Abstract
Purpose
The sealing of wide-open apex roots is still a challenge and requires the use of apical barrier techniques. The aim was to evaluate ex vivo the sealing and the apical morphology of 3 commercial calcium oxide (CaO)-containing sealers - namely, 2 zinc oxide-based (CRCS and Sealapex) and a calcium silicate MTA-based (TechBiosealer Endo) - placed in wet root canals with artificial wide-open apices.
Methods
Thirty human single-rooted teeth were shaped with Protaper and an artificial open apex (diameter size 110) was created. Each root was inserted in a custom-designed support containing simulated body fluid (Hank's balanced salt solution, HBSS) at the bottom simulating the presence of periapical fluid in the canal. Each sealer (TechBiosealer Endo, CRCS, Sealapex) was compacted to the apical 5 mm and the filled roots stored in HBSS at 37°C. The sealing was evaluated as micro-infiltrated fluid since 24 hours up to 6 months using a high-precision digital fluid flow meter. The sealers were also studied for setting time in HBSS, calcium releasing (statistical analysis by 2-way ANOVA followed by Student-Newman-Keuls test, P<0.05) and surface morpho-chemistry by environmental scanning electron microscope with energy dispersive X-ray (ESEM-EDX) and optical microscopy (OM) analysis.
Results
All sealers showed a stable seal. TechBiosealer Endo maintained a better seal than the other materials (P<0.05) and ESEM-EDX and OM analyses showed the presence of apatite deposits.
Conclusions
The clinical use of hydraulic hydrophilic MTA-based sealers can be recommended to stop/reduce the fluid flow rate through the apex. The artificial apical barrier in wet wide apices is a suitable technique able to seal wet root canals.
Keywords
Introduction
Many clinical conditions such as immature teeth, apical root resorption and endodontic retreatments are responsible for apical constriction loss and for a wide-open apex, which are frequently contaminated by fluid or blood.
In these conditions, the chance to obtain an adequate seal is compromised by the presence of root canal fluids, the large area to be sealed and an unfavorable canal shape. Nevertheless, a stable apical seal is crucial in endodontics to limit bacteria outflow and ensure the periapical tissues healing (1).
Zinc oxide eugenol–based as well epoxy resin–based sealers have several limitations to ensure apical sealing in wet root canals, therefore the application of any water-sensible endodontic materials, such as epoxy-based resin sealers (2-4), is to be avoided. For these reasons, the application of a bioactive, biocompatible and hydrophilic endodontic sealer may play a positive role to seal a wet or humid root canal apex and to promote a fast apical barrier formation (5) and the regeneration of periapical damaged tissue (6).
Several procedures using different materials have been recommended to induce root-end apical barrier formation. The application of calcium hydroxide is the most commonly advocated therapy for immature teeth with nonvital pulp and open apex, to produce apical tissue healing (1, 5, 7). Unfortunately, a number of applications are necessary, so the risk of reinfection and cervical root fractures is high (7-9).
Calcium silicate cements, commonly referred to as mineral trioxide aggregate (MTA) cements, are biocompatible bioactive hydrophilic materials (10-13) and are able to release calcium and hydroxide ions, showing in many in vivo studies better biological properties than other materials (14-16). MTA-based materials have been suggested for 1-step apexification (17) and have been recommended as a good alternative to conventional calcium hydroxide cements, showing interesting clinical results (18-22), but there are no studies confirming their long-term sealing ability in wide apex conditions (23).
The aim of this study was to evaluate the in vitro sealing ability of 3 different CaO-containing commercial sealers – 2 zinc oxide–based (Calcibiotic Root Canal Sealer [CRCS] and Sealapex) and 1 calcium silicate MTA–based (TechBiosealer Endo) – plugged in moist conditions at the apical third of single-rooted extracted teeth with artificially prepared wide apexes. The apical flow rate, as an index of sealing ability, was tested over time (from day 1 to 6 months after the obturation) using a digital fluid flow meter. The apical surface of the cements was studied after 6 months of immersion, with an environmental scanning electron microscope with energy dispersive X-ray (ESEM-EDX) analysis. Setting time, calcium releasing and internal morphology were also investigated.
Materials and Methods
Root preparation
Thirty human single-rooted teeth extracted for periodontal reasons were thoroughly cleaned and stored in distilled water at 4°C for less than 3 months prior to use. All teeth were sectioned at 14±1 mm from the apex with a water-cooled diamond bur (FG Intensiv n. D2; Lugano-Grancia, Switzerland). The canals were preliminarily shaped using a crown-down technique with nickel titanium rotary instruments (Protaper; Dentsply Maillefer, Ballaigues, Switzerland). The root canals were then widened using a Gates Glidden drill no. 4 (Maillefer, Ballaigues, Switzerland) to obtain an apical root canal diameter of size 110. The canals were irrigated between each instrument with 0.5 mL of ethylenediaminetetraacetic acid (EDTA, Ogna, Muggiò, Italy) and 1 mL of 5% NaOCl (Ogna, Muggiò, Italy), delivered from a 3-mL syringe with a 27-gauge needle, and finally rinsed with deionized water and dried with sterile paper points (Mynol, Milwaukee, WI, USA).
Root obturation
To reproduce the clinical condition (apical stop and canal wet environment), a tailored device (wet chamber) was designed. A special silicon support (Optosil; Heraeus Kulzer, Hanau, Germany) was prepared, and each tooth was positioned in the silicon to create an artificial alveolar chamber position in which to include the root. A simulated body fluid solution (0.02 mL of Hank's Balanced Salt Solution [HBSS]; Lonza, Verviers, Belgium) was added to the bottom of the support to simulate the presence of periapical fluid. Each root was placed inside the chamber so the periapical space and the entire root canal were filled by HBSS. The presence of fluid solution inside the root canal was assessed using paper points. The apical stop (i.e., silicon support) was 1 mm after the apical foramen.
The roots were randomly divided into 3 groups and obturated using commercial Ca-containing endodontic sealer: TechBiosealer Endo, Sealapex or CRCS (Tab. I). Each cement was prepared according the manufacturer's instructions, and using a micro spatula, the mixed cement was placed on the root canal orifice then carried up to the apex with a 50 K-file. The material was gently compacted with endodontic pluggers sizes 3 and 4 (LM-Dental, Parainen, Finland) into the first 5 mm of the apex (23). Each filled root was radiographed to verify the right placement of the filling material, the formation of an apical plug and the absence of deficit or X-ray detectable porosities. No back-filling was performed, to leave a fluid reservoir for the sealing ability test.
TESTED MATERIALS AND COMPONENTS

Fluid filtration test
An 18-gauge needle inserted across a plexiglas support was introduced into the coronal third of the filled root and the coronal side of the root was fixed to the plexiglass support with cyanoacrylate (Rocket, Corona, CA, USA). The external surface of root was coated with nail varnish to seal the root surfaces except for the apical orifice (2-mm apical surface free from varnish).
For controls, additional root canals were shaped as previously described and connected to a plexiglas support. In positive control (n=1), only the root walls were sealed with the nail varnish except for the apical orifice; in negative controls (n=1) the apex orifice also was sealed with a bonding agent (Scotchbond 1; 3M ESPE, St. Paul, MN, USA) and nail varnish.
All samples were stored in 10 mL of HBSS (used as simulated body fluid solution) at 37°C and submitted to the fluid filtration test after 1 and 7 days and 1, 3 and 6 months from the filling.
The composition of the HBSS was (g/L) 0.4 KCl, 0.06 KH2PO4 anhydrous, 0.35 NaHCO3, 8.0 NaCl, 0.05 Na2HPO4 anhydrous and 1.0 d-glucose.
A high-precision device (digital fluid flow meter) able to detect micrometric fluid movements through the apical system was designed for fluid flow rate measurements. The micrometric forward movement of an air bubble inside the microcapillary was converted into a fluid flow measurement through the filled root. Each sample was connected to the device (through the 18-gauge needle) working at a hydraulic pressure of 6.9 kPa (1 psi) and contained deionized water with chlorhexidine 0.03% to prevent bacterial contamination inside the system.
The fluid filtration rate was measured after different storage of the sample in HBSS (at 1 and 7 days and 1, 3 and 6 months), 3 times for each sample over a period of 4 minutes, and the mean was calculated. The results were expressed as microliters per minute. Positive and negative controls were examined at the beginning of each experimental session.
ESEM-EDX analysis
An environmental scanning electron microscope (ESEM, Zeiss EVO 50; Carl Zeiss, Oberkochen, Germany) connected to a secondary electron detector for energy dispersive X-ray analysis (EDX; Oxford INCA 350 EDS; Oxford Diffraction, Abingdon, UK) with the computer-controlled software Inca Energy Version 18, using 20-25 kV accelerating voltage, was used.
The roots were placed directly onto the ESEM stub (without any previous sample preparation) and examined in wet conditions.
ESEM-EDX analysis was used to evaluate the external cement–apex morphology of the samples, the morphology of the apex, the margin and the surface of the restoration after 6 months of soaking in HBSS. Elemental analysis (weight % and atomic %) of materials was performed applying the ZAF correction method.
Optical microscopy (OM) analysis
Additional filled roots (n=15) were soaked in 10 mL of HBSS at 37°C for 24 hours, removed from the medium, dried and inserted in epoxy resin (Hardrock; Remet, Bologna, Italy).
The roots were sectioned through the coronal-apex axes using a water-cooled diamond saw (Micromet M; Remet, Bologna, Italy) and analyzed through an optical microscope (Motic; Motic Incorporation Ltd., Hong Kong). Optical microscope analysis was performed to observe the inner side of the restoration and to detect the presence of voids or porosities.
Setting times under immersion in simulated body fluid
Materials were prepared according to the manufacturer's directions. Discs (10 mm in diameter and 2 mm high) were prepared and immediately immersed in 10 mL of HBSS. The initial and final setting times of the materials were evaluated using Gilmore needles according to ASTM C266-03 and ADA specifications (24, 25). The Gilmore initial setting time was the elapsed time (minutes) between the mixing of the cement and the first penetration measurement that does not mark the specimen surface with a complete circular impression. The initial setting time needle was 113.4 g in weight and 2.12 mm in tip diameter. After the initial setting time was measured, the specimens were tested every 5 minutes with the final setting time needle with a tip diameter of 1.06 mm and a weight of 453.6 g. Their exact evaluation was performed on 3 replicates for each material.
Calcium ions released in soaking water
The materials were mixed according to the manufacturer's directions, and cement discs (n=5, for each tested material) were prepared. Each disc was immediately immersed in 10 mL of deionized water in polypropylene-sealed containers stored at 37°C.
The amount of calcium ions leaked in the soaking water was assessed under constant stirring using a selective calcium ion probe (Calcium ion electrode; Eutech Instruments Pte. Ldt., Singapore) connected to a multiparameter laboratory meter (inoLab 750; WTW, Weilheim, Germany) after the addition of 0.200 mL (2%) of ionic strength adjuster (ISA, 4 mol/L KCl; WTW, Weilheim, Germany). The test was performed after 3 hours and 1, 7, 14 and 28 days of soaking, and the medium was refreshed with new deionized water after each test.
Statistical analysis
Descriptive statistics (mean, standard error) were used for the evaluation of the characteristics of the material groups. Normal distribution of the data was checked, and the significance of the differences between the groups was examined by 2-way ANOVA followed by the Student-Newman-Keuls test (significance P value cutoff of P<0.05).
Results
Fluid filtration test
Table II reports the mean values of the fluid filtration rate (μl/min) of all groups at different storage times (1 and 7 days and 1, 3 and 6 months). Statistically significant differences (P<0.05) were found among the groups.
APICAL FLUID FILTRATION
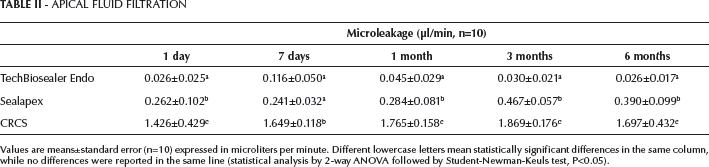
Values are means±standard error (n=10) expressed in microliters per minute. Different lowercase letters mean statistically significant differences in the same column, while no differences were reported in the same line (statistical analysis by 2-way ANOVA followed by Student-Newman-Keuls test, P<0.05).
TechBiosealer Endo maintained a low fluid filtration rate from day 1 to 6 months. CRCS showed the highest fluid flow rate compared with the other sealers. Sealapex showed lower values of fluid flow with respect to CRCS, but statistically higher than TechBiosealer Endo. The negative control exhibited little or absent fluid flow (0.004 μl/min), while the positive control demonstrated a high level of leakage.
ESEM-EDX analyses
ESEM-EDX analysis allowed the inspection of the apical external surface as restored, to detect its morphology and marginal adaptation, both conditions that may influence the fluid flow rate.
In the TechBiosealer Endo group (Fig. 1) the root end restoration surface showed an irregular morphology with calcium phosphate deposits which covered the marginal gaps and masked the obturation margins. EDX revealed high calcium (Ca) and phosphorous (P) peaks (Ca/P ratio approximately 2.9).
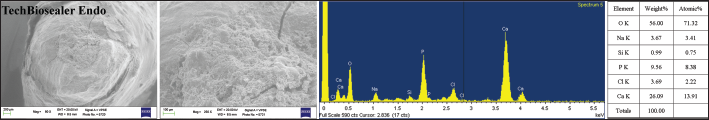
Apical morphology and chemical composition as shown with ESEM-EDX analysis of TechBiosealer Endo–filled wide-open apex after 6 months of soaking in HBSS at 37°C. EDX revealed high Ca and P peaks (Ca/P ratio approximately 2.9) beside traces of silicon (Si), chlorine (Cl) and sodium (Na). The cement surface is covered by a thick layer of Ca-P. Phosphorous is not in the composition of calcium silicate sealer, but it originates from the simulated body fluid and correlates with the apatite-forming ability of the calcium silicate sealer.
In the Sealapex group (Fig. 2), several voids and limited marginal gaps were detected at the cement–dentine interface. No external deposits were detected, either on the root surface or on the material surface.
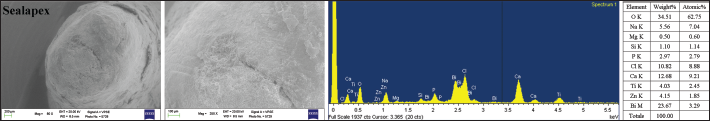
Apical morphology and chemical composition on ESEM-EDX analysis of Sealapex-filled wide-open apex after 6 months of soaking in HBSS at 37°C. EDX detected zinc (Zi), bismuth (Bi), Ca and titanium (Ti) not originating from the material composition; Na, Cl and traces of P came from the HBSS.
Also in the CRCS group (Fig. 3), a marginal gap at the cement–dentine interface was present.
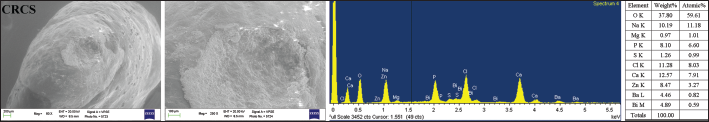
Apical morphology and chemical composition on ESEM-EDX analysis of CRCS-filled wide-open apex after 6 months of soaking in HBSS at 37°C. EDX showed Zi, Bi, sulphur (S), Ca and barium (Ba) not originating from the material, together with Na, Cl and P derived from the HBSS.
Optical microscopy analysis
Optical microscopy analysis showed the morphology of the internal structure of the cements after 24 hours in HBSS. A number of porosities were detected on the internal side of the Sealapex group. A thick layer of white precipitates well adherent to the cement surface was observed only on the apex of the TechBiosealer Endo–sealed roots (Fig. 4). No evident porosity or voids were detected on CRCS-sealed roots.

Apical morphology on optical microscopy of sectioned filled roots after 24 hours of soaking in HBSS at 37°C. The apatite layer on the TechBiosealer Endo–filled apex is shown (arrow), as is the large discrepancy between the CRCS and dentinal wall in the apical portion.
Setting times in simulated body fluid
Table III shows the initial and final setting times of tested materials after soaking in HBSS. Sealapex showed the longest setting time (more than 5 days for initial, and more than 8 days for final setting). In contrast, CRCS showed the shortest setting time (46±1 minutes for initial and 85±8 for final setting). TechBiosealer Endo showed 140±7 minutes for the initial and 351±10 for the final setting.
INITIAL AND FINAL SETTING TIMES IN SALT SOLUTION HBSS

Values are means±standard error (n=3).
HBSS=Hank's balanced salt solution.
Calcium ions released in soaking water
The amount of calcium ions leaked is reported in Table IV. All of the tested materials showed a release of calcium during the tested time. Sealapex showed the greatest amount of calcium release at all tested times.
CALCIUM IONS (PPM) RELEASED IN SOAKING WATER
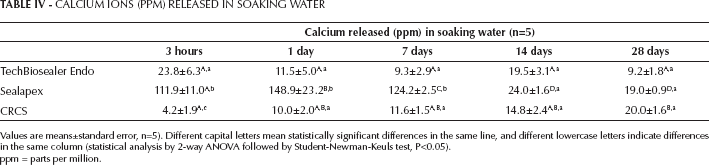
Values are means±standard error, n=5). Different capital letters mean statistically significant differences in the same line, and different lowercase letters indicate differences in the same column (statistical analysis by 2-way ANOVA followed by Student-Newman-Keuls test, P<0.05).
ppm = parts per million.
Discussion
In an attempt to create a simple and safe clinical procedure, this study proposed an in vitro method to simulate the orthograde obturation of typical wide-open apices in the presence of biological fluid (i.e., blood, periapical fluid and plasma) inside the root canal system.
The fluid filtration technique employed in this study to test sealing ability has been described previously and has proven to be sensitive and reproducible (17, 26-31). The main advantage of this technique is the possibility of preserving the specimens after each assay, allowing analysis at different study periods. The digital flow meter used was specifically designed to detect with micro measurement precision the fluid flow through the filled root.
The study demonstrated that the use of a calcium silicate MTA sealer such as TechBiosealer Endo in an open root apex is advisable to stop and prevent any apical fluid movement and to obtain a stable and durable seal better than the conventional ZnO CaO-containing sealers.
TechBiosealer Endo is a hydrophilic and hydraulic cement that sets in the presence of moisture (i.e., oral fluids and blood) and for this reason may create an apical barrier in wet/humid root canals. This apical barrier is bioactive thanks to the release of calcium ions and the apatite deposit formation (10-14).
CRCS is a zinc oxide–based sealer containing eugenol and eucalyptol, and it showed the highest fluid filtration rate at all tested times. This endodontic sealer demonstrated a fast setting time in HBSS, and for this reason the fluid present at the apical third may cause an incomplete sealer adaptation to the dentinal walls. The incomplete setting is responsible for the void and gaps detected at ESEM-EDX analysis, and caused the higher fluid filtration rate. For these reasons, CRCS is not adequate for wide-open apex obturation.
Despite its long setting time (more than 5 days for the initial setting), Sealapex showed a fluid filtration rate statistically lower than CRCS. Sealapex undergoes volumetric expansion during the long setting reaction, due to water absorption (31, 32) which probably ensures a stable seal in a short time.
TechBiosealer Endo showed a statistically significant better sealing ability and displayed a homogenous fluid filtration rate over time. As described, TechBiosealer Endo is a calcium silicate MTA sealer designed for orthograde endodontic applications. Previous linear variable displacement transformer studies demonstrated a linear expansion of calcium silicate cements during the setting reaction (33, 34). Moreover, the presence of NaF (11, 27, 34) in the composition of TechBiosealer Endo delayed the setting time and increased the expansion of the sealer from 0.10% to 6.72% compared with other MTA-based cements (34). This expansion is likely responsible for the low fluid filtration.
As showed by EDX analysis, the TechBiosealer Endo group displayed calcium and phosphorous peaks on the external apical root surface (Fig. 1). According to previous studies (10-13, 35, 36), calcium silicate MTA cements are able to produce a surface layer of apatite when in contact with simulated body fluids such as HBSS. This apatite layer may help to improve the biological activity at the bone–periapical level, promoting barrier formation and inducing the activation and differentiation of the apical cells (12, 13, 37, 38). Moreover, the apatite deposits may fill the porosity and residual gaps, providing an additional external sealing.
Conclusions
In conclusion, this study proposes and recommends the application of calcium silicate MTA cements when biological fluids (i.e., blood) are present in the apical third of the root canal and their fluid contamination may compromise sealer setting and adaptation.
