Abstract
Objective:
To evaluate the antitumor and antimicrobial properties of an alginate-based membrane (ABM) loaded with bismuth lipophilic nanoparticles (BisBAL NPs) and cetylpyridinium chloride (CPC) on clinically isolated bacteria and a pancreatic cancer cell line.
Material and methods:
The BisBAL NP-CPC ABM was characterized using optical and scanning electron microscopy (SEM). The antimicrobial potential was measured using the disk-diffusion assay, and antibiofilm activity was determined through the live/dead assay and fluorescence microscopy. The antitumor activity was analyzed on the pancreatic cell line (Panc 03.27) using the MTT assay and live/dead assay with fluorescence microscopy.
Results:
After a 24-h exposure (37°C, aerobic conditions), 5 µM BisBAL NP reduced the growth of K. pneumoniae by 77.9%, while 2.5 µM BisBAL NP inhibited the growth of Salmonella, E. faecalis and E. faecium by 82.9%, 82.6%, and 78%, respectively (p < 0.0001). The BisBAL NPs-CPC ABM (at a ratio of 10:1; 500 and 50 µM, respectively) inhibited the growth of all isolated bacteria, producing inhibition halos of 9.5, 11.2, 7, and 10.3 mm for K. pneumoniae, Salmonella, E. faecalis, and E. faecium, respectively, in contrast to the 6.5, 9.5, 8.5, and 9.8 mm obtained with 100 µM ceftriaxone (p < 0.0001). The BisBAL NPs-CPC ABM also reduced bacterial biofilms, with 81.4%, 74.5%, 97.1%, and 79.5% inhibition for K. pneumoniae, E. faecium, E. faecalis, and Salmonella, respectively. Furthermore, the BisBAL NPs-CPC ABM decreased Panc 03.27 cell growth by 76%, compared to 18% for drug-free ABM. GEM-ABM reduced tumoral growth by 73%. The live/dead assay confirmed that BisBAL NPs-CPC-ABM and GEM-ABM were cytotoxic for the turmoral Panc 03.27 cells.
Conclusion:
An alginate-based membrane loaded with BisBAL NP and CPC exhibits dual antimicrobial and antitumoral efficacy. Therefore, it could be applied in cancer treatment and to diminish the occurrence of surgical site infections.
Keywords
Introduction
Pancreatic cancer ranks as the fourth leading cause of cancer-related death globally, 1 with over 495,700 diagnoses recorded in 2020. 2 Following diagnosis, a mere 20% of patients qualify for surgery. 3 Even with adjuvant chemotherapy, post-operative survival rates are generally low, often falling below 5 years. 4 In cases where surgery is not viable, alternative treatments such as photothermal therapy, immunotherapy, and chemotherapy are explored, although the prognosis typically remains unfavorable. 5
While immunotherapy has shown success in treating leukemia and melanoma, pancreatic ductal adenocarcinoma poses unique challenges due to low immunogenicity and its immunosuppressive microenvironment. Typically detected at advanced stages, pancreatic cancers exhibit resistance to chemotherapy due to the dense stroma and disorganized vasculature. Overcoming obstacles such as inefficient drug penetration, poor absorption, and drug resistance is essential for effective antitumor treatment. 6
By 2030, pancreatic cancer is projected to become the second-leading cause of cancer-related deaths in the United States. 7 The urgent need for innovative treatment alternatives and diagnostics is evident. Applying nanomedicine to cancer treatment enables the controlled delivery of target-specific antitumor agents with minimal collateral cytotoxicity.8–10 Various nanoparticles designs, such as urokinase plasminogen activator receptor-targeted magnetic iron oxide nanoparticles (uPAR-IONPs), 11 electrospun poly(L-lactide)-hyaluronan (PLA-HA) scaffolds, 11 and poly(lactide)-co-glycolide-polyethylene glycol diamine (PLGA-PEG) immunonanoparticles, 12 have demonstrated successful controlled delivery and release of the anticancer drug gemcitabine (GEM) in preclinical cell cultures and xenograft tumor models of pancreatic cancer.
Moreover, fibrous membranes loaded with anti-tumor and antibacterial agents offer an innovative treatment alternative for pancreatic cancer. 13 Our group has reported the potent antimicrobial properties of bismuth lipophilic nanoparticles (BisBAL NPs) embedded in a chitosan-based membrane. 14 In vitro studies have shown that BisBAL NPs loaded within the biodegradable hydrogel also inhibited growth of human prostate, cervical, and colorectal cancer cell lines. 15 Furthermore, the combination of BisBAL NPs with cetylpyridium chloride (CPC) demonstrated a cumulative antitumor effect on human breast cancer cells 16 and lung cancer cells, in vitro. 17
Surgical site infections (SSI) are common complications in cancer patients and significantly increase morbidity. In a referral cancer center in Mexico, SSI occurred in 46% of patients (n = 219) following a pancreaticoduodenectomy. 18 The main causative pathogens were enterobacteria and enterococci, exhibiting high levels of multidrug resistance. 18 Pancreatic necrosis is a severe complication due to infection by gram-negative bacteria such as Klebsiella spp and Salmonella spp, which belong to the order Enterobacterales and display high antibiotic resistance. 19 Gram-positive bacteria, particularly Enterococcus spp., have also been isolated from SSI. 20 Previous studies have reported that BisBAL NPs inhibit the growth of both gram-positive and gram-negative bacteria, with minimal inhibitory concentrations (MIC) of 5 μM. 21 Furthermore, AH Plus root canal sealer supplemented with BisBAL NP has shown bactericidal and antibiofilm activity against Enterococcus faecalis, a bacteria commonly found in endodontic infections. 22
In this study, the authors describe the synthesis and characterization of an alginate-based membrane (ABM) loaded with both BisBAL NPs and CPC (BisBAL NPs-CPC-ABM), as well as the evaluation of the antimicrobial effectiveness of BisBAL NPs-CPC-ABM against clinical bacterial isolates and its antitumor activity against a pancreatic tumor cell line.
Materials and methods
Synthesis and characterization of BisBAL NPs
BisBAL NPs were synthesized using the colloidal method, as previously described. 21 Briefly, a 50 mM Bi3+ solution was prepared by dissolving Bi(NO3)3·5H2O in propylene glycol, with heating to 80°C and intermittent vortexing for 2 h. BisBAL NPs were generated by combining the Bi3+ solution with 2,3-dimercapto-1-propanol (BAL) in a 2:1 molar ratio. Scanning electron microscopy (SEM; FEI Tecnai G2 Twin, Hillsboro, Oregon, USA; 160 kV accelerating voltage) was employed to assess morphology, size, and distribution of the BisBAL NPs.
CPC stock solution
A 1 mM CPC stock solution was prepared by dissolving 0.339 g of CPC (Sigma-Aldrich; St. Louis, MO) in 1 mL sterile bidistilled water and stored at room temperature (RT).
Synthesis of ABM loaded with BisBAL NPs and CPC
To prepare a 2% (w/v) sodium alginate solution, 1 g of sodium alginate was dissolved in 50 mL sterile deionized water. The mixture was vortexed and continuously stirred using a magnetic stirrer for 2 h at RT until no clumps were visible. Once fully dissolved, BisBAL NPs and CPC were added at a ratio of 10:1, with final concentrations of 500 and 50 μM, respectively.
For the preparation of BisBAL NPs-CPC-ABM, 1 mL glycerol was mixed with 9 mL of the 2% sodium alginate solution supplemented with BisBAL (500 μM) and CPC (50 μM). The mixture was then left to dry at 40°C for 18 h. Subsequently, 50 mM CaCl2 was added to induce cross-linking.
For comparative purposes, an ABM was prepared using a 100 μM GEM solution (GEM-ABM; GEM; Sigma-Aldrich St. Louis, MO).
Microscopy and spectroscopic characterization of BisBAL NPs-CPC-ABM
Optical microscopy and scanning electron microscopy (SEM; FEI Tecnai G2 Twin, Hillsboro, Oregon, USA; 160 kV accelerating voltage) were employed to analyze the BisBAL NPs-CPC-ABM, GEM-ABM, and drug-free ABM. The chemical composition was verified by energy-dispersive X-ray spectroscopy (EDS) SEM (EDS; Oxford INCA X-Sight, Tubney Woods, UK).
Fourier-transform infrared spectroscopy (FTIR) spectra of BisBAL NPs-CPC-ABM, GEM-ABM, or drug-free ABM were obtained by using a Nicolet 7800 FTIR spectrometer equipped with a ZnSe-attenuated total reflectance (ATR) accessory. Scans in the spectral range of 4000 to 650 cm−1 were acquired, averaging 200 scans with a resolution of 4 cm−1. Additionally, Raman spectroscopy was used to analyze the structural quality of BisBAL NPs-CPC-ABM, GEM-ABM, and drug-free ABM. Spectra for each membrane were obtained using a Renishaw InVia spectrometer equipped with a 532 nm laser and a 10-s exposure.
Clinical specimen isolates
Clinical isolates of Klebsiella pneumoniae, Salmonella spp., Enterococcus faecium, and Enterococcus faecalis were collected from January 2021 to December 2022 at the Hospital Universitario Dr. José Eleuterio González, a third-level academic hospital located in Monterrey, Mexico. The hospital is a complex consisting of a medical school and two separate hospitals. The main building is a 600-bed hospital for the treatment of non-COVID-19 cases, with an average of 25,000 admissions and around 200,000 emergency room visits per year. It also receives referrals from neighboring public and private state hospitals. The second building is an 85-bed hospital designated for the diagnosis and treatment of suspected or confirmed COVID-19 cases. This study was conducted with approval from the Ethics Committee of the Hospital (approval number IF23-00004). The committee granted permission to waive written consent from patients because of the anonymization of clinical information.
To rapidly identify bacterial species, their cellular protein patterns were analyzed using matrix-assisted laser desorption ionization time-of-flight (MALDI-TOF) mass spectrometry (Microflex LT system, Bruker Daltonics, Bremen, Germany) according to the manufacturer’s instructions. Briefly, a single colony from cultures grown on blood agar plates at 37°C for 24 h was placed into a well of a 96-spot stainless steel target plate (Bruker Daltonics, Bremen, Germany). Next, 1 µL of 70% formic acid was added, and after drying, 1 µL of an alpha-cyano-4-hydroxycinnamic acid matrix solution (Sigma-Aldrich, Toluca, Mexico) was added. The plate was then analyzed by the MALDI Biotyper 3.0 software for spectra profile database matching. The isolates were classified according to the manufacturer’s recommended score identification criteria: a score range of 2.000−2.299 allows acceptable genus-level identification, and a score range 2.300–3.000 allows acceptable species-level identification. The mass spectra obtained from each isolate were further processed using flexAnalysis software (Bruker Daltonics) after applying spectra smoothing, top-hat baseline subtraction, and normalization.
Bacterial culture
Isolates from patients with infectious diseases (K. pneumoniae, Salmonella spp, E. faecium, and E. faecalis) were grown in tryptic soy broth agar (TSB; BD DIFCO, Sparks, MD, USA) at 37°C for 24 h under aerobic conditions.
Antimicrobial susceptibility testing
Antimicrobial susceptibility testing (AST) was performed using the VITEK 2 Compact automated system (Biomérieux, Craponne, France) following the manufacturer’s instructions. Initially, bacteria were cultured overnight at 35°C on plates containing tryptic soy broth agar with 5% sheep blood. Subsequently, a bacterial suspension in a 0.45% sodium chloride solution was adjusted to the 0.5 McFarland Standard. The suspension was then introduced into the automated system, which inoculated the AST-GP75 VITEK card for Gram-positive microorganisms or AST-N271 and AST-N272 VITEK cards for Gram-negative microorganisms. The cards were incubated at 35°C until the test was completed. Based on the MIC, isolates were categorized as susceptible, intermediate, or resistant using the current breakpoint criteria from the 2023 Clinical and Laboratory Standards Institute (CLSI).
Disk diffusion assay
The antimicrobial activity of BisBAL NPs-CPC-ABM on K. pneumoniae, Salmonella spp, E. faecium, and E. faecalis was assessed using the disk diffusion assay. Each bacterium was cultured individually in TSB agar (TSB, BD DIFCO, Sparks MD, USA) under aerobic conditions at 37°C for 18 h, with standard inoculums (0.5 McFarland). A bacterial culture (100 µL) was plated on TSB agar in which a central, 5 mm well had been punched. The central well was filled with any of the following growth inhibitors: 100 µM BisBAL NPs, 100 µM ceftriaxone as a positive control for inhibition, or bidistilled water as a negative control. After a 24-h incubation at 37°C under aerobic conditions, the inhibition halos were measured using a Vernier caliper. The experiment was conducted in triplicate.
Antibiofilm assay and fluorescence microscopy
Following the establishment of the bactericidal potential of BisBAL NPs-CPC-ABM against multidrug-resistant bacteria, its antibiofilm activity was assessed using the live/dead Bac-Light assay and fluorescence microscopy. 23 Suspension cultures of K. pneumoniae, Salmonella spp, E. faecium, and E. faecalis (1 × 105/100 µL/well) were allowed to form biofilms at 37°C for 24 h in a 96-well plate. Subsequently, the culture medium was removed, and the bacterial biofilms were exposed for 24 h (37°C; aerobic conditions) to BisBAL NPs-CPC-ABM (100 µM each) or drug-free ABM as a negative control. After washing with 10 mM phosphate-buffered saline (PBS) at pH 7.3, the bacterial biofilm was stained with 100 µL of SYTO 9 and propidium iodide (6 and 30 μM, respectively) at RT for 30 min in the dark, according to the supplier’s instructions. After a wash with sterile water, the biofilms were observed using an EVOS Cell Imaging System (Thermo Fisher Scientific, CA, USA) equipped with FITC and rhodamine filters.
Antitumor activity
The human pancreatic adenocarcinoma cell line Panc 03.27 (ATCC, CRL-2549; ATCC, Rockville, MD, USA) was cultured in Dulbecco’s Modified Eagle Medium/Ham’s Nutrient Mixture F-12 (DMEM/F12) supplemented with 10% fetal bovine serum (Gibco-Invitrogen, Carlsbad, California, USA), 100 U/mL penicillin, 100 µg/mL streptomycin, and 0.25 µg/mL amphotericin B (Sigma-Aldrich; St. Louis, MO) at 37°C and 5% CO2. Confluent monolayers were detached with trypsin (Gibco-Invitrogen, Carlsbad, California, USA) and washed with PBS at 500 ×g.
The cell viability of Panc 03.27 cells exposed to the various ABMs was assessed with the MTT test (Biotium, Hayward, CA, USA). ABMs were applied to the bottom of the wells of a 96-well plate using the casting/solvent evaporation method. Specifically, the following ABMs were tested: (i) BisBAL NPs-CPC-ABM (500 and 50 µM, respectively), (ii) GEM-ABM (100 µM) as a positive control for antitumor activity, and (iii) drug-free ABM as a negative control. The Panc 03.27 cell suspension (1 × 105/150 μL DMEM/F12) was added to the wells and allowed to be exposed to the ABMs for 24 h at 37°C and 5% CO2. The MTT test was performed following the manufacturer’s instructions and the OD570 was determined after MTT reduction. The test was performed in triplicate. Cell viability was expressed as a mean percentage of the negative control.
The cell morphology of Panc 03.27 cells was analyzed using the live/dead assay and fluorescence microscopy after a 24-h exposure to ABM loaded with BisBAL NPs-CPC or GEM. Panc 03.27 cells were observed with an EVOS Cell Imaging System (Thermo Fisher Scientific, CA, USA) equipped with FITC and rhodamine filters.
Statistical analysis
A one-way analysis of variance (ANOVA) was employed to analyze the data among the groups. A significance level of α = 0.05 was considered.
Results
Characterization of BisBAL NP
Consistent with previous reports, 21 BisBAL NPs were spherical with an average diameter of 19.6 nm and formed electron-dense aggregates, a common characteristic of this kind of metal nanoparticles (Figure 1). The EDX-SEM spectrum and X-ray diffraction pattern were indicative of bismuth. A size distribution histogram of BisBAL NPs informs about the dimension distribution of BisBAL NPs.
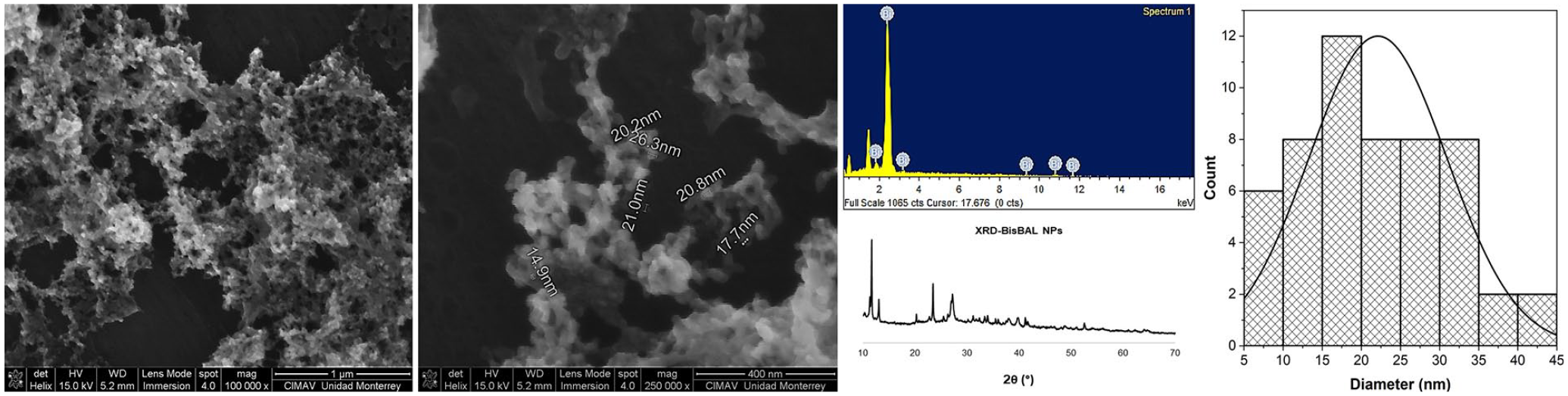
Characterization of BisBAL NP by Scanning Electron Microscopy (SEM). The size, morphology, and distribution of BisBAL NP were determined through SEM analysis. The EDX-SEM spectrum corroborates the specific presence of bismuth in the samples. The X-ray diffraction pattern confirms bismuth identity. A size distribution histogram of BisBAL NPs informs about particle dimension distribution.
Characterization of BisBAL NPs-CPC-ABM
Optical microscopy revealed a homogeneous arrangement of alginate in BisBAL NPs-CPC-ABM, contrasting with the uneven distribution observed in GEM-ABM and drug-free ABM (Figure 2, first column). SEM micrographs further confirmed the even distribution of BisBAL NPs within the ABM (Figure 2, second column). EDS-spectra of the different ABMs showed similar elemental composition (Figure 2, third column; the first two peaks represent C and O). In the case of BisBAL NPs-CPC-ABM, the presence of BisBAL NP was evident (seems that Bi replaces Ca that is present in the other ABMs) as supported by the strong peak observed in the SEM micrographs.
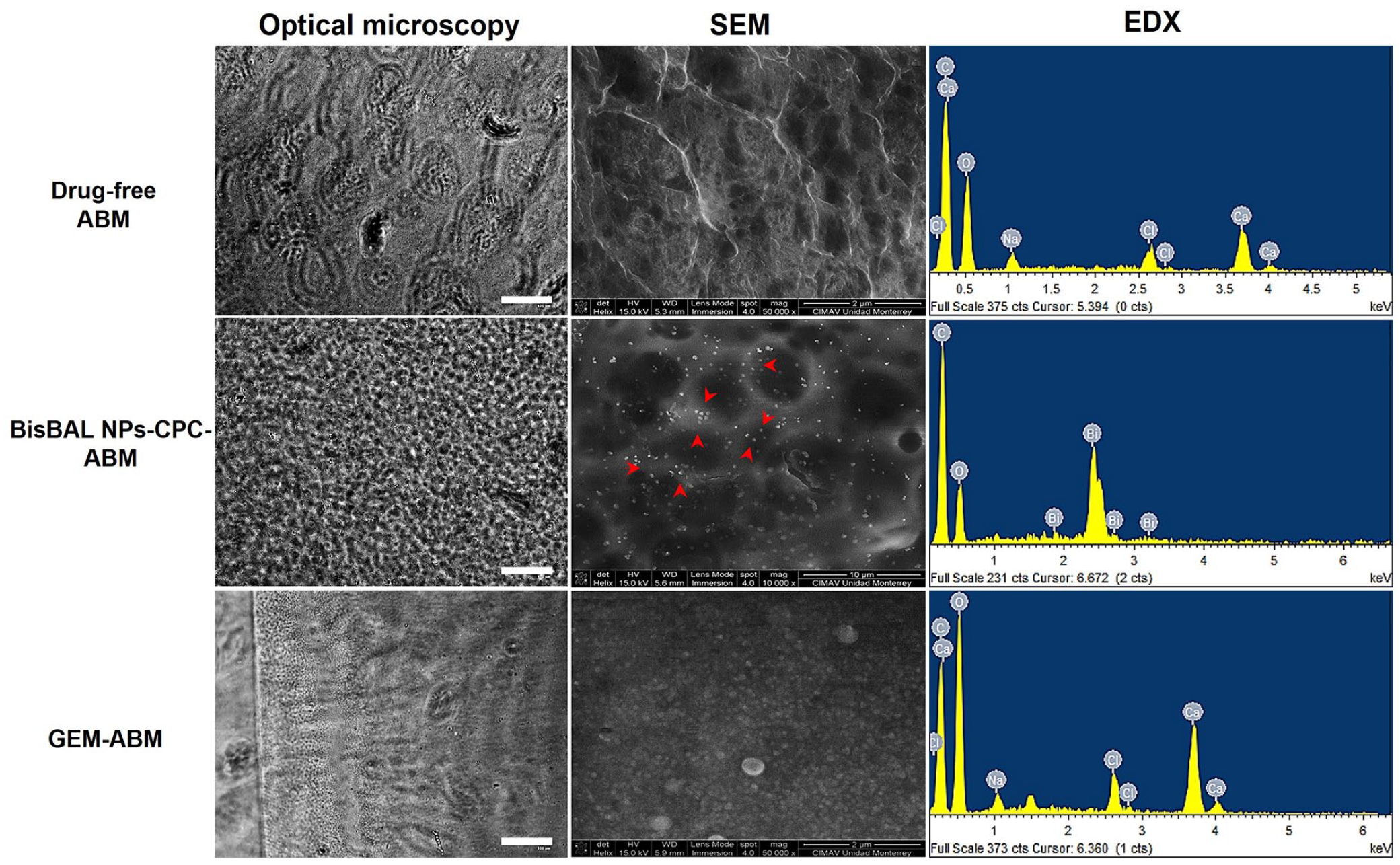
Characterization of an BisBAL NP-CPC-ABM by optical and scanning electron microscopy (SEM). The morphology of ABM varies depending on the incorporated substance. SEM image magnifications vary: BisBAL NPs-CPC-ABM at 10,000x; GEM-ABM and drug-free ABM at 50,000x.
FTIR spectra of unmodified alginate (Figure 3) showed typical absorptions at 3256 (OH), 2938 (CH), 2884 (CH), 1600 (asymmetric COO-), 1415 (symmetric COO-), 1327 (CH2 bend), 1031 (C-O-C in alcohol), and 925 cm−1. Additional absorptions were identified at 1235, 1209, and 1109 cm−1 in the spectrum of GEM-ABM, while no discernible differences were noticed in the one of BisBAL-NPs-CPC-ABM.
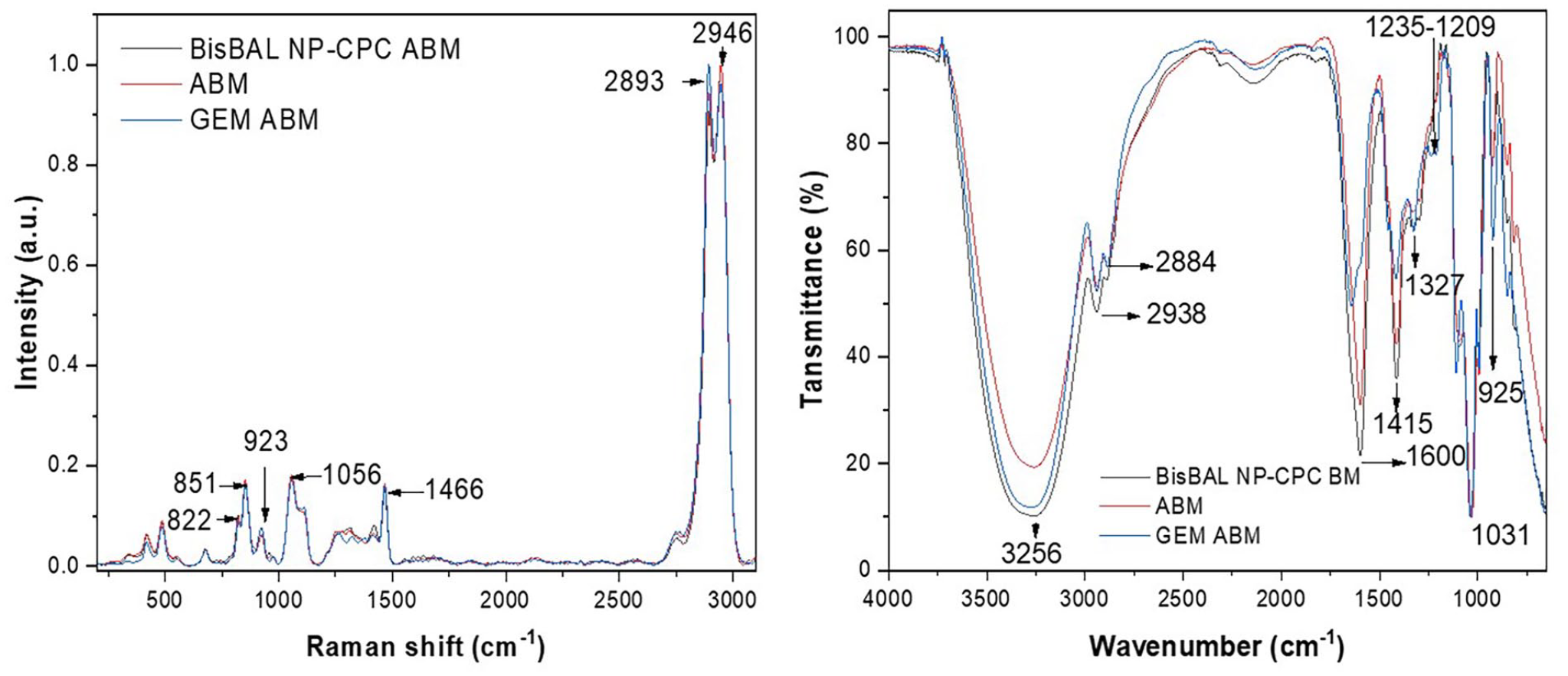
Raman and FTIR spectra of BisBAL NPs-CPC-ABM, GEM-ABM, and drug-free ABM.
Consistent with previous results, Raman spectra (Figure 3) of pristine alginate showed absorptions at 2946, 2893, 1466, 1056, 923 (C-O-C in polyguluronate) and 851 (C-C-H or skeletal C-C) and 822 (C-H) cm−1. Samples containing BisBAL NP and GEM did not show additional absorptions due to their low concentrations.
Characteristics of clinical bacterial isolates
The K. pneumoniae isolate 21-1076 was obtained from an endotracheal aspirate of a male patient in 2021 within the COVID-19 building (Figure 4). The E. faecium isolate was obtained from abdominal fluid from a male patient in 2022 from the internal medicine ward in the main hospital building (Figure 4). The Salmonella spp. isolate 21-0829 was obtained from a secretion sample of a male patient in 2021 from the internal medicine ward in the main hospital building (Figure 4). The E. faecalis sample was isolated from a patient with an endodontic infection in 2022.
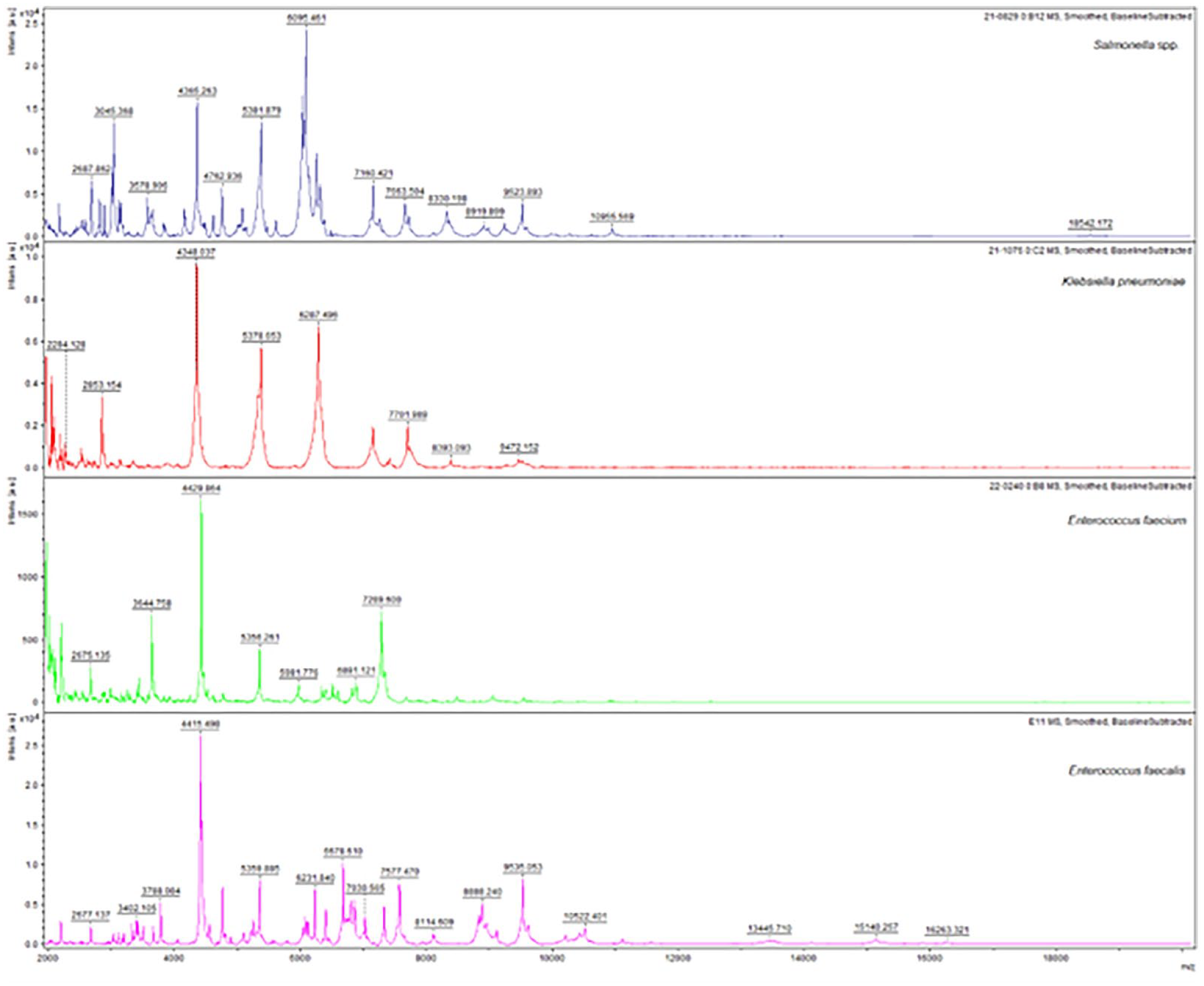
MALDI-TOF mass spectra of clinical isolates. The spectrum of each species (Salmonella spp., K. pneumoniae, E. faecium, and E. faecalis) was visualized after processing the spectra with flexAnalysis software (Bruker Daltonics).
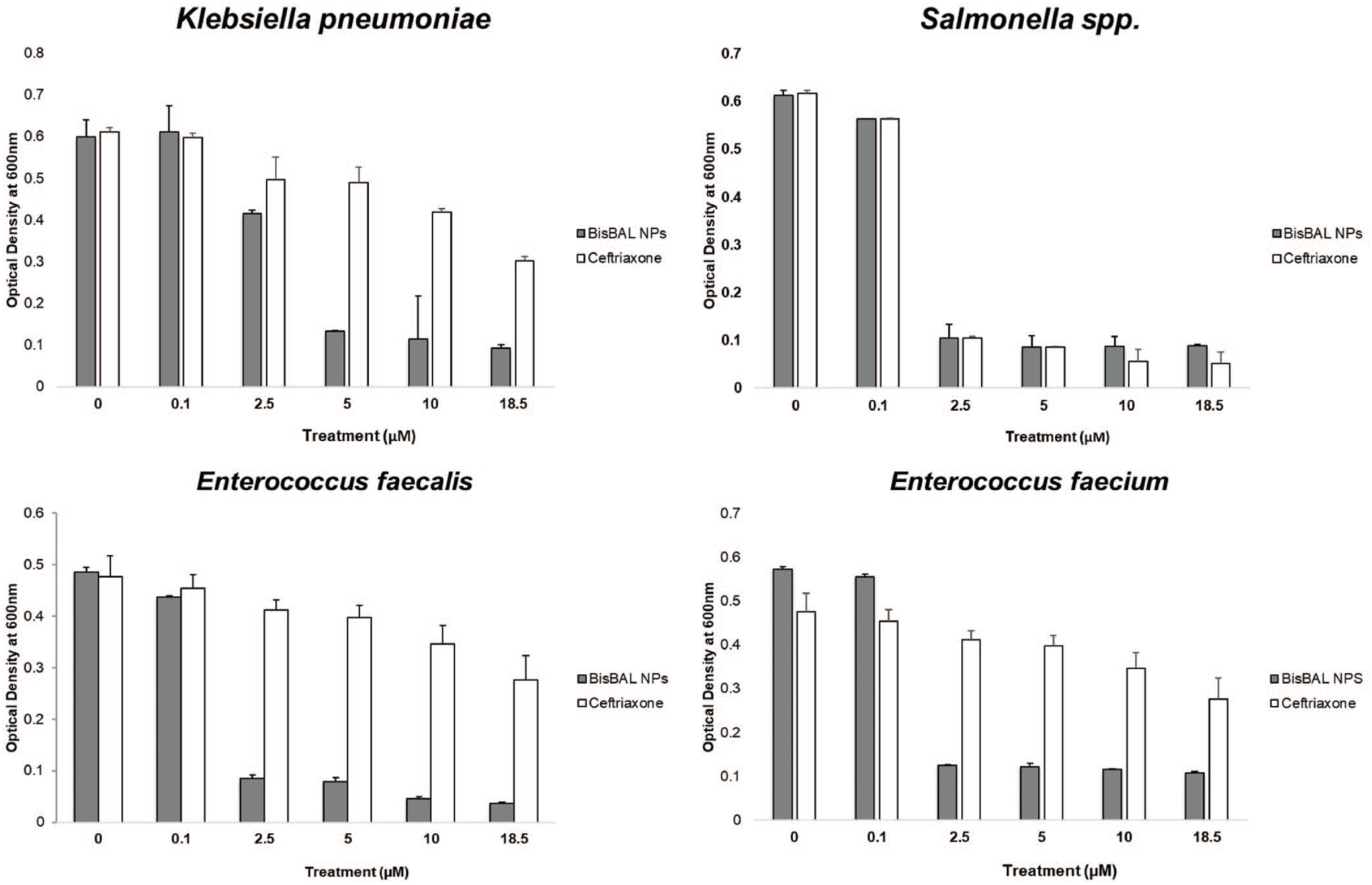
The AST drug resistance profiles showed K. pneumoniae and E. faecium were multi-drug resistant (Table 1).
Drug resistance profiles of K. pneumoniae and E. faecium clinical isolates.
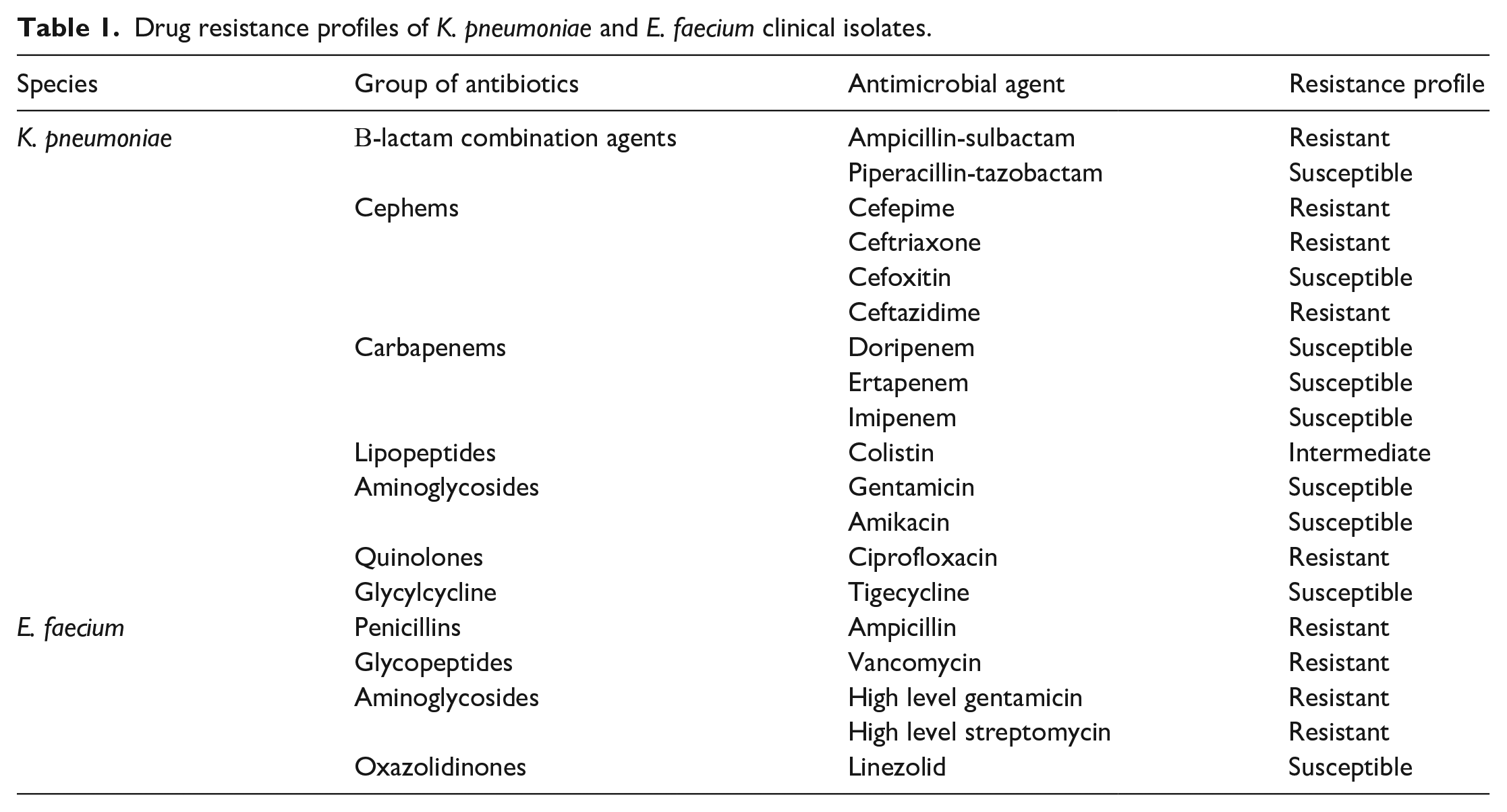
Bactericidal activity of BisBAL NPs alone and BisBAL NPs-CPC-ABM
BisBAL NPs effectively inhibited the bacterial growth of all tested microorganisms, exhibiting a superior antibacterial profile compared to the positive control ceftriaxone (Figure 5). A 24-h exposure at 37°C under aerobic conditions to 5 µM BisBAL NP resulted in a 77.9% growth reduction in multi-drug resistant K. pneumoniae, while 2.5 µM BisBAL NP was sufficient to reduce the growth of Salmonella by 82.9%, E. faecalis by 82.6%, and E. faecium by 78%; p < 0.0001 (Figure 5).
Determination of Minimal Inhibition Concentration (MIC) of BisBAL NP on multidrug-resistant isolates. Cultures of Klebsiella pneumoniae, Salmonella spp., Enterococcus faecalis, and Enterococcus faecium were exposed to BisBAL NP at concentrations ranging from 0.1 to 18.5 µM for 24 h. OD560 assessed cell suspension density. Drug-free cultures served as the growth control reference and ceftriaxone was used as a positive control of growth inhibition.
Once the antimicrobial activity of BisBAL NP alone had been demonstrated, the antimicrobial potential of BisBAL NPs-CPC-ABM was verified using disc diffusion assays. The growth inhibition halos due to BisBAL NPs-CPC-ABM (500 and 50 µM, respectively) were significantly larger (p range, 0.0004–0.0459) than the ones due to the growth inhibitor control (100 µM ceftriaxone) for all microorganisms tested, except for E. faecium (p, 0.0866) (Table 2). Altogether, these results suggest that BisBAL NP confers antimicrobial properties to the ABM.
Antimicrobial activity of BisBAL NPs-CPC-ABM.
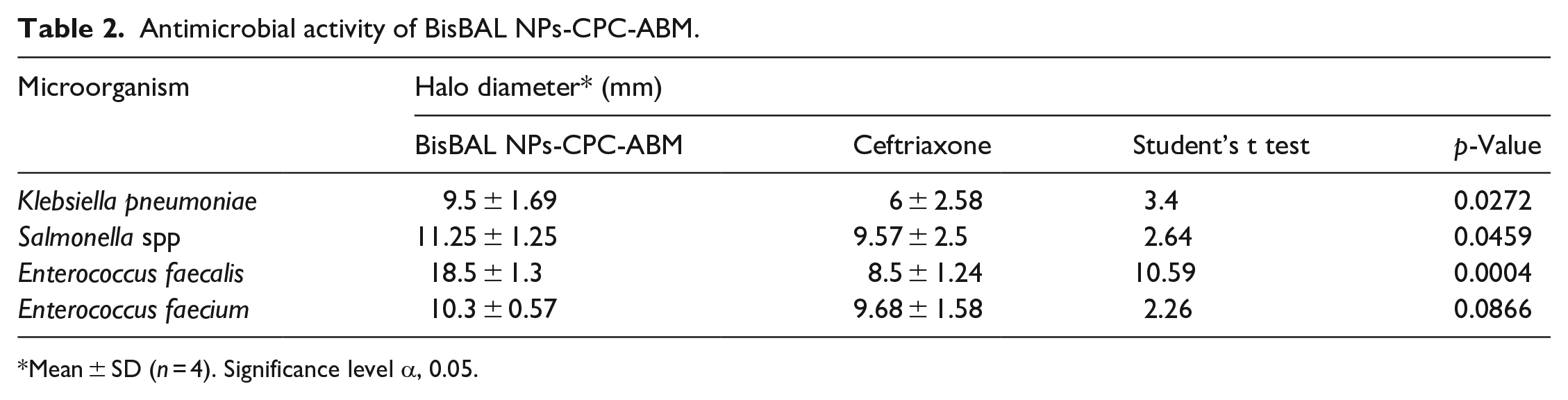
Mean ± SD (n = 4). Significance level α, 0.05.
Antibiofilm activity of BisBAL NPs-CPC-ABM
After a 24-h exposure to BisBAL NPs-CPC-ABM, most bacteria in K. pneumoniae, Salmonella, E. faecalis, and E. faecium biofilms had died (red), in contrast to the living bacteria (green) in biofilms exposed to drug-free ABM (Figure 6). Exposure to BisBAL NPs-CPC-ABM significantly diminished bacterial viability by 81.4% in the K. pneumoniae biofilm, 74.5% in the E. faecium biofilm, 97.1% in the E. faecalis biofilm, and 79.5% in the Salmonella biofilm (Figure 6). These results demonstrate that the antimicrobial activity of BisBAL NPs-CPC-ABM extends to biofilms.
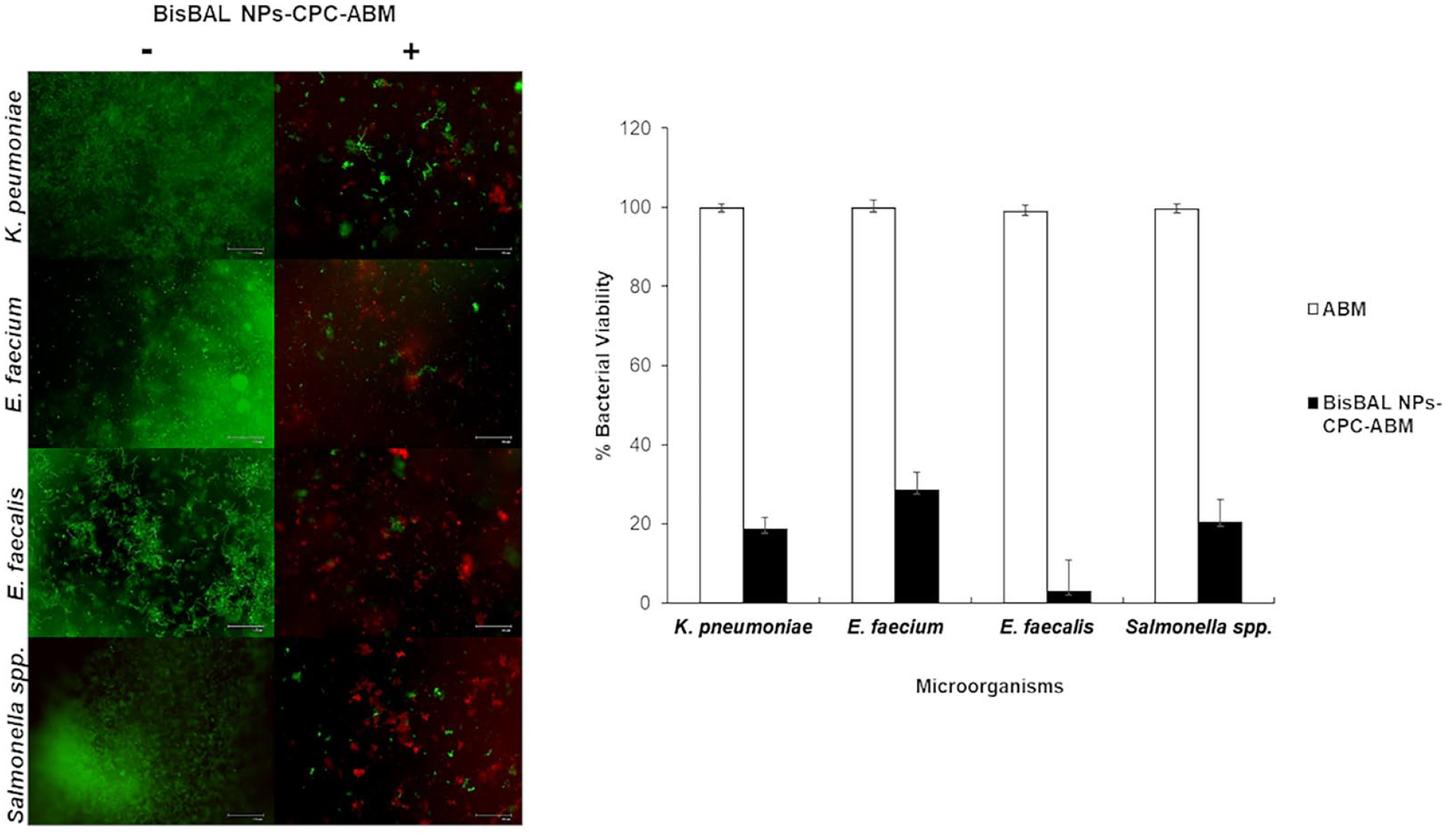
Antibiofilm activity of BisBAL NPs-CPC-ABM. BacLight viability assay in combination with fluorescence microscopy was employed to evaluate the antibiofilm activity of BisBAL NPs-CPC-ABM against Salmonella, E. faecalis, E. faecium, and K. pneumoniae biofilms in comparison to drug-free ABM.
Antitumor effect of BisBAL NPs-CPC-ABM
An MTT cell viability assay was used to evaluate the antitumor activity of BisBAL NPs-CPC-ABM on the human pancreatic adenocarcinoma cell line Panc 03.27. Solutions containing BisBAL NP-CPC (500 and 50 μM) and GEM (100 μM) exhibited equal effectiveness in inhibiting Panc 03.27 cell growth (97.3% and 97% inhibition, respectively; p < 0.0001) (Figure 7). When loaded into ABM, the antitumor activity diminished to 76% for BisBAL NPs-CPD-ABM and 73% for GEM-ABM, compared to 18% growth inhibition for drug-free ABM.
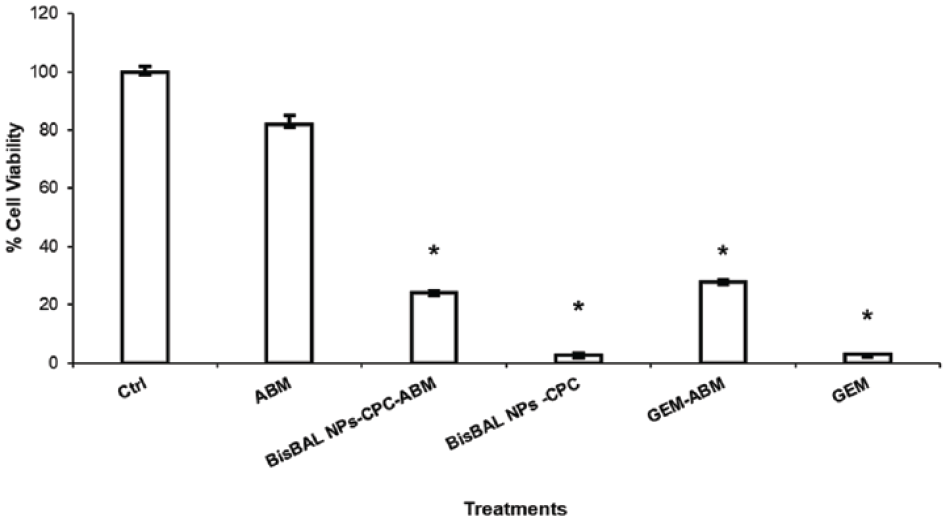
Efficacy of antitumor drugs (free or loaded within an ABM) on a pancreatic cancer cell line. Panc 03.27 cells were exposed for 24 h to BisBAL NPs-CPC solution (500 and 50 µM) and BisBAL NPs-CPC-ABM. Growth control: a drug-free culture; positive controls of inhibition: 100 µM GEM and GEM-ABM. Mean ± SD (n = 3); *, statistically significant (α = 0.05; p < 0.0001).
Fluorescence microscopy after a live/dead assay of Panc 03.27 exposed for 24 h to BisBAL NPs-CPC-ABM, GEM-ABM, drug-free ABM, and a culture not exposed to anything, revealed that the Panc 03.27 cells in the growth control and those exposed to drug-free ABM remained viable (green), while tumor cells exposed to BisBAL NPs-CPC-ABM and GEM-ABM had died (red) (Figure 8). Altogether, these results indicate a significant cytotoxicity of BisBAL NPs-CPC-ABM against tumor cells.
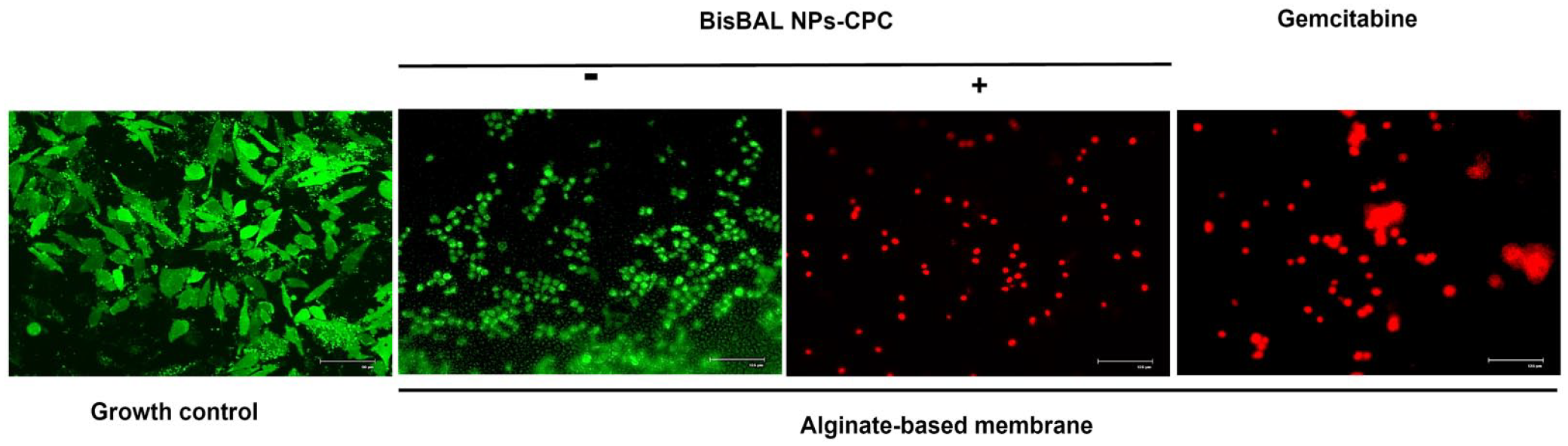
LIVE/DEAD assay and fluorescence microscopy of BisBAL NPs-CPC-ABM on pancreatic tumor cells. Panc 03.27 cells were exposed for 24 h to BisBAL NPs-CPC-ABM, GEM-ABM (positive control of inhibition), and drug-free ABM as growth control. Bar, 125 µm except for growth control: 90 µm.
Discussion
Pancreatic cancer poses challenges for conventional treatment methods. 24 Surgery is often the initial treatment option after a pancreatic cancer diagnosis. 25 However, not all patients are suitable candidates for surgery due to factors such as their overall health, cancer stage, or presence of metastasis. 26 Unfortunately, some patients who undergo surgery develop SSI. 27 SSI highly contributes to postoperative morbidity and mortality. 28 Risk factors for SSI include patient health status, procedural factors, and, notably, nosocomial bacteria. 29 Approximately 50% of pancreatic tumor extraction surgeries conducted in Mexican patients result in SSI. 18 Preventive measures against nosocomial infections, particularly SSI, include the use of biomedical items with antimicrobial and antibiofilm properties during surgery. Various nanoparticle-based delivery systems have been proposed for the diagnosis and treatment of pancreatic cancer.30,31 Shabana et al. developed a thermosensitive and biodegradable hydrogel for co-delivery of GEM and paclitaxel, which demonstrated antitumor effects on PANC-1 pancreatic cancer cells. 32 In this study, we describe the development of BisBAL NPs-CPC-ABM, a membrane with topical antimicrobial and antitumor properties, that could be applied as a coating for biomedical items to be used during cancer surgery. SEM analysis confirmed the homogeneous distribution of BisBAL NPs within the ABM. Similarly, BisBAL NPs exhibited homogeneous distribution when incorporated into the AH Plus root canal sealer and the addition of BisBAL NPs increased the antimicrobial and antibiofilm activities of AH plus-BisBAL NPs, in contrast to AH plus alone without alter the sealing properties of AH plus. 22 With respect to antimicrobial activity, BisBAL NP demonstrated growth inhibition of nosocomial isolates with MIC values of 2.5–5 μM. We previously reported a MIC value of 5 μM of BisBAL NP for both gram-positive and gram-negative bacteria, and 10 μM for Candida albicans. 21 Therefore, BisBAL NP may possess higher antimicrobial potency than silver nanoparticles, which exhibited a MIC value of 29 μM against methicillin-susceptible and methicillin-resistant Staphylococcus aureus. 33 Some of the nosocomial isolates employed in this study displayed multidrug resistance, including resistance to ceftriaxone and vancomycin. Notably, vancomycin-resistant enterococci are considered a serious threat in nosocomial septicemia. 34 The results of the disc diffusion assays showed that BisBAL NPs-CPC-ABM induced larger growth inhibition halos than the ones of the positive control of growth inhibition, also against the multidrug-resistant isolates such as K. pneumoniae and E. faecium. The difference was statistically significant for all isolates, except E. faecium. Previously it was reported the antimicrobial effect of BisBAL NPs against gram-positive and gram-negative bacteria, 21 however in this study is presented evidence about growth inhibition of multiresistant isolated bacteria by alginate-based membrane supplemented with BisBAL NPs. The presence of BisBAL NPs in alginate membranes will help to minimize the appearance of SSI.
Bacterial biofilms tend to increase bacterial resilience to stress, including exposure to antibiotics, compared to individual bacterial growth forms. Importantly, BisBAL NPs-CPC-ABM also exhibited bactericidal activity against biofilms formed by all isolates. Altogether, these findings support the broad-spectrum microbicidal activity of BisBAL NPs, which is preserved when incorporated into the BisBAL NPs-CPC-ABM membrane. Furthermore, the ABM appears to be suitable vehicle for delivering BisBAL NP.
When evaluating the antitumor effect of BisBAL NPs-CPC-ABM on pancreatic tumor cells, BisBAL NPs-CPC-ABM, it was found that BisBAL NPs-CPC-ABM exhibited effectiveness equal to the positive control of inhibition GEM-ABM, whereas ABM alone was hardly antitumoral. This demonstrates that ABM can serve as an effective vehicle for delivering antitumor drugs. Alginate membranes and other natural polymers, such as chitosan and its derivatives, show promise as drug delivery vehicles in cancer treatments.35–38 Recently, biomimetic gold nanorods (AuNRs) modified with erythrocyte membranes were successfully used as vehicles for siRNA delivery to inhibit pancreatic cancer cell growth. 39
Our group has previously reported on the antitumor activity of a biodegradable hydrogel loaded with BisBAL NP on human cervicouterine, prostate, and colon cancers. 15 Additionally, we have described the cumulative antitumor effect of BisBAL NP and CPC (in a 1:1 ratio; 50 μM mixture) against human lung cancer cell (A549). 17 The antitumor activity of BisBAL NPs-CPC-ABM was confirmed through a live/dead assay in combination with fluorescence microscope, which showed that Panc 03.27 cells exposed to BisBAL NPs-CPC-ABM died, while cells exposed to drug-free ABM did not. In this study the action mechanism was not tackled, but we have previously described that both BisBAL NP and CPC alter cell membrane permeability and alter the microtubule network of tumor cells.17,40 Collectively, these results provide evidence of the potential of BisBAL NPs-CPC-ABM as an effective antitumor treatment.
Conclusion
ABM appears to be a promising drug delivery vehicle, enabling a uniform distribution of the incorporated drugs. BisBAL NPs-CPC-ABM demonstrates broad-spectrum bacterial grown inhibition, including nosocomial multidrug-resistant, and demonstrates antitumor efficacy. This makes BisBAL NPs-CPC-ABM a potential candidate for topical antimicrobial and antitumor treatment. Consequently, it holds potential to reduce the occurrence of SSI in cancer patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors want to thank the Consejo Nacional de Humanidades, Ciencias y Tecnologías (CONAHCYT) for the grant provided.
