Abstract
Chronic osteomyelitis causes serious injury to patients. Antibiotic delivery systems based on poly(lactide-co-glycolide) (PLGA) have great potential for treatment of chronic osteomyelitis. However, PLGA has a glass-transition temperature that is higher than physiological temperatures, resulting in a lack of flexibility for implantation into the bone marrow cavity. As an alternative, poly(d, l-lactide-co-glycolide-co-ε-caprolactone) (PLGC) presents good flexibility due to the introduction of poly(ε-caprolactone) segments. To develop a new strategy for treatment of chronic osteomyelitis, a ciprofloxacin delivery system was prepared using PLGC as carriers, the antibacterial effects of which were evaluated both in vivo and in vitro. The in vitro release behavior showed that the average release reached 268.5 μg/days on day 33, with a cumulative release rate of 56.01%. A bacteriostatic ring, with a diameter of 26.83 ± 0.83 mm, was produced by ciprofloxacin against Staphylococcus aureus after 30 days of release via our ciprofloxacin-PLGC system. After 4 weeks of treatment in vivo, chronic-osteomyelitis-model rats had a bodyweight of 385.83 ± 17.23 g and a normal white-blood-cell count, as well as a lower number of bacterial colonies per gram of bone tissue of (10.6 ± 3.0) × 101 CFU/g. Furthermore, no inflammatory cells were observed via hematoxylin-and-eosin staining, and normal bone structure was observed via X-ray. Taken together, our findings indicate that our novel ciprofloxacin-PLGC system yielded noteworthy antibacterial effects both in vitro and in vivo, suggesting that it may be useful for treating patients with chronic osteomyelitis.
Keywords
Introduction
Osteomyelitis is bone inflammation caused by infectious microorganisms. 1 Additionally, osteomyelitis can also spread from infected tissues, such as following surgery, open fractures, and joint replacement. 2 After entering into the body, most bacteria adhere to the surface of necrotic soft tissue and bone tissue in a static state and form a bacterial biofilm, which has a protective effect on bacteria and greatly enhances bacterial resistance to antibiotics, making it difficult for bacteria to be completely removed.3,4 Therefore, chronic osteomyelitis is difficult to cure and easily relapses. Strategies for treating chronic osteomyelitis include surgical removal, bone reconstruction, and combined application of antibiotics. Unfortunately, long-term systemic application of antibiotics has deleterious side effects, and it is often difficult to achieve sufficiently high antibiotic concentrations locally at the lesion site. Local application of antibiotics can circumvent such disadvantages of systemic antibiotic treatments5,6 and, consequently, it has become an important component for treating chronic osteomyelitis. Polymethyl methacrylate (PMMA) beads (commonly known as bone cement) have been used to carry and release antibiotics for the treatment of osteomyelitis with some success. Since it was first applied clinically in the 1970s, 7 PMMA beads have become a standard for local drug release in the bone marrow cavity. However, some disadvantages of PMMA beads have also been found in clinical use, including the following. First, due to a lack of biodegradation, they need to be removed via a second surgery, which increases the patient’s physical discomfort/inconvenience and medical expenses. Second, after the initial release of antibiotics, the subsequent drug concentration is insufficient, which increases the resistance of pathogenic bacteria. Third, bacteria easily form biofilms around PMMA beads, preventing drug sterilization. Finally, bone-cement monomers, such as PMMA, are moderately toxic. To circumvent these shortcomings, absorbable biomaterials8–14 have been used to replace bone cement (e.g. PMMA) in the treatment of chronic osteomyelitis.
Poly(lactide-co-glycolide) (PLGA), a copolymer of lactide and glycolide, is the most common polyester used as a topical antibiotic carrier to fabricate biodegradable drug-delivery systems for treating osteomyelitis.15–17 For example, Jeong et al.18,19 prepared and evaluated the antibacterial activities of ciprofloxacin-encapsulated PLGA nanoparticles and microspheres. The results indicated that encapsulated ciprofloxacin maintained antibacterial activity after microencapsulation, and that ciprofloxacin-encapsulated PLGA particles yielded a substantial efficacy in inhibiting the growth of bacteria in vivo. Although PLGA holds considerable potential as a carrier for biodegradable drug-delivery systems,20,21 some noteworthy drawbacks limit its widespread application for treating chronic osteomyelitis. It is difficult to smoothly implant large PLGA particles into the bone marrow cavity due to their high glass-transition temperature and high crystallinity. Furthermore, PLGA produces acidic products during degradation, which may affect the activity of antibiotics and reduce their antibacterial efficacy. Therefore, it is necessary to improve the elasticity of PLGA and reduce its acid-degradation products, which may promote its application in the treatment of osteomyelitis. In our previous work, ε-caprolactone (ε-CL) was introduced into either PLGA chains or poly(d, l-lactide-co-glycolide-co-ε-caprolactone) (poly(DLLA-co-GA-co-CL), abbreviated as PLGC), and PLGC was found to exhibit better elasticity and controlled degradation 22 ; hence, PLGC represents a promising carrier for biodegradable drug-delivery systems.23–25 However, no studies have yet reported the use of a PLGC-based drug-delivery system for the treatment of osteomyelitis.
Ciprofloxacin has been investigated as a drug for local administration because it is effective against a broad spectrum of bacteria associated with osteomyelitis.26,27 In the present study, PLGC was used as a carrier to prepare a ciprofloxacin-loaded drug-delivery system for treating models of osteomyelitis in vitro and in vivo. Specifically, we evaluated the in vitro release behavior and antibacterial activity of the ciprofloxacin-PLGC system. Furthermore, a tibia chronic osteomyelitis model in Wistar rats was established to evaluate the therapeutic effects of our PLGC-based drug-delivery system in vivo.
Materials and methods
Reagents and equipment
d, l-lactide (DLLA) and glycolide (GA) (Daigang Biomaterial Co., Ltd., China) were recrystallized from acetic ether and dried at 37°C under reduced pressure before polymerization; ε-caprolactone (CL) was purchased from Sigma-Aldrich and was freshly distilled over CaH2 under reduced pressure prior to use. Ciprofloxacin HCl was purchased from Dalian Meilun Biotechnology Co., Ltd. Fetal bovine serum (FBS) was purchased from HyClone Laboratories. DMEM/F12 medium was purchased from Shanghai Yuan Wildlife Technology Co., Ltd. Rat bone marrow mesenchymal stem cells (BMSCs) were purchased from Guangzhou Sanye Company. Staphylococcus aureus was purchased from Shanghai Benoy Biotechnology Co., Ltd. SPF Wistar rats were purchased from Liaoning Changsheng Biotechnology Co., Ltd.
Synthesis of poly(d,l-lactide-co-glycolide-co-ε-caprolactone)
PLGC was prepared as reported in our previous study. 22 Briefly, a mixture of DLLA (40 mol%), GA (40 mol%), and CL (20 mol%) was added to glass ampoules under a nitrogen atmosphere, and Sn(Oct)2 was added to the solution in anhydrous toluene (2 × 10−4 mol of Sn(Oct)2 per mol of monomer). The toluene was removed by evacuation. The ampoules were purged three times with dry nitrogen and were then heat-sealed using a vacuum. The ampoules were placed in an oil bath and reacted at 130°C for 24 h while being vigorously shaken to obtain a homogeneous mixture of the monomers and the catalyst. After the copolymerization, the copolymer was discharged, dissolved in CHCl3, precipitated in excess methanol, washed in methanol, and dried under reduced pressure at 37°C until there was a constant weight.
Preparation of a novel ciprofloxacin-PLGC delivery system
First, 5 g of PLGC copolymer was accurately weighed and dissolved in chloroform. Then, 0.5 g of ciprofloxacin HCl was added according to a drug-to-carrier weight ratio of 1:10. The mixture was magnetically stirred and poured into a PTFE dish. After the complete evaporation of chloroform, the film was peeled and dried to a constant weight. The film was then cut into small pieces by scissors for later use, without any standardization in terms of the sizes of the individual pieces. Subsequently, the pieces of ciprofloxacin-loaded PLGC were weighed as needed, packed in sealed bags, and then irradiated with 25 kGy of 60Co for sterilization.
In vitro release behavior
First, 100 mg of ciprofloxacin-loaded PLGC was immersed in a glass vessel containing 10 mL of distilled water, which was then shaken at a frequency of 62 times/min in a constant-temperature air bath at (37 ± 1)°C. The distilled water was changed every 24 h. At different time intervals, 5 mL of sample was withdrawn from the vessel and the concentration of drug in each sample was measured using an ultraviolet (UV)-visible spectrophotometer at 271 nm, which was then used to estimate the amount of drug release The procedure was conducted in triplicate at each time point, and the averaged results are expressed as the mean ± standard deviation (SD) throughout the manuscript. The in vitro release behavior of ciprofloxacin-loaded PLGC was also investigated in acidic media with a pH of 4.5.
Cytotoxicity assay
For indirect cytotoxicity evaluations, 0, 1, 5, 10, or 50 mg of our ciprofloxacin-PLGC system (n = 3) was weighted and incubated, at 37°C with 5% CO2, with 1 mL of DMEM for 24 h to extract any leachable components. Then, the concentrations of leachable components were determined at 0, 1, 5, 10, and 50 mg/mL. The MTT method was used to detect the effects of different concentrations of leachable components from our drug-delivery system on the proliferation of BMSCs. After 3 days of cell culturing, the medium was replaced with leachable-component-containing DMEM. The cells were also incubated with either pure ciprofloxacin (10 mg/mL) or blank PLGC at a concentration range of 0–50 mg/mL each, and the mean value obtained for cell cultures incubated with DMEM only (negative control) was standardized as 100% cell viability.
Antibacterial activity in vitro
Activated Staphylococcus aureus was transferred to a nutrient broth and cultured at 37°C for 24 h, and then the bacteria were appropriately diluted with this nutrient broth to obtain a bacterial solution of 108 colony-forming units (CFUs)/mL. Subsequently, the Staphylococcus aureus bacterial solution was removed with a sterile cotton swab and was spread evenly onto a nutrient agar plate. Holes were punched into the nutrient agar plate and 100 μL of ciprofloxacin-containing release solution was added (after the release following 2 h, 4 h, 15 days, or 30 days; n = 3 samples for each time point). Then, the solution was incubated at a constant temperature for 24 h. A vernier caliper with an accuracy of 0.1 mm was used to measure the diameter of the bacteriostatic ring, and an area without obvious bacterial growth was visually observed as the edge of the bacteriostatic ring. Staphylococcus aureus was also incubated with blank PLGC carriers. In addition, a series of pure ciprofloxacin with concentrations of 15.6 μg/mL, 0.24 μg/mL, and 0.004 μg/mL were prepared to simulate the residual concentrations of pure ciprofloxacin (1 mg/mL) in vivo after several half-lives at 24 h, 48 h, and 72 h, respectively. Then, Staphylococcus aureus was incubated with these ciprofloxacin solutions.
Establishment and evaluation of a rat model of chronic osteomyelitis
All surgical procedures were approved by the Animal Ethics Committee of China Medical University. The model was established according to the method of Karau et al. 28 Twenty-four rats were weighed and anesthetized via intraperitoneal injection of chloral hydrate (3 mg/kg). A 1.5-cm-long skin incision was made under the right knee joint along the medial tibial anterior condyle to expose the proximal one-third of the tibia. The tibial bone marrow cavity was exposed using a 1.0-mm-diameter electric drill, after which the cavity was washed with saline and injected with 50 μL of morrhuate sodium. Then, 50 μL of sterile saline containing Staphylococcus aureus was injected into the exposed cavity. Subsequently, the bone marrow cavity was closed with bone wax and was then sutured. After the operation, the rats were placed in cages to allow them to move freely, and they were provided food and water ad libitum.
Four weeks later, three Wistar rats were sacrificed, and their right-lower extremities were examined via lateral X-ray to confirm the formation of chronic osteomyelitis. X-ray films were evaluated by two independent radiologists in a double-blind manner, following the evaluation method of Norden et al. 29 Four evaluation indicators were used, as follows: (1) the formation of dead bone (yes = 1 or none = 0); (2) periosteal responses (yes = 1, ambiguous = 0.5, or none = 0); (3) bone destruction (severe = 2 (involving multiple areas), moderate = 1 (only one location), mild = 0.5 (ambiguous), or none=0); and (4) the extent of disease extension to the proximal, middle, and distal done (yes = 1, ambiguous = 0.5, or none = 0). The other three Wistar rats were sacrificed, and hematoxylin-and-eosin (HE) staining was performed for the right tibia specimens to confirm the formation of chronic osteomyelitis. The results of pathological sections were evaluated according to the method described by Petty et al. 30
Treatment of chronic-osteomyelitis-model rats
Eighteen Wistar rats were randomly divided into an experimental group, control group, and blank group (n = 6 per group). The rats were anesthetized by intraperitoneal injection of chloral hydrate (3 mg/kg). Then, the opening of the bone marrow cavity was exposed again through the original incision and opened with a 1.5-mm diameter drill. Necrotic, hardened, and infected tissues were removed with a self-made spatula, and the bone marrow cavity was repeatedly washed with 2% hydrogen peroxide and physiological saline. The rats in the experimental group were implanted with 400 mg of our ciprofloxacin-PLGC system, the rats in the control group were implanted with 400 mg of ciprofloxacin-free PLGC polyester, and rats in the blank group were treated without any implantation. A fourth group of rats were treated with 40 mg of pure ciprofloxacin. Finally, the bone marrow cavity was closed with bone wax and was then sutured.
Observational indicators
The body weights and white blood cell (WBC) counts of rats in the three groups were measured at 0, 7, 14, 21, and 28 days after the operations. Additionally, the condition of the wound was observed to determine whether there was an inflammatory reaction, pus, and/or a sinus infection. Microbiological examination was also performed. After 4 weeks of treatment, the rats were sacrificed in each group and the right tibia specimen was harvested, placed into a sterile test tube, and sent to our microbiology laboratory. The bone tissue was pulverized into a powder, 1 g of bone tissue was accurately weighed, and then 1 mL of physiological saline was added to make a suspension. After a 10-fold serial dilution with saline, 0.1 mL of the diluted sample was inoculated onto blood agar plates and incubated at 37°C for 48 h. The number of bacterial colonies in 0.1 mL of each diluted sample was manually counted. Finally, the number of bacterial colonies per gram of bone tissue was calculated and expressed as 10 times the number of bacterial colonies in the 0.1-mL diluted sample (i.e. multiplied by the dilution factor). All tests were conducted in triplicate and under aseptic conditions.
Statistical analysis
SPSS 19.0 statistical software was used for statistical analysis of the obtained experimental data. The measurement data are expressed as the mean ± SD. Comparisons among experimental groups were determined via t-tests. A p < 0.05 was considered statistically significant.
Results
Preparation of a novel ciprofloxacin-PLGC delivery system
DSC curves were used to provide information on the thermal behavior of ciprofloxacin HCl in our prepared drug-delivery system (Figure 1). The DSC curve of ciprofloxacin HCl (Figure 1, curve A) showed the first endothermic curve at 146.2°C, which was suspected to be caused by dehydration. The second endothermic curve at 318.3°C showed the melting temperature of the crystal that was formed during decomposition. In contrast, the blank PLGC only displayed a glass transition at 21.8°C and did not exhibit any other peak up to 350°C (Figure 1, curve B). The curve related to the physical mixture of ciprofloxacin HCl and PLGC (ciprofloxacin-PLGC system) also displayed a glass transition at 21.0°C and showed the endotherms at 142.5°C and 315.6°C corresponding to the loss of water of crystallization and melting of the pure drug, respectively (Figure 1, curve C).

DSC curves of PLGC, ciprofloxacin HCl, and our PLGC-ciprofloxacin system: (A) ciprofloxacin HCl, (B) ciprofloxacin-PLGC system, and (C) blank PLGC.
The structure of the ciprofloxacin-PLGC system was analyzed using FTIR spectroscopy, as shown in Figure 2. The spectrum was recorded in the range of 4000–400 cm−1 for each sample. For ciprofloxacin, one prominent characteristic peak was found between 3500 and 3450 cm−1, which was attributed to the stretching vibration of the OH group and intermolecular hydrogen bonding. For PLGC, the absorption peak at 2948 cm−1 was due to C-H stretching vibrations, and the strong absorption peak at 1753 cm−1 was attributed to C=O stretching vibrations of the DLLA, GA, and CL segments. The peaks at 1183 cm−1 and 1090 cm−1 were attributed to C-O-C stretching vibrations, and the peaks at 1453 cm−1 and 1385 cm−1 were assigned to C-H bending vibrations of DLLA, GA, and CL segments. Collectively, these findings demonstrate that all of the characteristic peaks of ciprofloxacin and PLGC appeared in the spectra of our ciprofloxacin-PLGC system.
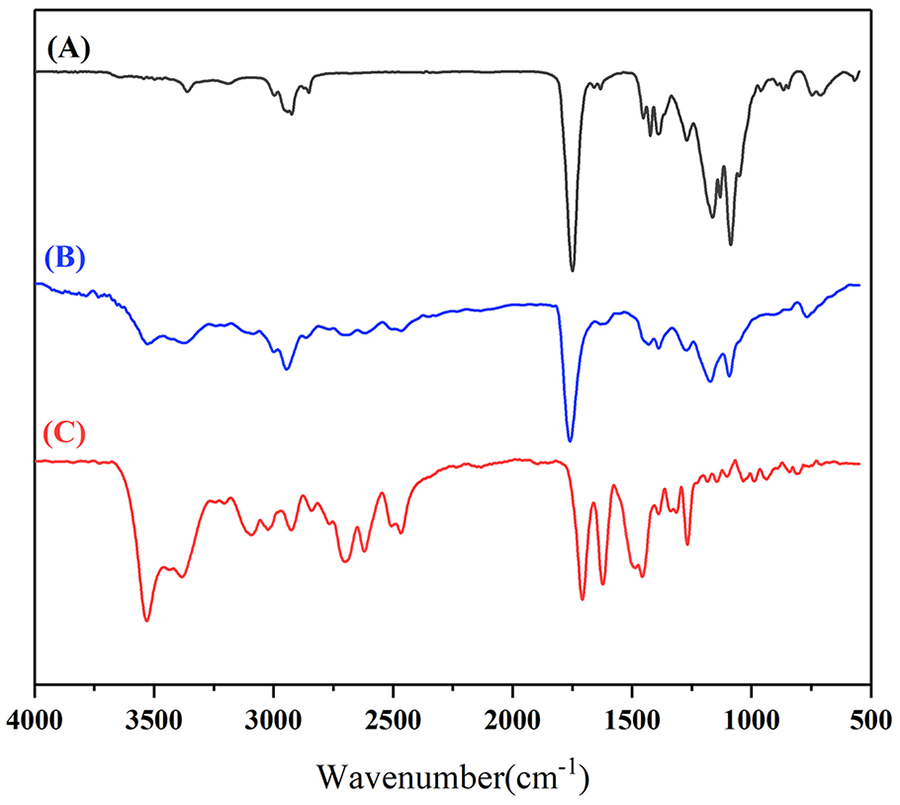
FTIR spectra of PLGC, ciprofloxacin, and our PLGC-ciprofloxacin system: (A) ciprofloxacin, (B) ciprofloxacin-PLGC system, and (C) blank PLGC.
In-vitro release
Figure 3 shows the in vitro release behavior of our ciprofloxacin-PLGC delivery system, indicating that it had a relatively high release rate in the early stages in the form of a burst release. The cumulative release rate on the first day was 19.34%, and the average release amount from 8 to 24 h was 82.4 μg. After 3 days, the drug release rate had subsided, with an average release amount of 80.0 μg/days on day 27. After day 30, a burst release of the drug occurred again. The average release amounts on days 30, 31, 32, and 33 were 105.3, 168.8, 229.7, and 268.5 μg/days, respectively, and the accumulative release rates were 48.68%, 50.53%, 53.06%, and 56.01%, respectively.

Cumulative release rate of our ciprofloxacin delivery system.
Furthermore, scanning electron microscopy (SEM) micrographs of ciprofloxacin-PLGC were monitored (VEGA 3, TESCAN) before and after drug release, and the sample surfaces were sputter-coated with Au to avoid charging before the test. The results showed that ciprofloxacin-PLGC was white and fragile before release, and the sample surface was flat and scattered with drug particles, while the inside was loose with ciprofloxacin drug crystals dispersed therein (Figure 4). After 30 days of release, the ciprofloxacin-loaded PLGC was more fragile, and pits and holes of various sizes appeared on the surface and inside of the PLGC.

SEM observations of our ciprofloxacin delivery system: (a) before and (b) after release.
The drug-release results of our ciprofloxacin-PLGC delivery system in acidic media is shown in Figure 5. The time to reach 57% of drug release at pH 4.5 was 15 days, compared to 33 days at pH 7.0. The cumulative release rate of ciprofloxacin was rapid under acidic conditions and reached 93% on day 17.

Cumulative release rate of our ciprofloxacin delivery system under acidic conditions.
Cytotoxicity
Figure 6 shows the cytotoxicity results of the leachable components from the ciprofloxacin-PLGC system in BMSCs. The results showed that the cell viabilities of were above 90% across the investigated range of concentrations of leachable components. Similar cell viabilities were found for BMSCs incubated in pure ciprofloxacin or blank PLGC at different concentrations.

Cytotoxicity results of different concentrations of leachable components from our ciprofloxacin delivery system (a) and PLGC carriers (b).
Antibacterial effects in vitro
Antibacterial effects in vitro were evaluated semi-quantitatively by measuring the diameter of the antibacterial ring (Figure 7). The inhibition zones were recorded from two or three parallel samples rather than as a triplicate of the same sample at each test point. The results showed that at 2 h after release, the diameter of the bacteriostatic ring of ciprofloxacin released by the ciprofloxacin-PLGC system against Staphylococcus aureus was 34.0 ± 0 mm. After 4 h, 15 days, and 30 days of release, the diameters of the ciprofloxacin bacteriostatic rings were 18.33 ± 1.25, 13.33 ± 2.33, and 26.83 ± 0.83 mm, respectively. In contrast, the untreated group and blank PLGC group did not present any bacteriostatic rings. For the group of pure ciprofloxacin, the diameter of the bacteriostatic ring was 29.2 ± 0.86 mm as the concentration was 15.6 μg/mL, and there no bacteriostatic ring was found for the pure ciprofloxacin with the concentration of 0.24 μg/mL and 0.004 μg/mL.
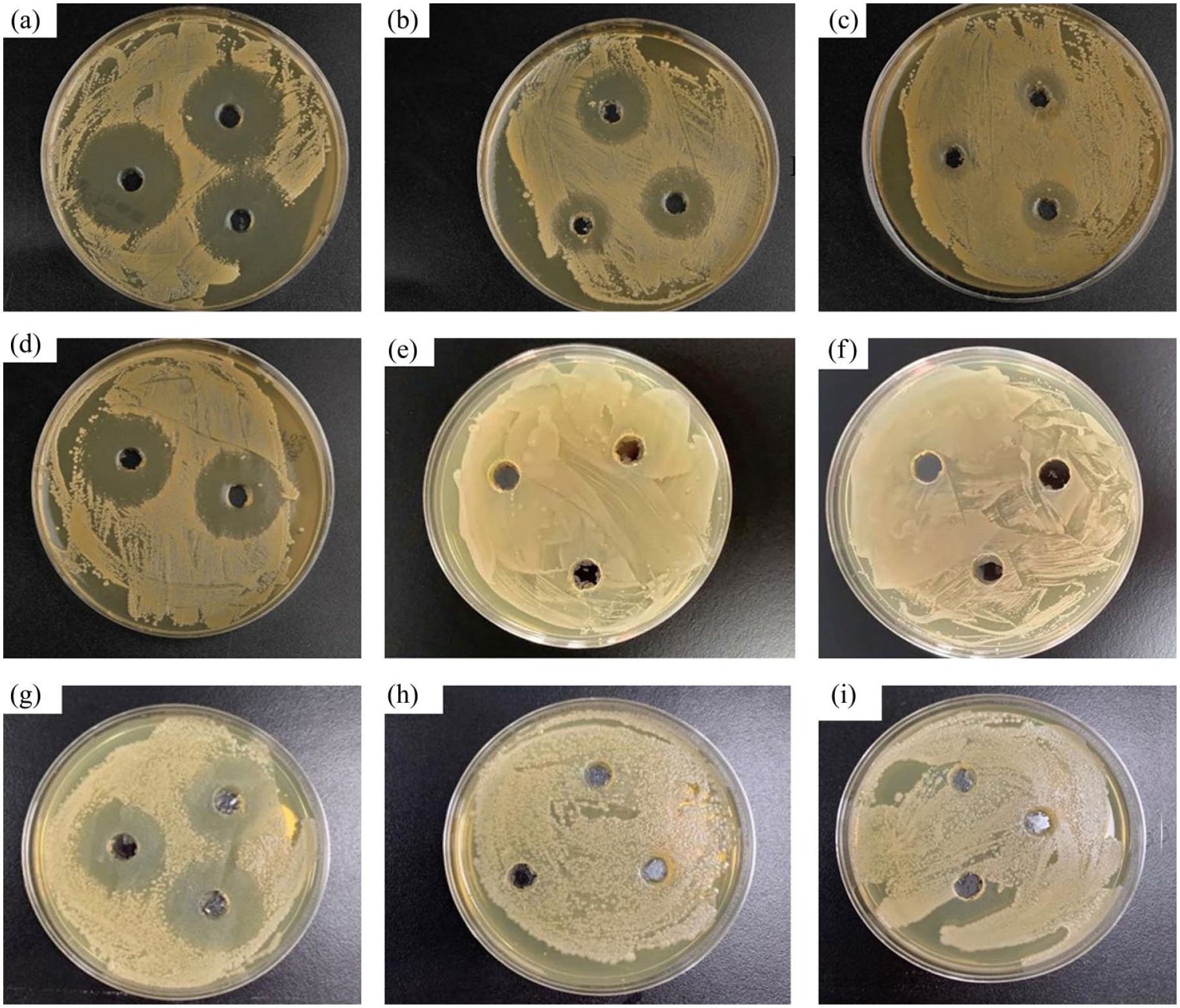
Bacteriostatic rings produced by our ciprofloxacin delivery system at 2 h (a), 4 h (b), 15 days (c), and 30 days (d) after release. The bacteriostatic rings produced by the blank-PLGC and untreated groups are shown in (e) and (f), respectively. The bacteriostatic rings produced by pure ciprofloxacin at 15.6 μg/mL (g), 0.24 μg/mL (h), and 0.004 μg/mL (i) are shown, indicating the antibacterial effects of pure ciprofloxacin after different number of half-lives at 24, 48, and 72 h.
Establishment of a rat model of chronic osteomyelitis
No rats died during the establishment of our rat model of chronic osteomyelitis. At 4 weeks after the induction of chronic osteomyelitis, all rats showed obvious signs of osteomyelitis at the lateral position of the tibia. X-ray imaging (Figure 8) revealed the following: decreased bone density; the bone trabecula was thickened, thinned, or even disappeared; the cortical bone was damaged; dead bone was formed; and the proximal end of the medullary cavity was widened and a periosteal reaction occurred. Based on the scores from Norden et al., one rat scored five points and two rats scored six points. Taken together, these results confirmed the successful establishment of our rat model of chronic osteomyelitis.
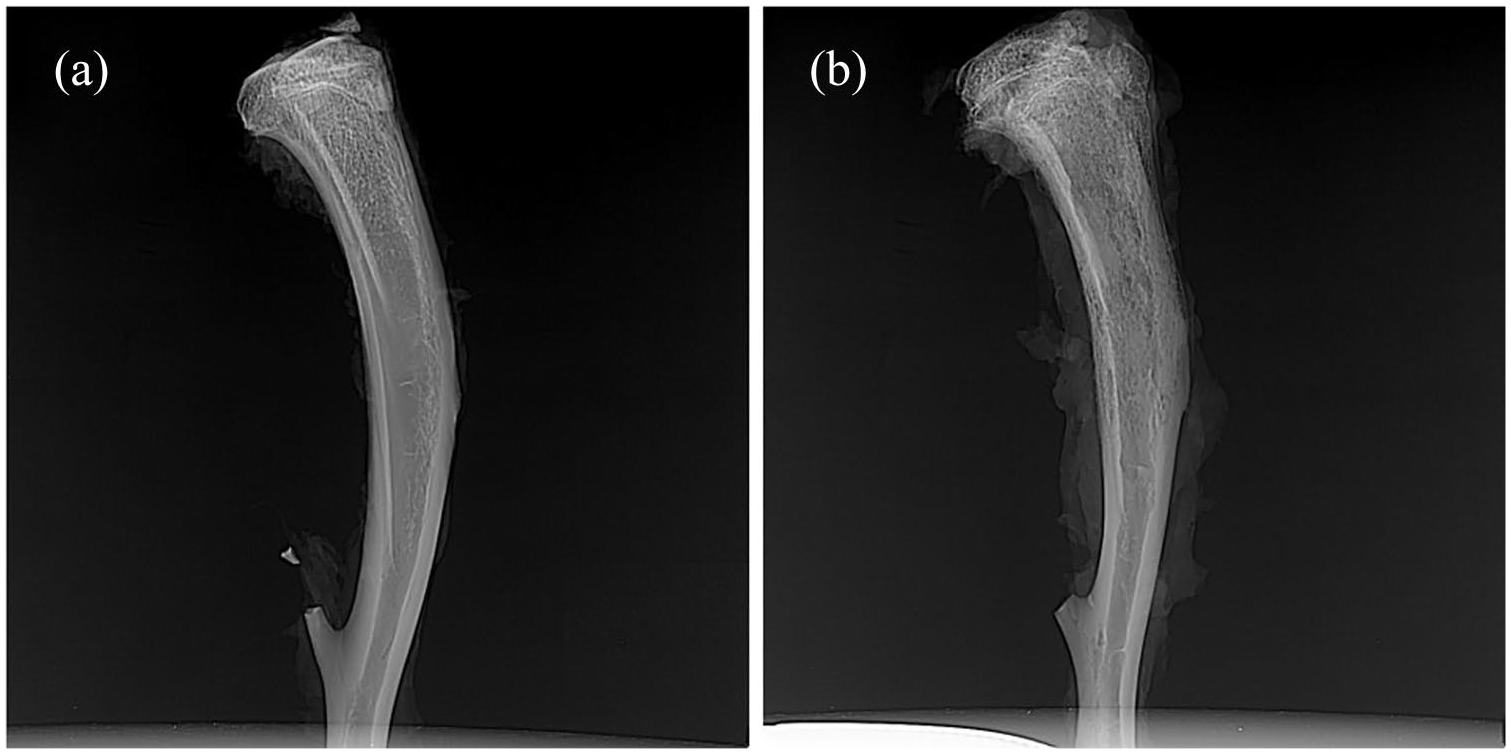
X-rays of normal rat tibia (a) and that after 4 weeks of S. aureus infection (b). Panel (b) shows obvious bone destruction in the bone marrow cavity, the formation of dead bone, unclear bone trabecular structure, and periosteal proliferation.
In addition, there were a large number of inflammatory cells, mainly plasma cells and lymphocytes, in the microcavity lesions, and dead bone formation and fibrous tissue proliferation were observed in HE sections (Figure 9). According to the method described by Petty et al., one rat had moderate osteomyelitis and two rats had severe osteomyelitis. These results further confirmed the successful establishment of our rat model of chronic osteomyelitis.

HE-stained sections of right-tibia specimens after 4 weeks of S. aureus infection. 40× (a), 100× (b). Black arrows: dead bone; green arrows: inflammatory cells; blue arrows: fibroproliferative tissue.
Treatment of chronic-osteomyelitis-model rats via our ciprofloxacin-PLGC delivery system ameliorates pathophysiology
Within 7 days of establishing our rat model of chronic osteomyelitis, each rat’s right surgical limb fell to the ground and the surgical incision became red and swollen. At approximately 14-days post-surgery, wounds, and sinus tracts in the proximal tibia gradually formed in 5/24 rats. After 7 days after implantation of our ciprofloxacin-PLGC delivery system, the swelling of the surgical limbs in the experimental group of rats disappeared and their walking became more flexible. At 14 days after implantation, the wounds of the rats had healed completely and their gaits had returned to normal.
At 4 weeks after implantation, the radiographic results in model rats showed that the proximal and middle tibia had mostly returned to normal structures in the experimental group; in contrast, bone destruction and periosteal reactions were still found in the control group, blank group, and the pure-ciprofloxacin group (Figure 10). At 4 weeks after treatment in the experimental group, inflammatory cells were not observed, and new bone, trabecular bone, and cartilage had formed. In contrast, inflammatory cells were clearly visible in other groups (Figure 11).

X-rays of chronic-osteomyelitis-model rats after four weeks of treatments: (a) Ciprofloxacin-PLGC system, (b) pure ciprofloxacin, (c) blank PLGC, and (d) no treatment.

HE-stained sections of right-tibia specimens from chronic-osteomyelitis-model rats after four weeks of treatments: (a) Ciprofloxacin-PLGC system, (b) pure ciprofloxacin, (c) blank PLGC, and (d) no treatment. Long red arrow, chondrocytes; short red arrow, new bone; yellow arrow, trabecular bone; black arrow, dead bone; green arrow, inflammatory cells.
After treatment, the body weights of the model rats were also evaluated. As shown in Figure 12, the body weights of the experimental group of rats mostly returned to their pre-implantation levels at 2 weeks of implantation. After 4 weeks of treatment, their body weights continued to increase to 385.83 ± 17.23 g, exceeding their pre-implantation body weights (278 ± 12.90 g). At four weeks after treatments, the body weights of rats in the control group, blank group, and pure-ciprofloxacin group also increased to 329.5 ± 16.82, 327.5 ± 13.69, and 333.6 ± 14.57 g, respectively, but these increases were significantly smaller than that of the experimental group (i.e. ciprofloxacin-PLGC delivery).

Changes in body weights of chronic-osteomyelitis-model rats during and following implantation.
After treatment, the WBC counts of rats in the experimental group were 14.05 ± 1.18 × 109 cells/L and were further increased at 3 days of implantation to 15.87 ± 1.78 × 109 cells/L, after which these counts decreased slowly and returned to normal after 4 weeks of treatment. A similar trend for changes in WBC count was found for each of the other groups (Figure 13), but the downward trend was much slower and WBC counts were still higher than normal after 4 weeks of treatments.

Changes in WBC counts of chronic-osteomyelitis-model rats during and following implantation.
Next, the therapeutic effects of our ciprofloxacin-containing PLGC drug-delivery system on osteomyelitis were evaluated in vivo, and the results are reported as the number of bacterial colonies per gram of bone tissue (Table 1). The number of bacterial colonies in the experimental group was (10.6 ± 3.0) × 101 CFU/g, while the numbers of bacterial colonies in the control group, blank group, and pure-ciprofloxacin group were (6.2 ± 0.8) × 105, (6.8 ± 0.4) × 105, and (6.5 ± 0.4) × 105, respectively.
Counts of bacterial content in each group of bone tissue after 4 weeks of treatments (CFU/g, x ± s, n = 6).

Discussion
In the present study, a drug-delivery system was prepared by a physical mixture of PLGC and ciprofloxacin HCl, using PLGC as a carrier. To investigate the effects of this physical mixture on the drug during the loading process, the thermal properties of ciprofloxacin HCl, blank PLGC, and the obtained system were determined by DSC (Figure 1). As shown in Figure 1, an absence of any significant change in the positions of the peaks of ciprofloxacin HCl in the drug-loaded system indicated that there were no significant interactions between the drug and PLGC during the preparation of this drug-delivery system. The lower intensity of the melting peak in the physical mixture might be related to the dilution effect of the carrier. Furthermore, the results of FTIR (Figure 2) showed that all of the characteristic peaks of ciprofloxacin and PLGC appeared in the spectra of our ciprofloxacin-PLGC system, and no appreciable shifts of these peaks were observed. The FTIR analysis also supported the observation that the mixing process did not cause detectable changes in the chemical structure of ciprofloxacin HCl when incorporated into our drug-delivery system.
The antibacterial activity of our ciprofloxacin-PLGC system was evaluated via both in vitro and in vivo experiments. During in vitro release of our ciprofloxacin-PLGC system, a significant burst effect was observed in the early stages (Figure 3), which was due to the fast release of the free ciprofloxacin enriched on the surface of the PLGC, resulting in an cumulative release ratio of 19.34% within 24 h. Then, the ciprofloxacin HCl contained inside of the delivery system was slowly released under the control of PLGC, resulting in a subsequent linear-release profile over the course of 4 weeks. It is well known that the degradation behavior of carriers plays an important role in the release of a drug-delivery system. During the degradation of PLGC, lactic acid and acetic acid were formed. However, it has been demonstrated in our previous studies that the introduction of CL content in PLGA polymers can reduce the formation of acidic products, and that the catalytic degradation effect still works and causes a significantly increased degradation rate of PLGC in the later stage during this process. 22 Furthermore, during the release process of ciprofloxacin, pores were formed after the drug release, promoting the release of drugs inside the system, which in turn led to the formation of more channels, thereby increasing the porosity of the system (Figure 4). This process also increased the contact area between the carriers and the release medium and accelerated the degradation of PLGC. Therefore, the degradation of the ciprofloxacin-PLGC system occurred simultaneously with drug release. The increase in porosity caused by the release and the autocatalysis of degradation products directly led to the rapid degradation of PLGC at the later stage and subsequently promoted rapid drug release, as indicated by cumulative release rates of 50.53%, 53.06%, and 56.01% at days 31, 32, and 33, respectively (Figure 3). The higher cumulative release rate of ciprofloxacin under acidic conditions, which reached 93% on day 17 (Figure 5), confirmed that acidic pH conditions affected burst release from PLGC. Low-pH-catalyzed breakage of the ester linkage of polymer backbones enhances polymer erosion. This behavior was indicative of autocatalysis accelerating PLGC degradation that presented as acceleration in the release behavior of our ciprofloxacin-PLGC system in the present study.
Proper biocompatibility is a prerequisite for the use of pharmaceutical carriers in vivo. Our cytotoxicity results showed that BMSCs in the control group grew well. As shown in Figure 6, the growth of cells in leachable components at different concentrations from 1 to 50 mg/mL was not different from that in the blank PLGC group or the control group, indicating that our ciprofloxacin-PLGC delivery system had good biocompatibility and could be used safely in vivo. The blank PLGC carrier also exhibited excellent cytocompatibility at a concentration up to 50 mg/mL, suggesting that blank PLGC was non-toxic in BMSCs.
Owing to its non-biodegradable characteristics, PMMA has the disadvantage of a low rate of drug release. While biodegradable polymers can be completely degraded after a sufficient time, the contained drug can also be completely released. The in vitro release of our ciprofloxacin-PLGC delivery system showed a release amount of 268.5 μg at day 33, indicating a concentration of 26.85 μg/mL. This result indicated that the concentration of the ciprofloxacin released by our PLGC system after 33 days exceeded the minimum inhibitory concentration of ciprofloxacin (0.4 μg/mL), which demonstrated that the concentration of the amount of the antibiotic released was sufficient for treating osteomyelitis. This result was similar to that of a previous study. 31 However, a sufficient concentration does not necessarily indicate that an antibiotic exhibits bactericidal activity. The preparatory process of our drug-delivery system involved drug loading and sterilization, which may have resulted in a decrease in antibiotic activity. To more directly measure the antibacterial efficacy of our drug-delivery system, we measured the size of the bacteriostatic ring produced by the system-released antibiotics at different time points. The presence of a bacteriostatic ring indicated that ciprofloxacin was biologically active in our samples. The results confirmed that our ciprofloxacin-PLGC delivery system inhibited S. aureus growth for up to 30 days (Figure 7). Furthermore, Figure 7 also shows that the diameter of the bacteriostatic ring in the pure-ciprofloxacin group was decreased significantly as the concentration of ciprofloxacin decreased; when the concentration of ciprofloxacin was decreased to 0.004 μg/mL, no bacteriostatic ring was found. These results imply that after being directly administered in vivo at a concentration of 1 mg/mL, ciprofloxacin was almost completely metabolized after 3 days due to its short half-life. Hence, the growth of bacteriostasis could not be inhibited at this time, demonstrating the controlled-release effect that PLGC played in the antibacterial effects of ciprofloxacin.
Owing to their advantages in terms of their abundant sources and easy feeding, Wistar rats are widely used to establish animal models. In the present study, the success of our rat model of chronic osteomyelitis was confirmed by X-ray and HE staining results. X-ray examination showed that there was obvious bone destruction and sequestrum formation in the bone marrow cavity (Figure 8). The results of HE staining showed that there were many inflammatory cells (mainly plasma cells and lymphocytes) in the lesions of the bone marrow cavity, and the formation of dead bone and fibrous tissue proliferation were also observed (Figure 9). These results confirmed the successful establishment of our rat model of chronic osteomyelitis.
Antibacterial activity was also examined in vivo after treatments. After the establishment of our rat model of chronic osteomyelitis, the rats were treated with one of the following: our ciprofloxacin-PLGC delivery system; a PLGC blank; pure ciprofloxacin; or no treatment (control). After 4 weeks of treatment, bone destruction and periosteal reaction were found in the control group, blank group, and pure-ciprofloxacin group via X-ray, which were not found in the experimental group (Figure 10), indicating the efficacious antibacterial effects of our ciprofloxacin-PLGC delivery system in vivo. HE staining of bone tissue lesions was performed and no inflammatory cells were observed in the experimental group (Figure 11). However, inflammatory cells were clearly visible in the other groups. These results directly showed that the ciprofloxacin-PLGC system eradicated bacterial infection.
After treatment, the bodyweights of the rats in the experimental group gradually increased after 7 days. After 14 days, the body weights of the rats returned to normal and reached their preoperative values. The weight gain of rats in the experimental group was considered to be due to an increase in appetite caused by decreased inflammation. The body weights of the rats in all other groups increased slightly, but were still lower than their normal body weights because these groups did not have significantly reduced inflammation due to insufficient antibiotic treatment. The results of the changes in body weights of each group indicated the significant antibacterial effect of our ciprofloxacin-PLGC delivery system.
The WBC count of normal male Wistar rats was 7.0 × 109 cells/L. In contrast, before implantation of our ciprofloxacin-PLGC delivery system, the WBC count of chronic-osteomyelitis-model rats was significantly higher than normal, at 14.05 ± 1.18 × 109/L, which was the result of chronic bacterial inflammation. Within 3 days after implantation, the WBC count was increased to 15.87 ± 1.78 × 109 cells/L due to the postoperative stress response, whereas there was no statistical difference in this parameter for any of the other three groups (p > 0.05). Thereafter, the WBC counts of all the groups showed a downward trend, but the decrease in the experimental group was more obvious and returned to normal after 4 weeks, while the WBC counts of the other groups were still significantly higher than normal. Statistical results showed that after 4 weeks of treatment, the WBC changes between the experimental group and the other groups were statistically significant (p < 0.05), while there was no statistically significant change between the control group, blank group, and pure-ciprofloxacin group (WBCs, p > 0.05). These results of WBC counts indicated the noteworthy efficacy of our ciprofloxacin-PLGC delivery system.
Finally, microbial testing was used to evaluate the therapeutic effects of our drug-delivery system in ameliorating chronic osteomyelitis. The results showed that the number of bacterial colonies in the experimental group was significantly lower than that in the other groups. Statistical results showed that the difference between the experimental group and the other groups was statistically significant (p < 0.05), while there was no statistical difference between the control group, blank group, and pure-ciprofloxacin group (p > 0.05). These results indicate that our ciprofloxacin-PLGC system achieved effective antibacterial effects via local application.
Conclusion
In our present study, a ciprofloxacin delivery system was prepared using biodegradable PLGC as a carrier for the treatment of chronic osteomyelitis. In vitro, the results of drug release and bacteriostatic rings revealed that our ciprofloxacin-loaded PLGC system sustainably released ciprofloxacin with sufficient concentrations and maintained antibacterial effects for 30 days. In vivo, treatment in chronic-osteomyelitis-model rats showed considerable antibacterial effects of our ciprofloxacin-loaded PLGC system, as indicated by changes in body weights and WBC counts that gradually returned to pre-implantation levels. The outstanding antibacterial effects of our drug-delivery system were also indicated by the lower number of bacterial colonies per gram of bone tissue. Through both in vitro and in vivo antibacterial experiments, our present study confirmed that our PLGC-based ciprofloxacin delivery system offers great potential in the treatment of chronic osteomyelitis.
Footnotes
Authors’ contributions
YL performed the experiments, analyzed the data and wrote the manuscript. XB contributed to data analysis. LA contributed to study design and manuscript editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Project of Health Committee of Shenyang and the Science and Technology Fund Project of Shenyang Medical College (20205043).
