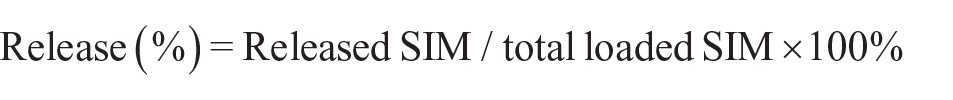Abstract
Simvastatin (SIM) has been documented to induce the osteogenic differentiation of periodontal ligament stem cells (PDLSCs). To establish an efficient release system for periodontal regeneration, a polycaprolactone (PCL) membrane scaffold containing SIM was electrospun and evaluated. The obtained PCL–SIM membrane scaffold showed sustained release up to 28 days, without deleterious effect on proliferation of PDLSCs on the scaffolds. PDLSCs were seeded onto scaffolds and their osteogenic differentiation was evaluated. After 21 days, expressions of collagen type I, alkaline phosphatase and bone sialoprotein genes were significantly upregulated and mineralized matrix formation was increased on the PCL–SIM scaffolds compared with the PCL scaffolds. In a heterotopic periodontal regeneration model, a cell sheet–scaffold construct was assembled by placement of multilayers of PDLSC sheets on PCL or PCL–SIM scaffolds, and these were then placed between dentin and ceramic bovine bone for subcutaneous implantation in athymic mice. After 8 weeks, the PCL–SIM membrane showed formation of significantly more ectopic cementum-like mineral on the dentin surface. These findings demonstrated that the PCL–SIM membrane scaffold promotes cementum-like tissue formation by sustained drug release, suggesting the feasibility of its therapeutic use with PDLSC sheets to improve periodontal regeneration.
Keywords
Introduction
Statins, the commonly prescribed cholesterol-lowering drugs in patients with hypercholesterolemia, have been shown to possess anti-inflammatory, antioxidant, and bone anabolic effects.1–3 Simvastatin (SIM), at a low dose, is believed to facilitate periodontal hard tissue engineering by having an osteogenic effect in periodontal ligament stem cells (PDLSCs). 4 Multiple local applications of SIM are capable of strongly decreasing the amount of inflammation and bone loss in an experimental periodontitis rat model.5,6 Despite these positive effects, SIM also has possible systemic side effects such as abnormalities in liver enzymes. After oral administration of SIM, first-pass hepatic metabolism limits the drug amounts in the periodontal tissue and has hindered its clinical application in periodontal regeneration.5,7 A sustained release scaffold needs to be established so that SIM can be applied locally to periodontal tissue. Local delivery systems such as a drug-eluting scaffold might provide advantages of overcoming possible adverse effects and delivering sufficient concentrations of drug to the target disease site.8–11
Electrospinning is an established method to create small diameter fibers and has become widely used in biomedical applications including tissue regeneration and drug delivery.9,11 In previous studies, hydrophobic small molecule drugs (300–700 Da) were incorporated into electrospun fibers at low drug loading of typically < 1 wt% to produce the sustained release, which is practical for the local injection of SIM into the tendons.9,12,13 An electrospun nanofibrous rosuvastatin-loaded poly(lactic-co-glycolic) acid stent has been shown to produce sustained drug release for 4 weeks. 14 Electrospun scaffolds are also emerging as promising tissue engineering scaffolds to promote periodontal regeneration, which is characterized by the formation of cementum, bone, and periodontal ligament.15,16 An electrospun microfibrous membrane was designed for simultaneous engineering of periodontal complex and it was appropriate for harvesting periodontal ligament cell sheets to form deposited hard tissue on the tooth dentin surface. 16 Accordingly, we hypothesized that a SIM-loaded electrospun membrane might guide periodontal hard tissue regeneration as a drug-releasing scaffold.
Cell sheet engineering, a strategy for seed cell delivery to the periodontal defect area, has been introduced to regenerate periodontal attachment and restore health.17,18 In the present study, multiple layers of PDLSC sheets were harvested using a SIM-loaded electrospun membrane scaffold. The obtained cell sheet–scaffold construct was placed between the surface of the root dentin and ceramic bovine bone (CBB), simulating dental matrix under physiological conditions, for heterotopic regeneration experiments. The drug release of the SIM-loaded scaffold, PDLSC cell viability, and osteogenic differentiation in vitro, as well as the capacity for periodontal regeneration in vivo, were investigated.
Materials and methods
Fabrication of SIM-loaded membrane scaffolds
To provide sustained release, SIM-loaded membrane scaffolds were fabricated by the electrospinning method (Figure 1(a)). Polycaprolactone (PCL, M = 60,000, Esun Industrial, Shenzhen, China) (300 mg) was first dissolved in 3.75 mL chloroform to prepare a polymer solution at a concentration of 8% (w/v). SIM (567020; Merck KGaA, Darmstadt, Germany) was mixed with the polymer solution at a concentration of 0.5% (w/w) with respect to the polymer used. After stirring at room temperature, the homogenous PCL–SIM mixture was transferred to a 10-mL syringe attached to a steel needle of 0.6 mm diameter. A positive high voltage of 15 kV was introduced to the needle and a fixed grounded plate for electrospinning. The emitting rate of the polymer solution was 0.7 mL/h controlled by a syringe pump. 10 The electrospun fibers were trapped with the aluminum foil collector located 15 cm from the needle tip. After 5 h, the obtained PCL–SIM membrane scaffolds were dried under vacuum to evaporate the solvent before cutting into 10-mm diameter sample pieces. PCL membrane scaffolds without SIM loading were prepared as control. All PCL and PCL–SIM scaffolds were sterilized in ultraviolet C light for 4 h.

(a) The principle of electrospinning to produce SIM-loaded membranes. Overall appearance (b) and scanning electron microscopy image (c) (original magnification, 2000×) of PCL–SIM membrane scaffolds. (d) The calibration curve of SIM was plotted using known drug concentration (µg/mL) on the x-axis and average OD value at 238 nm measured by ultraviolet/visible spectrophotometry on the y-axis. The equation of linear regression was y = 0.0056x + 0.0027, R2 = 0.9938, in this experiment. (e) Cumulative release of SIM was expressed as a relative percentage from PCL–SIM membranes. The released buffer was measured by ultraviolet/visible spectrophotometry at 238 nm. The drug concentration in the released buffer was calculated from the calibration curve of SIM. The percentage of SIM release was calculated and plotted versus time. All experiments were conducted in duplicate with three independent sets. Bars represent mean ± SD (n = 3).
The morphology of electrospun SIM-loaded fibers was observed by scanning electron microscopy (SEM) (S-3400N, Hitachi, Japan) at the accelerating voltage of 15 kV. After critical drying with a critical point dryer (E-1010, Hitachi, Tokyo, Japan), the samples were gold-coated and observed.
In vitro release of SIM
The release profile of SIM from the electrospun PCL–SIM membrane was evaluated in phosphate-buffered saline (PBS) (pH 7.4) using an elution method. Each sample was accurately weighed and placed in a glass test tube containing 1 mL PBS, then incubated at 37°C in a plate shaker for up to 28 days. At specific time intervals of 0, 1, 2, 3, 4, 5, 6, 7, 10, 13, 17, 20, 24, and 28 days, the release medium was collected for measurement and replaced with 1 mL fresh PBS. The collected buffer absorbance was measured by ultraviolet/visible spectrophotometry (Cary 50 spectrophotometer; Varian, Agilent Technologies, Santa Clara, CA, USA) at 238 nm. The drug concentration in the released buffer was calculated from the calibration curve of SIM in the same buffer. This analysis was performed in three replicates for each time point. The percentage of SIM release was calculated as per the following equation and plotted versus time
where the total loaded SIM is the theoretical weight of SIM loading during the electrospinning process.
Isolation and characterization of PDLSCs
All experimental protocols were reviewed and approved by the Experiment and Ethics Committees of Shanghai Stomatological Hospital, Fudan University (20170007). PDLSCs were isolated from healthy premolars extracted from two donors (aged 11 and 13 years) for orthodontic reasons. Briefly, the periodontal ligament tissues were minced and seeded in growth medium consisting of α-minimum essential medium (GIBCO, Carlsbad, CA, USA), 10% fetal bovine serum (GIBCO), 1%
Cell viability of PDLSCs on scaffolds
PDLSCs (2 × 105) within 20 µL of medium were seeded onto the PCL and PCL–SIM scaffold samples in 24-well plates. The cells were allowed to adhere for 2 h before 2 mL of growth medium was added. PDLSCs on scaffolds were further cultured for 1, 3, and 5 days and the proliferation was measured using a cell counting kit-8 (CCK-8; Dojindo, Kumamoto, Japan). The culture medium was removed from each well and 800 µL of 10% CCK-8 solution was added. After incubation for 2 h, 200 µL aliquots were transferred to a 96-well plate for measurement. Cell viability was determined from the optical density value measured at 600 nm with a multidetection microplate reader (Epoch 2; Biotek, Winooski, VT, USA). PDLSCs cultured on scaffolds at day 5 were stained using a live/dead cell imaging kit (R37601, Invitrogen), and then imaged with a laser scanning confocal microscope (A1, Nikon, Melville, NY, USA).
In vitro osteogenic differentiation of PDLSCs on scaffolds
To evaluate osteogenic differentiation, PDLSCs (2 × 105) were seeded onto PCL or PCL–SIM scaffolds, and further cultured in osteogenic differentiation medium containing 5 mM β-glycerophosphate, 50 µg/mL ascorbic acid, and 10 nM dexamethasone (all from Sigma-Aldrich, St Louis, MO, USA) for 21 days. The medium was changed twice a week. Cells on PCL scaffolds cultured in growth medium were used as a negative control.
Quantitative real-time polymerase chain reaction
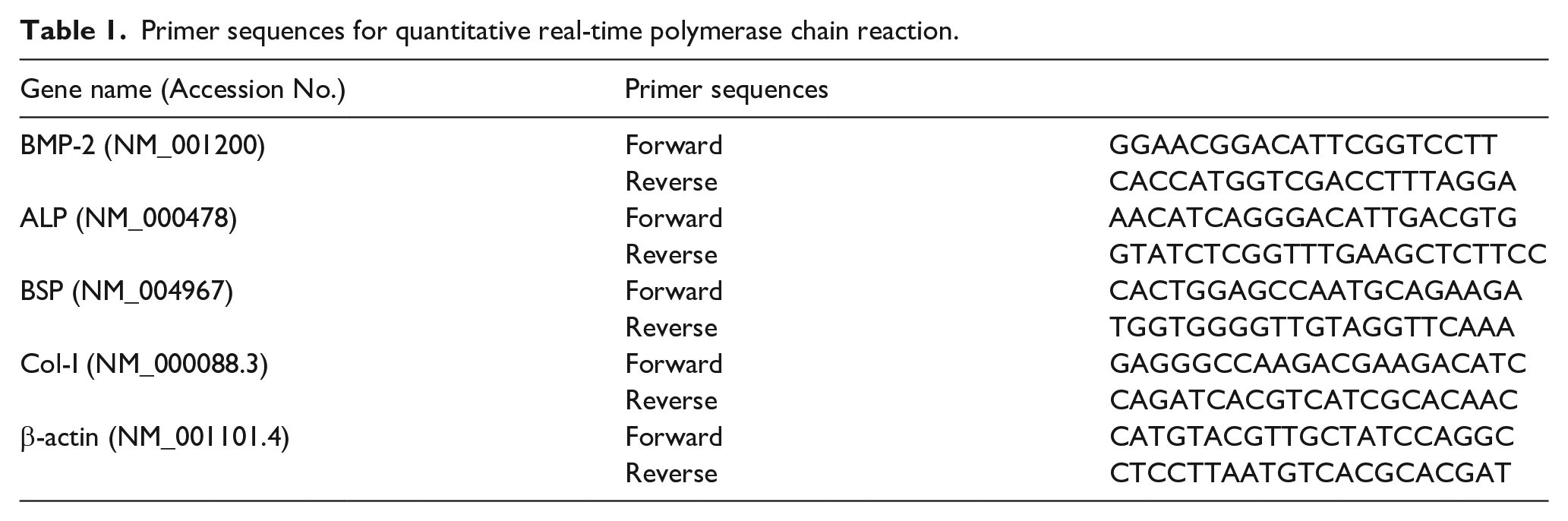
Total RNA was extracted from PDLSCs with TRIzol solution (Invitrogen) and quantified by NanoDrop 2000 (Thermo, USA), followed by cDNA synthesis. To evaluate the gene expression level of osteogenic markers, quantitative real-time polymerase chain reaction (PCR) was carried out in triplicate with SYBR Green reagent (TaKaRa Bio Inc., Shiga, Japan) using a specified thermal cycler (LightCycler 96, Roche, USA). The osteogenic marker genes evaluated were bone morphogenic protein-2 (BMP-2), alkaline phosphatase (ALP), bone sialoprotein (BSP), collagen type I (Col-I), and the endogenous control gene β-actin. The PCR program was 40 cycles of amplification at 95°C for 10 s, followed by 60°C for 30 s. The primer sequences are listed in Table 1. The results were calculated and normalized to the level of β-actin.
Primer sequences for quantitative real-time polymerase chain reaction.
Alizarin Red S staining
The osteogenic differentiation of PDLSCs was evaluated by Alizarin Red S (ARS) staining of mineral deposition. Samples were fixed with 4% paraformaldehyde for 10 min and washed with distilled water. Calcium accumulation on the scaffolds was stained with 2% ARS solution (Sigma-Aldrich) for 10 min, washed five times with distilled water and then photographed. For the quantitation of ARS staining by colorimetric assay, the deposition was dissolved in 10% cetylpyridinium chloride (Sigma-Aldrich). The solution was transferred to a 96-well plate and the absorbance at 562 nm was measured using a microplate reader.
In vivo transplantation of PDLSC sheets on scaffolds
To evaluate the potential of the SIM-releasing membrane for simultaneous periodontal regeneration in vivo, we prepared a periodontal regeneration model according to previous reports.16,17
Construction of PDLSC sheets with the scaffold
At first, cell sheets were harvested using an electrospun PCL or PCL–SIM membrane (Figure 2(a)). PDLSCs (1 × 105) were seeded into 12-well plates and cultured in growth medium supplemented with 100 µg/mL ascorbic acid. After 10 days, cell sheets matured and were harvested. A PCL–SIM membrane was placed in a well and the cell sheet was pulled towards the edge of it using sterile forceps. The membrane was then placed with the harvested cell sheet facing upwards, allowing the cell sheet to adhere to the membrane. Another cell sheet was harvested after 30 min. The procedure was repeated three times until three layers of cell sheets were assembled with the PCL–SIM membrane. An electrospun PCL membrane without SIM loaded onto it was used as the control.

Harvesting of multiple periodontal ligament stem cell (PDLSC) sheets with a polycaprolactone or polycaprolactone–simvastatin membrane scaffold. (a) Schematic representation of PDLSC sheets assembling with the membrane scaffold. (b) Representative scanning electron microscopy (SEM) image indicates morphology of the top view of the cell sheet–membrane construct. (c) SEM image of the cross-sectional view of the cell sheet–membrane construct. (d) Immunofluorescence staining of the PDLSC sheets showed a well-developed network of collagen fibers. DAPI staining is shown in blue, collagen type I in red. (e) Hematoxylin–eosin staining of the cross-sectional view of the cell sheet–membrane; multiple cell sheets adhere onto the membrane scaffold. In (b) and (c), the scale bar represents 10 µm; in (d) and (e), the scale bar represents 50 µm.
The obtained cell sheet–scaffold construct was immunostained for Col-I (ab34710, Abcam) to visualize the extracellular matrix of PDLSC sheets. The top view and cross-sectional view of the cell sheet–scaffold construct were observed using SEM. The cell sheet–scaffold construct was immersed in liquid nitrogen and crosscut with scissors. The samples were then fixed in 2.5% glutaraldehyde at 4°C overnight, dehydrated through an acetone dilution series. The cell sheet–scaffold construct was embedded in paraffin and sections were stained with hematoxylin–eosin (HE) to observe the cohesion between the membrane and harvested cell sheets.
Implantation and histological examination
To prepare a periodontal regeneration model, the cell sheet–scaffold construct was placed between the surface of root dentin and CBB (Fourth Military Medical University, China) for heterotopic regeneration experiments (Figure 3(a)). Samples of root dentin (5 mm × 3 mm) were prepared by mechanically removing the cementum from the tooth root. Under a stereoscope (M80, Leica, Wetzlar, Germany), a cell sheet–scaffold construct was placed onto the surface of root dentin and incubated in medium for 2 h to ensure cell sheets adhered to the dentin surface. A CBB block was assembled onto the scaffold and sutures were used to keep the stability of the periodontal regeneration model. The model was incubated in medium for transplantation subcutaneously on the dorsal of athymic mice. The implants were retrieved after 8 weeks and fixed in 4% paraformaldehyde for longer than 48 h. The samples were decalcified in 10% EDTA with weekly changes of solution for 12 weeks and embedded in paraffin. Sections (5 µm thickness) prepared from the central portion of the implants were stained with HE and Masson’s trichrome. To quantify the newly formed mineralized deposition area, ImageJ2X software (National Institutes of Health, Bethesda, MD, USA) was used on four representative microphotographs, and the percentage of regenerated hard tissue area on the dentin surface was calculated.

(a) Schematic representation of the periodontal regeneration model. The stereoscope image indicates that the cell sheet–membrane construct was able to accurately adapt to the root dentin surface. The scale bar represents 2 mm. (b) Histological analysis of periodontal regeneration on the tooth root surface. Compared with the control group, PCL–SIM membranes facilitated regeneration of the cementum-like tissue. White dashed lines represent the borderlines of the newly formed mineralized layer on the dentin surface. At high magnification, small blood vessels (thin arrows) and embedded cementoblast-like cells (big arrows) could be seen in both groups. The scale bar represents 100 µm. (c) Semi-quantitative analysis showed that the area of newly formed mineralized layer was higher in PCL–SIM membranes compared with PCL membranes. (d) Masson’s trichrome staining showed the orientation of organized collagen fibrous tissue (white dashed arrows) in both groups.
Statistical analysis
The data of each group are shown as means ± standard deviations. Statistical analysis was performed by one-way analysis of variance using the software SPSS 20.0, and a P-value < 0.05 was considered statistically significant.
Results
SIM release profile from the electrospun membrane scaffold
Using the electrospinning procedure, membranes with SIM-loaded fibers were successfully fabricated. SIM-loaded scaffold samples with a diameter of 10 mm (5.86 ± 0.78 mg in mass) were prepared (Figure 1(b)). According to the loaded drug concentration, the theoretical weight of SIM loading in each sample was approximately 300 µg. From the SEM image with a magnification of 2000×, the surface of the SIM-loaded fibers was smooth without drug crystals (Figure 1(c)). With the aid of the standard curve of SIM at 238 nm (Figure 1(d)), the release pattern over 28 days was evaluated. As shown in Figure 1(e), a total of 39.1% of the loaded SIM was released from the membrane over the first 5 days; on day 28, drug release could still be detected and the final cumulative released amount was 82.2%. In drug delivery area, one of the advantages of using electrospun fibers is high drug encapsulation efficiency (up to 100%). 9 Considering that the half-life of SIM is rather short (3–4 h), it is conceivable that the majority of SIM was incorporated in the electrospun fibers during preparation.
Cell viability of PDLSCs on scaffolds
The primary periodontal ligament cells from extracted teeth were capable of forming clonogenic cell clusters when cultured at a low density, as reported in other studies19,20 (Figure 4(a)). Based on the colony-forming ability, PDLSCs were purified using the limiting dilution technique. The expanded PDLSCs were positive for vimentin, confirming their mesodermal origin (Figure 4(b)), and for STRO-1, confirming their early mesenchymal stem cell status (Figure 4(c)). To identify the osteogenic differentiation potential, PDLSCs were cultured in osteogenic conditions for 21 days and formed abundant ARS-positive mineralized nodules (Figure 4(d)). Both PCL and PCL–SIM scaffolds had no effects on the proliferation of PDLSCs (P > 0.05). PDLSCs were found to proliferate on the scaffolds, regardless of SIM loading in the scaffold (Figure 4(e)). After 5 days of culture, live-dead fluorescence assays showed that most cells were stained green as viable and only a few were stained red as dead. PDLSCs were capable of adhering to the scaffolds and exhibited a healthy spindle morphology (Figure 4(f)).

Isolation of periodontal ligament stem cells (PDLSCs) and cell viability on scaffolds. (a) Representative image of colony-forming assay stained with Crystal Violet after 10 days of culture. (b) PDLSCs were positive for vimentin by immunohistochemical staining. (c) PDLSCs expressed STRO-1, shown by immunofluorescence staining. (d) PDLSCs formed Alizarin Red S–stained mineralized nodules after 21 days of osteogenic induction. (e) Absorbance of CCK-8 assay showed no difference in any of the groups after 5 days of culture. All experiments were conducted in duplicate with three independent sets. Bars represent mean ± SD (n = 3). (f) Representative confocal images of the live/dead assay at day 5. Green fluorescence indicates viable cells and red fluorescence indicates dead cells on the scaffolds. In (b), (c), and (d), the scale bar represents 100 µm.
Osteogenic differentiation of PDLSCs on scaffolds
Osteogenic differentiation of seeded PDLSCs was upregulated by SIM released from the PCL–SIM scaffolds. After 21 days in culture, the intensity of mineralized extracellular matrix was assessed by ARS staining and quantitation. PDLSCs seeded onto either PCL or PCL–SIM scaffolds showed mineralized nodule formation, with the calcium-rich matrix being observed on the scaffolds (Figure 5(a)). In comparison with the control PCL scaffolds, SIM-releasing scaffolds showed significantly more accumulation of mineralized matrix (Figure 5(b)). The gene expression levels of osteogenic markers were analyzed. In comparison with the control, gene expression of Col-I, BSP, and ALP were significantly increased (Figure 5(d) to (f)). However, the gene expression of BMP-2 by cells on the PCL–SIM scaffold was significantly lower (Figure 5(c)).

Osteogenic differentiation of periodontal ligament stem cells (PDLSCs) on scaffolds. (a) Representative Alizarin Red S–stained images of mineralized nodule deposition at day 21. The scale bar represents 30 µm. (b) Alizarin stain intensity was evaluated by densitometry. PDLSCs cultured on PCL–SIM scaffolds showed significantly increased mineralized nodule deposition than cells on PCL scaffolds. (c) BMP-2, (d) Col-I, (e) BSP, and (f) ALP mRNA gene expression of PDLSCs on scaffolds.
Assembly of the cell sheet–scaffold construct
As the incorporation of ascorbic acid into the growth medium resulted in a mature PDLSC sheet after 10 days of culture, PDLSC sheets were harvested with an electrospun membrane scaffold as shown in Figure 2(a). PDLSCs secreted significant amounts of the extracellular matrix which resulted in a collagenous network, and the well-developed network of collagen fibers was confirmed by immunostaining of Col-I (Figure 2(d)). After successfully delivering three layers of PDLSC sheets, we obtained the PDLSC sheet–scaffold construct. Under SEM imaging, the topical PDLSC sheet remained intact and the cells were densely packed, which displayed abundant matrix deposition (Figure 2(b)). The cross-sectional view of the cell sheet–scaffold construct showed that different layers of PDLSC sheets appeared to be integrated and a multilayered cellular tissue was formed (Figure 2(c)). Corresponding with the cross-sectional SEM imaging, representative HE staining of the construct showed that multiple layers of cell sheets appeared to be integrated, and cohesion between the membrane and seeded cell sheets was stable (Figure 2(e)).
SIM-releasing scaffolds promoted cementum-like tissue formation in vivo
For the ectopic regeneration of periodontium, the PDLSC sheet–scaffold construct was placed between root dentin and a CBB block. As shown in the stereoscope images (Figure 3(a)), the PDLSC sheet–scaffold construct adapted accurately to the dentin surface. Eight weeks after implantation into immunodeficient mice, the scaffolds loaded with/without SIM exhibited good tissue integration. All samples developed ectopic cementum-like hard tissue layers on the tooth root dentin surface. Compared with the membrane without loading of SIM, the SIM-releasing membrane facilitated the deposition of mineralized layers on the dentin surface (Figure 3(b)). Quantitatively, the PCL–SIM scaffold significantly promoted formation of the cementum-like mineralized tissue layer at comparable levels to the control group (P < 0.01 ) (Figure 3(c)). Masson’s staining showed that an organized collagen fibrous tissue was closely attached to the cementum-like calcified tissue layer (Figure 3(d)). Thus, both PCL and PCL–SIM membranes appeared to not only adapt multilayer PDLSC sheets to the tooth root surface but also promote cementum-like tissue formation with fibrous directionalities to form a tooth-supportive structure. The SIM-releasing membrane significantly enhanced regeneration of the cementum-like mineralized tissue layer in comparison with the control.
Discussion
To develop sustained release fibers for periodontal regeneration, SIM-loaded PCL electrospun membrane scaffolds were fabricated in our study. In general, drug release from an electrospun polymer depends on many factors, including the drug solubility in the polymer solution, and the diffusion and biodegradability of the polymer matrix.21–23 Drug–polymer compatibility, the drug solubility in the drug/polymer/solvent system, can correlate with the ability to fully encapsulate drugs and accomplish sustained release. 9 The solvent evaporated quickly when the electrospinning solution was elongated rapidly, thus phase separation was avoided and the lipophilic drug was likely to remain inside the electrospun fibers.9,13 As a lipophilic drug, SIM is highly soluble in the PCL/chloroform solution. The SEM image revealed that the SIM-loaded fibers were smooth and there was no drug crystal on the surface. It was postulated that SIM was embedded well in the electrospun fibers. 22 The drug-loading content of PCL–SIM was about 3.8 µg/mm2, which was similar to a previous study. Although it was difficult to measure exactly the dose of a locally released drug to cells adhering directly to the scaffolds, rosuvastatin delivery at a concentration of 5 µg/mm2 was sufficient to reduce the inflammatory reaction. 14 The drug release from the PCL–SIM scaffolds continued for a sustained period of 28 days. The long-lasting SIM releasing promoted the osteogenic differentiation of MC3T3-E1 cells, as evidenced by increased expression of the osteogenic mRNA and protein, and more accumulation of mineralized nodule. 24 It should be noticed that, because of the alteration of pH value and protein enzymes in gingival crevicular fluid, drug release might be accelerated in vivo with severity of the periodontal disease. 25
In addition to consideration of the drug release system, the delivery of seeded cells with excellent activity should also be emphasized in periodontal regeneration. As demonstrated in our study, PDLSCs were identified by their postnatal stem cell characteristics such as colony-forming capacity, expression of mesenchymal stem cell markers, and capacity for multipotent differentiation.4,26 PDLSCs were capable of adhering to and proliferating on the PCL and PCL–SIM scaffolds, being stained green as viable and exhibiting typical spindle shape. This suggested the good biocompatibility of the membrane scaffolds. This good biocompatibility was attributed to the low SIM content of the scaffold, since SIM released from high drug dose (> 1 mg/mL) hydrogels displayed significant cytotoxic effect on MC3T3-E1 cells. 24 The cytotoxicity of high-dose SIM (> 0.1 μM ) has been reported in other studies.1,8 PCL is a clinical-grade product, which would facilitate future transplantation.15,27,28 When cultured on scaffolds in osteogenic medium for 21 days, PDLSCs formed abundant mineralized nodules. A significantly greater amount of mineralized matrix was observed in the presence of SIM release, with higher gene expression of ALP, Col-I, and BSP, such as observed in other investigations.24,29 It was found that SIM released from the hydrogels maintained its osteogenic effect, as evidenced by the increased MC3T3-E1 cell mineralization. 24 SIM-loaded scaffolds increased the osteogenic potential of MC3T3-E1 and dental pulp cells, in a pathway that involves expression of the osteogenic mRNA gene, such as Col-I, ALP, BSP, and osteopontin.8,29
PDLSC-based periodontal engineering has been extensively studied, and cell sheet strategy has been applied as a promising option to improve the regeneration of periodontal tissue.30,31 Continuous cell sheets, with the preservation of cellular junctions and endogenous extracellular matrix, could provide a mimicking cellular microenvironment and showed the increased expression of cementogenic genes.26,30 Cementum is the hard tissue layer on the dentin surface in which the periodontal ligament functions by anchoring the collagenous fibers. It is still difficult to enable the functional regeneration of periodontal tissue and integration with the tooth root surface. 32 The cell sheet engineering was developed by utilizing a thermo-responsive surface which enabled cell sheet harvesting without enzymatic detachment. 33 Nevertheless, it is possible to successfully harvest cell sheets with non-treatment culture plates by utilizing electrospun PCL membranes.16,28,34 This method is also used in the present study. The electrospun PCL–SIM membranes were shown to provide a suitable texture for harvesting cell sheets and accurately adapting to the root surface to prevent cell sheet dislocation. SEM and HE staining showed that PCL–SIM membranes were desirably flexible for stable cohesion of PDLSC sheets, which could reduce the risk of cell sheet displacement and enable the formation of cell sheet–membrane constructs.
The periodontium is a unique structure that consists of two mineralized tissues, cementum and alveolar bone, as well as the fibrous interface, periodontal ligament. 35 As a model of periodontal regeneration for in vivo transplantation, the cell sheet–scaffold constructs were assembled with root dentin and CBB, forming a periodontal microenvironment. Results of HE and Masson’s trichrome staining demonstrated that cementum-like hard tissue was successfully formed on the root dentin surface by the use of both PCL and PCL–SIM membranes. As an integrated part of the periodontal biomimetic architecture, the cementum-like tissue was indicated by an abundant deposited tissue layer on the dentin surface, embedded cementoblast-like cells, and organized connective collagen fibrous tissue.35,36 Collagen is the major constituent of the extracellular matrix in cementum, and the predominant collagen form is type I. 36 Higher magnification revealed that small blood vessels were present in both groups, indicating the good biocompatibility of PCL and PCL–SIM membranes. Based on some experimentally induced periodontitis trials, a meta-analysis revealed that no apparent effect was observed on cementum formation after local or systemic use of statin. 7 As far as the current research is concerned, the PCL–SIM membrane had critical significance to development of the cementum-like mineralized tissue on dentin surface. This might be attributed to two main factors: the sustained drug-releasing scaffolds and the cell sheet therapy. The PCL–SIM membranes presented some features of both an osteogenic factor carrier and a tissue regeneration scaffold. On the other hand, multilayered PDLSC sheets served roles as both the source of regenerative cells and the natural extracellular matrix.
Despite the present encouraging in vivo results, the limitations of scaffold design were noticed. Adjusting scaffold porosity to the requirements of tissue regeneration should be the main focus to allow cell penetration and hold the extracellular matrix. Precisely designed microchannels with pores of diameter between 300 μm and 800 μm had been suggested as the optimal size for cell adhesion and nutrient exchange. 37 The scaffold geometry played a pivotal role, since the highly oriented microchannels could drive the regeneration of ligament fibers between bone and dentin tissue.35,38 Optimization of the drug-releasing scaffold by anatomical design should be taken into consideration for further improvements.
Conclusions
Development of an appropriate scaffold being capable of both SIM release and acting as a cell sheet graft is the main target of the current study. We proposed a PCL–SIM membrane which would promote cementum-like tissue formation in vivo, by simultaneously functioning as a long-term drug release system and a suitable scaffold for cell sheet assembly. Compared to recombinant proteins or genetic approaches to induce periodontal regeneration, SIM-releasing membranes and cell sheet technology were safer, more effective, and inexpensive for future clinical cell-based periodontal regeneration. The biomimetic architecture of cementum-like tissue on the tooth root surface was regenerated with a heterotopic regeneration model in immunodeficient mice. Further orthotopic transplantation experiments will be performed in a surgically created periodontal defect model, which may lead to the application of such a cell sheet–membrane tissue-engineered construct for clinical periodontal regeneration.
Footnotes
Acknowledgements
We would like to thank Prof YH Liu and Dr WX Li for their assistance and guidance in this research.
Contributorship
QL and BZ researched literature and conceived the study. BZ wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81701011), the Shanghai Sailing Program (17YF1416500), and the Shanghai Nature Science Foundation (15ZR1435900).
Guarantor
Q Li.