Abstract
This study reported about the fabrication of dentin non-collagenous proteins (dNCPs) polyelectrolyte multilayers and evaluated its osteogenic potential. The composite sandwich structure of dNCPs polyelectrolyte multilayers was generated on the surface of polycaprolactone electrospinning membranes by the Layer-by-Layer self-assembly technique. The dNCPs-coated membranes comprised the experimental group and the non-coated membranes acted as the control. Nanofiber morphologies of both membranes were observed under scanning electron microscope. The release of dNCPs was evaluated by ELISA kit. Periodontal ligament stem cells (PDLSCs) were seeded on both membranes. The morphology changes and proliferation of cells were tested. The expressions of osteogenic-related genes and proteins were evaluated by RT-PCR, alkaline phosphatase (ALP) activity assay, and immunofluorescence staining. dNCPs-coated membranes displayed significantly different fiber morphology than the non-coated membranes. A stable release of dentin phosphoprotein was maintained from day 4 to day 15 in the experimental group. Cells on dNCPs-coated membranes were found to have cuboidal or polygonal shapes. The proliferative rate of cells was significantly lower in the experimental group from day 4 to day 9 (p<0.05). However, cells on the dNCPs-coated membranes demonstrated a significantly higher ALP content and expression levels of osteogenic gene and proteins than the controls (p<0.05). These results indicated that dNCPs polyelectrolyte multilayers could induce the osteogenic differentiation of PDLSCs in vitro.
Keywords
Introduction
Periodontal tissue destruction caused by inflammation usually leads to tooth loss. Periodontal ligament-derived stem cells (PDLSCs) have recently been widely studied in periodontal tissue regeneration because of their capacity for a high proliferation rate and multi-lineage differentiation.1 -3
To provide an optimal microenvironment for periodontal tissue regeneration, scaffold surfaces modified with biomolecules were developed by various methods.4 -6 Layer-by-Layer self-assembly technology (LbL technology) is an ideal strategy for surface modification of materials, and has been applied to assemble multilayers with bioactive factors for local delivery.7,8 Proteins incorporated into the multilayers by LbL technology were proved to be able to maintain their activities and promote stem cell differentiation.9 -11 Positively charged poly(allylamine hydrochloride) (PAH) and negatively charged poly(sodium 4-styrenesulfonate) (PSS) are commonly used to fabricate the self-assembly functional multilayers. BMP-2 loaded in PAH/PSS multilayer films was confirmed to induce early osteogenic differentiation of bone mesenchymal stem cells. 9 However, synthetic polymers exhibit poor biodegradation and biocompatibility. Collagen (COL) is the main constituent of cementum and is conducive to tissue regeneration. As it is the main protein of connective tissue in animals and the most abundant protein in mammals, COL can enhance cellular attachment because of its more natural surfaces than synthetic polymers. 12 Alginate (ALG) is able to optimize drug release in a controlled fashion and thus is considered as an ideal drug delivery vehicle.13 -15 Our previous study fabricated ALG/COL nano-films with high stability and cytocompatibility, which could promote cell adhesion to the scaffold surface. 16 The present study added natural polymers (COL and ALG) to the PAH/PSS films to address the limitations of poor biocompatibility.
Dentin non-collagenous proteins (dNCPs) comprise mixed proteins and growth factors extracted from dentin.17,18 These proteins are easily collected and considered to provide a suitable environment for osteogenesis and cementogenesis.19 -22 PDLSCs, treated with non-collagenous protein, were proved to present increased expression levels of osteogenic genes. 19
This work aimed to fabricate the composite sandwich structure of {[(dNCPs/PSS)/PAH]/[ALG/COL]}20 polyelectrolyte multilayers and evaluate its potential in the osteogenic differentiation of PDLSCs in vitro.
Materials and methods
Materials
Polycaprolactone (PCL, Mw: 1×105), poly-ethyleneimine (PEI), PAH and PSS were all obtained from Sigma-Aldrich (USA). Collagen (COL, 07CBPE2, Symatese, France) and ALG (Pronova LVG-10, NovaMatrix, USA) were prepared. dNCPs were kindly provided by Professor A.J. Smith from University of Birmingham, UK. Water for the salt solutions was prepared by a Milli-Q water system.
Fabrication of dNCPs-coated PCL membrane
PEI, PAH, and PSS polyelectrolyte solutions were prepared at a concentration of 1 mg/ml. The COL solution was positively charged and the ALG solution was negatively charged. The COL was dissolved in PAH solution at the concentration of 0.3 mg/ml and ALG was dissolved in PSS solution at 0.3 mg/ml. All these aqueous solutions contained 0.15M NaCl. The dNCPs solution was negatively charged and dispersed into the PSS solution at a final concentration of 0.3 mg/ml (pH=6.5).
PCL nanofiber membranes were prepared by the electrospinning methods. Briefly, a solution of PCL in trichlormethane solvent (8 wt%) was gently mixed. Then, the solution was delivered through a needle at a constant flow rate of 0.7 ml/h by a pump (LSP01-1A, Longer Pump Inc., Baoding, P.R. China). The voltage for nanofiber generation was 15 kV (ZS-60 kV/2 mA, Rixing Electric Inc. Shanghai, P.R. China). An aluminum foil plate located at a distance of 15 cm from the needle tip was applied for nanofiber collection. The spinning environment was set at 38°C and 18% humidity. The surface of the PCL membrane was negatively charged, and PEI was deposited on its surface to form a highly positive surface. The PEI-prepared PCL membrane was then placed in (dNCPs/PSS) solution for 10 min to generate the (dNCPs/PSS) layer. The activated membrane was afterward washed with deionized water to remove the polyelectrolyte that was not closely attached to the membrane. Next, the PCL membrane was immersed in PAH aqueous solution for 10 min and rinsed again by deionized water to produce a (dNCPs/PSS)/PAH layer. Then, the same membrane was immersed into ALG and COL solution for 10 min sequentially, followed by deionized water rinse. In total, (dNCPs/PSS), PAH, ALG and COL were alternately assembled onto the PCL membrane for 20 cycles (Figure 1(a)). Finally, the PEI-{[(dNCPs/PSS)/PAH]/[ALG/COL]}20 multilayers were incorporated onto the surface of the PCL membrane (dNCPs-coated membrane). Then, the dNCPs-coated membrane was immersed in 1-ethyl-3-(3-dimethylpropyl)-carbodiimide (EDC) and N-hydroxy-succinimide (NHS) solution for 30 min, and rinsed with PBS (containing 1% V/V antibiotic-antimycotic) (HyClone) and frozen at −20°C. The dNCPs-coated membranes were considered as the experimental group. The pure PCL membranes without coatings (the non-coated membranes) were the controls.

Characteristics of dNCPs-coated membranes.
Scanning electron microscopy
dNCPs-coated membranes and non-coated membranes were critical-point dried and coated with gold (JEOL JFC-1600 Auto Fine Coater, Tokyo, Japan). Surface morphologies of both were observed by scanning electron microscopy (SEM, LEO982, Germany).
Release of dentin phosphoprotein (DPP)
For in vitro release study, the dNCPs-coated membranes (S= 4 mm × 6 mm) were dipped into 1 ml PBS in EP tube at 4°C. A volume of 200 μl solution was collected every day and another 200 μl PBS was added into the original solution in the EP tube. The collection was performed every day for 21 days. The release amount of DPP, which was the major constituent of dNCPs, was detected by DPP ELISA Kit (Abcam, USA). The daily amount of sustained DPP release was converted by the standard curve.
Morphological observations
The isolation and culture of PDLSCs was performed by a technique as previously described. 23 PDLSCs (8000 cells per well) were seeded onto the dNCPs-coated membranes and non-coated membranes in 48-well plates. After incubation for the set times (4 d and 12 d), the samples were washed, fixed with 2.5% glutaraldehyde, and dehydrated. The cells were observed under SEM (LEO982, Germany) for morphological analysis.
Cell viability and metabolic assay
After 8-day culture, the cell viability of PDLSCs seeded on the coated and non-coated membrane was analyzed using a Calcein-AM/PI Viability kit (NO.C326, DOKINDO, Japan). Following the manufacturer’s instructions, cells were washed with PBS three times, and then incubated with working solutions for 30 min. Samples were viewed under an immunofluorescent confocal laser scanning microscopy. Viable cells stained green whereas dead cells appeared red. The morphology of cells in both groups was 3D reconstructed and plotted (NIS-Elements AR software). The thickness of the cell layers were measured by software (NIS-Elements AR).
The metabolic activity of PDLSCs on the dNCPs-coated and non-coated membranes was evaluated by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay (MTT assay). PDLSCs of the 5th passage were seeded on the membranes of two groups in 96-well plates at a density of 3×103 cells per well. The MTT assay (FMGBio, Shanghai, China) was performed after co-culture for 36 h, 4 d, 7 d, 9 d, and 11 d, according to the manufacturer’s instructions. Attenuance values for every well were measured by reading the absorbance at 490 nm with a spectrophotometer (MK3, Thermo, USA). Three independent experiments were carried out, and each contained triplicate samples.
Real-time polymerase chain reaction (RT-PCR)
After 7 days of co-culture, the osteogenic/odontogenic-related genes of the PDLSC in the coated and non-coated groups were examined. The acid guanidine method by a TRIzol_reagent (Invitrogen) was applied to extract the total cellular RNA from the culture. cDNA was then generated by PrimeScript RT Master Mix (TaKaRa, Dalian, China). The expression level of osteogenic/odontogenic genes, including Collagen-I (COL-I), Bonesialoprotein (BSP), Osteonectin (ON), Osteocalcin (OC), Dentin matrix protein (DMP-1) and Dentin Sialophosphoprotein (DSPP), was quantitated by qPCR with SYBR Premix Ex Taq (TaKaRa, Dalian, China). Glyceraldehyde-3-phosphate dehydrogenase (GADPH) was used as an endogenous control gene. The samples were subjected to 40 cycles of amplification with the specific primers (Table 1). All the samples were analyzed in triplicate, and the experiments were repeated three times. The analysis was performed by 7500 Software v2.0.4 (Lifetechnologies) with the comparative CT method (ΔΔCT).
The seqences of primers used for PCR amplification.
Alkaline phosphatase activity assay
PDLSCs were seeded on the membranes of two groups in the 24-well plates at a density of 1.2 × 104 cells per well. The alkaline phosphatase (ALP) activity in both groups was evaluated at 36 h, 4 d, 7 d, and 11 d. Cells were washed with ice-cold PBS twice and solubilized with lysis buffer (10 mM Tris/HCl, 2 mM MgCl2 and 0.05% Triton X-100, pH 8.2) and ice-sonicated for three pulses of 9 s. Subsequently, the solution was centrifuged for 10 min at 12,000 rpm at 4°C. A volume of 30 μl of the supernatants was generated from each well and ALP assay buffer (Beyotime Biotechnology, China) was added. After incubation in the dark for 2 min, the chromogenic fluid was added to each well. Then, the mixture was incubated at 37oC for 2 min and the absorbance was measured by spectrophotometry at a 520 nm wavelength.
Immunofluorescence staining
The expressions of osteogenesis-associated proteins including osteopontin (OPN) and osteocalcin (ON) were evaluated by immunofluorescence staining. Cementum attachment protein (CAP), a specific marker of cementoblast lineages,24,25 was also tested. After culturing on the dNCPs-coated or non-coated membranes for 8 days, PDLSCs were fixed with 4% polyoxymethylene for 20 min after washing. The specimens were incubated in PBS with 0.1% Triton X-100 for 20 min and then blocked with 5% BSA (Calbiochem, USA) at room temperature for 20 min. Primary antibodies, mouse anti-human OPN (Abcam, UK), mouse anti-human CAP (Santa Cruz, USA), and mouse anti-human ON (Abcam, UK), were applied and incubated in the dark at 37°C for 1 h. Subsequently, the samples were incubated with secondary antibody (rabbit anti-mouse IgG-FITC, Pierce, USA) in the dark at 37°C for 2 h. The cells were counter-stained with DAPI (NO.C1002, Beyotime Biotechnology, China) and observed under a confocal laser scanning microscope (Olympus FV100, Germany).
Statistical analysis
Statistical analyses were performed using the Student’s unpaired t-test with SPSS 17.0 and Excel software. The difference was considered to be statistically significant at p<0.05.
Results
Characteristics of dNCPs-coated PCL membrane
Representative images of dNCPs-coated membranes were observed under SEM (Figure 1(b)). dNCPs-coated membranes displayed significantly different fiber morphology compared with the coarser and threaded fiber of non-coated membranes.
DPP release kinetics in vitro
As shown in Figure 1(c), the in vitro release behavior of DPP from the dNCPs-coated membrane was characterized by ELISA kit. An initial burst release was found (from 355 ± 49.20 ng/ml to 772 ± 53.37 ng/ml per day) during the first 4 days. A stable release was then maintained from day 4 to day 15 (from 724 ± 53.19 ng/ml to 835 ± 76.23 ng/ml per day). Afterward, it the results suggest that the release of DPP gradually decreased from day 16 to day 21.
Morphology changes of PDLSCs cultured on dNCPs-coated membranes
The SEM images showed that PDLSCs cultured on dNCPs-coated membranes exhibited distinct morphological changes compared with those on non-coated membranes (Figure 2). After 4-day culture, cells on dNCPs-coated membrane adopted significantly cuboidal or polygonal shapes, whereas those on the non-coated membrane were fibroblastic in shape (Figure 2(a)). Compared with cells in the control group, PDLSCs seeded on the dNCPs-coated film demonstrated a coarse surface with several mineralization secretions or secretory vesicles after 8 day culture (Figure 2(b)). On day 12 culturing, cells in the non-coated group covered almost 90% of the membrane, whereas those in the dNCPs-coated group covered nearly 80%. The SEM observation showed that cells seeded on the dNCPs-coated membranes appeared flatter and more cuboidal than cells on the non-coated membranes (Figure 2(c)).

Morphology of PDLSCs on the dNCPs-coated and non-coated membranes under SEM.
Effect of dNCPs multilayers on the viability of PDLSCs
The staining results suggested that the majority of cells seeded on the surfaces of both membranes were viable after 8-day incubation (Figure 3(a)). No significant cell death appeared within the two groups. However, PDLSCs on the dNCPs-coated membranes appeared cuboidal or polygonal in shape, whereas cells on the non-coated membranes were fibroblast-like and of distinctly elongated, spindle shape. It was found that the average thickness of cell layer in the non-coated group was 50.25 ± 6.31 μm, as compared with the cell layer in the dNCPs-coated group of 26.47 ± 4.55 μm (*p<0.05) (Figure 3(b)). Overall, these results suggest that the cell layer of the non-coated group was significantly thicker than that of the dNCPs-coated group (p<0.05).

Viability of PDLSCs on dNCPs-coated and non-coated membranes after 8-day culture.
Effect of dNCPs multilayers on the proliferation of PDLSCs/MTT assay
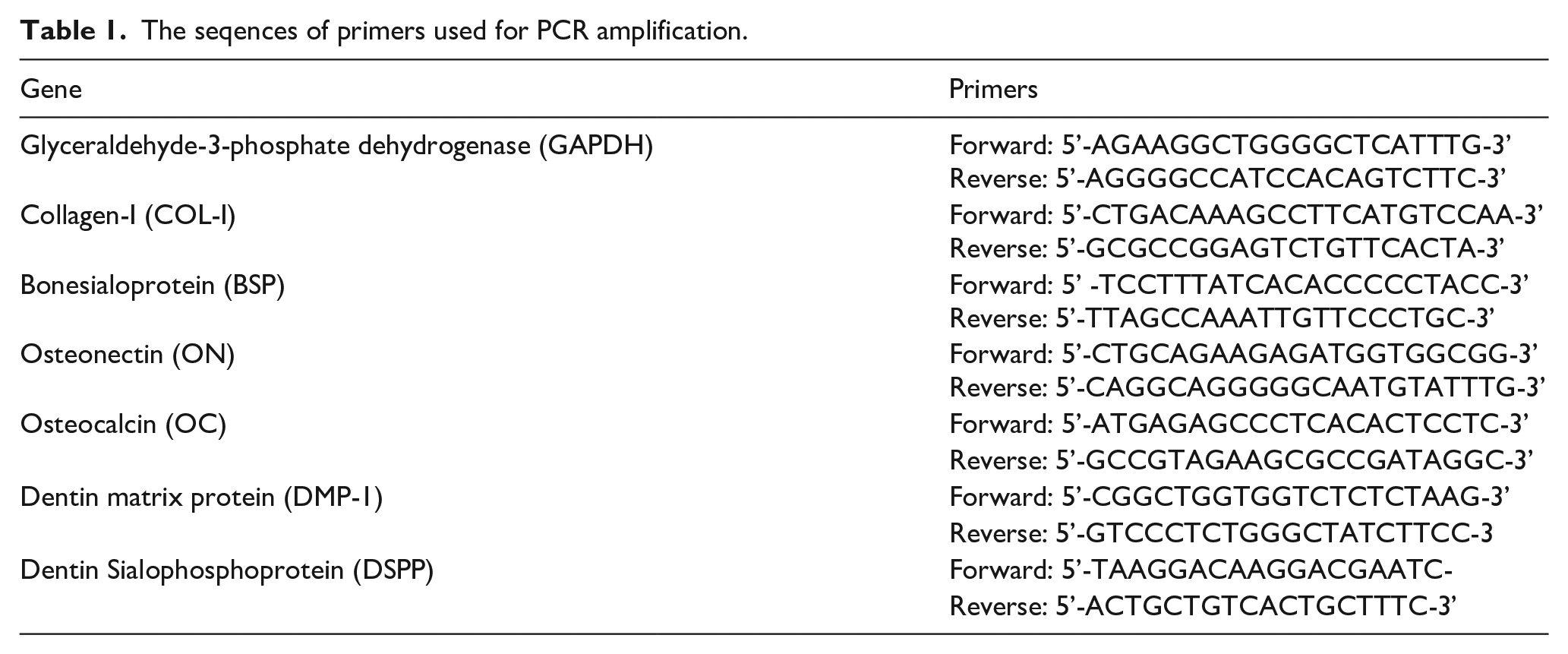
MTT assays showed no distinct difference in the initial proliferation amount of PDLSCs between the experimental group and the controls at the beginning of the culture. Afterward, PDLSCs on the dNCPs-coated membranes exhibited a statistically significant lower proliferative rate from day 4 to day 9 as compared with the cells on the non-coated membranes (p<0.05) (Figure 4(a)).
The effect of the dNCPs multilayers on the proliferation, mineralization, and osteogenic/cementogenic differentiation of PDLSCs.
The mRNA expression of osteogenic/odontogenic gene
The expression of osteogenic gene (COL-I, BSP, ON and OC) was significantly higher in dNCPs-coated group, in comparison with the controls after 7 days of culture (p<0.05) (Figure 5). The results suggested no obvious expression transcript levels of DMP1 and DSPP genes in the dNCPs-coated group compared with the non-coated group on day 7 (Figure 5).
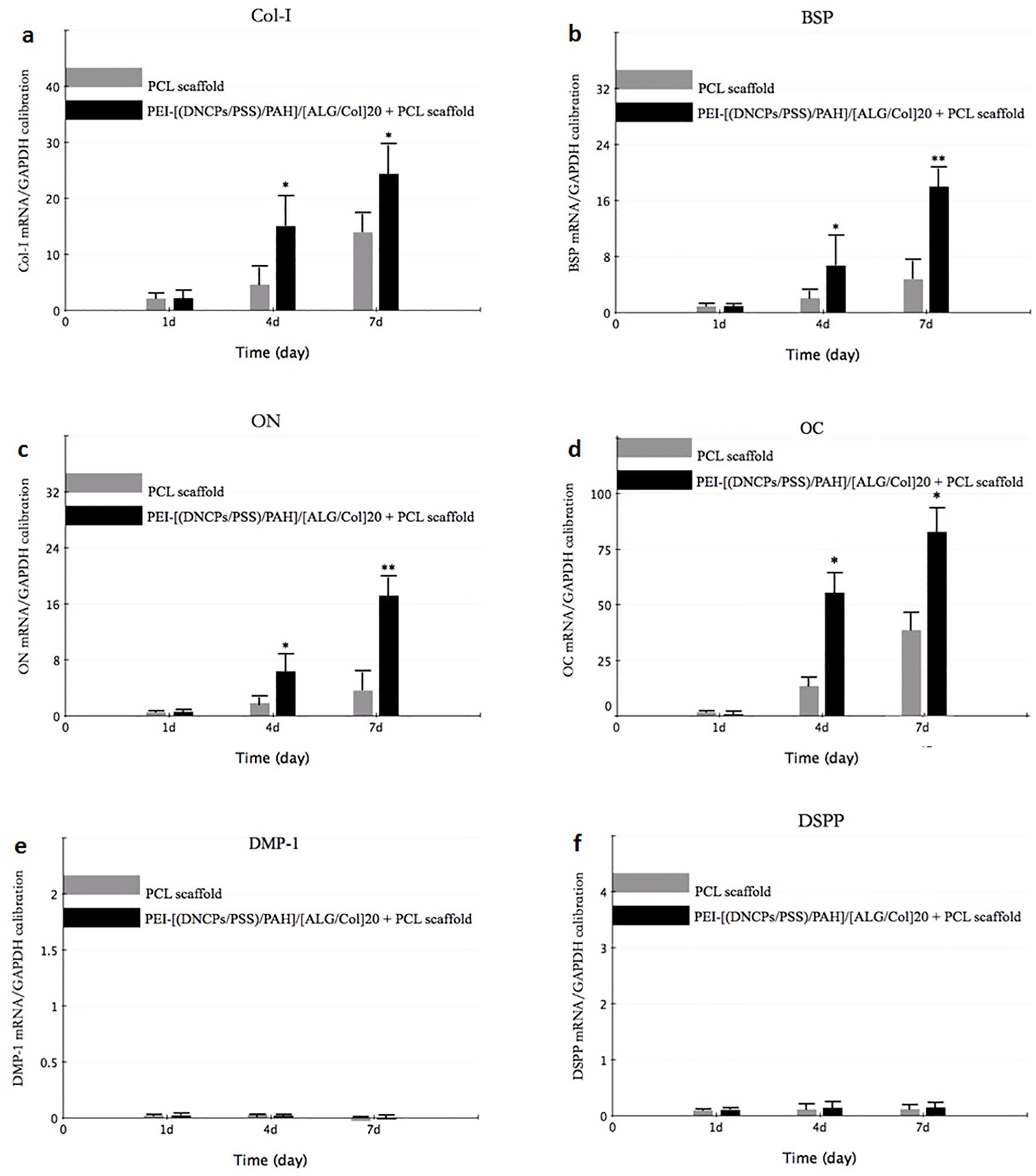
qRT-PCR measurements for osteogenic/odontogenic marker genes.
Effect of dNCPs multilayers on ALP activity of PDLSCs/ALP Assay
The ALP activity of PDLSCs in the two groups was detected on days 1.5, 4, 7, and 11 in this study (Figure 4(b)). In general, PDLSCs on the dNCPs-coated membranes demonstrated a significantly higher ALP content than the controls at the defined time points during the culture periods (p<0.05). The ALP activity of cells on the dNCPs-coated membranes increased more rapidly compared with that of the control group from the initial time to day 11 culture.
Immunofluorescence staining
As indicated in Figure 4(c), the representative image shows the PDLSCs co-cultured with dNCPs-coated membranes and non-coated membranes with OPN (green), CAP (green), and ON (green) immunofluorescence staining. The results showed that OPN and ON were strongly expressed in PDLSCs of the experimental group with respect to those in the control group. It was observed that CAP appeared obviously more highly expressed in cells treated with dNCPs-coated membranes as compared with the controls.
Discussion
In the paired system, synthetic polymers films are commonly applied in the fabrication of multilayers due to their property of strong charge.9,10 In our study, a PSS layer was applied to carry dNCPs due to the same charge, and paired with PAH as stable functional multilayers. The ALG/COL films were proved to enhance the cellular attachment and modify cell morphology on the surface of scaffolds. 16 In the present study, a ALG/COL multilayer was added into the [(dNCPs/PSS)/PAH] multilayer to improve the biodegradability and biocompatibility of coating films. The addition of ALG/COL-paired multilayers into another multilayered structure has not been reported in previous studies. It was shown that dNCPs-coated membranes exhibited significantly different fiber morphology compared with the coarser and threaded fiber of non-coated membranes. The modification of the nanofiber morphology might be due to the adsorption of COL onto the nanofibers.
Moreover, most multilayer coatings possess a dense structure, which is well suited for molecular separation. 26 Polyelectrolyte multilayers involving ALG have been used as barrier films to slow the release of encapsulated molecules.27,28 To prolong the release of dNCPs, the ALG/COL multilayer was used as the barrier layer. Thus, (dNCPs/PSS)/PAH and ALG/COL multilayers were deposited alternately to assemble composite sandwich structures (Figure 1(a)). This construction increased the thickness of the coating films and slowed down the release of dNCPs. The results indicated that the coating membranes displayed a relatively stable release concentration of dNCPs from day 4 to day 15. Obviously, this configuration involving functional multilayers and barrier multilayers deposited alternately could represent a promising carrier model to slow the release of biomolecules.
The present study demonstrated that dNCPs released from the coating films had the ability to promote the osteogenic differentiation of PDLSCs in vitro. Compared with the cells in the controls, cells seeded on the dNCPs-coated membranes adopted coarse surfaces with several secretory vesicles at the early stage after being seeded. The in vitro release study showed that a small amount of dNCPs was released from the polyelectrolyte multilayers in the initial 4 days. It was possible that the release of dNCPs had some positive effect on the active function of synthesis and secretion of cells in the experimental group. PDLSCs treated with dNCPs-coated membranes showed slight changes in their morphology after culture for 12 days. Cells in the experimental group were observed as having a flat, cuboidal, or polygonal appearance. However, cells on the non-coated membranes tended to maintain a fibroblast-like spindle shape, even when confluent. Obviously, there were significant differences in the morphology between these two groups. The results were consistent with the observations under immunofluorescent confocal laser scanning microscopy. In addition, it was confirmed that the thickness of cell layers in the non-coated group was significantly thicker than that of the dNCPs-coated group. The results further verified the differentiation of PDLSCs.
The expression of osteogenic genes, including COL-I, BSP, ON, and OCN, was obviously higher in the experimental group compared with that of the controls. In addition, the higher content of ALP in the experimental group demonstrated that the mineralization capacity of PDLSCs was significantly enhanced by the dNCPs multilayers in comparison with the controls. It was demonstrated that PDLSCs induced by the dNCPs multilayers had a mineral-forming capacity at the protein level. The results of immunofluorescence staining also showed that cells in the experimental group exhibited significantly higher expression of mineralization-associated proteins (OPN and ON) than that in the control group. Together, these data further suggested that PDLSCs cultured on the dNCPs-coated membranes indeed obtained the mineralization potential.
The MTT results reflected that dNCPs multilayers played a suppressive role on the proliferation of PDLSCs. However, both RT-PCR and ALP test results showed that dNCPs promoted the mineralization and induced osteogenic differentiation of cells. It suggested that dNCPs slowed down the proliferation of PDLSCs, resulting in osteogenesis. Proliferation and differentiation are considered inversely correlated with each other due to the dual-function regulators which control both processes. 29 These results were consistent with those of the previous studies which concluded that some components of dNCPs could promote cell differentiation as well as inhibit cell proliferation.20,21
Although it was demonstrated the dNCPs multilayers provided a suitable microenvironment for PDLSCs osteogenic differentiation, whether the restructure has the same effect in vivo remains unknown. An in situ model demonstrating bone regeneration in animals must be included in future research. The present study demonstrated that the composite sandwich multilayers provided a promising biomolecule carrier model. Regarding the periodontal complex involving three constitutions of cementum, periodontal ligament, and alveolar bone, more proteins and cytokine such as FGF-2 for periodontal ligament 30 are needed. The aims of further studies are to combine double or triple cytokines into the polyelectrolyte multilayers and deliver them spatiotemporally for regeneration of the whole periodontal complex.
Conclusions
We have designed and fabricated the composite sandwich structure of {[(dNCPs/PSS)/PAH]/[ALG/COL]}20 multilayers. The multilayers slowed the release of dNCPs and had the potential to induce osteogenic differentiation of PDLSCs in vitro. The barrier membrane is used for periodontal regeneration in the clinic work. The present study may provide a promising surface coating to modify the barrier membrane for the delivery of biomolecules in a localized and stabilized release way.
Footnotes
Acknowledgements
The authors acknowledge Professor A.J. Smith from University of Birmingham, UK for his support providing dNCPs proteins.
Contributorship
YueHua Liu and Jing Chen researched literature and conceived the study. Wenxing Li and Qiang Li were involved in protocol development, gaining ethical approval and data analysis. Jing Chen wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [81470768], Shanghai Sailing Program [17YF1416500] and the National Natural Science Foundation of China [81701011].
Guarantor
Yuehua Liu
