Abstract
Aim:
To analyse the influence of the degree of conversion (DC) and light curing residues of different bulk fills (BFs) composites on the inflammatory profile in the subcutaneous tissue of rats.
Materials and methods:
Resin disks of BF-resins and their active conventional resins (CR; 3M®, Ivoclar®, and Kerr®) were light-cured at 2 mm (BF-superficial) and 4 mm (BF-deep) thicknesses and analyzed by infrared spectroscopy (FTIR; n = 3/group; DC and light curing residues). Then, the disks were implanted in four quadrants in the subcutaneous tissue of Wistar rats (sham, CR, BF-superficial and RF-deep), and after 7, 14, and 28 days, the animals (n = 6/day) were euthanized for histological analysis of the intensity of the inflammatory process (scores 0–3). Kruskal–Wallis/Dunn and ANOVA/Bonferroni tests were used (p < 0.05, Graph Pad Prism 5.0).
Results:
The DC of CR 3M® did not differ significantly compared to BF-superficial and BF-deep resins (p = 0.235). The Ivoclar® and Kerr® resins showed a higher DC with CR and BF-superficial compared to the BF-deep (p = 0.005 and p = 0.011, rctively). Kerr® resins showed a higher Bis-GMA/UDMA ratio, especially in BF-deep resin (p < 0.05). 3M® and Ivoclar resins did not show high inflammation scores, but for Kerr® BF resins (superficial and deep), the inflammatory process was significantly higher than that in the CR and sham quadrants (p = 0.031).
Conclusion:
The tissue inflammatory response after resin inoculation depends on the DC and light curing residues of Bis-GMA.
Introduction
Composite resin is the primary material of choice for direct restorations, both for its mechanical and optical characteristics. 1 However, there are disadvantages, such as polymerization shrinkage, adhesion failures, and the need for incremental techniques, as well as the possibility of incomplete conversion of monomers to polymers, which may lead to mechanical problems and biological tissue damage. 2
Bulk fill (BF) resin composites have been proposed in recent years with the potentiality of being cured with thickness of 4mm or more, according of manufacturers. These materials are characterized by particular properties which allow the polymerization of layers of such a depth. 3 The technology employed in formulating these materials varies by manufacturer, but some mechanisms are the same for all manufacturers and have been developed to ensure adequate mechanical and biological properties, such as the use of new methacrylate-based monomers, the use of chemical modulators of the polymerization reaction, new photo-initiating systems, increased translucency and reinforcement of the inorganic phase by fiberglass. 4
One of the main characteristics of materials, in general, is biocompatibility. The biocompatibility of composite resin is mainly determined by its degree of conversion (DC), as this factor may determine the greater or lesser release of unpolymerized monomers during their curing process. 5 Total DC depends mainly on intrinsic factors such as the chemical structure of the monomer and polymerization initiator concentration and extrinsic factors such as the polymerization conditions. 6 It has been shown that a decrease in DC can lead to a decrease in the physical-mechanical properties and an increase in the release of monomers into the oral environment. 7 Some of these released substances have mutagenic and cytotoxic characteristics 8 and may cause inflammatory reactions in the gingival, pulp, and oral mucosa tissues. 9
Although the literature broadly addresses the mechanical characteristics of these materials, there is still no published data on the possible inflammatory tissue reaction caused by the release of non-fully polymerized compounds in living tissues. Published studies addressing such reactions are performed by inoculation into cell cultures, most commonly fibroblasts or pulp cells. 10
Thus, this study aims to evaluate the influence of the degree of conversion (DC) and light curing residues of different bulk fills (BFs) on the inflammatory profile in the subcutaneous tissue of rats.
Materials and methods
Materials and sample preparation
We used three different BF-composites and its analogs no-BF composites. The three different BF-composites used were a Tetric N-Ceram Bulk Fill (Ivoclar®) resin, a Filtek Bulk Fill One (3M®) resin and a Sonic Fill (Kerr®) resin. The control non-BF-composite respectively used in this study were a Tetric N-Ceram (Ivoclar®) resin, a Filtek Z-350 (3M®) resin and a Herculite Classic (Kerr®) resin. Their chemical compositions are shown in Table 1.
Chemical composition of BF and non-BF resin disks implanted subcutaneously in Wistar rats.

The specimens were prepared using two 2-mm-thick steel devices measuring 4×4 cm containing concentric circles (5 mm × 2 mm). The resins were inserted as a single insertion into each circle and were covered and separated by polyester tape and polymerized directly for 20 seconds (Valo®, Ultradent, South Jordan, UT-1000 mW/cm²).
After polymerization, the disks were removed by extrusion. Thus, two 5-mm-diameter × 2-mm-thick composite bodies were obtained with disk 1 with the surface radiating directly on the top of the device in direct contact with the photopolymer radiation. These specimens were called surface bulk fills (BF-superficial). Disk 2, located below Disk 1, was irradiated by the light filtered by Disk 1, called deep bulk fill (BF-deep).
Conventional resin (CR) specimens were obtained using only one device, obtaining 5 mm × 2 mm disks.
Analysis of the degree of conversion of Bis-GMA residues by FTIR spectroscopy
In order to analyse the degree of conversion, 27 resin disks were used, n = 3. The result was obtained by infrared spectroscopy (FTIR). The reading parameters used were as follows: 2 cm–1, 128 scans, in the range between 520 and 4000 cm–1, and power of 100 mW. As a means to obtain the unpolymerized spectrum, the specimens were obtained by inserting the material into 2-mm-thick steel plates with 5-mm-diameter perforations stabilized with glass slides and adhesive tape. The sets were taken to the Vertex Spectrophotometer (Bruker Optik GmbH). Baseline correction and curve normalization were performed using the Opus program (Bruker Optik GmbH), increasing its intensity, correcting, and limiting interference from other elements such as water and oxygen.
Then, the set was removed from the equipment and photoactivated according to the methodology described in the sampling protocol, and a new reading was performed. The DC was evaluated using aliphatic and aromatic = C-H bands located at 4743 and 4623 cm–1, respectively. After photoactivation, the band peak height values of each material were entered in the following formula: conversion degree = 100 * 1 – [(aliphatic / aromatic) photopolymerized / (aliphatic / aromatic) unpolymerized].
Additionally, the peak heights of the 1610 cm−1 (Bis-GMA) and 1640 cm−1 (UDMA) bands were measured and recorded after light curing to analyse the light curing residues of the composite resins.
Animal model of subcutaneous implant
This project was approved by the Christus University Center Ethical Committee on Animal Use (CEUA) under protocol number 12/17, and 54 female 4-month-old Wistar rats weighing 250–280 g were used. The animals were housed in temperature-controlled rooms (n = 6/cage) and received water and food ad libitum.
After trichotomy and antisepsis of the dorsum of the animals (2% chlorhexidine), the rats were anesthetized with xylazine (20 mg/kg) and ketamine (80 mg/kg). Four incisions were made on the backs of the animals that were divided into quadrants I (left front), II (right front), III (right posterior), and IV (left posterior) clockwise starting from the left front portion. The incisions were 3 cm from edge to edge (horizontally and vertically).
After dissection, quadrant I was sutured without material insertion (sham group), quadrant II was sutured with CR disk insertion, and quadrants III and IV were sutured with bulk-fill superficial and deep, respectively, resin disk insertion. The disks were prepared at the time of the surgical procedure, and the suture was performed with 4-0 nylon thread (Procare; n = 6 rats/group/day of euthanasia).
After 7, 14, and 28 days of inoculation, the animals were euthanized by anesthetic solution overdose, and the disks and adjacent tissues were removed and fixed in 10% buffered formalin at pH 7.0. After histological processing, 3-µm-thick histological slides were stained with haematoxylin-eosin.
Ten microfields (400×) from each quadrant were analyzed in the regions containing the resin specimens on a camera-attached microscope (BX43, Olympus®; U-TV0.63XC, Olympus®) with image capture software (Olympus Soft Imaging LC Micro software, Olympus®). Histological scores for inflammatory infiltrate according to Gomes-Filho were classified according to intensity as absent (score 0), mild (score 1, up to 25 inflammatory cells per field), moderate (score 2, between 26 and 125 inflammatory cells per field) and intense (score 3, more than 125 inflammatory cells per field). 11
Statistical analysis
FTIR spectroscopy data were expressed as the mean ± SEM and compared by ANOVA/Bonferroni test. Histological scores were expressed as the median (minimum–maximum) and compared by the Kruskal–Wallis/Dunn test. All analyses were performed using 95% confidence in GraphPad Prism 5.0 software (p < 0.05).
Results
Conversion degree rating
The average DC of 3M® resins did not differ significantly when comparing control resin (92.9 ± 2.0%), BF-superficial (91.6 ± 5.7%) and BF-deep (80.6 ± 4.4%; p = 0.235). The group of Ivoclar® resins showed a higher DC in the control (85.5 ± 5.5%) and BF-superficial (89.5 ± 5.4%) resins, unlike the deep region of its bulk fill resin (58.7 ± 1.2%; p = 0.005). Similarly, control (95.9 ± 2.4%) and BF-superficial (94.4 ± 5.8%) Kerr® resins showed a higher DC than their BF-deep resins (43.6 ± 9.6%; p = 0.011; Table 2).
Analysis of the degree of conversion (A) remover? and Bis-GMA/UDMA (B) remover? ratio of CR or BF-resin (surface or deep) disks.

ANOVA/Bonferroni, mean ± SEM; A,Bp < 0.05 intragroup analysis; a,bp < 0.05 intergroup analysis.
3M®-CR resin = Filtek Z350 XT; 3M-BF-resin = Filtek Bulk fill One; Ivoclar®-CR = Tetric N-Ceram; Ivoclar®-BF-resin = Tetric N-Ceram Bulk fill; Kerr®-CR-resin = Herculite Classic; Kerr®-BF-resin = Sonic fill Bulk fill.
There was no difference in the degree of conversion of control (p = 0.264) or BF-superficial (p = 0.611) resins. However, the 3M® BF-deep resin showed a higher DC than the same Kerr® resin while neither group presented a difference from the Ivoclar® brand (p = 0.015; Table 2).
Evaluation of the relation between Bis-GMA (1609.4 cm−1) and UDMA (1537 cm−1) residues
The 1609.4 cm−1/1537 cm−1 ratio of the 3M® group showed no difference between the control (0.83 ± 0.05) or BF-superficial (0.72 ± 0.07) and BF-deep (0.79 ± 0.05; p = 0.438). However, Ivoclar® resin showed a lower ratio of 1609.4 cm−1/1537 cm−1 in the control (0.54 ± 0.02) than in BF-superficial (0.83 ± 0.03) and BF-deep (0.82 ± 0.01; p < 0.001). In Kerr® resins, the ratio 1609.4 cm−1/1537 cm−1 was significantly lower in control (1.54 ± 0.48) and BF-superficial (1.79 ± 0.15) resins than in BF-deep resin (2.70 ± 0.18; p < 0.001; Table 2).
There was no difference in the 1609.4 cm−1/1537 cm−1 ratio in the control resins of the three brands (p = 0.108). However, Kerr® resin showed a significantly higher 1609.4 cm−1/1537 cm−1 ratio than the 3M® and Ivoclar® resins in both the superficial (p < 0.001) and deep (p < 0.001) BF types (Table 2).
Histological analysis of the inflammatory profile around the specimens
At day 7, all samples from all sites showed intense inflammatory infiltrate. At day 14, there was a significant reduction in the intensity of inflammatory infiltrate in all sham sites (p < 0.05; Table 2).
In the 3M® group, after 14 days, the conventional resin (CR) group presented moderate inflammatory infiltrate, and in the groups treated with BF resin, the inflammation was between moderate and intense (p = 0.076). After 28 days, the results were similar, and despite slight variations, there was no difference between the intensity of the inflammatory infiltrate in the surgical sites of the sham group, control resin, and superficial or deep BF resins (p = 0.099; Figure 1, Table 3).
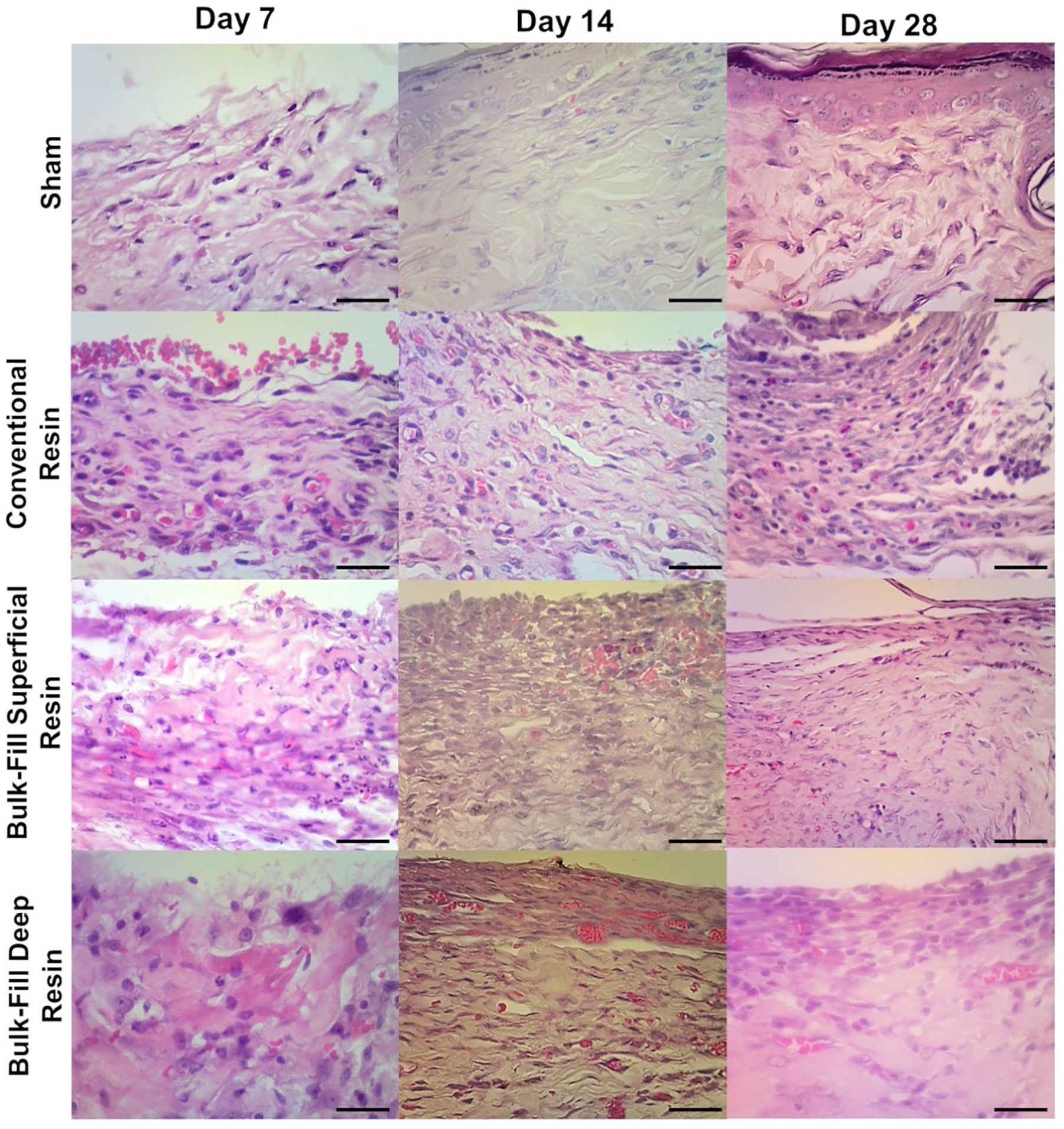
Histopathological analysis of CR-resin Filtek Z350 XT or BF-resin Filtek Bulk fill One resin disks (3M®) implanted subcutaneously in Wistar rats (HE, 400×; scale bar = 50 µm).
Histopathological analysis of CR or BF-resin disks implanted subcutaneously in Wistar rats.

Kruskal–Wallis/Dunn test (n = 6/group); Data showed as median (minimum–maximum); A,Bp < 0.05 intragroup analysis; a,bp < 0.05 intergroup analysis.
Histologic scores (11): 0 = 0 inflammatory cells/microscopic fields; 1 = 1–25 inflammatory cells/microscopic fields; 2 = 26–125 inflammatory cells/microscopic fields; 3 = >125 inflammatory cells/microscopic fields.
In the Ivoclar® group, all sites submitted to resins presented intense inflammatory infiltrate after 14 days, with values significantly higher than those of the sham group (p = 0.036). On day 28, all sites had mild or absent inflammation (p = 0.163; Figure 2, Table 3).

Histopathological analysis of CR-resin Tetric N-Ceram or BF-resin Tetric N-Ceram Bulk fill resin disks (Ivoclar®) implanted subcutaneously in Wistar rats (HE, 400×; scale bar = 50 µm).
In the Kerr® group, on day 14 and day 28, the inflammatory infiltrate intensity scores were significantly higher in both the superficial and deep BF-treated groups than those in the sham group (p = 0.028 and p = 0.031, respectively; Figure 3, Table 3).

Histopathological analysis of CR-resin Herculite Classic or BF-resin Sonic fill Bulk fill resin disks (Kerr®) implanted subcutaneously in Wistar rats. (HE, 400×; scale bar = 50 µm).
Discussion
The technique of inserting resins in incremental layers has been the standard for sufficiently converting monomers 12 and consists of placing increments of resinous material to a thickness of 2 mm or less, followed by exposure to light curing, and then repeating the increments until the cavity is fully filled. 13 A distinct advantage of this technique is the limited thickness of the resin to be penetrated by light. Therefore, limiting the thickness of the increments allows for adequate light penetration and subsequent polymerization. Despite these benefits, there are several disadvantages associated with incremental techniques, such as the possibility of blistering on incorporation or contamination between resin layers, adhesion failures between increments, placement difficulty due to limited access in small cavities, and increased clinical time. 14
BF compounds were introduced to overcome these disadvantages. These materials promise to promote light transmission to achieve a polymerization depth greater than 4 mm.3,15 There have been some investigations of the clinical performance of bulk-fill composites 15 including, marginal quality investigations, 16 cusp deflection, 3 cusp deflection in joints with microleakage, 17 adhesion to the lower dentine wall, 15 and DC. 18 However, no clinical study has investigated the elution of unreacted monomers from bulk-fill compositions. The present study aimed to identify the interactions between these materials and the tested tissues in rats.
In this study, the deepest Kerr® resin had a lower DC and a more intense inflammatory infiltrate for 28 days than the other groups. Thus, corroborating the literature, there is a direct relationship between DC and the intensity of the inflammatory infiltrate. 19 DC reduction impairs the chemical and mechanical proprieties of BF resins, 20 and the high bioavailability of monomeric residues is strongly toxic to several cell types, 21 such as fibroblasts 22 and odontoblasts. 23 The direct contact of monomeric residues active apoptosis regulatory caspases causes toxicity and an inflammatory process, 24 as shown in Kerr® BF resin disks, negatively affecting pulp tissue, 25 following unbound composite diffusion through the dentine to the pulp. 26 Thus, the degree of conversion of a composite is crucial for determining its biocompatibility.
Fibroblasts are more sensitive to monomer toxicity. The exposition of connective tissue of the rats treated with Kerr® BF resin disks showed a high bis-GMA proportion and prolonged the inflammatory process. Despite blood cell cultures not being influenced by monomeric Bis-GMA, this monomer leads to overexpression of proinflammatory cytokines due to cell toxicity,27–29 as shown in our study.
In Kerr® resin, which presented the highest inflammatory process, in addition to the poor degree of conversion, a high proportion of Bis-GMA was observed. Bis-GMA has the lowest degree of conversion according to the literature. The degree of conversion increases in the following order: Bis-GMA < Bis-EMA < UDMA < TEGDMA. 25 Similar experimental models with subcutaneous inoculation of resin cements with a high concentration of bis-GMA also led to an increase in CD68 phagocytic cells and a prolonged inflammatory process, 19 as shown in our data.
Bis-GMA is considered the most viscous and least flexible monomer owing to the strong intramolecular bond of hydrogen with its hydroxyl groups and the presence of rigid aromatic nuclei in its structure. 26 According to the results of this study, Kerr® and Ivoclar® bulk fill resins, in their deepest portion (4 mm), presented lower DC results compared to the other groups. This finding is related to the reactive characteristics of Bis-GMA that exist in their compositions. The bulk-fill 3M® resin, which has AUDMA and UDMA in its composition, showed higher degrees of conversion, confirming the literature data. 26
Adequate light curing dramatically attenuates cell damage induced by monomers. 30 Therefore, despite the BF resins allowing application in a single increment, 20 there is a gradual decrease in DC in the deeper layers of the material, 31 thus we suggest caution in the use of BF resin with a large concentration of bis-GMA in very deep cavities (>2 mm). UDMA and TEGDMA are also cytotoxic,22,24 however, light curing of the composite with higher UDMA content (Tetric N-Ceram, Filtek Z-350, Tetric N-Ceram Bulk Fill, Filtek Bulk Fill One) led to fewer monomer residues and toxicity.
It has been suggested that there is a significant relationship between unpolymerized monomers and cytotoxic effects. 32 Residual monomers, such as TEGDMA and bis-GMA, which are released from composite resins, have been shown to induce differential cytotoxicity and apoptosis in human dental pulp cells. TEGDMA, HEMA, and Bis-GMA have been shown to induce oxidative stress production in dental pulp cells. 33 After polymerization, residual monomers can be released into oral fluids or the diet and produce cytotoxic effects 34 including, cell imbalance, increased oxidation levels, due to the generation of ROS and subsequent cell death via apoptosis, as well as DNA damage. 35 The lower degree of conversion found in the deep portions of Sonic fill-Kerr® resins and the higher ratio of Bis-GMA/UDMA residues may be directly related to the longer perpetuation of inflammatory tissue levels (for 28 days). These monomers with a lower DC also activate T lymphocytes that greatly prolong the inflammatory process, 36 as shown in rats 28 days after the beginning of the protocol.
Another critical factor affecting the leaching of residual monomers is the nature and molecular size of the monomers in the resin. Smaller molecules are expected to leach faster than larger molecules. Small molecular monomers can be extracted in considerably larger amounts than large molecular monomers. 32 TEDGMA is a low-molecular-weight monomer that exhibits greater mobility and is eluted faster than large molecules, such as Bis-GMA, 21 which may justify the prolonged inflammatory response observed in this study in resins with high Bis-GMA (Kerr®) concentration. Another contributing factor is the fact that Bis-GMA is poorly water soluble, making it difficult to dissolve over time. 37
It is important to emphasize that in vitro studies on the biological effects of resin-based materials generally include several commonly used substances (monomers, oligomers, photo-initiators) acting separately 2 in specific cell types. However, the isolated findings observed in vitro have limitations compared to broad findings from experimental animal models, in view of the complexity of tissue and cellular interactions of an organism.
Thus, in this study, it was observed that the inflammatory process is significantly influenced by the low degree of conversion, mainly associated with the high concentration of Bis-GMA residues. Studies with tissues closer to dental tissues should be performed to observe the impact of this process on the dentin-pulp complex.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
