Abstract
Zirconia is the preferred material for dental restorations; however, dental restorations are usually affected by zirconia fractures due to chipping and delamination of the veneer ceramic. One effective solution for repairing chemically inert zirconia frameworks is to strongly chemically bond them with the composite resin via surface modification. Thus, the bonding strength between the zirconia and composite resin determines the performance of dental restoration. Herein, we investigate the shear bond strength between zirconia ceramic and two ceramic repair systems before and after thermal cycling based on different surface pretreatments, including air-abrasion and a novel silane coupling agent. When treated with combined sandblasting, novel silane and 10-methacryloyloxydecyl hydrogen phosphate act as a bonding agent for the zirconia surface, and the maximum shear bond strength achieves 27.5 MPa, as measured by a universal testing machine through the average of 16 separate measurements. The results show that the combined treatment resists the interface damage caused by expansion and contraction during thermal cycling. The long-term bond durability is due to the micro-mechanical bond force formed by resin and ceramic, and the chemical bonds of Zr-O-Si at the interface. Results indicate that selective pretreating the surface results in high bond strength between the zirconia and the composite resin, which is meaningful to optimize dental restoration.
Keywords
Introduction
Yttria-stabilized tetragonal zirconia polycrystals (Y-TZP), compared with dental silica-based ceramics, exhibit high flexural strength, fracture toughness, 1 and biocompatibility. As a result, zirconia is widely used as a key material for single crowns,2 the framework for fixed partial dentures (FPDs),2,3 orthodontic brackets, and implant abutments.2–9 Due to the high content and thickness of crystals, 10 zirconia is pure white and has low translucency. 11 Thus, surface modifications are commonly used to improve the color and translucency 12 for zirconia restorations.13,14 Generally, feldspar laminated ceramics sintered on the zirconia framework are commonly used. 15 The destruction of zirconia restoration is chipping and delamination of veneer ceramics, rather than the fracture of the zirconia framework itself.16–19 Compared with metal-ceramic FPDs, zirconia FPD veneers show a significantly higher fracture rate. 20 According to previous studies, the chip-off rate is in the range from 13% to 15.2% between 24 and 60 months.21,22 The restoration is replaced by the damaged porcelain decoration. However, for patients, the complete replacement is not necessarily a convenient choice due to various reasons.23,24 In clinic, an effective method to repair a small area of damage, which does not cause aesthetic or occlusal function problems, is to use composite resin to directly bond to zirconia in the oral cavity. 25
The bonding strength between the composite resin and zirconia ceramic is crucial for repairing zirconia frameworks via direct repair technology.26,27 Due to its high stiffness and chemical inertness to resist corrosion, it is very difficult to physically or chemically modify the surface of zirconia ceramic. Previous studies reported that air-particle abrasion or sandblasting effectively improved the wettability of the ceramic surface, increased the surface energy, and created a rough surface. As a result, the improvement of the bonding area leads to micro-interlocking retention of the composite resin or resin cement. 28 However, sandblasting might lead to crystal transition from the tetragonal phase to the monoclinic phase, which reduces the mechanical properties of the material. 29 Thus, some manufacturers suggest using alumina particles with a size of less than 50 μm for sandblasting, so as to minimize the crystal phase transition. 30 In order to improve the success rate of repairing zirconia ceramics with composite resin, the surface of zirconia is modified by many intraoral repair systems, such as the Ceramic Repair System (Ivoclar Vivadent, Liechtenstein) and Clearfil Repair (Kuraray, Okayama, Japan). Both systems pretreat the surface of the zirconia ceramic with a silane coupling agent that contains 3-methacryloxypropyletrimethoxysilan (3-MPTS) (Figure 1), and cohere the composite resin onto the surface with different bonding agents containing different adhesive monomers. The 10-methacryloyloxydecyl hydrogen phosphate (MDP) phosphate monomer with bonding agent is able to bond with the zirconia surface via a chemically stable covalent bond (-P-O-Zr-) 31 to enhance the bonding performance between the ceramic and resin cement or binder. However, some studies indicated lower bond strength was obtained between MDP-based resin cement and zirconia ceramic without sandblasting. 32 In addition, zirconia does not respond to common etching and silanization procedures due to its chemical inertness. Therefore, in this study, we develop a silane coupling agent to silanize zirconia ceramics. The experimental silane is a mixture of two silane solutions that contain 3-MPTS and 1,2-bis(trimethoxysilyl)-ethane (BTE) (Figure 2). The silane is used to test the effect on the bonding strength between the zirconia ceramic and composite resin.

Chemical structure of 3-methacryloxypropyletrimethoxysilane (3-MPTS).

Chemical structure of 1,2-bis(trimethoxysilyl) ethane (BTE).
The purpose of this study is to investigate the effects of different silane coupling agents and different repair systems on the bonding strength of zirconia ceramic to composite resin. The hypotheses are as follows: (a) the surface pretreatment of zirconia ceramic with a silane coupling agent can effectively improve the shear bond strength between the zirconia ceramic and composite resin; (b) a zirconia ceramic can repair a system that contains a MDP phosphoric acidic monomer as the bonding agent, resulting in a high shear bond strength.
Methods
Preparation of zirconia blocks
The materials used in this study are shown in Table 1. A total of 256 zirconia ceramic blocks with a side length of 6mm and height of 2mm (ZS-B70, KaVo Arctica, Germany) completed sintering in a vacuum furnace according to the steps specified by the manufacturer. The sintered zirconia ceramics were embedded by pouring resin (Shofu, Tokyo, Japan) so that the 6 mm square side was exposed. This exposed surface was used as a bonding surface, and was polished under flowing water in the order of 320, 600-, and 1000-grit of silicon carbide paper, respectively, to remove loose alumina particles and polluted organic impurities. The polished specimens were cleaned by ultrasonic concussion with 95% ethanol for 10 min.
The composition and manufacture of materials in this study.
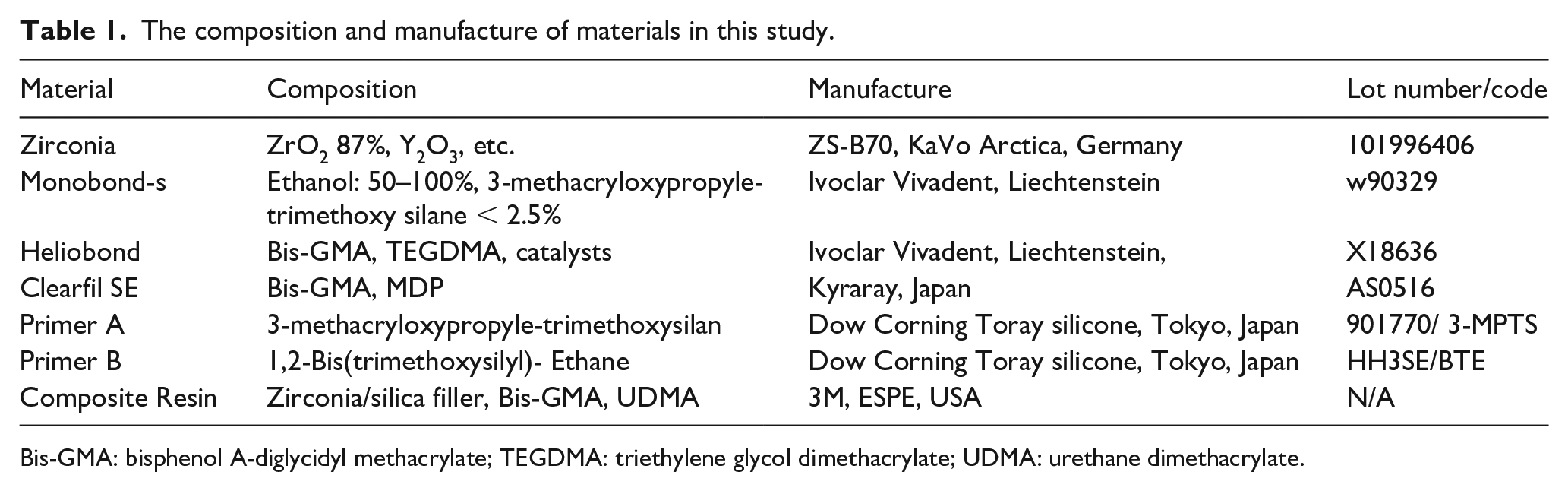
Bis-GMA: bisphenol A-diglycidyl methacrylate; TEGDMA: triethylene glycol dimethacrylate; UDMA: urethane dimethacrylate.
Preparation of the silane coupling agent
The silane coupling agent was double liquid, consisting of Primers A and B. Primer A was the mixture of 3-MPTS and BTE, and then 50 mg mixed silane was dissolved in 1 ml ethanol. The content of 3-MPTS and BTE in the mixed silane was 70 and 30 mol%, respectively. Hydrochloric acid solution was used (0.1 mol/L, Wako Pure Chemical Industries, Osaka, Japan) as a catalyst to accelerate the methoxyl hydrolysis of silanes. Firstly, the hydrochloric acid solution was diluted to 0.05 mol/L by deionized distillation until the pH was 1.65. Secondly, the diluted hydrochloric acid aqueous solution was mixed with 50 vol% ethanol. This mixed solution was used as Primer B of the silane coupling agent in this experiment.
Preparation of the specimens after pretreatment
The 256 samples were randomly divided into eight groups (32 samples in each group) according to different surface pretreatment methods or repair systems, as shown in Figure 3.
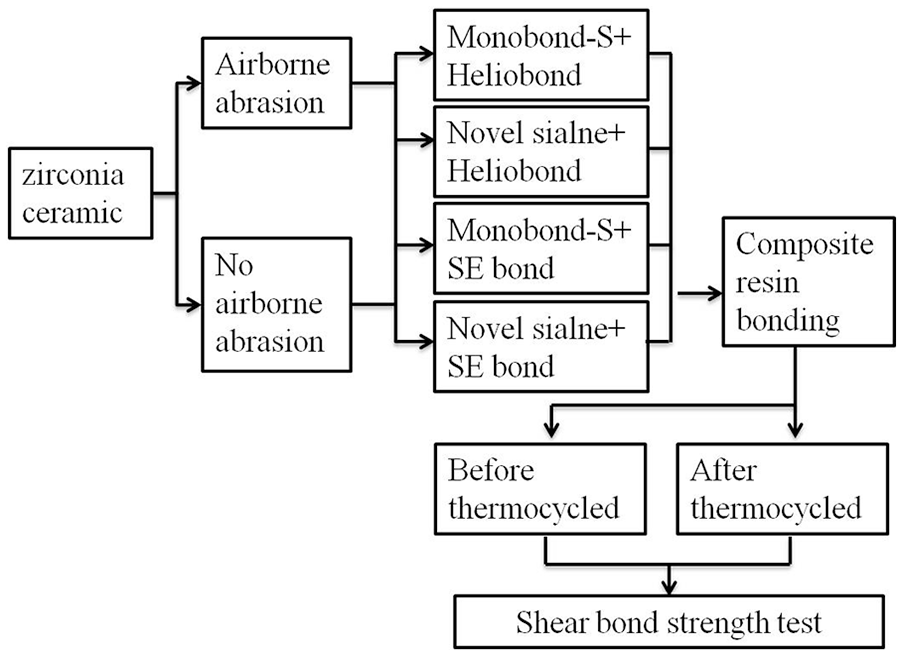
Schematic distribution of the test protocol.
For Groups 1–4, from a distance of 20 mm, the surface of zirconia was treated by airborne-particle abrasion with silica-modified Al2O3 particles (50 µm) using an oral Microblaster (Basic Quaattro IS; Renfert, Hilzingen, Germany) for 30 s at 0.25 MPa. After being cleaned with 95% ethanol, the test sample was air dried to remove the remnants of sand particles. The remaining Groups 5 and 6 remained without sandblasting pretreatment. As per the manufacturer’s instructions, one bottle type of commercial ceramic primer (monobond-s) was applied to the zirconia surface of Groups 1, 2, 5, and 6 for 60 s.
The designed silane coupling agent was utilized for treating chemically the surface of zirconia in Groups 3, 4, 7, and 8. The amount of each application of silane was 0.3 mg, which was composed of equal amounts of Primers A and B, and the actuation duration was 60 s. After the silane was evaporated basically in 60 s duration, the silanized zirconia ceramic was dried at room temperature (23 ± 1°C) for another 2 min.
After drying in air, the surface was treated with Heliobond for Groups 1(AMH), 3(ANH), 5(NMH), and 7(NNH) or SE bond for Groups 2(AMS), 4(ANS), 6(NMS), and 8(NNS) as the bonding agent and lightly cured (Coltolux LED, Coltene, OH, USA) for 20 s, respectively. In order to ensure the output power density (350–500 mW/cm2) of the light source, a 3 Light Checker (3M Health Care, Tokyo, Japan) was used.
Bonding and testing
After surface pretreatment, we placed the prepared silicone rubber ring on the surface of the zirconia ceramic. The silicone ring had a round hole with an inner diameter of 4 mm and a thickness of 2 mm. The composite resin (Z350, 3M, ESPE) was filled into the circular hole and pressed with 50 g weight. The residual composite resin and silicone mold were carefully removed after the irradiation with visible light.
The above eight groups were random divided into two subgroups (n = 16): one subgroup was stored in 37°C water for 24 h (before thermocycling (TC)), while the other subgroup was treated for TC. The samples were thermocycled between two water baths at 5°C and 55°C for 10,000 cycles (Thermal Shock Tester, Thomas Kagaku, Japan). The retention time in each water bath was 60 s and the transfer period was 7 s.
A universal testing machine (Instron, 3367, Canton, MA, USA) with crosshead speed of 1.0 mm/min until failure was used to test the shear bond strength of the composite resin for the zirconia surface. The shear load was calculated in megapascals by dividing the load at failure by the surface area of each sample in square millimeters. If the composite resin fell off before the shear test, the bonding strength was defined as 0 MPa. A cross-sectional view of diagrammatic sketch of the shear bond test is illustrated in Figure 4.

Diagrammatic sketch of the shear bond test.
Failure mode analysis
After the shear load test, the failure modes for all specimens were determined by a digital optic microscope (MSV 330, Anyty, 3R, Japan) and are illustrated in Figure 5. The examination revealed several types: (a) cohesive failure arising from the fracture of the composite or zirconia, as shown in Figures 5(a) and (b); (b) mixed failure with both cohesive and interface failure, as shown in Figure 5(c); and (c) interfacial failure at the interface between the zirconia and composite resin, as shown in Figure 5(d).

Optical images of the typical fracture surface: (a), (b) cohesive failure: the fracture of the composite or zirconia; (c) mixed failure: both cohesive and interface failure; and (d) interfacial failure between the zirconia and composite resin.
Statistical analysis
The shear bond strength and standard deviation of each group before and after TC were calculated. The effects of different surface treatments and fatigue tests were performed using two-way analysis of variance (ANOVA) combined with Tukey’s least significant difference post-hoc test (SPSS Software, San Diego. CA, USA), and p < 0.05 is considered significant. 33
The complex chi-square (χ2) test was used to compare and determine the significant differences of the three types of fracture mode. The level of 0.05 was considered statistically significant.
Scanning electron microscopy observation
The sample surface was highly polished and coated with gold. A scanning electron microscope (SEM; Hitachi S-4800) was used to observe the change of surface morphologies of the zirconia samples before and after airborne-particle abrasion and TC.
Results
The shear bond strength (Table 2) between the zirconia ceramic and composite resin is controlled by the silane treatment (experimental or monobond-s) and the two kinds of bonding agents. Before TC, lower shear bond strengths of 7.5, 6.8, 9.8, and 8.4 MPa are obtained for the NMH, NMS, NNH, and UTPC groups, respectively. This result suggests that without the sandblasting treatment, the zirconia surface exhibits weak bonding with the composite resin. The groups treated with air-abrasion, that is, AMH, AMS, ANH, and ANS, exhibit relatively higher shear bond strengths of 17.4, 21.5, 30.4, and 31.2 MPa, respectively. Treatment with the novel silane agent results in an increase in the bonding strength (Groups 3 and 4) compared with treatment with the commercially available silane (Groups 1 and 2). The bonding effect between the zirconia ceramic surface and composite resin can also be demonstrated by the fracture mode when different treatments are applied. Without the sandblasting treatment, most of the specimens show mixed failure (Figure 5(c)) or interfacial failure (Figure 5(d)). Although there is no significant difference in the fracture mode, the specimens treated with the novel silane agent (Groups 7 and 8) show more mixed failures than those treated with the commercial silanes (Group 5 and 6). It is noteworthy that Groups 7 and 8 show few cohesive failures. When treated by sandblasting, most of the specimens (Groups 1–4) show cohesive failure (Figures 5(a) and (b)), with significant differences compared to the samples without sandblasting treatment.
Shear bond strength (mean value and standard deviation in brackets) between the zirconia ceramic surface and composite resin before and after thermocycling; type of fracture mode.

The different superscript numbers for the horizontal row: different (p < 0.05). The same subscripts (a–e) for vertical column: p > 0.05). [1/2/3]: cohesive/mixed/interfacial failure. n = 16.
TC: thermocycling.
The zirconia surface morphologies after the treatment, shear bond strength measurement and TC are also observed via the SEM. In comparison of the surface before the treatment (Figure 6(a)), the air-abrasion improves the roughness of the surface (Figure 6(b)) and creates an irregular surface, which is useful for both chemical bonding and the interaction with the composite resin. After the shear bond strength, the treated zirconia surface shows cohesive failure (Figures 6(c) and (d)), including structure damage of the composite or its framework, indicating high bonding strength between the zirconia and resin cement. The treated (designed silane coupling agents) zirconia surface (Figure 6(f)) shows few cracks after TC compared with that treated with commercial silane (Figure 6(e)), which shows an excellent bond durability in TC. The fracture mode controlled by different treatment indicates that the surface air-abrasion and silane treatment lead to the formation of a micro-retention structure and favors the chemical bonding at the interface for high bonding strength.

Scanning electron microscopy images of the zirconia surface: (a) before and (b) after air-abrasion; (c) and (d) cohesive failure of the zirconia surface after the shear bond strength test; (e) zirconia surface treated with the commercial silane after thermocycling (TC); (f) zirconia surface treated with the designed silane coupling agents after TC.
To investigate the influence of silane treatment and the two kinds of bonding agents on the bond durability, the bonding strength between the zirconia ceramic surface and composite resin after 10,000 TCs is shown in Table 2. It can be seen that the lowest bonding strength is obtained in the groups without the sandblasting treatment (Groups 5–8), and these groups show obviously reduced bonding strength after TC. In particular, for Groups 5 and 6, treated with commercial silane coupling agents (monobond-s), a bonding strength of 0 MPa is obtained, with a standard deviation of 0. The fracture mode for Groups 5 and 6 changes from mostly interfacial failures to all interfacial failures, whereas that of Groups 7 and 8 changes from mostly mixed failures to mostly interfacial failures. The former shows no significant differences, but the latter exhibits significant differences. After TC, the highest bonding strengths of 27.5 and 23.9 MPa are obtained in groups ANS and ANH, respectively, which are higher than those of the other groups. Groups 3 and 4 are treated with sandblasting and the novel silane agent but with different bonding agents (Helio-bond and Clearfil SE). Treatment with the novel silane coupling agent results in a more than two-fold increase in the bonding strength compared with that of treatment with the commercial silane coupling agents (11.8 MPa for AMH and 14.5 MPa of AMS, Figure 6(e)). The type of fracture mode for Groups 1 and 2, which are treated with the commercial silane coupling agents, changes obviously from cohesive failures to mixed failures and interfacial failures. This result shows significant improvement compared with Groups 5 and 6 without the sandblasting. In contrast, most of specimens in Groups 3 and 4 show good fracture modes and cohesive failures.
Discussion
The purpose of this study is to demonstrate the effect of different zirconia surface pretreatment and repair systems on the bonding strength of zirconia ceramic to composite resin. From the initial shear bond strength, the results indicate that sandblasting is crucial for increasing the bonding strength between the zirconia ceramic surface and composite resin. Treatment with silane (experimental or monobond-s) or selectively using a bonding agent does not enhance the bonding strength between the zirconia ceramic and composite resin. The treatment by silane agent effectively increases the bonding strength between the zirconia ceramic and composite resin. Moreover, there is no significant difference in the bonding strength between two kinds of bonding agents (Heliobond and Clearfil SE) when the other experimental methods are kept the same. By observing the fracture mode of the bonding interface, the novel silane agent treatment has the potential for improving the bonding effect of zirconia ceramic and composite resin. This result arises from a low initiation efficiency in silane hydrolysis of the methacryloyl organic acid monomers of commercial silanes (monobond-s). As a result, some incompletely hydrolyzed 3-MPTS molecules are adsorbed physically on the ceramic surface, competitively occupying the Zr-O positions on the ceramic surface and hindering the formation of Zr-O-Si bonds.33–35 For example, the carbonyl groups of 4-methacryloxyethyltrimelliticacidanhydride (4-META) are bonded to the silanol groups on the ceramic surface by hydrogen bonding, which hinders the chemical adsorption of 3-MPTS molecules, thus leading to a reduction in their condensation efficiency. This effect influences the bonding strength between the zirconia ceramic surface and composite resin. When the experimental novel silane agent is initiated by aqueous hydrochloric acid, the C-H groups in ≡Si-O-CH3 (or ≡Si-O-CH2CH3) become significantly weaker, while the ≡Si-OH group becomes stronger and, finally, a very strong -Si-O-Zr group appears. 35 These results indicate that hydrochloric acid effectively promotes the hydrolysis of 3-MPTS, resulting in the adherence of hydrolyzed 3-MPTS to the ceramic surface by chemical bonds. The SEM observation (Figure 5(b)) reveals that air-particle abrasion with alumina particles is a suitable zirconia surface treatment method for increasing the surface energy of ceramics and the wettability between resins and ceramics.36,37
When TC is applied, most of the experiment groups exhibit a significant decrease in bonding strength. For Groups 5 and 6, in spite of being treated with the novel silane coupling agent, the bond between the ceramic and composite resin fails before the shearing test. Groups 7 and 8 also show obviously reduced bonding strength compared with that of before TC. In addition, the ceramic surface in Group 7 shows a higher strength than that in Group 8, indicating that the treatment increases bond durability. This result is consistent with the observation that there are few crack in Figure 6(f). In addition, few bonding failures occur. The difference between the ANH and ANS arises from the different types of acidic adhesive monomers comprising the bonding agents, TEGDMA (ANH) and MDP (ANS), respectively. MDP, an acidic phosphate ester monomer, forms a stable chemical bond with the zirconium oxide in zirconia ceramics

The formation of a stable chemical bond is effective to improve the bonding strength between the zirconia ceramic surface and composite resin and to resist the damage caused by expansion or contraction during thermal cycling.38,39 Treatment with the novel silane coupling agent is an effective way to improve the bonding strength and bond durability. The high performance with the experimental silane coupling agent is due to the formation of an interpenetrating polymer network (IPN) (Figure 7) by polymerization at the interfaces using siloxane and polymer after adding BTE to 3-MPTS. The polymerization is effective to graft covalently composite resins to the silane surface.
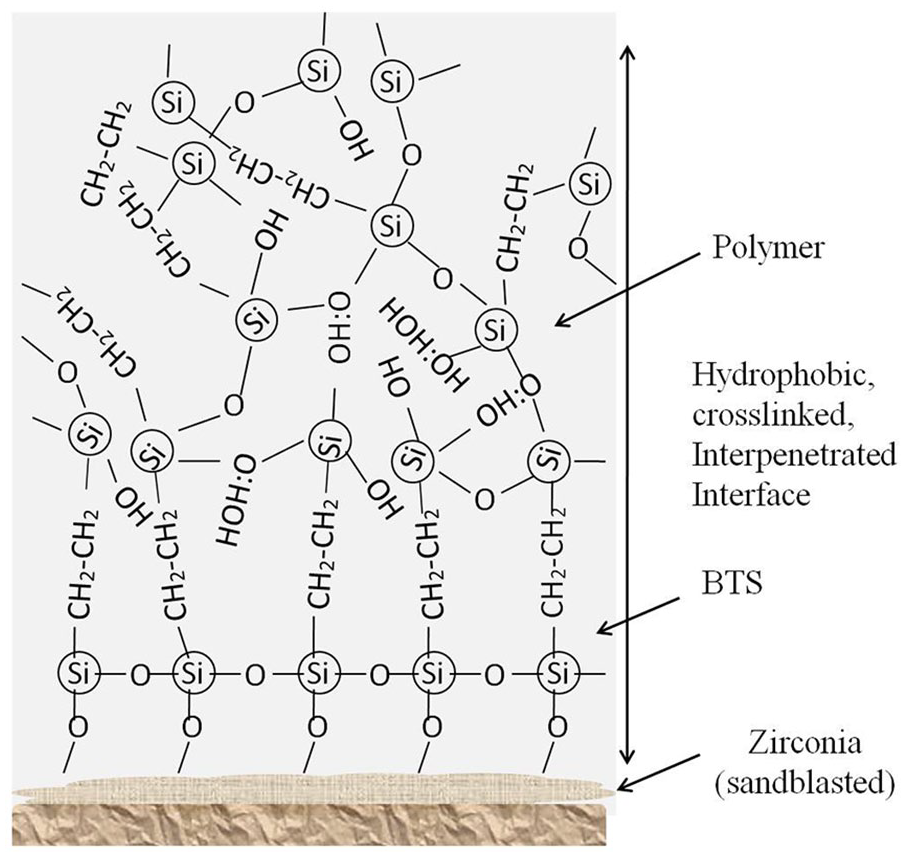
Chemical bonding between the composite resin and zirconia ceramic through the cross-linked interpenetrating interface. BTE: 1,2-bis(trimethoxysilyl)-ethane.
By observing the different changing of fracture modes between Groups 1, 2 and 5, 6, it can be seen that sandblasting is effective to improve the bond durability. However, many interfacial failures occur in Groups 1 and 2, which indicates that the treatment with both sandblasting and the commercial silane coupling agents is insufficient to resist the damage caused by expansion or contraction during thermal cycling. The significant difference with Groups 1, 2 and 3, 4 indicates that the novel silane coupling agent is an effective treatment to resist the bonding layer damage caused by the thermal-cycled fatigue test. This may be due to the formation of an IPN in the novel silane coupling agent, which has a certain elasticity for resisting the internal stress caused by the different thermal expansion rates of ceramics and resins during TC. 40 It is noteworthy that the result exhibit significant differences compared to ANH and ANS (Groups 3 and 4). This result indicates that treatment with the bonding agent containing MDP has a positive effect on the bond durability.
Conclusion
We investigated the effect of two zirconia ceramic repair systems and silane coupling agents on the bonding strength between zirconia and composite resin before and after TC. Air-particle abrasion with alumina particles is suitable to create an irregular zirconia surface and to increase the surface bonding, leading to the formation of a micro-retention structure. The combined treatment with the novel silane coupling agent and a MDP-containing bonding agent increases the bonding strength and bond durability. As a result, we obtain the maximum bonding strengths of 27.5 and 23.9 MPa after TC for ANS and ANH, respectively. The long-term bond durability arises from the micro-mechanical bond force formed by the resin and ceramic, and chemical bonds of Zr-O-Si at the interface. The improvement of shear bond strength between zirconia and composite resin is beneficial to dental restoration by selectively pretreating the surface of zirconia ceramic.
Footnotes
Acknowledgements
We would like to thank Ying Chun Sun for his assistance and guidance in this research.
Contributorship
Rui li and Ying Chun Sun researched literature and conceived the study. Rui li was involved in protocol development, gaining ethical approval, and data analysis. Rui li wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript
Guarantor
Rui li
Declaration of conflicting interests
Rui li is an employee of Stomatological Hospital, Tianjin Medical University.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an award from the Natural Science Foundation of Tianjin, China. (18JCYBJC95500) to Rui li.
