Abstract
The incorporation of drugs and bioactive compounds in the natural rubber latex (NRL) matrix has been an alternative for the development of transdermal release membranes. Ibuprofen (IBF) is known to be used to treat inflammatory diseases, but when administered orally, high concentrations can cause some adverse problems. In this work, the incorporation of IBF in the NRL membranes was evaluated by physical-chemical, in vitro permeation, hemocompatibility and molecular modeling assays. In addition, the in vitro release profile of IBF in acid and basic media was analyzed during 96 h. The IBF-NRL membrane exhibited the absence of intermolecular bonding that could hinder drug release and presented compatible mechanical properties for applications as a cutaneous adhesive (0.58 and 1.12 MPa to Young’s modulus and rupture tension, respectively). The IBF-NRL system did not present a significant hemolysis degree (1.67%) within 24 h. The release test indicated that in the first hours of the study, 48.5% IBF was released at basic pH and 22.5% at acidic pH, which is characteristic of a burst effect. Then, a stable release profile was observed until the end of the assay, with total IBF release of 60% in alkaline medium and 50% in acidic medium. The drug permeation results indicated that the IBF-NRL membranes can be used for the local skin treatment with permeation of 3.11% of IBF. Dynamic Molecular simulations indicated a pronounced electric dipole in the ionized form of IBF, which suggests a more effective interaction with water, explaining the efficient drug release in alkaline solutions. In general, the results demonstrate that the IBF-NRL membrane has great potential for a new adhesive that can be used for the treatment of inflammatory processes and injuries.
Introduction
Inflammation or inflammatory response can be defined as a complex set of physiological events which promote protection of the body tissues and organs. Such a cascade event acts by restricting damage at the site of aggression, although it can have deleterious effects when very intense.1–4 In the United States, inflammatory diseases and related syndromes affect approximately 60 million people. A study conducted by Wylezinski et al. 5 elucidated the prevalence, clinical characteristics, challenges and financial impacts of four inflammatory diseases and two related syndromes that had an estimated cost of $90 billion in 2018.
Treatment of inflammatory diseases consists of the administration of non-steroidal anti-inflammatory drugs (NSAIDs), such as ketoprofen, naproxen, celecoxib, indomethacin, mefenamic acid, etoricoxib, ibuprofen (IBF), and diclofenac, which are often used to reduce inflammation and relieve pain. In addition to this medicines class, there are many other options as corticosteroids (prednisone, hydroxychloroquine), oral drugs known as DMARDs (disease-modifying antirheumatic drugs), that including methotrexate, sulfasalazine, leflunomide, azathioprine, and cyclophosphamide, besides biologic drugs (infliximab, etanercept, adalimumab, certolizumab, golimumab, abatacept, tocilizumab, and rituximab).6–8
NSAIDs belong to a wide class of heterogeneous compounds, containing one or more aromatic rings attached to functional acids. The main mechanism of action of NSAIDs is associated with the inhibition of cyclooxygenase (COX) and a consequent reduction in the conversion of arachidonic acid into prostaglandin.9,10 These drugs have a broad range of therapeutic indications, such as analgesia, anti-inflammation, antipyresis, prophylaxis against cardiovascular diseases, chronic pain and have part of the most commercialized drugs around the world.11–14
Toxic effects of NSAIDs are well-known when administered over longer periods of treatment or at higher doses. Schwaiger et al. 15 observed hyaline droplet degeneration of tubular epithelial cells and the occurrence of an interstitial nephritis of the fish kidney when exposed to diclofenac. Lelièvre et al. 16 reported the development of cardiovascular shock, hypocoagulability and thrombopenia in a case of a 58-year-old woman who ingested 26 g of naproxen in a suicidal attempt. Walsh et al. 17 showed the increased prevalence of abomasal ulceration in Holstein calves when treated with IBF (10-day course). Ungprasert et al. 18 showed that IBF appears to be the preferred NSAIDs based on its favorable gastrointestinal and nephrotoxicity profiles.
IBF, (RS)-2-(4-(2-methylpropyl)phenyl) propanoic acid is derived from propanoic acid, being indicated for the treatment of inflammation, pain and fever, by inhibiting activity of the enzyme cycloxygenase. 19 However, when administered orally, it can trigger adverse problems such as stomach pain, vomiting, diarrhea and several other gastropathies. 20
In order to avoid these misfortunes, drug delivery systems (DDSs) are interesting strategies to deliver a compound more selectively to a specific site in the human body. DDSs can influence the pharmacokinetic profile, dose, 21 speed of release, 22 location and duration of the drug action, even influencing the resulting side effects.
Manoukian et al. 23 showed that supersaturations, microemulsions, nanosystems, and microneedles are drug delivery vehicles regularly employed to minimize these adverse problems. Moreover, chitosan, pectin, alginate, poly(ε-caprolactone), poly(methyl methacrylate), poly(lactic acid) can be used as matrices for DDSs due to biocompatibility and easy handle.24–29
Nowadays, natural rubber latex (NRL) membranes have been used as matrix due to their angiogenic activity and debridement potential, besides being biocompatible and effective for wound healing. This biopolymer is extracted from the rubber tree Hevea brasiliensis and contains 45% weight of dry rubber (cis-1,4-polyisoprene), 50% of water and 5% of proteins, carbohydrates, lipids and other non-rubber materials.30,31 The NRL membranes have been applied in chronic ulcers, 32 replacement of cervical esophagus in dogs, 33 bone repair, 34 repair diaphragmatic defects in dogs, 35 and tympanum repair. 36
Moreover, NRL has been used as a matrix in the release of drugs, peptides, molecules, proteins and vegetable extracts because with simple changes in the material morphology, it is possible to change the release time and the quantity released. Aielo et al. 37 demonstrated the release of sodium diclofenac in NRL membranes, increasing the typical release time of oral tablets from 2–3 to 74 h. Floriano et al. 38 showed that ketoprofen loaded NRL membranes have no hemolytic effects and released 60% of the ketoprofen incorporated in 50 h. Barros et al. 39 developed diclofenac potassium transdermal patches based on latex matrix, where the drug was released for up to 216 h. However, to the best of our knowledge, there are no reports of the incorporation of IBF in latex matrix for sustained release.
In this work, IBF was incorporated into the NRL membranes aiming to define a new biocompatible adhesive for the treatment of inflammatory processes with reduction of adverse effects. For this purpose, hemocompatibility in red blood cells and drug release processes were evaluated. In addition, physical and chemical characterizations of the membranes were carried out via mechanical resistance tests, Fourier Transform Infrared Spectroscopy and Scanning Electron Microscopy. As a novelty of this research, molecular modeling studies will be presented for the first time, aiming to investigate relevant interactions and local reactivities of IBF and the NRL-model system. The results evidence the potential of the new adhesive for sustained drug release, the membrane also presented low toxicity and appropriate mechanical properties that allow its application as cutaneous adhesives.
Methods
Materials
NRL was purchased from BDF Rubber Latex Co. Ltd. (producer and distributor of concentrated NRL, Guarantã, Brazil). Standardization of this material is obtained from the mixture of two clones: RRIM 600 and PB 235. After extraction, the latex is kept in a liquid phase through the addition of ammonium hydroxide (pH 10.2). The samples were centrifuged at 8000 rpm to eliminate high molecular weight proteins responsible for undesired allergic and cytotoxic reactions. 40
The raw material of IBF (powder, C13H18O2, and 99.1% purity) was obtained from Vasudha Pharma Chem Limited. The IBF was macerated for homogenization and solubilized in NaOH (Sigma-Aldrich®, Germany) 0.1 mol/L (pH 12.7) at 25°C.
Membrane preparation
The drug was incorporated by mixing 4 mL of NRL with 1 mL of alkaline IBF solutions (20 mg/mL). The resulting solutions were deposited in Petri dishes and kept at room temperature for the drying process during a period of 72 h to guarantee full polymerization (IBF-NRL membranes). The chosen IBF concentration is compatible with previous studies involving anti-inflammatory release systems. The NRL membrane sample without incorporating IBF was also prepared under the same conditions.
Membrane characterization
The presence of functional groups and possible interactions between NRL and IBF compounds were investigated by Fourier Transform Infrared spectroscopy (FTIR). The samples were measured by Attenuated Total Reflection method (ATR) using a TENSOR 27 (Bruker®, Germany), equipped by diamond crystal, in the region between 4000 and 400 cm−1 (source: HeNe laser; detector: DLaTGS) with 4 cm−1 resolution and 32 scans.
The mechanical properties of the rectangular membrane samples with 60 length, 25 width, and 1 mm thickness were evaluated by tensile tests, carried out on a DL-2000 (EMIC®, Brazil) testing machine. For this purpose, it was employed a 50 kgf load cell at a speed of 500 mm/min (according to ASTM D412) until the failure at room temperature (25°C). The Young’s modulus was calculated from the initial linear response (0%–5% elongation). The experiments were performed in triplicate.
Membranes surfaces were examined at 1000X and 5000X magnifications using High-Resolution Scanning Electron Microscope (FEG-SEM; JEOL®, model 7500F, Japan) with 2 kV, take-off angle of 35° and gold as conductor material. The evaluated areas were randomly selected.
In vitro release of ibuprofen
To evaluate the IBF release, the IBF-NRL membranes were submerged in 100 mL of phosphate buffered saline (PBS) solutions (pH 7.4 and 5.6), from which aliquots were collected during an interval ranging from 0 to 96 h. After each measurement, the aliquots were returned to the solutions in order to keep the total volume of the samples. Analyses at different pH were conducted to mimic the release profile in injured (pH 7.4) and healthy (pH 5.6) skin. 41 The drug released was monitored by spectrofluorometer (λexc = 263 nm; λemi = 288 nm; 20 kW; PerkinElmer®, model LS 45, USA). The experiments were carried out in triplicate. The values of the constants A1 and A2 and the characteristic times t1 and t2 were calculated using the OriginPro8® software (Massachusetts, USA) from the ExpDec 2 fitting function.
Franz diffusion cell assays
In vitro permeation assays of IBF-NRL membranes (content 35.38 µg/cm2 of drug) were conducted in a Franz’s cells with diffusion area of 1.77 cm2 (Marte Mag Multi 6). Acetate cellulose membranes (0.45 mm, UNIFIL, São Paulo, Brazil) were placed between donor compartment (7 mL) and Franz’s cell receptor, keeping IBF-NRL membranes in contact with receptor solution (PBS). Different aliquots (2 mL) were collected during an interval of 0 to 24 h and the permeation values of IBF were determined in a spectrofluorometer (PerkinElmer®, model LS 45, USA) at λemi = 288 nm. PBS solution (pH 7.4) was utilized as blank in this experiment. The experiments were conducted in triplicate, at 37°C and constant magnetic stirring (300 ± 0.2 rpm). Subsequently, the equation below was used to calculate the amount of permeated IBF.
Where Qreal, t is the amount of permeated drug at time t, Ct is the concentration obtained relative to time t, Vr represents the receptor solution volume (7 mL), Cc is the concentration of previous sampling and Vc is the volume of the collected sample plus the dead volume. 42
Molecular modeling
To interpret the interaction between IBF molecules and NRL, local reactivity parameters of IBF and a NRL model system were evaluated. Given the pKa of IBF (between 4.91 and 5.20), the calculations were conducted considering non-modified and dissociated (ionized) structures of the drug. Preliminary conformational studies were carried out to define an oligomeric model for polyisoprene containing 10 units (NRL model system). For this purpose, 50 distinct conformers were generated via molecular dynamics (MD) simulations at high temperature (1000 K) 43 using AMBER classical force field 44 with the aid of Gabedit computational package. 45 All the structures were pre-optimized employing a PM6 semiempirical Hartree-Fock method 46 and the most stable conformer was selected for subsequent studies.
The ground state geometries and electronic properties were evaluated in a DFT approach using Becke, 3-parameter, Lee-Yang-Parr exchange-correlation functional (B3LYP)47–50 and the 6-31G valence double zeta basis set on all the atoms. The presence of the solvent (water) was simulated via polarizable continuum method (PCM). 51 All the calculations were carried out with the aid of the Gaussian 09 computational package. 52
The reactivity of the systems was evaluated via condensed-to-atoms Fukui Indices (CAFIs)53,54 and molecular electrostatic potentials (MEP) maps. CAFIs define relevant molecular sites for soft-soft interactions, involving the frontier molecular orbitals of the compounds and allow to identify the most reactive sites in relation to electrophilic (f–) and nucleophilic (f+) agents.55–57 MEP maps show electron rich and electron deficient regions on the molecules, which are relevant for hard-hard interactions via electrostatic effects.
The CAFIs were estimated via finite differences of the atomic electron populations of neutral and charged systems.55–57 To avoid negative indices, the electronic populations were estimated via Hirshfeld’s partition charge method.58,59 The MEP color maps were designed via atomic charges coming from Chelp scheme. 60
Hemocompatibility assay
Hemolytic activities of the NRL and IBF-NRL membranes were investigated according to Cesar et al. 61 using sheep blood cells with modifications. 5 mL of sheep blood was used with 3.8% (w/v) of sodium citrate (Sigma-Aldrich®, Germany) as an anticoagulant (in the proportion of 9:1, blood:citrate). The mixture was twice washed with 0.9% saline solution (w/v) and centrifuged at 1000 rpm for 10 min, discarding the supernatant.
Membrane samples (6 mm diameter) were incubated in direct contact with 1 mL of 5% erythrocyte solution (v/v, in 0.9% saline solution (w/v)) for 24 h at 37°C. Negative and positive controls (NC and PC) were designed by considering solutions without membranes (NC – 0% hemolysis) and with Triton X-100 detergent (PC – 100% hemolysis). After centrifuging (1000 rpm for 10 min), 100 µL of the supernatant was transferred into a 96-well microplate and the optical densities (OD) at 540 nm were measured in an Epoch Microplate Spectrophotometer (BioTek®, USA). Less than 5% hemolysis was regarded as nontoxic effect level in the experiments. The experiment was carried out in quintuplicate and hemolysis rate (%) was calculated using the equation below:
Where OD (sample) is the optical density of the samples, OD (NC) is the optical density of the negative control and OD (PC) is the optical density of the positive control.
Statistical analysis
Descriptive statistics were performed using OriginPro8® software (Massachusetts, USA), where one-way ANOVA analysis followed by Bonferroni post-test (p < 0.05) was used to determine variations between groups.
Results
Membrane characterization
The FTIR spectroscopy was used to characterize the membrane surface in terms of interaction between ions from IBF and the NRL membrane. Figure 1 illustrates the FTIR results obtained for the IBF-NRL membrane, NRL membrane and IBF powder. It was possible to verify in the NRL spectrum (blue line) the characteristic bands of poly (cis-1,4-isoprene) such as 2960–2852 cm−1 (CH2 symmetric and CH3 asymmetric stretching), 1446 cm−1 and 1375 cm−1 (CH2 and CH3 deformation), 1662 cm−1 (C=C stretching) and 835 cm−1 (=CH out of the plane bending). 62

FTIR spectra of the NRL, IBF powder and IBF-NRL.
The IBF spectrum (red line) showed the main bonds observed in this compound: intense and characteristic stretch band of carboxylic acid carbonyl (1718 cm−1), aromatic CH stretching (3089 cm−1), and CH3 and CH2 stretching bands (2954 cm−1 and 2924 cm−1, respectively). 63 Bands around 1721.5 cm−1 can be attributed to C=O stretching of the carbonyl group, commonly observed in recovered IBF. 64 It is interesting to highlight the signal corresponding to the anti-inflammatory in the IBF-NRL spectrum, evidenced by the intense stretching band at 1718 cm−1.
In order to evaluate the influence of IBF on the elasticity of the NRL membranes, tensile tests were performed in the IBF-NRL system. As a control, tests were also performed with the NRL membrane without IBF.
In Figure 2, the IBF-NRL membrane showed low elastic modulus and high elongation compared to the NRL membrane. In particular, different regimes can be identified at regions a, b, and c, which are associated with elastic regime, plastic regime and the rupture of the material, respectively. The elastic regime is the operational region of a biomaterial.
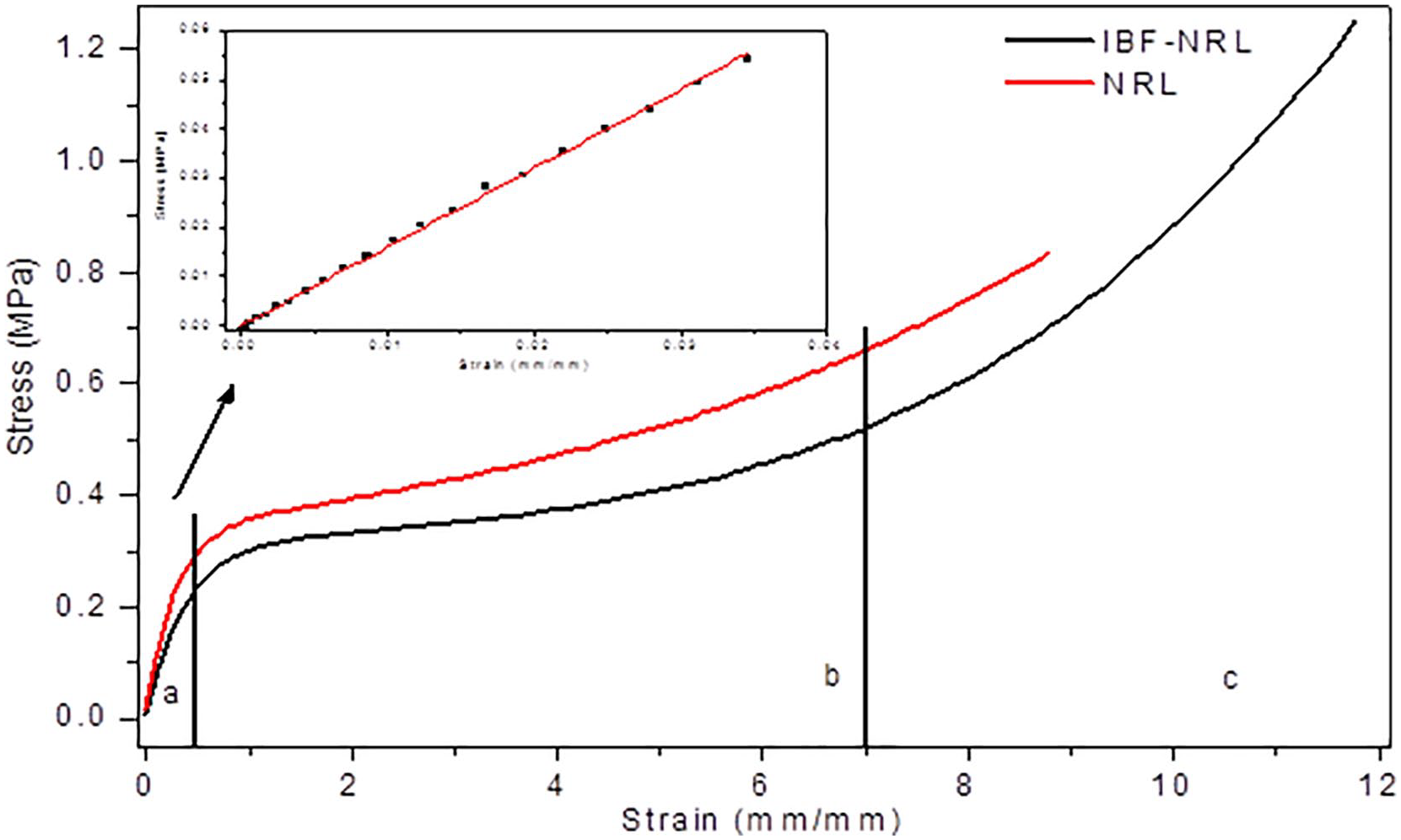
Stress-strain curves of the NRL and IBF-NRL membranes with three different regions (inserted graphical: expansion of the elastic region (a)).
The mechanical results obtained for the NRL membrane were 1.55 ± 0.09 MPa, 0.78 ± 0.08 MPa and 786.07 ± 95.01% for Young’s modulus, rupture tension and deformation, respectively, while for the IBF-NRL membrane, it is obtained 0.58 ± 0.14 MPa (Young’s modulus), 1.12 ± 0.15 MPa (rupture tension) and 1046.39 ± 132.42% (deformation). It was noticed that the IBF-NRL membrane presented an increase of 33% in the deformation and 43% in the rupture tension, and a decrease of 62% in the Young’s modulus in relation to the NRL membrane, showing that the IBF-NRL membrane is more rigid. Through statistical analysis, it was possible to observe that the membranes were statistically similar (p < 0.05) in terms of strain. However, they are statistically different in terms of rupture tension and Young’s modulus.
Figure 3 shows a comparison between the surfaces of NRL membranes with and without IBF. However, both NRL and IBF-NRL membranes did not exhibit pores on their surfaces (Figure 3(a) and (b)). Drug particles in three-dimensional forms on the IBF-NRL membrane surface (Figure 3(c)) demonstrate that the IBF absorption was dispersed in reticules (crystals).

SEM images of IBF-NRL (a, c) and NRL (b, d) membranes at magnifications 1000X (a, b) and 5000X (c, d).
In vitro release of ibuprofen
The analysis of in vitro IBF release was conducted by optical fluorescence, according to Radwan et al. 65 and Damiani et al. 66 Figure 4 demonstrates the release profile of IBF in different media (basic and acid). An interesting result of better effective drug release was verified in basic medium (pH 7.4). Usually, drug release kinetics through the membranes in the first hours (0–4 h) evidence a burst release process, which is associated with the presence of IBF on the material surface. In this same period, the IBF-NRL membrane released around 9.7 mg/mL or 48.5% (in basic pH) and 4.5 mg/mL or 22.5% (in acid pH). Note that the release kinetics obeys a bi-exponential function (ExpDec2), according to the equation below:
Where y(t) is the amount of IBF amount released by the NRL membranes as a function of the time (t), y0 is the initial drug amount in the polymeric matrix, A1 and A2 are constants, and t1 and t2 are the characteristic times.

Drug release as a function of time in different media: acid and basic.
The fastest exponential is associated with the IBF release on the membrane surface, while the slowest one can be linked to the drug located inside the membranes.37,39 Figure 4 also illustrates that the release process occurs during a period of 96 h, with the total liberation of 12 mg/mL approximately in alkaline medium (60%) and 10 mg/mL, in acidic medium (50%). The values of the constants A1 and A2 and the characteristic times t1 and t2 are also observed in Figure 4.
In vitro permeation assay
In vitro permeation was carried out to analyze IBF diffusion across synthetic membranes. The drug contents incorporated in the NRL membranes to permeation assay were of 35.38 µg/cm2. Figure 5 shows the cumulative IBF diffusion per unit area across the acetate cellulose membrane over 24 h. We observed that IBF incorporated into the NRL membrane starts the permeation after 30 min. The permeated value found into the receptor solution after 24 h was 1.10µg/cm2, in other words, 3.11% ± 0.50 (w/w) of the drug incorporated into the NRL membrane.

Permeation (g/cm2) of IBF incorporated into the NRL membrane as a function of the time in synthetic membrane (acetate cellulose).
Molecular modeling
Figure 6(a) illustrates color maps (CAFI and MEP descriptors) associated with IBF, ionized IBF and the NRL model system. Red regions define the most reactive and negatively charged sites for CAFI and MEP, respectively, while blue regions define non-reactive or positively charged sites. A representation of the electric dipoles is shown in Figure 6(b).

(a) Color maps of the local reactivities of IBF, ionized IBF and NRL model system: CAFIs and MEPs and (b) representation of the electric dipoles of IBF.
As expected, the most reactive sites of the NRL model system are centered on C=C bonds, mainly on the carbon atoms attached to hydrogen (C–H), for both electrophilic and nucleophilic interactions. It was also noticed that negative charges are centered on the C–H region, while positive charges are spread out over other molecular sites. Non-dissociated IBF presented charge concentrations at the hydrogen (positive) and oxygen (negative) atoms of COOH group, which can lead to a more effective interaction with NRL local charges.
Hemocompatibility assay
Figure 7 illustrates the hemolytic activity of the IBF-NRL in comparison to the NRL, positive and negative control. As can be seen, the IBF-NRL membranes did not cause significant red blood cell damage within 24 h of application, exhibiting a hemolytic rate of 1.673 ± 2.390%. For the NRL membrane, the result was statistically similar, with a rate of hemolysis of 2.131 ± 2.323%. Both membranes were considered non-toxic, once the obtained values are below 5%. Furthermore, the positive control showed 100 ± 0% hemolysis and the negative control, 0 ± 0%.

Samples employed for the evaluation of hemolytic activity (%): positive control (PC), negative control (NC), and systems containing the NRL and IBF-NRL membranes.
Discussion
In the present study, the IBF-NRL membrane was characterized by FTIR spectroscopy, mechanical tests and SEM analysis. As can be seen in Figure 1, the anti-inflammatory signal and characteristic bands of poly (cis-1,4-isoprene) were observed in the IBF-NRL spectrum. Furthermore, the absence of new bands in the IBF-NRL membrane allowed us to infer that there was no effective chemical interaction between the drug and membrane components. Thus, the integrity of IBF and maintenance of its active principle have been preserved. These results are in agreement with other works, which indicated the drug structural integrity (sodium diclofenac, glycerol, moxifloxacin, ciprofloxacin, and ketoprofen) after incorporation into the NRL.37,38,41,62,67 Therefore, it can be concluded that the characteristic bands of IBF and the NRL were preserved in the FTIR spectrum of the IBF-NRL membrane, as it was not observed the appearance of new bands or the disappearance of others.
It is known that mechanical tests on the NRL membranes are extremely important in the health area, due to their applications in regeneration of diaphragms, 35 esophagus, 33 wound,32,68 and bones, 69 in addition to other prophylactic uses. Moreover, it is necessary to verify if the mechanical behavior of the biomaterial has been preserved after the incorporation of the drug, indicating that the elasticity and rupture tension of membranes is sufficient for biomedical application.70–72 In any application, the intensities of the mechanical efforts to which the membrane must be subjected must guarantee that the total treatment time is completed without exceeding the limits of this phase, guaranteeing the material integrity.
The mechanical results showed that the IBF-NRL membrane is more rigid than the NRL membrane. This fact is confirmed by an increase of 33% and 43% in the deformation and rupture tension, respectively, and a decrease of 62% in the Young’s modulus in relation to the NRL membrane.
The outcomes obtained are similar to those reported by Garms et al. 62 They incorporated moxifloxacin in the NRL membranes. So far, it was observed that the mechanical resistance obtained for this biomaterial is suitable for biomedical applications once the Young’s modulus (0.84 MPa) and rupture tension (1.12 MPa) are in accordance with the values reported in the literature associated with NRL application in human skin (0.01 to 50 MPa for Young’s modulus and 1 to 40 MPa for rupture tension).70–72
It is reported in the literature that the addition of components to the NRL membranes alters the sample stiffness, leading to a consequent modification of the elasticity, 61 which is compatible with our results. Resende et al. 73 analyzed the variation of Young’s modulus of NRL incorporated with clay nanocomposites. Murbach et al. 74 reported in their experiments that the incorporation of ciprofloxacin into a latex membrane altered its mechanical behavior, making it more rigid and fragile. Floriano et al. 38 showed that the incorporation of ketoprofen in NRL membranes led to a breaking force of 0.61 MPa, that is, a value 10% lower in relation to the NRL membrane. In this context, the behavior of the IBF-NRL membrane follows the reported patterns, making the sample less elastic than the pure NRL without losing flexibility.
The SEM analysis (Figure 3) revealed smooth surfaces, without cracks or pore formation in both membranes (without and with IBF). Similarly to these results, some authors have also observed the absence of pores on the NRL membranes surface.75,76 In addition, the 3D IBF particles were dispersed in the form of crystals on the membranes, as reported in other works, which indicates that significant amount of the pharmaceutical assets can be positioned along the membrane surface.62,77,78
The presence of IBF on the membrane surface suggests that the drug release can occur very quickly in the solutions, giving rise to the burst release effect, which consists of the almost spontaneous release of some fraction of the compound to the external system. Previous studies have also reported that the drugs, which have crystalline structure on the NRL membranes surfaces, have influenced the burst release.38,39,77–79
The in vitro analysis of IBF release demonstrated that a greater amount of the drug was released in the basic medium (60% in pH 7.4) than in the acid medium (50% in pH 5.6). It is known that during the healing process the pH of the skin varies from basic with wounds (pH 7.4) to acid in healthy skin (pH 5.6). Therefore, this result favors the use of the membrane as a dermal adhesive in the treatment of inflammatory processes, since the greatest amount of IBF was released in a basic environment. Moreover, the burst release process is associated with the presence of IBF on the material surface, while the slower release is explained by the drug presence inside the membranes.
Garms et al. 41 observed that the membrane released 65.44% (pH 7.4) and 53.13% (pH 5.6) of moxifloxacin until 295 h through two mechanisms: (a) diffusion, which is responsible for the initial burst release, where the drug is released by the surface pores, and (b) polymer erosion, where a stable release profile is observed due to the matrix degradation and the drug dispersion in the middle. Trecco et al. 80 verified the influence of the pH on the release of Casearia sylvestris extract until 200 h. The highest release rates of the extract were found in the basic solutions (pH of 7.4 and 7.6) due to the breaking of the crosslink bonds of the NRL membranes, destabilizing them. Furthermore, Floriano et al. 38 showed that 60% of ketoprofen was released by the NRL membranes up to 250 h. The authors explained that the burst release process was caused by the fact that the drug was adsorbed on the NRL membrane surface. After that, they observed a stable profile (24–50 h) which was related with the slow diffusion of the drug through the matrix. Therefore, IBF carried by the NRL membrane was released in greater amounts in the first hours through diffusion and, later, by the polymer erosion mechanism, according to the cited literature.
Once IBF is released, it must be able to cross the skin barrier. In this sense, the in vitro permeation capacity of the IBF-NRL membranes was determined. Franz cells are a widely used methodology to evaluate in vitro drug permeation. We demonstrated that after 24 h, 3.11% of the IBF managed to cross the cellulose acetate membrane (Figure 5). Carvalho et al. 81 obtained similar results (3.96% of permeation (w/w)) on pig skin using NRL incorporated with Casearia sylvestris. Already, Pichayakorn et al. 82 showed 10%–60% of permeation (w/w) of nicotine incorporated into the NRL membrane, because the nicotine is highly lipophilic, its absorption is facilitated through skin layers. Ng et al. 83 studied the IBF drug diffusion from saturated solution across 13 different types synthetic membranes. The authors observed that porous membranes derived from various polymers demonstrated different degrees of diffusional resistance to IBF. In general, the porous membranes can be categorized into high-flux (8–18 mg/cm2/h) and low-flux (0.1–3 mg/cm2/h) membranes. Our results demonstrated that the IBF-NRL membranes can be employed for the local skin treatment because the permeation was minimal (3.11% of IBF). Moreover, this membrane is inexpensive, easy to handle and simple fabrication.
From molecular modeling studies (Figure 6), it was observed that chemical reactions involving IBF should be hindered by steric effects for f– (reactive sites located at non accessible regions of the ring) and by electrostatic repulsion for f+ (negative charges on the nucleophilic center), which can explain the absence of new vibrational bands in Figure 2. Finally, a clear charge separation was noticed for dissociated IBF (IBF COO–), with the formation of an effective electric dipole that enhances its interaction with water and consequently increases the drug release. The higher reactivity on the oxygen atoms in relation to f– can be linked to IBF mechanism of action, which is still not clarified.84,85
In fact, given the IBF pKa, its dissociated form (IBF COO–) is supposed to be present in both the investigated pHs, being dominant in basic solutions. 42 The pronounced electric dipole and charge separation noticed for this structure suggests a more effective interaction with water, explaining the efficient drug release in alkaline solutions with very effective burst release processes (Figure 4).
To assess whether the components released by the IBF-NRL membrane are compatible with the hematic system, hemolytic activity tests were conducted. According to standard procedure ASTM F 756-00, this test evaluates the acute in vitro hemolytic properties of materials intended for use in contact with blood, such as transdermal adhesives. 86 This assay is one of the most used for the evaluation and identification of the biomaterials biocompatibility. Moreover, hemolysis is regarded as an especially significant screening test, once it provides quantification of small levels of plasma hemoglobin, which may not be measurable under in vivo conditions.87,88
The NRL and IBF-NRL membranes presented a hemolytic rate of 2.131 ± 2.323% and 1.673 ± 2.390%, respectively, indicating that they are non-toxic, as they did not damage the red blood cell membrane.
These outcomes have also been observed in other studies that reported hemolytic activity below 5%. Miranda et al. 75 analyzed the pure NRL membranes and did not observe hemolytic activity in their samples (hemolysis around 0%–1.8%). On the other hand, some authors evaluated the hemolytic effects using the NRL membranes loaded with peptides (oxytocin, desmopressin acetate), drugs (scopolamine, ketoprofen, gentamicin) and other (calcium phosphate, polylactic acid)38,61,77–79,89,90 and also no changes were noted in red blood cells. The interaction of blood with polymeric adhesives is one of the most important criteria for its success for in vivo applications. Hence, our results showed that the IBF-NRL membrane is a promising dressing in the treatment of inflammatory processes.
Conclusion
A novel natural latex-based polymeric adhesive for sustained IBF releasing has been developed and characterized. It was shown that there is no chemical interaction between the drug and the polymer matrix. The values of Young’s modulus (0.58 ± 0.14 MPa), ultimate rupture tensile (1.12 ± 0.15 MPa), and elongation (1046.39 ± 132.42%), were compatible with the mechanical properties required for cutaneous adhesives. Preliminary biocompatibility studies showed the IBF-NRL system can be considered a promising biomaterial, with no significant degree of hemolysis within 24 h of use. The in vitro drug release test showed an increased release in the first few hours of the study, followed by a slower process that continued until the end of the assay. The amount of IBF released was higher in alkaline media (60%), which may be associated with distinct action on injured skin condition (pH 7.4). Based on the IBF permeation tests, the IBF-NRL membranes are can be employed for skin treatment with permeation minimal of 3.11% of IBF. The local reactivities and electronic properties of IBF and the NRL-model system, estimated via DFT calculations, suggest that the drug release is driven by the presence of IBF ionized forms in the membrane. These results indicate that the IBF-NRL system can be a simple and inexpensive alternative for production biocompatible adhesives, which can be used in the treatment of inflammatory processes and injuries. Moreover, it is expected that the polymeric adhesive will decrease the high concentrations of the drug consumed orally, reducing their side effects.
Footnotes
Acknowledgements
The authors thank to Paulista Medicine Company (CPM-Pharma), EMS Pharma Group Coordination for the Improvement of Higher Education Personnel (CAPES), and Advanced Microscopy Laboratory (LMA) located at UNESP Chemistry Institute in Araraquara. We would also like to thank NR Barros for his assistance in hemolysis activity and results discussion in this manuscript.
Author contribution
This work was carried out in collaboration between all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the FAPESP, São Paulo Research Foundation, (Processes: 2011/17411-8, 2014/17526-8, 2017/19603-8), and Brazilian Council for Scientific and Technological Development (Process: 470261/2012-9).



