Abstract
Background:
The main microorganism associated with the failure of endodontic treatments is Enterococcus faecalis. Although several endodontic therapeutics have demonstrated antimicrobial activity against E. faecalis, the antimicrobial effectiveness of chitosan (CsNPs) and silver nanoparticles (AgNPs) included into conventional endodontic sealers for endodontic therapies is still unclear.
Aim:
The objective of this study was to evaluate the antibacterial activity increment (AAI) of endodontic sealers containing CsNPs and AgNPs as well as some chemical components against E. faecalis by direct contact assays.
Methods:
CsNPs and AgNPs were synthesized by reduction and ionic gelation methods, respectively. Nanoparticles were characterized by dynamic light scattering and energy dispersive X-ray analysis. The bactericidal activity was tested on monolayers on agar plates and collagen membrane surface assays against E. faecalis.
Results:
The size of CsNPs was 70.6±14.8 nm and zeta potential was 52.0±5.4 mV; the size of AgNPs was 54.2±8.5 nm, and zeta potential was –48.4±6.9 mV. All materials, single or combined, showed an AAI, especially when CsNPs, chlorhexidine (Chx), and the combination of CsNPs-Chx were added. However, the combination of CsNPs-Chx showed the highest (55%) AAI, followed by Chx (35.5%) and CsNPs (11.1%), respectively. There was a significant statistical difference in all comparisons (p < 0.05). Tubliseal (40%) and AH Plus (32%) sealants showed a higher AAI on E. faecalis in the monolayer test and collagen membrane assay analyzed by scanning electron microscopy.
Conclusions:
Tubliseal and AH plus sealers combined with nanoparticles, especially CsNPs-Chx, could be used for conventional endodontic treatments in the control of E. faecalis bacteria.
Introduction
In endodontics, the complete cleaning, shaping, and the hermetic sealing of the root canal system are crucial. Therefore, the main objective of endodontic therapy is the elimination of microorganisms from the root canal system and the prevention of subsequent reinfection. However, it is impossible to eliminate all bacteria from the root canal system during treatment; so it is essential that endodontic materials have specific components that release substances for antibacterial activity.1,2 An endodontic sealer is a material used to fill up the gap between the root canal system of the teeth and the obturating materials used in root canal infections. Besides, it is important to guaranty the absence of microorganisms in primary infections, but unfortunately, radiographic evaluation fails to detect the presence of microorganisms either before or after the endodontic treatment. 3 Besides, refractory infections due to persistent apical periodontitis after an endodontic treatment may be due to poor sealing related to the presence of accessory canals and microorganisms in the root canal system.
On the other hand, in the last few years, Enterococci have received increasing attention because of the development of multiple antimicrobial drugs resistance after antibiotic therapies that include β-lactams penicillin, amoxicillin, and clindamycin. 4 One mechanism added to its natural resistance, Enterococci may develop plasmid-and-transposome-mediated resistance to several antibiotics.5–7 Also, since endodontic infections have a biofilm structure that includes several bacterial species, it is possible that bacteria develop antibiotic resistance after therapy during endodontic infections episodes. 8 Another mechanism of Enterococcus faecalis implicated in endodontic reinfections are the virulence factors that permit the adherence to host cells and extracellular matrix that facilitate tissue invasion, 9 and the capability to aggregate with other bacteria and produce biofilms. Furthermore, it has been reported that a 37 kDa plasmid-encoded adhesion protein is located in the surface; 10 this adhesion mediates the cell–cell contact, which facilitates the plasmid exchange between recipient and donor bacterial strains. In this way, the genetic material that produces antibiotic resistance can be transferred between E. faecalis and other bacteria species. 11
Besides, it has been reported that a biofilm infection is rarely eliminated entirely, even in individuals with a competent innate and adaptive immune response. 12 Therefore, it is important to explore new alternatives and substances to eradicate E. faecalis infections during the different steps that are included in root canal treatment. However, the complexity of the anatomy of the root canal system provides the niche for bacteria remaining where endodontic sealers cannot reach such as canal irregularities or seal minor spaces between the dentinal wall and the core filling material. 13 One possible solution for this is the improvement of endodontic sealers by adding nanoparticles or any other chemical components, which could help to neutralize microorganisms remaining in the root canal system, improving prognosis and endodontic success. Chitosan (CsNPs) and silver nanoparticles (AgNPs) are two of the most important nanomaterials proposed for applications in the biomedical field due to excellent physicochemical and biological properties.8,14,15 Chitosan is a non-toxic, inexpensive, and highly biocompatible biopolymer that can be easily biodegraded through different hydrophilic enzymes, promoting positive biological effects such as bactericidal, anti-inflammatory, antioxidant, antitumor, and healing properties.16–19 Nanomaterials based on chitosan have been widely used in the regeneration of different types of tissues, especially skin and bones, and have been used in many other biomedical and pharmaceutical applications.20–25 On the other hand, AgNPs are the most common metallic nanomaterials used for the control of several types of microorganisms due to their very well-known antimicrobial properties even in drug-resistant microorganisms,15,18,26,27 including the E. faecalis strain.18,28–32
Although several studies have determined the antimicrobial activity of chitosan and silver compounds as nano-scaled materials against E. faecalis, mainly used in combination with irrigant agents or endodontic sealers during endodontic therapies. There are not available works that have evaluated, through standardized methods based on microbiological assays by using direct contact procedures, the synergistic bactericidal activity of CsNPs and AgNPs. The aim was to assess, in vitro, the antimicrobial effectiveness of conventional endodontic sealers containing CsNPs or AgNPs, compared with chlorhexidine (Chx), calcium hydroxide with propylene glycol, and CsNPs+Chx against E. faecalis on direct contact assays.
Materials and methods
Endodontic sealers
The following sealers were evaluated in this in vitro study: AH Plus (Dentsply, Konstanz, Germany); Endosequence (Brasseler, USA); MTA Fillapex (Angelus, Londrina, PR, Brazil); Sealapex and Tubliseal (Kerr, Romulus, MI, USA). All sealers were prepared in strict compliance with the manufacturer’s recommendations (50 mg of base and 50 mg of catalyst were used for each sealer). Each sealer was evaluated alone and mixed with different substances: AgNPs and CsNPs; calcium hydroxide with propylene glycol (Ca(OH)2 + propygly); Chx ( at 0.3%); and CsNPs containing Chx (CsNPs-Chx). All experiments were carried out in triplicate.
Synthesis of AgNPs and CsNPs
The AgNPs were prepared with 100 mL of a 0.01 M AgNO3 (silver nitrate, 209139-25G, Aldrich, St. Louis, MI, USA) solution placed in a 250 mL glass reaction vessel. Under magnetic stirring, 10 mL of deionized water containing gallic acid (0.1 g) was added to the Ag+ solution. Then, the pH was adjusted to 11 with NaOH 1.0. M. Finally, AgNPs were dialyzed for 24 h in a 6 cm dialysis membrane tubing (MD44×100CLR RC, diameter 22 mm, MEMBRA-CEL®, USA).
The CsNPs were synthesized by an ionic gelation method. Merck chitosan solution (Chitosan low molecular weight, 448869-50G, Aldrich, St. Louis, MI, USA) was prepared at 0.3% w/v in 25 mL of 1% acetic acid under constant magnetic agitation until completely dissolved. Then, Chx was added at 3% v/v and allowed to shake for 10 minutes. Subsequently, a solution of sodium tripolyphosphate was added to 0.1% to the chitosan solution with Chx and mixed for another 10 min.
Dynamic light scattering (DLS), energy dispersive X-ray analysis (EDX), and transmission electron microscopy (TEM)
Molecular size and zeta potential values of CsNPs and AgNPs were analyzed by DLS (SAXSpace, Anton Paar, Ashland, VA, USA) and EDX (EDAX, Mahwah, NJ, USA) to identify the elemental composition of each nanoparticle sample. For EDX, samples were deposited on a silicon substrate by drop-coating. Then, the solution was left to dry and characterized by the EDAX system installed in a FEI-Inspect F50 (FEI Company, Hillsboro, OR, USA) field-emission scanning electron microscope. Also, TEM analysis (Phillips CM-200) at an accelerating voltage of 25 kV was used to determine the shape of particles.
Bacterial growth conditions and bacterial monolayer assay
The antibacterial activities of five different endodontic sealers were evaluated against a reference strain of E. faecalis (ATCC 19433), which was previously confirmed by polymerase chain reaction assay. The bacteria strain was cultivated in both agar and broth media; all samples were adjusted to 0.5 on the McFarland scale. Bacteria were grown aerobically to late logarithmic or early stationary phase from frozen stock cultures in brain heart infusion broth (BHI, BD, Benton Dickinson, USA) and incubated at 37°C for 24 h. E. faecalis was inoculated into tubes containing 5 mL of BHI and incubated for 24 h at 37°C. Then, a bacterial suspension containing 1.5 × 108 CFU/mL (200 µL was prepared in 3 mL of phosphate buffer solution (PBS)) was spread on agar plates to make a monolayer of E. faecalis. Freshly mixed specimens from each tested material were prepared into uniform size (approximately 20 µL of each material to achieve a 4 mm diameter), then activated sealers containing 20 µL of CsNPs or AgNPs were placed onto agar plates and incubated at 37°C for 48 h under aerobic conditions. The diameter of the inhibition zone formed on the bacteria monolayer was measured in millimeters according to a previous report. 32
Collagen membrane assay
Sterile fragments of 4 × 4 mm2 of collagen membranes (OsseoGuard-Biomet 3i LLC, Oakland, NJ, USA) were placed on 12-well plates and were inoculated with 200 µL of E. faecalis (containing 3.5 × 107 CFU/mL) on sterile PBS for 24 h at 37°C. Later, each sample was washed four times with PBS, and 100 µL of the different test solutions were added in triplicate (propylene glycol with calcium hydroxide, Chx 0.3%, and CsNPs-Chx), as well as positive (20 µL of 64 µg of amoxicillin/clavulanic acid solution) and negative (distilled water) controls for 2 h. Finally, all samples were fixed in 2% glutaraldehyde solution for 4 h at 4°C and washed with distilled water, and dehydrated in gradual series of ethanol (35, 50, 70, 90, and 100% for 15 min) to do scanning electron microscopy (SEM) analysis. 33
Statistical analysis
All data are expressed as mean, SD, and range. Qualitative data are expressed as frequency and proportion. Non-parametric tests analyzed the difference between groups, Kruskal–Wallis and Dunn’s multiple comparison tests were used. JMP program version 9.0 (SAS Institute, Cary, NC, USA) and Stata Intercooled version 11.0 (Stata Corp LP, College Station, TX, USA) were used for statistical analysis. Statistical analysis significance was set at p < 0.05.
Results
DLS, EDX, and TEM of CsNPs and AgNPs
The physical and chemical characteristics of CsNPs and AgNPs are shown in Figure 1. Uniform sizes, spherical shapes, and good particle distributions were consistently observed for both types of the nanoparticles used (Figure 1). The zeta potential results indicate that CsNPs had positive values (52.0±5.4 mV), while AgNPs showed consistently negative electrical charges (−48.4±6.9 mV). According to the DLS results, single, centered and thin peaks were found in CsNPs (70.6±14.8 nm) and AgNPs (54.2±8.5 nm) samples, respectively (Figure 1). EDX analysis of CsNPs and AgNPs, respectively, show a conventional distribution of oxygen, carbon, phosphorus, sodium, and silica for CsNPs and silver, oxygen, sodium, and silica for the AgNPs. The prominent peaks of silica and sodium could be highly expressed by the presence of the silicon merged with the substrate used (data not shown).
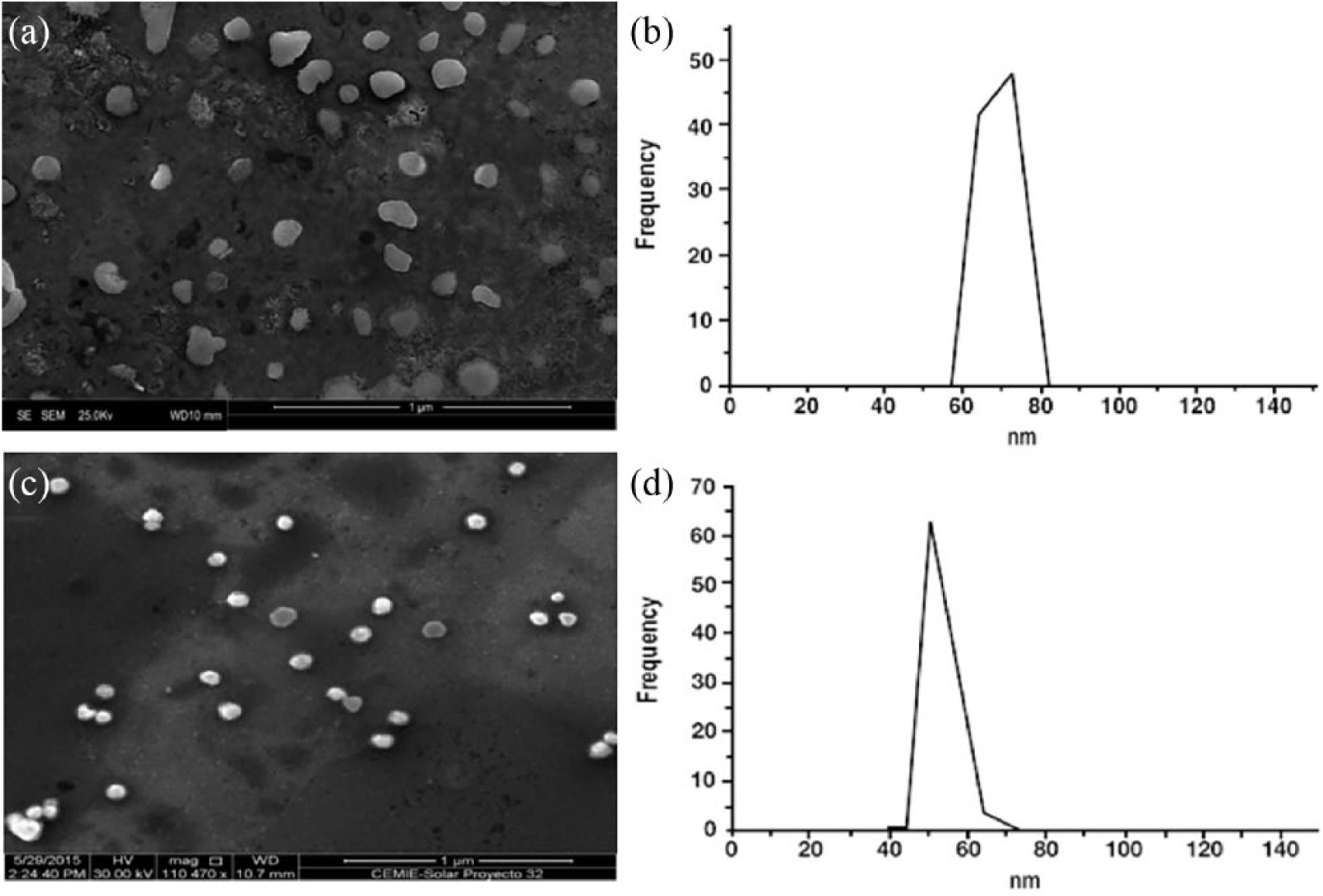
SEM micrographs and size determination of chitosan (CsNPs) and silver (AgNPs) nanoparticles.
Antimicrobial evaluation of monolayer assay
Table 1 shows the antimicrobial activity of endodontic sealers expressed as the range of zones of inhibition in millimeters. The inhibition zone by single sealers was as follows: Sealapex cement (8.9 mm), followed by MTA Fillapex (8.7 mm) and Endosequence (8.6 mm) cements. All cements showed an increased inhibition activity when mixed with different substances, but especially the bactericidal activity increased when the cement included CsNPs-Chx (13.1 mm). The bactericidal activity was followed by Chx (11.3 mm), Ca(OH)2+propylene glycol (10.7 mm), AgNPs (9.2 mm), and CsNPs (9.0 mm), respectively.
Antibacterial activity of CsNPs and AgNPs included in different sealers against Enterococcus faecalis.
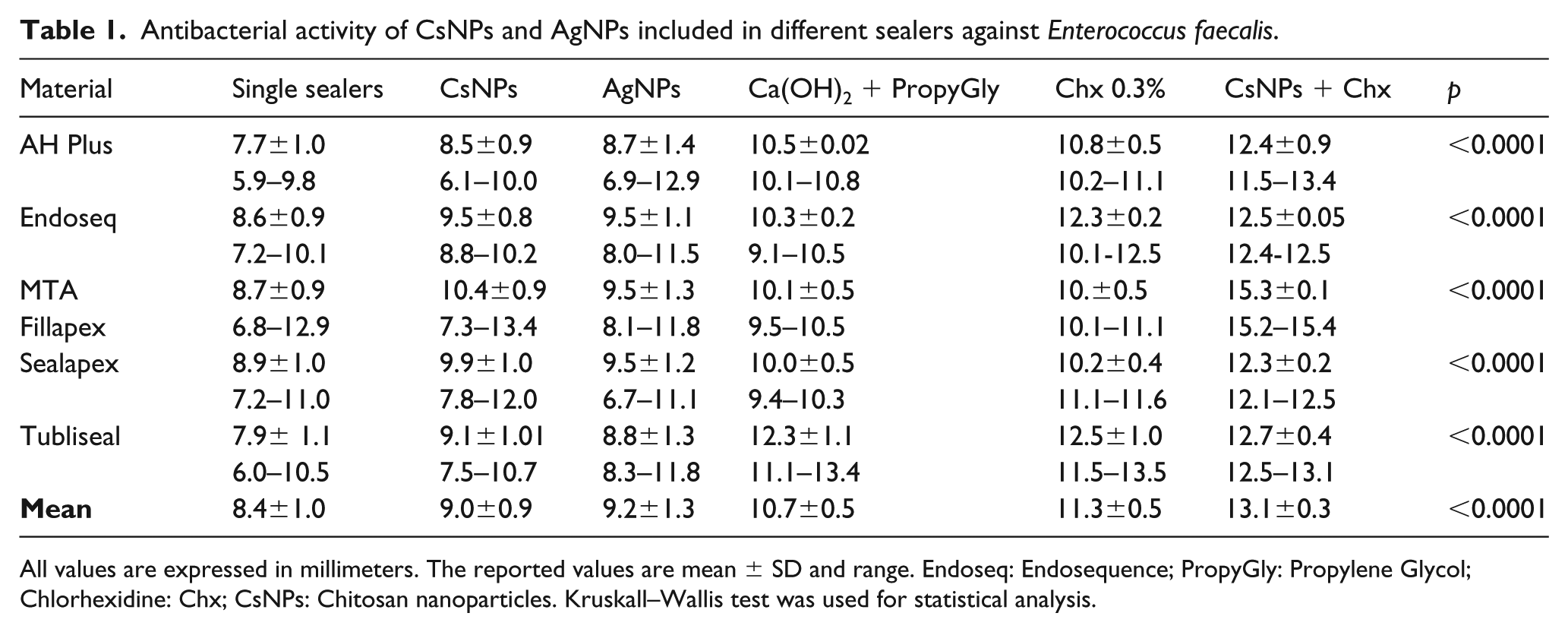
All values are expressed in millimeters. The reported values are mean ± SD and range. Endoseq: Endosequence; PropyGly: Propylene Glycol; Chlorhexidine: Chx; CsNPs: Chitosan nanoparticles. Kruskall–Wallis test was used for statistical analysis.
All sealers containing CsNPs+Chx showed the highest values of antibacterial activity increment (AAI) compared to all the different substances. AAI was 55.9%, followed by Chx 0.3% (34.9%), Ca(OH)2+propylene glycol (27.3%), CsNPs (11.1%), and AgNPs (9.9%). Tubliseal cement showed higher AAI (40%) in all combinations, followed by AH Plus (32.7%), Endosequence (26.6%), MTA Fillapex and Sealapex both with 21.6% of AAI (Table 2).
Bacterial inhibition areas of the endodontic sealers with CsNPs and AgNPs compared with sealers containing different substances.
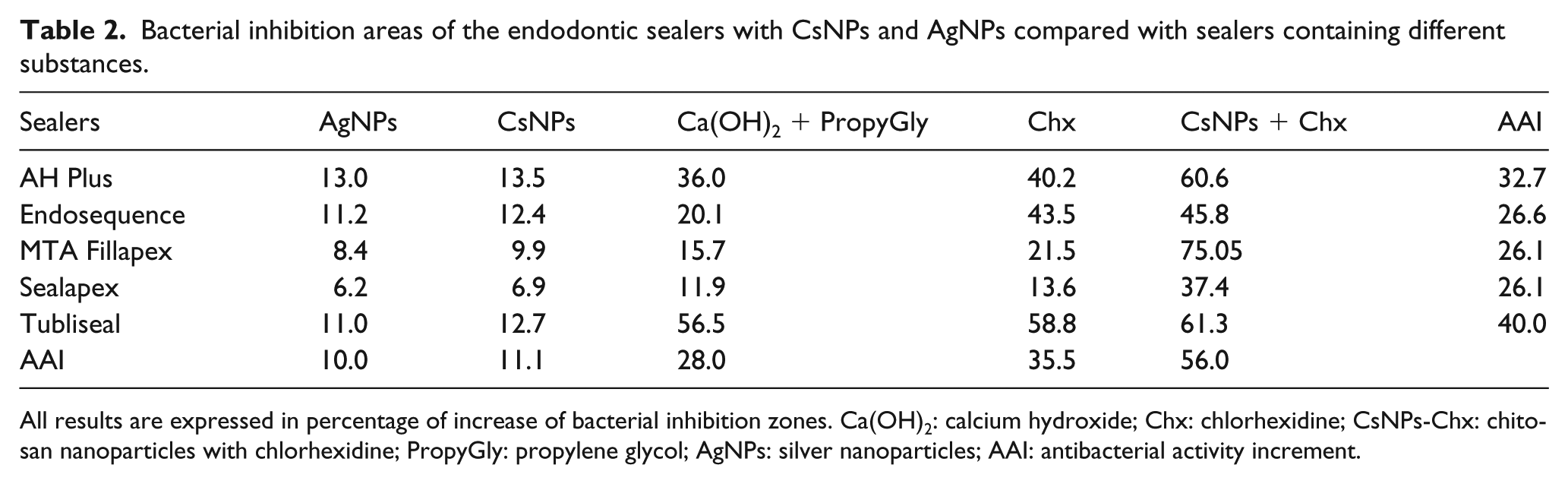
All results are expressed in percentage of increase of bacterial inhibition zones. Ca(OH)2: calcium hydroxide; Chx: chlorhexidine; CsNPs-Chx: chitosan nanoparticles with chlorhexidine; PropyGly: propylene glycol; AgNPs: silver nanoparticles; AAI: antibacterial activity increment.
The addition of the different tested solutions to the sealers increased the antibacterial activity, but CsNPs containing Chx presented the highest value of all sealers. In addition, all sealers used in this study have a statistically difference in antimicrobial activities (Table 3).
Comparison of the antimicrobial activity between the different sealers on monolayer test.
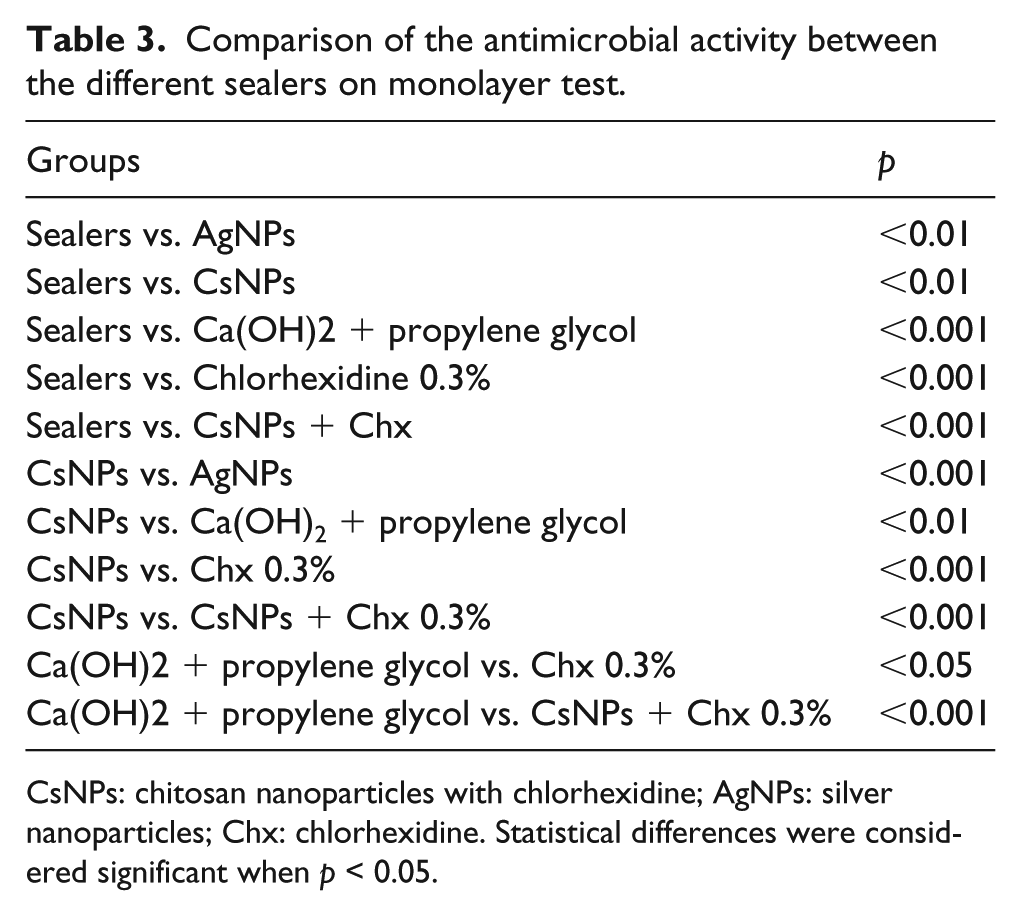
CsNPs: chitosan nanoparticles with chlorhexidine; AgNPs: silver nanoparticles; Chx: chlorhexidine. Statistical differences were considered significant when p < 0.05.
Antimicrobial evaluation of the collagen membrane assay
SEM was used to analyze the antibacterial effect on E. faecalis of a single substance on the collagen membrane model; representative images are shown in Figure 2. There was a gradient of the antibacterial activity of single material, sealers holding propylene glycol combined with calcium hydroxide, cement with Chx, and sealers containing CsNPs-Chx. Figure 2(a) (control positive without any solution) shows a high number of E. faecalis, while the sealers holding calcium hydroxide produced aggregates clusters creating fragmented inhibition areas (Figure 2(b)). Figure 2(c) (0.3% of Chx) shows a high bactericidal effect. However, the best inhibition areas were shown in the sealer that contains CsNPs-Chx (Figure 2(d)).
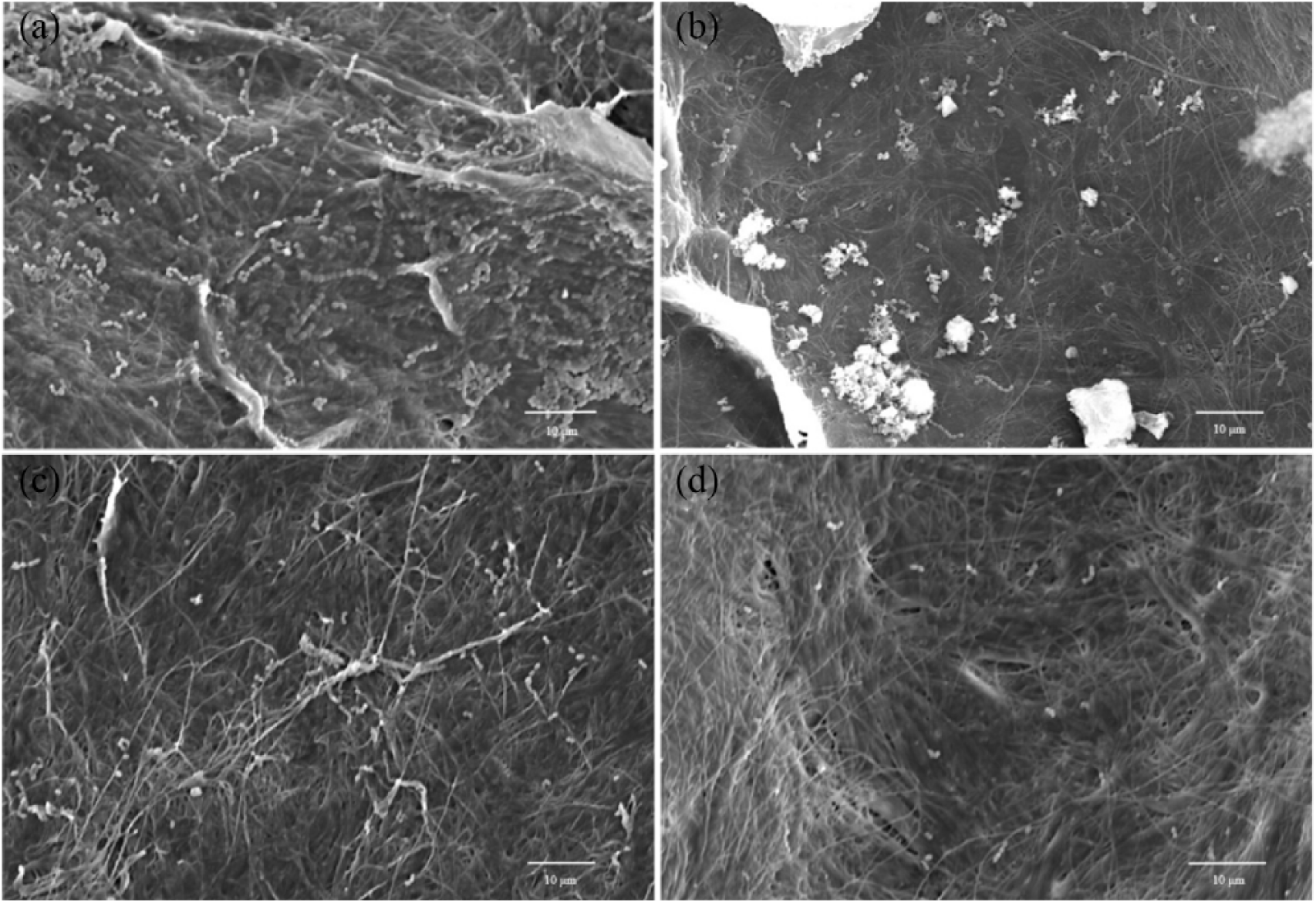
SEM micrographs of collagen membranes infected with E. faecalis and exposed to different sealers. (a) Control (no agent); (b) propylene glycol with calcium hydroxide; (c) 0.3% of chlorhexidine; (d) CsNPs containing 0.3% of chlorhexidine.
Discussion
This study determined that CsNPs and AgNPs included into endodontic sealers improve their antimicrobial properties against E. faecalis on monolayer assay. Also, the inclusion of conventional bactericidal endodontic irrigators, such as Chx and propylene glycol/calcium hydroxide (intracanal medication), increased the antimicrobial properties of sealers/NPs; however, those samples with CsNPs/Chx were demonstrated to have the best bactericidal properties (p < 0.001). Results from SEM micrographs confirm that the CsNPs samples combined with 0.3 % of Chx evidently promote a low number of microorganisms loaded on the surfaces of collagen membranes compared to control, propylene glycol/calcium hydroxide, and Chx groups, respectively. Besides, the bactericidal activity, for both CsNPs and AgNPs, was tested into broth cultures media (BHI and Tryptic Soy broths), but results were unreliable as the tested sealers reacted with the different components of the broth cultures; some sealers agglutinate or dissolve at different rates of time.
AgNPs and CsNPs are attracting increased attention in the biomedical field due to their bactericidal activity against some bacteria and fungi.34,35 The physicochemical properties of AgNPs and CsNPs have developed to analyze biocompatibility, biodegradability, bacteriostatic and bactericidal activity, aggregation capability, solubility, and precipitation range. To analyze some of these properties, nanoparticle size and zeta potential properties are important issues to provide insights into the different applications. Besides, this information is important to apply biological reducing agents ranging from bacteria, fungi, and natural extracts that have emerged as a biological route to synthesized nanoparticles. 36 Nowadays, AgNPs are involved in biomedical applications such as antibacterial agents, drug delivery vectors, theragnostic agents, and cancer treatment. 37 Thus, it is very important to provide information about physic-chemical properties to know the pharmaceutical and adverse effects for the different applications in the biomedical field. It has been reported the importance of the antibacterial properties of root canal lubricants and irrigants that contain Chx and cetrimide, showing antibacterial activity in vitro studies against E. faecalis. 38 Also, some sealers have significantly demonstrated antimicrobial efficacy against E. faecalis, Candida albicans, and Staphylococcus aureus. 39 This antibacterial property could prevent residual and recurrent infections and contribute to endodontic therapy, providing a hermetic seal between dentin walls and gutta-percha. 40 In this study, all endodontic sealers (AH Plus, Endosecuence, MTA Fillapex, Sealapex, and Tubliseal) showed some degree of bactericidal activity against E. faecalis, but Tubliseal and AH Plus showed the highest AAI when mixed with the different substances tested in this study. Probably these two sealants are an excellent choice for further research, since it is possible to combine them with other chemical solutions. In cases where Chx is selected as the main bactericidal agent, MTA Fillapex should be considered as well. It is important to notice that in this study, two standardized methods to test the antimicrobial activity providing direct contact with E. faecalis were used. 40 Also, SEM illustrations confirmed the antibacterial gradient pattern in the collagen membrane assay, showing that the combination of CsNPs-Chx had the strongest bactericidal activity, followed by Chx and propylene glycol with calcium hydroxide. Medical chitosan applications require, as a starting material, a low molecular weight, high solubility, and low viscosity in water at physiologically acceptable pH values.41,42 However, CsNPs has demonstrated variable bactericidal activity against different bacterial species and fungal pathogens (e.g. Streptococcus mutans, Escherichia coli, Staphylococcus aureus, C. albicans, etc.). Unfortunately, in this research project, CsNPs showed only a slight bactericidal activity against E. faecalis, but CsNPs could be used as a carrier for bactericidal substrates due to its biocompatibility. Recently, the combination CsNPs–AgNPs has been useful in in vitro and in vivo tests (skin injuries in rats) by avoiding microbial contamination.43,44 It has been reported in in vitro studies that antibacterial nanoparticles have higher activity because of the higher surface area that enables to achieve a greater degree of interaction with the negatively charged surface of bacterial cells. 45 Furthermore, it has been reported that some nanoparticles using different sizes, well-defined zeta potentials, and other specific physicochemical properties can play an important role in the antimicrobial activity against several bacterial species.18,19,27,46,47 The CsNPs and AgNPs used in this study were indicated to have good distribution and uniform sizes and shapes, while zeta potentials suggest a low risk to be agglomerated due to high electrical charge in CsNPs (52.0±5.4 mV) and AgNPs (−48.4±6.9 mV) samples promoting better particle stability.48–50 It is possible that CsNPs and AgNPs can produce a wider contact area with particular electrical charge, creating synergistic antimicrobial properties and promoting more contact area on the bacteria cell membrane and, in some cases, having the ability to penetrate into the cell leading to bacterial cell death.18,19,48 It is necessary to do additional experiments to explore the changes in physicochemical properties, cytotoxicity, and biocompatibility.
In this study, AH Plus, Tubliseal, and Endosequence showed an enhanced bactericidal activity; these sealers have a material base of mineral trioxide aggregate, zinc oxide eugenol, and bioceramic, respectively. This means that further research should be carried out with different sizes and morphologies of CsNPs that could provide a slow release of Chx in some of the sealers mentioned above. This in vitro model could be useful to test materials, chemical, or organic components against different bacterial species that are important in different biomedical fields (dentistry, orthopedics, dermatology, rheumatology, and others) where infections after surgery are a danger for the clinical success of treatment. This is an in vitro study where endodontic sealers combined with different substances showed antibacterial activity against E. faecalis in direct contact assays.
It is important to note that those sealers containing CsNPs-Chx showed a statistical difference (p < 0.001) when compared with Chx. AgNPs, CsNPs, Chx, and propylene glycol have shown antibacterial activity in different studies carried out in the biomedical field.44,45 When these substances were added to the sealers, all of them increased its antibacterial activity, but the best combination was CsNPs-Chx (p < 0.05). Previous studies have reported a significant antibacterial effect of CsNPs and zinc oxide nanoparticles against planktonic E. faecalis.49,50 It is important to note that there are reports about cytotoxicity in animal models (especially in small intestine, liver, brain, and kidneys) followed by repeated oral exposition of AgNPs,51,52 while CsNPs are used in pharmaceutical drug delivery systems due to their biocompatibility. 53
Conclusions
All single endodontic sealers tested were demonstrated to have good antibacterial properties; however, their antimicrobial effectiveness against E. faecalis was especially enhanced when Chx or the combination CsNPs-Chx was added. Since CsNPs have a wide spectrum of activity and high killing rate against Gram-positive bacteria, they could be an excellent material to be added to endodontic sealers. The collagen membrane method was used to confirm the antibacterial activity from different substances and analyze the mechanisms involved in the antibacterial activity from different solutions tested. This research was an effort to avoid the use of antibiotics since antibiotic-resistant bacteria is a major issue in dental infections. 7
Future research is needed to observe the behavior of endodontic sealers combined with CsNPs-Chx on biocompatibility with peri-radicular tissues, biodegradability, the ability to conserve the original physical and chemical properties, and assess the antibacterial effect on bacterial biofilms and its products. It represents a challenge for the scientific community to develop new bioactive compounds for endodontic sealers to avoid refractory infections.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
