubber dams, cleaned with 3% hydrogen peroxide, and disinfected with 2.5% sodium hypochlorite. Endodontic access was completed by employing a sterile high-speed bur. The operative field was cleaned by irrigation with 2.5% sodium hypochlorite which was subsequently inactivated with sterile 5% sodium thiosulfate. Once a cavity could be accessed, the first dam was removed and the coronal gutta-percha was eliminated using sterile Gates Glidden drills. The apical tissue was exposed after removing contaminated gutta-percha with solvent-free Hedstrom-type hand files. Next, the canal was filled with phosphate-buffered saline of pH 7.3 (PBS), and the dental piece was extracted with a # 15 type K file (Dentsply Maillefer, Ballaigues, Switzerland), which was introduced at a level approximately 1 mm below the apex of the dental piece. The sample was recollected in an Eppendorf microtube containing 1 mL brain-heart infusion (BHI; BD DIFCO, Sparks, MD, USA), and incubated for 7 days at 37°C under aerobic conditions.
For microleakage analysis, the dental pieces were incubated in 5.25% hypochlorite for 30 min to eliminate organic debris and stored in PBS at room temperature until use.
Materials development and characterization
The production of AH Plus supplemented with BisBAL NP
BisBAL NPs were synthesized and analyzed as previously reported.15–17 Briefly, bismuth nitrate in propylene glycol (Bis) was mixed 2:1 with 2,3-dimercapto-1-propanol (BAL) to produce BisBAL nanostructures. The distributions of BisBAL NP size and shape were analyzed with scanning electron microscopy (SEM; FEI Tecnai G2 Twin, Hillsboro, OR; 160 kV accelerating voltage). Energy-dispersive X-ray spectroscopy spectroscopy SEM (EDS-SEM; Oxford INCA X-Sight, Tubney Woods, UK) was used for bismuth identification.
AH Plus was freshly prepared according to the manufacturer’s instructions (Dentsplay de Trey, Konstanz, Germany) and mixed with 10–100 µM of BisBAL NPs. AH Plus alone was used as negative control. The mixture of AH Plus-BisBAL NP was analyzed with SEM and EDX spectroscopy as described above.
Bacterial culture and antimicrobial/antibiofilm properties
Bacterial culture and clinical isolates
E. faecalis strain 11420 was purchased from the American Type Culture Collection (ATCC 11420) and cultured according to the provider’s instructions. Clinical isolates, obtained from patients with endodontic infections, were grown in trypticase soy broth agar (TSB; BD DIFCO, Sparks, MD, USA) at 37°C for 24 h under aerobic conditions. The specific presence of E. faecalis was confirmed with real-time polymerase chain reaction (PCR) (see below).
E. faecalis identification by real time-PCR
Genomic DNA was extracted from clinical isolates using the Isolate II Genomic DNA kit (Bioline, UK) according to the provider’s instructions. Extracted DNA (5 µL) was amplified in a final volume of 20 µL using the E. faecalis genesig® Easy kit (Primer Design, UK) 18 and the genesig q16 real-time thermocycler (Primer Design, UK) according to the provider’s instructions. 19
Antimicrobial activity of BisBAL NP-supplemented AH Plus
The antimicrobial activity of AH Plus-BisBAL NPs on E. faecalis growth was analyzed with the disk diffusion assay.16,20,21 E. faecalis and bacteria isolated from endodontic patients were cultured in TSB agar (BD DIFCO, Sparks MD, USA) at 37°C, for 18 h with standard inoculums: 100 µL bacterial culture, 0.5 MacFarland, was spread on TSB agar plates using a sterile cotton swab. Wells were punched with a previously sterilized 5-mm diameter polystyrene ring and filled with AH Plus-BisBAL NPs or AH Plus alone (freshly prepared and in semi-solid state. Growth inhibition was measured with a Vernier after a 24-h exposure. The experiment was carried out in triplicate.
Antibiofilm activity of BisBAL NP-supplemented AH Plus
The antibiofilm activity of AH Plus supplemented with BisBAL NPs was analyzed with a microbial viability stain (SYTO 9) and fluorescence microscopy on E. faecalis biofilms. Hereto, a 12-well plate, coated with (~35 mg) of AH Plus with or without BisBAL NPs were seeded with 1 × 106 cells/well in 100 µL of TSB (ATCC strain or a human isolated specimen) and incubated at 37°C for 48 h. The biofilms of E. faecalis strain 11420 and clinical isolates cultured in non-coated wells served as growth controls. After incubation, cells were stained with 20 µM SYTO 9 (Invitrogen, Carlsbad, CA, USA)22–24 for 30 min at 37°C in the dark. After staining, the biofilm was washed with sterile water and observed at 485 nm by fluorescence microscopy (EVOS, Floyd, Thermo Fisher Scientific, MA, USA).
Microleakage evaluation of BisBAL NP-supplemented AH Plus
To determine whether BisBAL NPs provide resistance to the passage of bacteria through the entrance orifice to the root canals, a split-chamber bacterial penetration test was used. Hereto, 50 single-rooted dental pieces with a straight root, a closed apex, and devoid of cracks and root caries were decoronated to obtain 16-mm long specimens. The working length was established visually (under a microscope) by subtracting 1 mm from the length of a K-file size # 15 (Dentsply Maillefer, Ballaigues, Switzerland) placed at the apical foramen. The instrumentation included ProTaper Universal rotatory files S1 (3 mm short of working length), SX file (with a circular movement), and S1, S2, F1, F2, F3, F4, and F5 files (Dentsply Maillefer, Ballaigues, Switzerland until their working lenghts. During canal preparation, 1 mL 2.5% sodium hypochlorite was used for irrigation. Toward the end of canal preparation, root canals were irrigated with 1 mL 17% EDTA after each file. The smear layer was removed by irrigation with 5 mL 17% EDTA and 5 mL 2.5% sodium hypochlorite. Finally, sodium hypochlorite was washed out with 10 mL sterile water and the specimens were autoclaved in glass containers with 5 mL of distilled water to avoid dehydration. After sterilization, the specimens were sealed with conventional gutta-percha (Hygienic, Coltene, Lanenau, Germany) and obturated employing lateral compaction with standardized and non-standardized gutta-percha cones in a laminar flow hood. The teeth, thus obturated with gutta percha, were randomly assigned to one of the following experimental groups: AH Plus-BisBAL NPs (n = 20), AH Plus alone as negative controls (n = 20), and sealer-free positive controls (n = 10). To set up the split-chambers, obturated pieces were placed inside 1.5-mL Eppendorf tubes, from which the taper ends had been cut off. The junctions between the root and the tube were sealed with cyanoacrylate and three layers of varnish. The Eppendorf tube samples were placed inside sterile polypropylene tubes containing 11 mL of culture medium, so that at least 3 mm of the root apex was inside the medium. The junctions between the Eppendorf and polypropylene tubes were sealed with cyanoacrylate and varnish. Samples were incubated at 37°C for 5 days to verify sterility of the set-up. In case of turbidity, that is, contamination, the set-up was sterilized and re-tested. Finally, 10 µL of E. faecalis culture were inoculated daily for 12 days, and leakage was analyzed by measuring optical density at 600 nm by employing a 96-well microplate absorbance reader (BioTek, Winooski, VT, USA).
Cell culture and biocompatibility
Cell culture
Human gingival fibroblasts (HGFs) were kindly donated by Dr. Higinio Arzate’s laboratory at the Autonomous National University of Mexico. HGFs were grown in DMEM supplemented with 10% fetal bovine serum (Gibco-Invitrogen, Carlsbad, California, USA), 100 U/mL penicillin, 100 µg/mL streptomycin, and 2.5 µg/mL amphotericin at 37°C with 5% CO2. 25 HGF were maintained until reaching confluency.
Cytotoxicity assay
The cytotoxicity on HGFs was analyzed with the MTT cell viability assay. Hereto, 1 × 105 cells/100 µL were cultured in a 96-well plate of which the wells had been coated with a layer (~35 mg) of AH Plus alone or supplemented with 10 or 50 µM BisBAL NP. After a 24-h exposure, the MTT assay was performed according to the provider’s instructions (Biotium, Hayward, California, USA). Non-coated wells served as a growth control. Additional non-coated controls were incubations with 10 and 50 µM BisBAL NP, and 2.5% sodium hypochlorite. The absorbance at 570 nm (A570) was measured with a microplate reader (BioTek, Winooski, VT, USA).
Statistical analysis
One-way ANOVA with Tukey correction was used to compare among groups. A significance level of α = 0.05 was considered for all tests.
Results
Materials development and characterization
Characterization of BisBAL NPs, AH Plus, and the mix by SEM
BisBAL NPs were spherical in shape, with an average diameter of 24 nm (Figure 1(a)). Also, BisBAL NPs formed electrodense clusters that are common among this kind of nanoparticles. The presence of bismuth was confirmed by EDS-SEM in pure BisBAL NP samples (Figure 1(a)) and AH Plus supplemented with BisBAL NP, but not in AH Plus alone (Figure 1(b)).

Characterization of BisBAL NP alone and supplemented AH Plus by SEM. (a) The surface structure, diameter, and organization of BisBAL NPs were determined by SEM. Bismuth identification was corroborated by the element composition in EDX spectrum. (b) Characterization of AH Plus added with BisBAL NP by SEM and EDX spectroscopy. Arrows indicates the presence of bismuth nanoparticles into AH plus sealer.
Interestingly, the typical clusters of spherical BisBAL NPs were observed at the surface of AH Plus (red arrows), which would facilitate direct contact with microorganisms. Altogether these data strongly suggest that BisBAL nanostructures were only present in the BisBAL-NP supplemented AH Plus.
Bacterial culture and antimicrobial/antibiofilm properties
E. faecalis detection from human clinical samples
The E. faecalis identification from clinical isolates obtained from patients with endodontic infection was done by real time-PCR employing specific primers and probe for E. faecalis. E. faecalis was identified the positive control E. faecalis, ATCC strain 11420 and within all clinical isolates (Figure 2). No amplification was detected in the negative control (water; blue line) supporting the veracity of our results. Altogether, these results are relevant because these samples represent the biofilm attached to root canals and probably play an important role in endodontic infection.

E. faecalis identification from clinical isolates by real time-PCR. E. faecalis ATCC strain 11420 (red line) and a clinical isolate from an endodontic patient (black line) were amplified by qPCR. Negative control, water (blue line); the kit’s positive control (green line).
Antimicrobial and antibiofilm activity of BisBAL NP-supplemented AH Plus
The antimicrobial property of AH Plus supplemented with BisBAL NPs was analyzed by disc diffusion assays on E. faecalis ATCC strain and clinical isolates from endodontic patients). BisBAL NP-supplemented AH Plus was 4.9 times more potent than AH Plus alone. AH Plus supplemented with BisBAL NPs yielded significantly larger halos than AH Plus alone (p, 0.0001) on both the E. faecalis ATCC strain and the clinical isolates from endodontic patients (Table 1).
Antimicrobial activity against E. faecalis ATCC 11420 and clinical isolates determined by disc diffusion.

Likewise, BisBAL NP-supplemented AH Plus, but not AH Plus alone, inhibited the formation of E. faecalis biofilms of both the ATCC strain (Figure 3). Thus, it seems that BisBAL NP supplementation confers antimicrobial and antibiofilm potential to the sealer AH Plus.

Antibiofilm activity of BisBAL NP-supplemented AH Plus. Biofilms of E. faecalis (ATCC strain 11420 or clinical isolates) were exposed for 48 h to AH Plus alone or BisBal NP-supplemented AH Plus. The non-exposed cultures of E. faecalis (ATCC strain and human clinical isolates) served as controls. Bar, 65 µm.
Microleakage evaluation of BisBAL NP-supplemented AH Plus
The split-chamber test revealed that after 12 days of constant inoculation, none of the sealing cements presented leakage (Figure 4). Thus, it seems that the incorporation of BisBAL NPs to AH Plus does not modify its sealing properties, while it does provides antimicrobial activity.
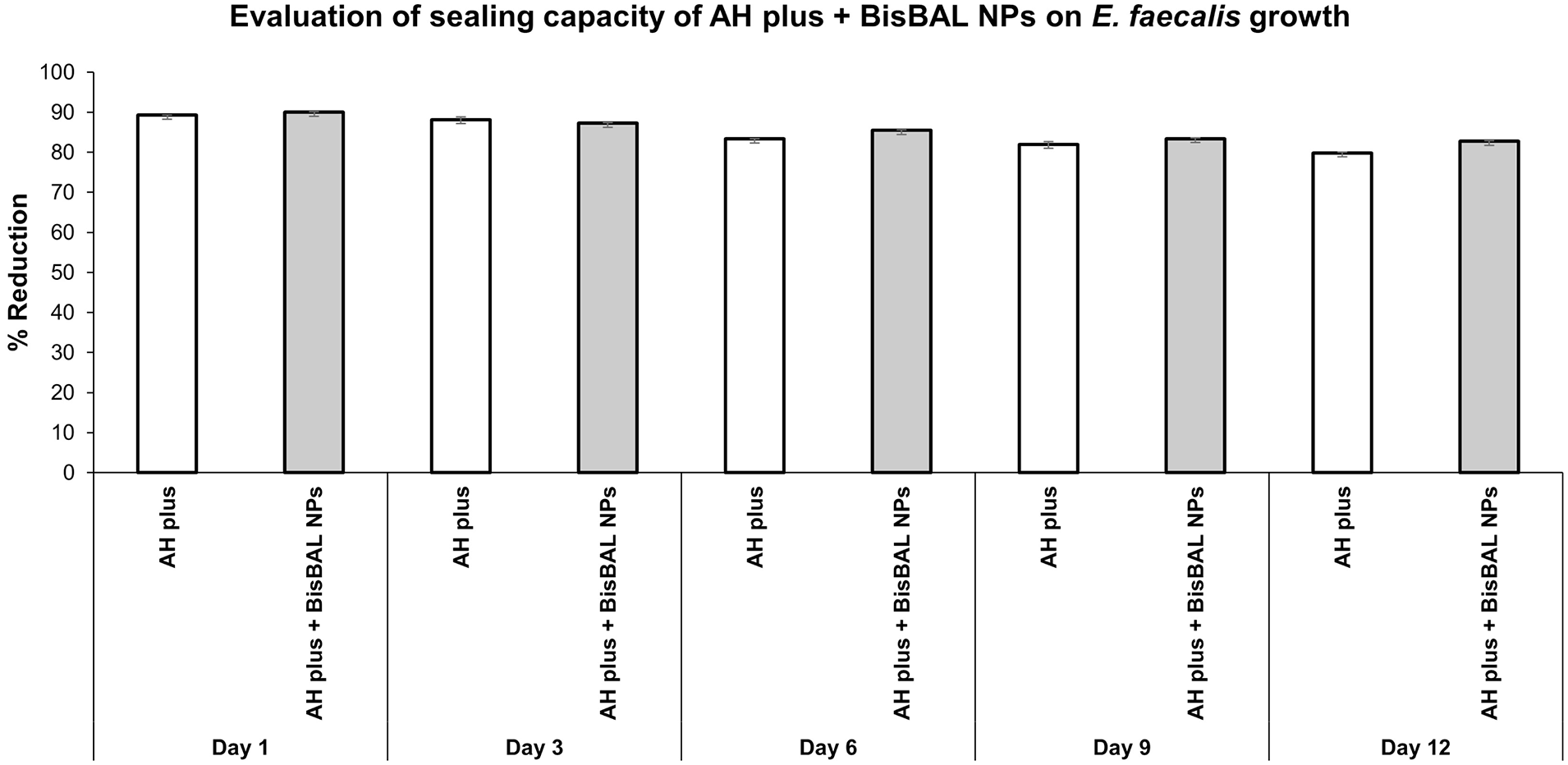
Sealing capacity of AH plus, alone and supplemented with BisBAL NPs. The split-chamber bacterial leakage test was used to evaluate the possible filtration of E. faecalis after 12 days of constant inoculation. Optical density was measured at 600 nm employing a microplate absorbance reader (BioTek, Winooski, VT, USA). An OD600 of 0.1 was considered as positive (i.e. contamination). Data obtained are expressed as reduction percentage.
Cell culture and biocompatibility
Cytotoxicity of BisBAL NP-supplemented AH Plus on human cells
The possible cytotoxicity of BisBAL NP-supplemented AH Plus was verified with the MTT assay on HGFs. After a 24-h exposure, the survival rates in comparison to a growth control were: AH Plus, 92.2%; AH Plus supplemented with 10 or 50 µM BisBAL NP, 89.8% and 87.3%, respectively; 10 or 50 µM BisBAL NP alone, 89.8% and 83.7%, respectively; and 2.5% sodium hypochlorite, 6.7% (Figure 5). There were no significant differences among the groups, except for sodium hypochlorite 2.5%. These results suggest that the incorporation of BisBAL NP into AH Plus has an insignificant to limited cytotoxicity for HGF.

Cytotoxicity of BisBAL NP-supplemented AH. Human gingival fibroblasts (1 × 105 cells) were exposed for 24 h to AH Plus supplemented with 0, 10, or 50 µM BisBAL NP. Controls: growth control (culture medium), 10 or 50 µM BisBAL NP, and 2.5% sodium hypochlorite. Cell viability (% respect to untreated cells considered 100%) was evaluated with an MTT assay. Bars and error bars indicate means ± SD respectively; (n = 6); *, statistically significant (p < 0.0001; α = 0.05).
Discussion
The cleaning and disinfection of the root canal system during endodontic treatment continues being a huge challenge. The goal of removing bacterial biofilms adhering to portions of the root canal and surrounding structures is not always achieved. 26 The most common strategy is the use of liquid disinfectants. Sodium hypochlorite is the main treatment option worldwide. 27 Interestingly, the effectivity of silver nanoparticle to remove biofilms from root canals was more effective when applied as a gel than as a suspension. 28
Several approaches have been proposed to improve the bactericidal and antibiofilm activities of dental materials employed in endodontic treatments.28,29 So far, innovative dental materials are not available in clinical practice. Although there are several reports on antibiotics and antibiofilm solutions, the option of antibacterial root canal sealers could provide innovative alternatives. Root canal sealers should seal the prepared canal and avoid contamination by microorganisms. Therefore, an ideal root canal sealer should have antimicrobial potential and kill all microorganisms to avoid infections in case of leakage.30,31 The supplementation of endodontic sealers with antibacterial nanostructures has enhanced clinical success rates. 32 The use of metal in dentistry is not new; gold, silver, zinc, platinum, titanium, nickel, and zirconium, have all been employed in the oral cavity because of their hardness and mechanical properties.33–35 Nanotechnology has emerged as a new discipline that may generate “smart drugs” with improved properties in biological systems. 36 Typically, spherical nanoparticles have a diameter <100 nm, and their reactivity associates with their size. 37 Although there are several reports on the usage of nanotechnology in the dental area, root canal sealers have been hardly explored.38,39 The employment of nanoparticles in dental materials to improve their properties has become more common in the past 5 years.30,40,41 Herein, we describe the antimicrobial and antibiofilm potential of AH Plus root canal sealer supplemented with BisBAL NPs. Previously, we reported on the excellent antimicrobial activity of BisBAL NP against gram-positive and gram-negative bacterias, and against fungi. 17 The addition of BisBAL NP to MTA conferred antimicrobial properties to the cement, including against E. faecalis . 16 Similarly, the incorporation of quaternary ammonium polyethylenimine (QPEI) nanoparticles to AH Plus conferred antimicrobial activity to the root canal sealer. 31 We found that the addition of 50 µM BisBAL NP to AH Plus potentiates antimicrobial properties against both E. faecalis strain ATCC 11420 and clinical isolates of the same species. Recently, a novel new root canal sealer, composed of dimethylaminohexadecyl methacrylate (DMAHDM) and silver nanoparticles, decreased the colony forming units of E. faecalis six times as compared to AH Plus. 42 We found that, in contrast to AH Plus alone, the incorporation of BisBal NPs completely inhibited biofilm formation by both the E. faecalis strain ATCC11420 and clinical isolates. We hypothesize that BisBAL NPs kill the bacteria during their adherence process to the BisBAL NP-supplemented root canal sealer. Similarly, the incorporation of DMAHDM-silver nanoparticle into AH Plus provided bactericidal activity against E. faecalis . 43
To analyze whether the incorporation of BisBAL NP into AH Plus modified its sealing properties, a split-chamber bacterial leakage test was performed. In contrast to other studies, where leakage of the AH Plus occurred in 40% of the groups by day 7 or 12,44,45 in our study there was no bacterial penetration during the observation period of 12 days. Thus, it seems that the addition of BisBAL NP to AH Plus improved the antimicrobial/sealing capacity of AH Plus. Song et al. 46 analyzed the in vitro microleakage of several new sealers by staining with a 1% methylene blue solution for 2 weeks. There were no significant differences among the sealers tested.
Unfortunately, many studies on the innovation of dental materials in order to improve antimicrobial potential have gone away when verifying the cytotoxicity for human tissues. Here we show that a 24-h exposure to BisBAL NP-supplemented AH Plus was hardly to not cytotoxic to cultured HGFs. Our previous studies showed repeatedly the high biocompatibility of BisBAL NP or BisBAL NP-supplemented cements both in vitro and in vivo.16,47–49 Histopathological studies in mice revealed no signs of tissue damage in any of the analyzed organs. 49 In contrast, silver, gold, and copper nanoparticles have repeatedly been reported to be cytotoxic.29,50–53 In spite of its widespread usage, silver nanoparticles have many adverse effects, including acute toxicity and genotoxicity. 54 Furthermore, it has been reported that dental devices that contain nanoparticles of gold, silver, and titanium promoted neurotoxicity. 55 Thus, BisBAL NP combines antimicrobial/antibiofilm activity with a good biocompatibility.
In summary, we provide evidence on the antimicrobial and antibiofilm potential of BisBAL NP-supplemented AH Plus root canal sealer. BisBAL NP-supplemented AH Plus inhibited the development of biofilms of the E. faecalis ATCC strain 11420 and clinical isolates. The sealing properties of AH Plus were not affected by supplementation with BisBAL NP. BisBAL NP-supplemented AH Plus was not cytotoxic to HGFs. Altogether, our data suggest that BisBAL NP-supplemented AH Plus is an innovative root canal sealer alternative for endodontic treatment.
Conclusions
BisBAL NP-supplemented AH Plus constitutes an innovative nanomaterial to prevent re-infection in endodontic patients without cytotoxicity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Sectorial Fund for Education Research of the CONACyT for grant A1-S-20148, and Irene Meester for critical reading and edition.
