Abstract
The effect of grain size of dental zirconia on the shear bond strength of composite resin cement was newly studied. Disc-shaped dental zirconia with small (sample S) and large (sample L) grains were made by sintering of pre-sintered dental zirconia at 1450°C for 0.5 h and 40 h, respectively. After the sintering, the average grain size of sample S was 1.37 ± 0.15 µm, while that of sample L was 3.74 ± 0.50 µm. The sintered discs were successively polished with different grades of diamond and alumina slurries. The interfacial free energies were 63.5 ± 4.2 dyne/cm for sample S and 52.1 ± 5.5 dyne/cm for sample L. Stainless steel cylinders, previously sandblasted with 50 µm alumina powder, were bonded to the zirconia discs using composite resin cement. Next, samples were kept in an oven for 7 d at 36.5°C. The shear bond strength of sample S was 23.0 ± 4.5 MPa while that of sample L was 17.5 ± 4.6 MPa. After the fracture, the areal % values of composite resin cement remaining on the zirconia surfaces were 89.7 ± 5.9% for sample S and 61.6 ± 5.5% for sample L. The results suggest that grain size reduction has a potential to enhance the degree of bonding between a composite resin cement and a dental zirconia due to the increase of interfacial free energy.
Introduction
Zirconia is one of the most representative structural ceramics due to its good mechanical properties. Its high fracture strength and toughness originate from a phase transformation toughening mechanism. The stress intensity factor at a crack front is effectively reduced by absorbing the crack propagation energy by transforming the tetragonal phase of zirconia into a monoclinic phase. Additionally, volumetric expansion of the grains surrounding the crack surface, which is caused by phase transformation, also hinders crack propagation. The energy consumption of crack propagation due to phase transformation and consequent volumetric expansion of the surrounding grains produces a high fracture strength, toughness, and resultantly Weibull modulus. Furthermore, the color of zirconia can be controlled using various rare earth additives. Thus, zirconia is prevalently used for dental crowns, 1 bridges,2–4 brackets, 5 and abutments. 6
However, time-dependent degradation may occur in the zirconia structure influencing the mechanical properties of the zirconia.7,8 The low adhesive capability of composite resin cement on its surface is also a problem in some dental applications. The low-temperature degradation problem can be somewhat overcome by decreasing its grain size (to slow the phase transformation) and adopting suitable sintering aids. 9 However, the low adhesive capability of composite resin cement on the zirconia surface remains a problem.
There have been many studies to enhance the adhesion between composite resin cement and zirconia. These can be divided into two groups depending on bond type: mechanical10–15 or chemical bonding.10,12,16–21 The surface roughening method using sandblasting11,12,15 and/or grinding11,12 has been developed to improve mechanical bonds. Increased bond strength is achieved through mechanical locking of a composite resin cement on zirconia surface. Some studies reported that the decrease of mechanical properties of the zirconia body due to the occurrence of surface flaws during the airborne particle abrasion.11,22,23 However, global results based on the meta-analysis reported that airborne particle abrasion rather improved flexural strength of zirconia 24 by residual surface compressive strength caused by volume expansion of the grains occurred during phase transformation.25,26 Thus, the decrease in fracture strength by airborne particle abrasion cannot be a problem.
The purpose of the selective infiltration etching (SIE) method is similar to that of the airborne particle abrasion. The difference between them is the method used to create a rough surface. In the selective infiltration etching method,18,21 channel-like structures among the zirconia grains are produced by selectively dissolving a silica glass phase, which was previously infiltrated by molten glass through a heat-treatment.
Many studies have been conducted to coat a silica phase on the zirconia surface by abrading with SiO2 or SiO2-coated Al2O3 particles.10,14,17 This is done to enhance the degree of bonding through a reaction between the Si-OH groups coated on the zirconia surface and those in the composite resin cement.
Treating the zirconia surface with silane is another attempt used to increase the degree of bonding between a composite resin cement and a dental zirconia. This process is divided into two main strategies according to method of silanization. One uses a silane solution,12,16 while the other uses a plasma 16 or after-plasma treatment. 20
The usage of phosphate ester primers and phosphate-modified composite resin cements is another way to utilize chemical bonding between the composite resin cement and zirconia.27–30 The negatively charged phosphate groups in phosphate ester primers (P-O− groups) are expected to bond chemically with the positively charged zircon ions.30,31 The hydrogen bonding is also expected to occur between the Zr-OH and P=O (oxo group).30,32 There have been many attempts to increase the degree of bonding between the composite resin cement and zirconia, but there has been no effort to increase it simply by controlling grain size of zirconia.
The interfacial free energy (surface energy) is the amount of broken interatomic bonds that occurs when a surface is created. Thus, the thermodynamic stability of the surface is low compared to the bulk of a material. This creates surface reactivity with foreign materials and consequently reduces the interfacial free energy. In addition to the surface, the grain-boundaries and triple junctions of a ceramic are sites where many interatomic bonds are disrupted. At a fixed sample size, the number densities of grain-boundary and triple junction increase as decreasing grain size, and this also increases the interfacial free energy. Therefore, we can expect the improved adhesive capability of the composite resin cement on zirconia surface by decreasing the grain size of dental zirconia.
In this experiment, the effect of grain size of dental zirconia on the shear bond strength of the composite resin cement was examined with the main focus on interfacial free energy. Disc-shaped dental zirconia samples with small and large grains were prepared, and the shear bond strengths of composite resin cement on these zirconia surfaces were evaluated.
Methods
Preparation of zirconia samples
Disc-shaped zirconia samples (a diameter of 12 mm and a height of 2 mm) with small and large grains were prepared through sintering of pre-sintered dental zirconia (LAVA plus, 3M ESPE) at 1450°C for 0.5 h and 40 h, respectively.
Before the tests, the sintered discs were sequentially polished (Tegrapol-35, Struers) with different grades of diamond (9, 3, and 1 µm, DiaPro, Struers) and Al2O3 slurries (0.04 µm, OP-S, Struers) to exclude the effect of surface roughness. The polished discs were washed with a sonicator (SH-2100, Saehan) in deionized water, 70% ethanol, 99.8% ethanol, 99.8% acetone, 70% ethanol, and finally deionized water for 20 min per step. The washed samples were kept in a humidity-controlled desiccator (Super Dry Keeper, Sanplatec) at 10% relative humidity to protect from the contamination until the next experiments.
Characterization
A field emission-scanning electron microscope (FE-SEM; S-4700, Hitachi) was used to observe the microstructures. Image analyzing software (ImageJ, National Institutes of Health) was used to assess the grain size. The grain size was determined by the linear interceptive count method according to ASTM E112-96(2004)e2. Then, the zirconia discs with small and large grains by sintering at 1450°C for 0.5 h and 40 h, were referred to as samples S and L, respectively, in accordance with grain size.
The crystal phases were assessed with an X-ray diffractometer (XRD; D8 Discover, Bruker). A laser confocal scanning microscope (LSM 5 Pascal, Carl Zeiss) was used to assess the surface roughness of the two zirconia samples. Ten replicates were used for each sample. The interfacial free energy was calculated from the contact angle of a deionized water drop on the sample (SEO 300A, SEO Optics).33,34 Five different areas were measured on each sample.
Shear bond strength testing
The shear bond strength of the composite resin cement on the zirconia disc was evaluated with a general-purpose testing machine (LF plus 1 kN, LLOYD Instruments) after bonding the cylindrical stainless steel on the zirconia discs using the composite resin cement.
Before the cementation, the base of the stainless steel cylinder (3 mm in a diameter and 5 mm in a height) was sandblasted using 50 µm alumina for 15 s vertically at a distance of 10 mm. The purpose of sandblasting was to ensure that the fracture occurred between the resin cement and zirconia rather than between the stainless steel cylinder and resin cement. One coat of the metal primer (Alloy Primer, Kuraray) was applied on the stainless steel with a microbrush, left for 30 s, and then dried with air syringe. The zirconia surface was then treated with a MDP-containing primer (Z prime plus, Bisco). A composite resin cement (Panavia F2.0, Kuraray America) was applied to the stainless steel cylinder and then the stainless steel cylinder was bonded to the zirconia disc with a weight of 1 kg to standardize the thickness of the resin cement. Excess resin was immediately removed with a microbrush and Oxyguard® was applied. Each quadrisection were treated for 20 s with an LED curing light with approximately 1000 mW/cm2 (Elipar™ FreeLight 2, 3M ESPE). The samples were kept in an oven at 37°C for 7 d to complete the chemical cure and then the shear bond test was performed.35,36 The samples were tested with a parallel knife-edge blade touching the cement layer at a crosshead speed of 0.5 mm/min. The maximum load at fracture was measured, and the shear bond strength (σs) was calculated in MPa using the formula σs = F/A, where “F” is the stress at failure (Kgf) and “A” is the bonded area of resin cement (mm2). Ten samples for each group were used to evaluate shear bond strength. The areal percentage of resin cement remaining on the zirconia surface after the fracture test (the adhesion remnant index) was evaluated via stereotype optical microscopy. The fractured surfaces were categorized as cohesive, adhesive, or mixed fractures.
Statistical analyses were carried out using the SPSS v.25 software (SPSS/PC Inc.). Normality was tested with Kolmogorov–Smirnov test and equality of variances was tested with Levene’s test. Data were analyzed by Student’s t-test and Mann Whitney U test at a significance level of 0.05.
Results
The microstructures of the two sample types after sintering are shown in Figure 1, and their grain size distributions are shown in Figure 2. The average grain size of sample S was 1.37 ± 0.15 µm, and the grain size distribution ranged from 1.0 to 1.9 µm. The average grain size of sample L was 3.74 ± 0.50 µm, and the grain size distribution ranged from 2.6 to 5.1 µm. The average surface roughness of sample S was 1.65 ± 0.76 µm while that of sample L was 1.50 ± 0.55 µm. The surface roughness was not significantly different between the two groups (p = 0.661).

FE-SEM images of (a) sample S and (b) sample L after sintering of pre-sintered dental zirconia at 1450°C for 0.5 h and 40 h, respectively.
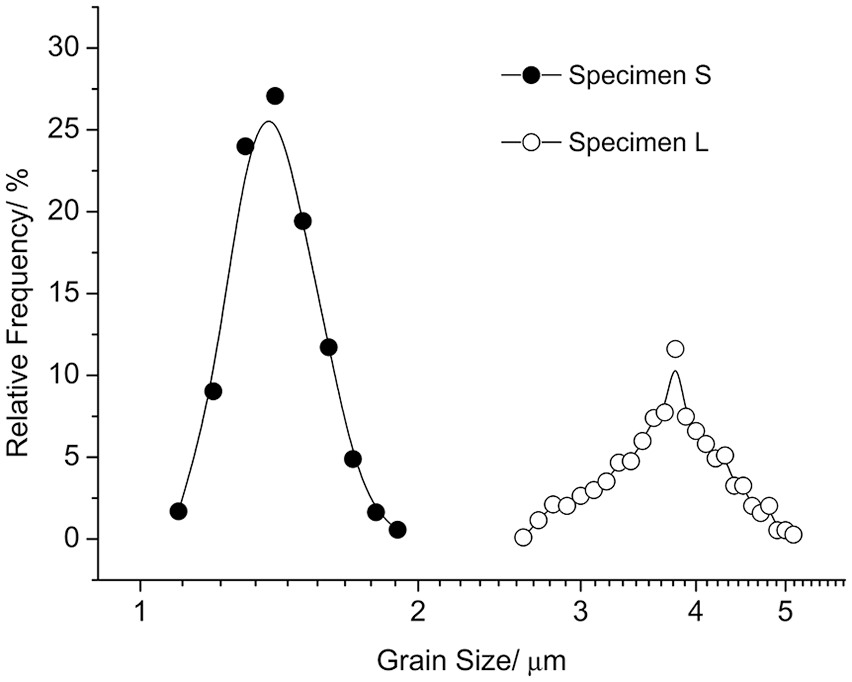
Grain size distributions of sample S and L after sintering of pre-sintered dental zirconia at 1450°C for 0.5 h and 40 h, respectively.
Phase analyses were carried out after sintering of pre-sintered dental zirconia at 1450oC for 0.5 and 40 h, respectively (Figure 3). The major and minor phases of the two samples were yttrium-stabilized tetragonal (t-ZrO2) and monoclinic zirconia (m-ZrO2), respectively.

XRD patterns of sample S and L after sintering of pre-sintered dental zirconia at 1450°C for 0.5 h and 40 h, respectively.
The interfacial free energies calculated from the wetting angles of a deionized water were 63.5 ± 4.2 dyne/cm for sample S and 52.1 ± 5.5 dyne/cm for sample L. The difference between the two groups was statistically significant (p = 0.013).
After bonding, the shear bond strength between the zirconia disc and composite resin cement was evaluated (Figure 4). The shear bond strength of sample S was 23.0 ± 4.5 MPa and that of sample L was 17.5 ± 4.6 MPa. The difference between the two groups was statistically significant (p = 0.014).
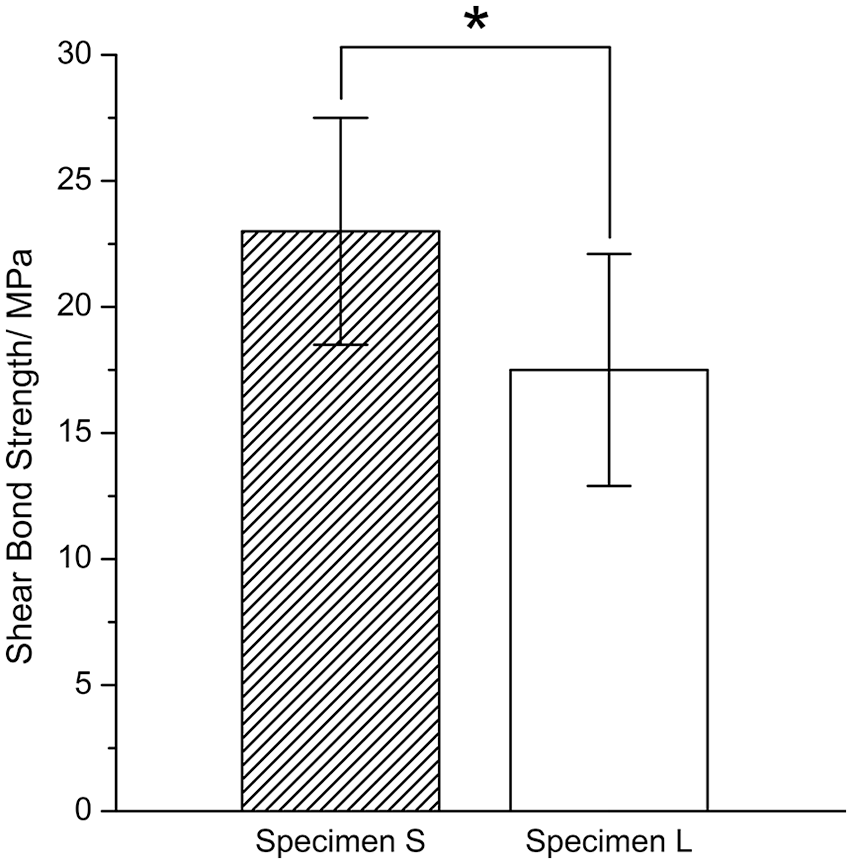
Shear bond strengths of the composite resin cement on samples S and L, respectively.
The areal percentage of composite resin cement remaining on the zirconia disc after the shear bond test was evaluated via stereotype optical microscopy (Figure 5). The areal percentages of composite resin cement remaining on the zirconia surface were about 89.7 ± 5.9% for sample S (Figure 5(a)) and 61.6 ± 5.5% for sample L (Figure 5(b)). The difference between the two groups was statistically significant (p = 0.000). The fracture types for both samples were a mixed mode of adhesive and cohesive fractures (Figure 5). In sample S, however, most of the fracture occurred in the composite resin cement itself, while a small part (about 10%) of the fracture occurred at the interface between the composite resin cement and zirconia disc (Figure 5(a)). In contrast, about 40% of the fractures in sample L arose at the interface between zirconia and composite resin cement (Figure 5(b)).

Photographic images of composite resin cements remaining on (a) sample S and (b) sample L after the shear bond test. Sample S showed less composite resin cement loss compared with sample L. The composite resin cements were assumed to be bonded in a round shape (white arrow) at the center of the zirconia disc (white triangle). The composite resin cement remained on zirconia was marked with white square and the composite resin cement loss was marked with white asterisk.
Discussion
The grain size effect of dental zirconia on the shear bond strength of composite resin cement was newly studied. Disc-shaped zirconia samples with small (sample S) and large (sample L) grains were prepared by controlling the sintering time.
The crystal phases were the same between two samples (Figure 3). The surface roughness was not significantly different between two samples after polishing (p > 0.05). The average grain size of sample S (1.37 ± 0.15 µm) was about 63% smaller than that of sample L (3.74 ± 0.50 µm) after sintering (Figures 1 and 2). As a result, the number densities of grain-boundaries and triple junctions were much higher in sample S than sample L (Figure 1). Indeed, the interfacial free energy of sample S (63.5 ± 4.2 dyne/cm) was about 21% higher than that of sample L (52.1 ± 5.5 dyne/cm).
The shear bond strength of the resin cement on specimen S (23.0 ± 4.5 MPa) was about 31% higher than that on specimen L (17.5 ± 4.6 MPa). The reported shear bond strengths of the same composite resin cement (Panavia F2.0) on zirconia, which was not treated with airborne particle abrasion, ranged from 10 to 23 MPa.12,19–21,37 Thus, the shear bond strengths of the composite resin cement on samples S and L were within the normal range, even though it is difficult to directly compare them with other reports because shear bond strength is sensitive to various processing parameters.
After shear bond test, a mixed mode of cohesive and adhesive fractures was found in both samples. However, the areal percentage of composite resin cement remaining on sample S (89.7 ± 5.9%) was about 46% higher than that on sample L (61.6 ± 5.5%). This indicates that the adhesive capability of the composite resin cement on zirconia was higher in sample S than in sample L, possibly due to the different grain sizes. This implies that shear bond strength of composite resin cement on zirconia is critically dependent on interfacial free energy.
The surface represents termination of the repeating unit of the crystal. Thus, interfacial free energy is produced by excess or deficient electrons that originate from broken bonds of the crystal. This means that the surface is an energetically unstable place. A grain-boundary and triple junction also contain a large number of broken bonds due to intersection of two or three grains with different crystal orientations. Thus, interfacial free energy originates from the surface, grain-boundaries, and triple junctions.38–40 Consequently, as grain size decreases at a fixed sample size, the interfacial free energy increases. This brings to increase the surface reactivity.
When the composite resin cement is applied to the zirconia surface, the coupling agents which contain a charged functional group such as phosphate 41 are believed to react with the surface, grain-boundaries, and triple junctions, which also have charges by broken interatomic bonds. 30 Therefore, the amount of adhesive bonding between the zirconia surface and the charged coupling agent in resin cement increases as the grain size decreases. This explains the higher shear bond strength of sample S than that of sample L. If the resin cement is applied to the unpolished zirconia surface, there is a possibility that the shear bond strength will be increased slightly because the resin cement can penetrate along the grooves of the grain-boundaries. However, the effect will be much smaller than that of airborne particle blasting abrasion because the surface irregularities treated by airborne particle blasting abrasion will be much higher than those of grain-boundaries.
Collectively, the improved shear bond strength of composite resin cement on dental zirconia could be achieved by reducing the grain size of zirconia. This could be possible by increased number density of grain-boundaries and triple junctions, which increased the interfacial free energy of zirconia. The evaluation of bonding durability after thermal aging, and the combination study of grain size reduction and airborne particle blasting are under investigation and will be published in near future.
Conclusion
The effect of grain size of dental zirconia on the shear bond strength of composite resin cement was studied. The interfacial free energy of zirconia could be increased simply by reducing the grain size and this increased the shear bond strength of composite resin cement on the zirconia surface. This means that the controlling the grain size of dental zirconia can be an alternative way to improve the adhesive capacity of composite resin cement on the zirconia surface crown.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic Science Research Program through a National Research Foundation of Korea [NRF-2020R1F1A1051208].
