Abstract
Infections of implants pose a severe problem in the field of orthopedic surgery, because they can cause bone degradation with subsequent loosening of the implant. The discrimination between septic implant loosening and aseptic loosening can be a challenge, and hence novel diagnostic methods have been introduced to improve the detection of bacteria. Because a major problem is their firm adherence to implants due to biofilm formation, sonication has been introduced, followed by identification of bacteria by culture or genetic methods. In this study, we compared the results obtained after sonication pretreatment with those of microbiological testing of tissue samples and histopathological evaluation of the same tissue. Furthermore, we related the results obtained following sonication to the clinical diagnosis of septic or aseptic implant loosening, respectively. Sonication of explanted devices also enhances the likelihood of detecting bacterial growth in patients who were considered “aseptic” based on the clinical evaluation.
Introduction
Implant-associated infections are among the most feared complications in the field of orthopedic surgery. Although the incidence of infection of an endoprosthesis is about 0.5% to 1.5% – increasing up to 5% in revision implants and up to 10% in tumor implants (1) – considering the high numbers of total joint replacements that are performed every year, infections pose a severe problem because patients usually require protracted and extensive treatment (reviewed in 2-8). This, above all, represents a tremendous psychological strain on patients and is also associated with high socioeconomic costs (9). Implant-associated osteomyelitis is the prototype of an infection caused by bacterial biofilms, which colonize the implanted materials. Because the bacteria are relatively resistant to numerous antibiotics, the condition is difficult to treat (10-15). The infection elicits a profound and persistent local inflammatory immune response, which is thought to eventually participate in tissue degradation and bone resorption (reviewed in (16)).
Despite clinical signs of infection, bacteria are not always detected by the standard microbiological tests, making it difficult to differentiate between aseptic or septic loosening. In implant-associated osteomyelitis, about 40% of the cases test “negative” for bacteria (4, 17-19). Therefore, improved methods for the detection of bacteria have been introduced. Based on the observation that bacteria adherent to implants can be removed in a vital form by ultrasonication (20), this method was applied to the diagnostic of bacterial colonization on the surface of implants (21, 22). In our study, we evaluated the diagnostic benefit of the sonication method compared to the routine microbiological testing of tissue samples and to histopathological examination of the same samples.
Materials and Methods
In vitro experiments
To pretest the sonication procedure,

Biofilm formation on titanium discs. After being cultivated with bacteria, biofilm formation was seen after 24 h and after staining with Mira-2-Ton® (left). The disc without biofilm and after staining with Mira-2-Ton® is shown on the right.
Patients and Patient-derived Material
Explanted devices of 87 patients undergoing surgery because of aseptic or septic loosening were included in the study (35 knee prostheses, 49 hip prostheses, 3 shoulder prostheses). There were 44 males and 43 females. Diagnosis of a loosening was based on patient's complaints, clinical examination, and by conventional x-ray and/or CT-scan. The diagnosis of septic loosening was based on clinical evaluation (reddening, swelling, hyperthermia, pain, pus intraoperatively, existence of a sinus tract) and laboratory results (elevated CRP levels and white cell count). The criteria for a septic loosening were either a sinus tract, pus intraoperatively or at least three positive signs mentioned above. During surgery, two to four tissue samples were collected from around the implant and sent to the microbiology department and the pathology department for examination. The study was approved by the local ethics committee, and informed consent was obtained from the patients.
Sonication of implants
The explanted prostheses were placed into sterile plastic bags in the operating room. Enough Ringer solution to cover the implant was added (400 mL). The plastic bag was sealed and placed in a second plastic bag. The implants were thus placed in the ultrasound bath (Ultrasonic TI-H 20; Elma, Singen, Germany) and treated with various settings of the ultrasonic power and time: with 100% (250 W) for 5 min, 100% (250 W) for 1 min, 70% (175 W) for 5 min or 10% (25 W) for 5 min. The ultrasonic frequency of 45 kHz was used at all times. From 50 samples, the entire Ringer solution was centrifuged at 5000 rpm for 10 min, resuspended in 20 mL Ringer solution and divided into an aerobic and anaerobic blood culture bottle (BD Bactec plus aerobic/anaerobic; Becton Dickinson, Franklin Lakes, NJ, USA). From 37 samples 20 mL were taken directly from the Ringer solution without adding a centrifugation step and divided into an aerobic and an anaerobic blood culture bottle. The bottles were incubated at 36°C until positive or for a maximum of 14 days in a BACTEC Fx (BD). Positive bottles were subcultured on Columbia 5% sheep blood agar (BD), chocolate agar, MacConkey agar, and SCS agar (all bioMérieux, Craponne, France). Bacteria were identified by MALDI-TOF mass spectrometer (Microflex; Bruker Daltonics, Billerica, MA, USA). Susceptibility testing was done using VITEK® 2 microbial identification system (bioMérieux).
Diagnostic procedures
Tissue samples were processed as follows. After arrival at the lab, the tissue was ground using a porcelain mortar, followed by the addition of 1 mL of 0.9% NaCl. This suspension was inoculated onto Columbia 5% sheep blood agar (BD), chocolate agar, MacConkey agar, SCS agar, SNVS Agar (all bioMérieux), Thioglycolate broth (BD) and then gram-staining was performed. Plates and broth were incubated until positive or for a maximum of 5 days at 36°C in 5% CO2 or under anaerobic conditions. Identification of bacteria was done with MALDI-TOF mass spectrometer (Microflex; Bruker Daltonics). Susceptibility testing was done using the VITEK® 2 microbial identification system (bioMerieux).
For the histopathological diagnostics, the samples were fixed in formalin, embedded in paraffin, and after cutting slices of 2 μm thickness, routine hematoxylineosin staining was performed. The criterion for an implant-associated infection was at least 23 polymorphonuclear neutrophils per 10 high-power fields (23, 24).
Statistical test
The concordance of the results of the three diagnostic methods was calculated using Cohen's Kappa test, and the interpretation of the test was as published in (25).
Results and Discussion
Establishing sonication conditions using in vitro-formed biofilms
To assess the efficiency of sonication procedures with regard to removal of bacterial biofilms and the survival of bacteria, a series of in vitro experiments was performed. Biofilms were grown on titanium discs using radiolabeled bacteria for quantification. Establishment of biofilm was confirmed by staining with Mira-2-Ton® (Fig. 1). Different settings were used and it was found that approximately 70% of the bacteria could be removed within 1 to 5 min of sonication with a setting of 100%, and that the efficiency of removal declined with lower settings. With a setting of 70%, 57% of the bacteria was removed, and at 10% only 25% was removed. Mere shaking of the implant removed only 5% (all values are the average of repeated experiments using a total of 5 discs). Following sonication, the bacteria were harvested from the fluid and plated on blood agar to assess their survival. Under all our experimental conditions the bacteria survived, which means that a failure to detect bacteria following sonication is not due to cell death or reduced growth rate.
Evaluation of the diagnostic methods for implant infection
Seventy-seven patients were studied for the evaluation of diagnostic methods for implant infection. For each patient, conventional microbial diagnostic of tissue samples was performed, as was analysis of bacterial growth after sonication of the removed implant, and histopathological examination of tissue samples. The agreement or non-agreement of the results was determined using Cohen's kappa test. The conventional microbial diagnosis and the results after sonication coincided in 19 cases that tested positive, and in 30 cases that tested negative. This is with κ=0.289 a fair agreement only (data summarized in Tab. I). A moderate agreement was seen between both the microbiological tests and the histopathological evaluation (κ=0.441 and κ=0.401, respectively) (Tab. I). In agreement with published data by others, the detection of bacteria was considerably better following sonication of the implant as opposed to conventional microbial testing and is in line with the presumption that bacteria forming biofilms on implants might be missed if not removed from the implant (20). Again, also in agreement with earlier data by others, the histological diagnostic was not very sensitive (26, 27). The slightly better agreement of the histopathological findings with the microbiology testing following sonication as opposed to testing tissue samples (κ=0.441 vs. κ=0.401) suggests that these bacteria cause inflammation and thus are likely to participate in implant loosening.
DIAGNOSTIC METHODS FOR IMPLANT INFECTION: ESTIMATION OF AGREEMENT (n=77)
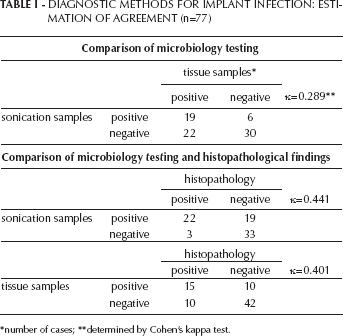
number of cases
determined by Cohen's kappa test.
In 48 patients, a joint aspiration was done pre-operatively. Of those, 15 tested positive, which was in substantial agreement with testing of tissue samples (κ=0.667), but only in moderate agreement with the results obtained following sonication of the implants (κ=0.443) (data summarized in Tab. II). These results again underline the fact that because of firm adhesion to the implant, biofilmforming bacteria escape conventional detection methods, and indicates that pre-operative joint aspiration, a commonly used procedure to distinguish between septic and aseptic loosening, is effectual.
AGREEMENT OF MICROBIAL TESTING OF PRE-OPERATIVE JOINT ASPIRATION WITH SONICATION SAMPLES AND TISSUE SAMPLES (n=48)
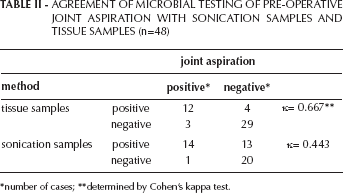
number of cases
determined by Cohen's kappa test.
Relationship of the diagnostic results to the clinical data
Next, we tested whether the three diagnostic methods were consistent with the clinical diagnosis of “septic” versus “aseptic implant loosening.” The conventional microbial testing was in best agreement with clinical data (κ=0.671), the results after sonication showed the least agreement, (κ=0.513, moderate agreement) (data summarized in Tab. III). The most likely explanation is that in some patients the bacterial biofilm on the implant causes only mild or atypical symptoms, and consequently the patients are falsely classified as “aseptic.” Persistent infections – even when mild –could still result in gradual loosening of the implant, because a proinflammatory environment is generated (see histopathological findings), which then promotes the generation of bone-resorbing osteoclasts. These findings also emphasize the difficulty of clearly differentiating between septic and aseptic loosening based on clinical data only.
AGREEMENT OF DIFFERENT DIAGNOSTIC METHODS WITH THE CLINICAL EVALUATION (n=77)
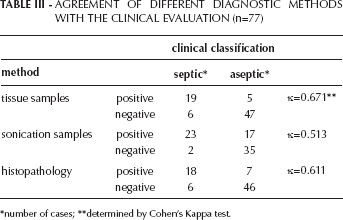
number of cases
determined by Cohen's Kappa test.
Of the patients included in the study, 24 had already previously suffered from a septic/infectious incidence of the joint prosthesis. In these patients, 17 tested positive following sonication, but only 13 by conventional methods. Moreover, when patients had received antibiotic treatment prior to surgery (n=11), bacteria were still detected after sonication in 10, but only 4 by conventional testing of tissue samples. This result is in line with the observation that bacteria organized in biofilms acquire resistance to numerous antibiotics.
Identification and location of the bacteria species
For this study, 90 explanted devices were used. Following sonication, in 48 cases bacteria of various species were found, whereas by conventional testing only 27 tested positive (Tab. IV). When the two methods both tested positive (in 19 samples) the same bacteria species was detected; only in 2 samples were different bacteria found. With regard to the species that were detected, there was no significant difference, which was not really unexpected, considering the wide variety of species versus the relatively small number of cases.
IDENTIFICATION OF BACTERIA SPECIES
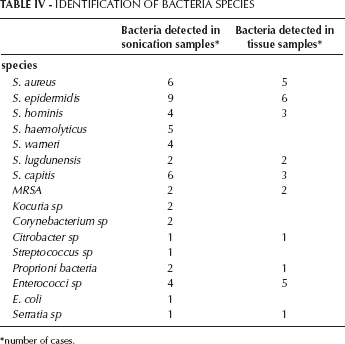
number of cases.
We also investigated whether there were differences in the bacterial species causing implant infections at knee, hip, or shoulders. As summarized in Table V, no significant differences were seen, which, again, is not surprising due to the small number of cases (Tab. V).
BACTERIA SPECIES AT DIFFERENT LOCATIONS
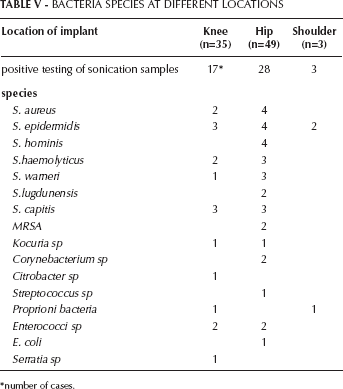
number of cases.
Conclusions
Sonication of explanted devices enhances the likelihood of detecting bacterial growth. The obvious explanation is that bacteria form biofilms on the implants. These sessile communities are difficult to detect in swabs or tissue samples, because they adhere strongly to the implant, and because they might not readily adjust to growing under culture conditions. Thus, as a diagnostic method, microbial testing after sonication allows a better discrimination between “septic” and “aseptic” loosening, and helps to decide whether the loosening is indeed aseptic (28). Even though the diagnosis is only possible “after the fact,” it is still worthwhile, because of the risk of re-infection for the newly applied device and the possibility of altering or prolonging the postoperative antibiotic treatment according to the detected bacteria. The relevance and significance of clinically unapparent infections with regard to implant loosening is difficult to assess. Because bacteria are also found on implants that are removed routinely – without any hint of infection or implant loosening (29, 30) – one might presume that these are harmless. We think that it is the persistence of the biofilm that eventually elicits an inflammatory response; as a self-perpetuating process, this response gradually alters the microenvironment toward a proinflammatory
Footnotes
Acknowledgements
The titanium discs were kindly provided by Adler Ortho, Milan, Italy.
