Abstract
Introduction:
We assessed the effect of different available denture cleansers on the roughness and hardness of polyetherketoneketone, thermoinjection-molded polyamide, and polymethylmethacrylate.
Materials and Methods:
A total of 150 disc-shaped specimens were fabricated (10 mm × 2 mm) from these three denture base resins, and divided into five subgroups (n = 10) according to immersion procedures. One of these groups subjected to distilled water served as control, whereas other groups were subjected to daily cleansing with four denture cleansers (Corega, Protefix, Curaprox, and Perlodent) for 8 h a day for 140 days. The surface roughness and hardness values of specimens were recorded by measuring twice at baseline, and again after application of chemical solutions. Topography alterations after treatments were assessed with scanning electron microscopy. The data were subjected to statistical analysis and comparison among groups was done using Kruskal Wallis and Wilcoxon Signed Ranks tests. P-value <0.05 was considered significant.
Results:
The surface roughness of polyetherketoneketone, polymethylmethacrylate, and polyamide dentures was increased significantly by chemical solutions of denture cleansers. While the hardness value of polyetherketoneketone was not affected significantly after immersion in denture cleansers, those of polymethylmethacrylate and polyamide decreased significantly. Compared with Curaprox, the effervescent tablets significantly altered the surface hardness and roughness of polyamide.
Conclusion:
Denture cleansers can considerably alter the surface roughness and hardness of denture base resins and should be used carefully depending on the material.
Introduction
Cleaning of dentures is important for maintaining oral mucosal health and the longevity of partial removable dentures. Bacterial and fungal colonization on dentures can cause denture stomatitis, angular cheilitis, and poor oral health.1,2 In order to maintain the hygiene of removable prostheses, various chemical, mechanical, and chemical+mechanical methods are recommended. 3 Among these, mechanical methods are used widely by patients for minimizing biofilm formation; however, several elderly patients find this difficult because of impaired motor function or physical limitations.4,5 Therefore, the additional use of a chemical cleanser for a certain period of time is recommended.6,7 Denture cleansers are classified into different groups according to their chemical composition, which may include alkaline peroxides, acids, enzymes, and alkaline hypochlorites. 8
Ideally, denture cleansers should reduce or remove the biofilm without altering the physical and mechanical properties of the denture base material.9,10 However, several studies claim that the prolonged use of denture cleansers can produce destructive undesirable effects on the physical and mechanical properties, including the color, surface roughness, and hardness, of denture base resins.11,12 Surface roughness is an important factor implicated in plaque formation and bacterial colonization on dentures. 13 The physical strength of the denture base resin determines the ease of finishing and polishing and the resistance to erosion during cleaning. 14
Over the years, polymethylmethacrylate (PMMA) has been preferred for denture fabrication because of the ease of manipulation, lower cost, and need for simple equipment. 10 In recent years, however, a new generation of polyamides known as “nylon” have been commonly used for this purpose. 15 Polyamide resins are thermoinjectable and easy to polish; in addition, they exhibit good flexibility and durability, and low porosity and cytotoxicity. Therefore, they are good alternatives to conventional PMMA-based acrylic resin. 16 However, they have several disadvantages, such as difficulties in reparability and smoothening, which can lead to microbial retention on the denture surface. 17
Polyaryletherketone (PAEK) belongs to the family of thermoplastic polymer materials and has been used for denture fabrication via computer-aided design and computer-aided manufacturing (CAD/CAM) systems. 18 Polyetheretherketone (PEEK) polymer, which belongs to the PAEK family, has been well recognized for its superior performance and is used in cardiovascular and craniomaxillofacial procedures as well as restorative dentistry. 19 According to the manufacturer’s recent declaration, polyetherketoneketone (PEKK) is the latest member of the PAEK family, with 80% higher compressive strength and superior performance when compared with PEEK. Because of its properties, it has been used as a restorative material in dental implant procedures, fixed prostheses, and healing caps. 20 PEKK is also considered an alternative to conventional materials for removable prostheses because of its white color and excellent mechanical and physical properties such as low plaque retention, high durability, and resistance to abrasion. 21 Several studies have documented the influence of denture cleansers on the mechanical and physical properties of heat cure acrylic resins (PMMA). However, to our knowledge, there is limited information about the influence of denture cleansers on the surface properties of PEKK and polyamide resins. Therefore, the aim of the present study was to evaluate the effects of different types of denture cleansers (effervescent tablet form or citric acid-based) on the surface roughness and hardness of three different denture base resins. Our hypothesis was that denture cleansers can alter the surface roughness and hardness of denture base resins.
Materials and methods
PEKK (Pkt group; Cendres+Metaux, Biel, Switzerland), thermoinjection-molded polyamide (DEFLEX, Nuxen SRL, Buenos Aires, Argentina; Dfl group), and PMMA (Heraeus Kulzer, Hanau, Germany; Mld group) were used in the present study. Table 1 lists all the materials used in the study. From each denture base resin, 50 disc-shaped specimens measuring 10 mm × 2 mm were prepared and divided randomly into five subgroups (n = 10) based on the denture cleanser used for treatment.
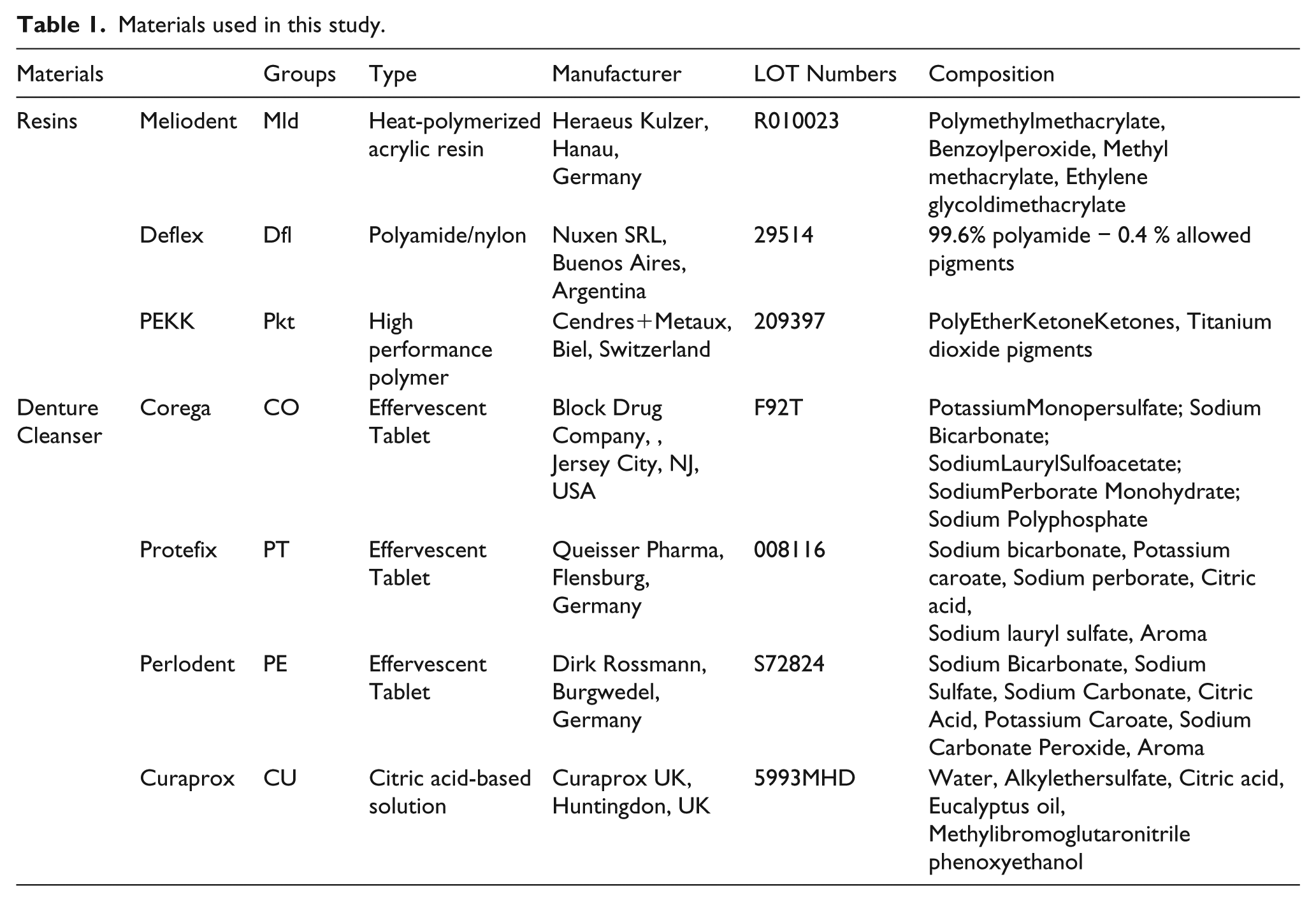
Materials used in this study.
Specimen preparation
For the Mld group, 50 disc-shaped wax patterns (Cavex Set Up Regular, Cavex, Haarlem, the Netherlands) were prepared with a 2-mm thickness and 10-mm diameter. These patterns were invested in Type IV gypsum (Ultrarock Klassek, Megadental, Büdingen, Germany) in metal flasks. After the dewaxing process, the metal molds were packed with heat-polymerized acrylic resin mixed and prepared according to the manufacturer’s instructions. The acrylic resin was then polymerized under pressure in a hot water bath at 100°C for 30 min. Subsequently, the specimens were allowed to cool and excess resin was removed using a handpiece.
For the Dfl group, 50 polyamide resin samples were melted at 270–288°C for 11 min, in accordance with the manufacturer’s recommendations. The melted resin was then injected into a muffle in 30 s under a pressure of 6 bar, which was applied for 3 min. Once the pressure was released, the specimens were allowed to cool before the muffle was opened.
For the Pkt group, 50 disc-shaped specimens (10 mm × 2 mm) were designed in the stereolithography (STL) format using AutoCAD (Autodesk, San Rafael, CA, USA). These STL files were transferred to a five-axis milling machine (Roland DWX50, Roland DGA, Irvine, CA, USA), and specimens were milled from a Pekkton ivory block (Cendres+Metaux, Biel, Switzerland) with the help of a CAD/CAM system (3Shape A/S, Copenhagen, Denmark). All specimens were prepared by a single dental technician and cleaned ultrasonically with distilled water.
For all specimens, one of the surfaces was finished with a sequence of 150–800-grit aluminum oxide abrasive sheets under running water, using a Metaserve2000 polishing machine (Buehler UK, Coventry, UK). This helped in achieving standardized, smooth, plane surfaces that allowed the positioning of the specimens for roughness measurements. A single investigator then used the same machine to polish the specimens with a felt disc. A digital caliper (Digimatic Caliper 0-40”, Mitutoyo Corporation, Kawasaki, Kanagawa, Japan) was used to ensure a specimen thickness of 2 mm for the surface roughness and hardness tests. Baseline measurements of roughness and hardness were performed after storage of the specimens in distilled water at 37°C for 48 h. 22
Treatment with denture cleansers
The five subgroups (n = 10 each) for each resin base were randomly subjected to treatment with distilled water (control), Corega (CO), Protefix (PT), Curaprox (CU), or Perlodent (PE). CO, PT, and PE are available as effervescent tables, and the same procedure was used for these three cleansers in accordance with the manufacturers’ recommendations. Briefly, a chemical solution was prepared by immersing a tablet in 200 ml of warm distilled water. 23 The citric acid-based cleanser CU was placed at the base of the individual denture box of the company until concentrated solution was covered according to the manufacturer’s recommendations. Then, 200 ml of warm water was added to the concentrated solution for preparation of the final chemical solution. All specimens were immersed completely in the chemical solutions or water. The control group specimens were immersed in distilled water throughout the experiment, while the other specimens were exposed to their respective chemical solutions for 8 h a day. Between treatments, the specimens were stored in distilled water at room temperature for 16 h. The experiment was repeated for 140 days, 24 and the chemical solutions were prepared fresh every day.
Measurement of surface roughness
A surface profilometer (Surftest 402 Analyzer, Mitutoyo Corporation, Tokyo, Japan) was used to measure the average surface roughness values for the different specimens. The 5-μm diamond tip, which was in contact with the measurement surface, horizontally scanned a 350-μm area at a speed of 0.5 mm/s, under a load of 0.75 mN. The roughness values were obtained in micrometers. The surface profilometer was calibrated after each measurement using the manufacturer’s reference specimen. For each specimen, roughness values were recorded at baseline (T0) and after treatment (T1). Measurements were obtained from three different locations in a clockwise direction, and the average of the three measurements was recorded as the mean value for that specimen. In total, 150 specimens (450 T0 and 450 T1 measurements) were evaluated, and all measurements were made and recorded by a single investigator.
Vickers hardness measurements
The surface hardness (Vickers Hardness Number; VHN) of the specimens was also measured at T0 and T1. A Vickers microhardness tester (Matsuzawa MHT2 High Quality, Matsuzawa Seiki, Tokyo, Japan) was used with a load of 100 gf and dwell time of 30 s for all groups. Measurements were made at three different locations on each specimen, and the average was recorded as the mean value for that specimen. In total, 150 specimens (450 T0 and 450 T1 measurements) were evaluated, and all measurements were taken and recorded by a single investigator.
Scanning electron microscopy
From each group, a specimen with a roughness value that was closest to the average value for that group was selected for scanning electron microscopy (SEM) examination. The specimens were dried at 37°C for 24 h. Then, 35% phosphoric acid was applied to the dried specimens for 10 s, and the surfaces were cleaned with air/water spray for 5 s before sputter-coating with gold–palladium. The prepared surfaces were then examined under a scanning electron microscope (Zeiss EVO LS10 VP, Oberkochen, Germany).
Statistical analysis
All statistical analyses were performed using SPSS version 23 (IBM, New York, NY, USA). The Kolmogorov-Smirnov test was used to determine whether the specimens showed normal distribution. Because the distribution was not parametric, hardness and roughness values were analyzed and compared between groups using Kruskal-Wallis and Mann-Whitney U tests. The Wilcoxon signed rank test was used to compare values before and after treatment in each group. A P-value of <0.05 was considered statistically significant.
Results
Surface roughness
The mean surface roughness values for each group are presented in Table 2. There were significant intergroup differences at baseline, with the lowest value observed for the Mld group and the highest value observed for the Pkt group (Table 2). At T1, there were no significant differences among the CO, PT, PE, and CU subgroups in the Mld and Pkt groups. However, surface roughness values were significantly higher for the CO, PT, and PE subgroups than for the CU subgroup in the Dfl group (P < 0.05); there were no significant differences among CO, PT, and PE (P > 0.05). All denture cleansers increased the surface roughness values relative to the baseline values in the Mld, Dfl, and Pkt groups (P < 0.05). Although the surface roughness at T1 was greater than that at T0 in the control group, the difference was not statistically significant (P > 0.05).
Mean ± SD roughness values (Ra, μm) of materials evaluated.
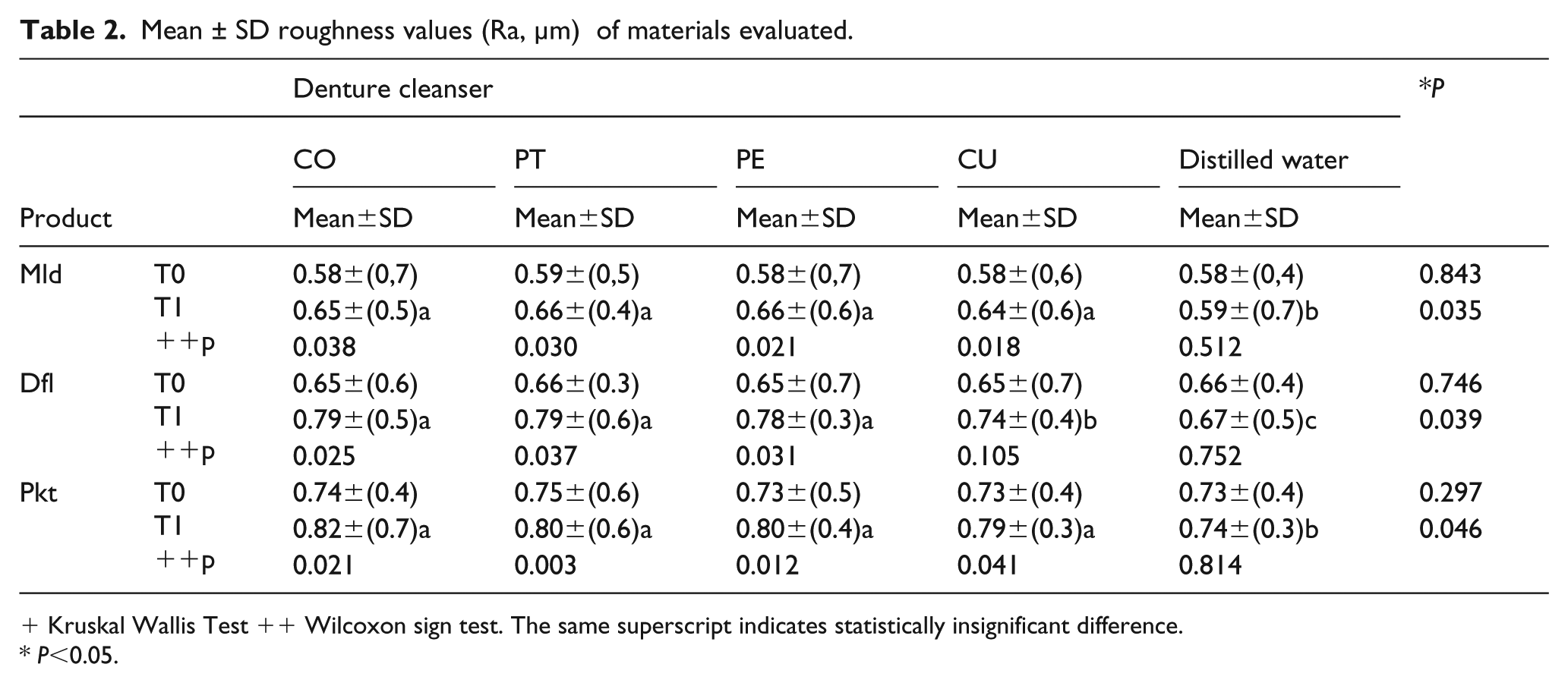
Kruskal Wallis Test ++ Wilcoxon sign test. The same superscript indicates statistically insignificant difference.
P<0.05.
Surface hardness
The mean VHNs for the different groups are presented in Table 3. Distilled water decreased the hardness values of all materials, although its influence was not statistically significant (P > 0.05). In the Dfl and Mld groups, but not in the Pkt group, all four denture cleansers significantly decreased the hardness values relative to the baseline values (P < 0.05). In the Dfl group, the hardness values after immersion in CO, PT, and PE were lower than that after immersion in CU. There were no statistically significant differences between the denture cleansers in the Pkt group. The highest and lowest hardness values were recorded for the Pkt and Dfl groups, respectively.
Mean ± SD hardness values (kg/mm2) of materials evaluated.
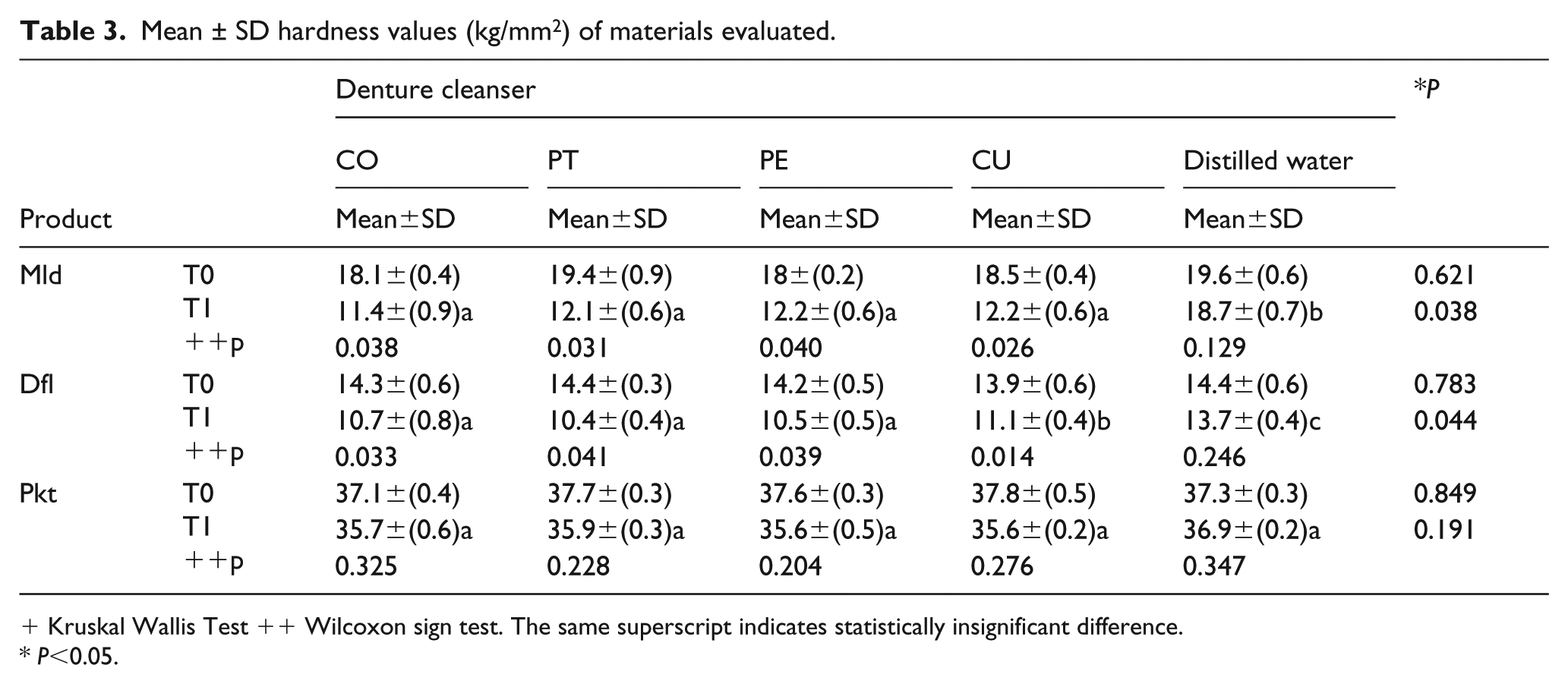
Kruskal Wallis Test ++ Wilcoxon sign test. The same superscript indicates statistically insignificant difference.
P<0.05.
SEM analysis
The SEM images obtained after treatment were consistent with the results of roughness and hardness measurements. The specimens in the Mld group showed the least roughness, and all denture cleansers affected the surface properties in a similar manner (Figure 1). Figure 2 shows the differences among the denture cleansers with regard to their effects on the specimens in the Dfl group. The alteration on the surface topography of the specimen in the CU subgroup was the lowest. The surface roughness of all specimens in the Pkt group increased after treatment with the denture cleansers, with the highest roughness value obtained for the CO subgroup (Figure 3).

SEM images of Mld surfaces (500×). (a) CO, (b) PT, (c) CU, (d) PE, and (e) control. Surface roughness was increased after the immersion of the denture cleansers. The lowest roughness values were observed in Mld group against Pkt and Dfl groups.

SEM images of Dfl surfaces (500×). (a) CO, (b) PT, (c) CU, (d) PE, and (e) control. Surface roughness was increased after the immersion of the denture cleansers. CU was the lowest roughness values among denture cleansers in Dfl group.

SEM images of Pkt surfaces (500×). (a) CO, (b) PT, (c) CU, (d) PE, and (e) control. Surface roughness was increased after the immersion of the denture cleansers. The highest roughness values were observed in Pkt group against Dfl and Mld groups.
Discussion
According to the results of the present study, the hypothesis that denture cleansers can affect the surface roughness and hardness of PEKK, thermoplastic polyamide, and PMMA denture base resins was accepted. We tested three different brands of sodium bicarbonate-containing cleansers and one citric acid-based cleanser, and the results revealed significant alterations in the surface roughness and microhardness of all three denture base resins after treatment with the different denture cleansers.
Sodium perborate turns into an alkaline peroxide solution by decomposing into sodium metaborate, hydrogen peroxide, and nascent oxygen when it comes in contact with water. 25 This peroxide solution combines alkaline detergents (which reduce surface tension) and agents such as sodium perborate or percarbonate (which release oxygen from the solution). Both mechanical and chemical cleaning are effected through oxygen bubbles. 26 The increase in the surface roughness after treatment with denture cleansers in the present study can be attributed to the active oxygen released by the solution containing hydrogen peroxide at a certain temperature. 12 Moreover, Davi et al. reported that treatment with alkaline chemical solutions altered the surface morphology of polymeric denture base resins by inducing the release of plasticizers. 27 We found that CU, a citric acid-based cleanser, resulted in roughness values lower than those observed with the other three effervescent cleansers; this could be attributed to the difference in the amount of sodium bicarbonate and the lack of a mechanical cleansing mechanism that is a characteristic of effervescent cleansers. 26 The polyamide specimens were expected to show an increase in the surface roughness, because polyamide shows greater water absorption than does PMMA. 28 However, we did not compare the materials in terms of water absorption in our study, because structural changes in the materials should be considered in that case; future studies can focus on this topic. PMMA denture base resin is resistant to surface changes under pressure, depending on the intensity, and it shows less water absorption. The higher water content of Cu explains the milder effects on PEKK and PMMA than on polyamide in the present study. The increased water absorption and dissolution of plasticizers seen with polyamide resin are responsible for the deterioration of its properties. 29
Machado et al. observed a significant increase in the surface roughness of denture base resins after repeated disinfection by immersion in a chemical solution containing sodium perborate added via effervescent tablets. 12 Peracini et al. also reported that, compared with distilled water, chemical solutions prepared from two different sodium perborate-based effervescent tablets significantly increased the surface roughness of heat-polymerized acrylic resin. 23 Durkan et al. claimed that there was a significant increase in the surface roughness of polyamide and PMMA denture base resins after immersion in cleanser solutions prepared from sodium bicarbonate tablets. 30 The results of the present study are consistent with those of these in vitro studies.
In the present study, surface roughness exhibited the maximum values after cleanser treatment in the Pkt group, followed by the Dfl and Mld groups (Table 2). One possible explanation is that we attempted to standardize all specimen surfaces by polishing and smoothening processes before initiation of the experiment. Bollen et al. reported that the surface roughness of the acrylic resin was affected by polishing abrasives used during the standardization process. 31 Because PMMA is easy to polish, its initial roughness is less. However, polyamide resins have a fibrous, semi-flexible structure and low surface hardness. Moreover, the high water temperature and oxygen-releasing solution used during the cleansing procedure may cause changes in the resin surfaces. 32 Because there are no reports on the surface roughness of PEKK, it was difficult to make comparisons for this material. We speculate that smoothening and polishing of PEKK with aluminum oxide abrasive sheets is difficult because of its high compressive strength. Moreover, it seems difficult to functionalize the PEKK surface with functional groups because of its intrinsic chemical inertness. 33 PEKK possesses a high degree of nanometer surface features; therefore, it exhibits a more nano-rough surface topography that significantly increases the surface area and exposes the novel chemical structure. 34 Consequently, the Pkt group showed the maximum surface roughness before and after treatment with denture cleansers. However, PEKK should be analyzed with regard to the content and distribution of the material in future studies. In the present study, immersion in distilled water increased the surface roughness of all materials, probably because water absorption can cause variations in dimensions and fatigue, with subsequent fractures in the resin material. 17
We found that surface hardness exhibited the maximum values after treatment in the Pkt group, followed by the Mld and Dfl groups (Table 3). This finding can be attributed to the different mechanical and physical properties and the specimen preparation methods. The hardness of PEKK, polyamide, and PMMA decreased after immersion in the denture cleansers, regardless of the type of cleanser. However, this decrease was not statistically significant in the PEKK group. Once again, there are no studies on the effects of alkaline peroxide solution on the hardness of PEKK, so comparisons were not possible. However, one theory could be that the diphenylketone moiety in the PEKK backbone generates active radicals on its surface under heat and ultraviolet irradiation. The radical species subsequently induce functional monomer (such as vinyl monomer containing target functional groups) polymerization, resulting in a functional group-decorated PEKK surface. 35 The decrease in the hardness of the PEKK specimens can be attributed to the combination of monomers with free radical species via oxygen bonding. However, because it exhibits a crystalline as well as an amorphous structure (which means strength and increased durability), the decrease was not statistically significant. Furthermore, the material has a slow rate of crystallization, so it is resistant to the effects of corrosive materials. 36 Meanwhile, the difference between PMMA and polyamide was anticipated, because PMMA is a conventional resin without flexible agents such as plasticizers. Furthermore, polyamide resins have been shown to have a greater fibrous content and lower modulus of elasticity. 37 In a study that compared the effects of denture cleansers on the surfaces of polyamide and PMMA, Durkan et al. reported that the hardness of polyamide was lower than that of PMMA. 30 They also observed that treatment with sodium perborate decreased the hardness values for both polyamide and PMMA. Neppelenbroek et al. reported that sodium perborate solution decreased the hardness of acrylic resins, 38 while Pinto et al. reported that disinfectant solutions promoted a statistically significant decrease in the hardness of hard reline resins compared. 39 The latter authors also claimed that the mechanical resistance of polyamide resins is higher than that of conventional heat cure resins; therefore, the former exhibit a lesser decrease in the surface hardness. Another reason for the lesser decrease in hardness is the absence of residual monomer in polyamide resin. 13 Our findings are in agreement with the findings of these previous studies. The decrease in the hardness of conventional heat cure acrylic resin specimens can be attributed to the continuous polymerization reaction, monomer release, and the combination of monomers with free active radicals via bonding with liberated oxygen. 40 With regard to the effects of the various denture cleansers, we found that the effect of CU on the hardness of polyamide was not as significant as the effect of CO, PT, and PE. The amount of sodium bicarbonate in CU, which has a high water content, is less than that in the other effervescent cleansers. Polyamide was affected to a lesser extent because the water absorption volume was higher than that with PMMA and PEKK.
The three effervescent cleansers used in the present study showed similar results and effects in terms of both surface roughness and hardness, even though we expected different results from all three solutions. This finding indicates that the mechanisms of action of the three cleansers are similar. Briefly, exposure of denture base resins to chemical solutions containing alkaline peroxide results in the generation of free radicals from the peroxide. Small molecules are obtained from large molecules via oxidation and conversion by free radicals.
This study has some limitations. First, the effects of different temperatures of the prepared chemical solutions, exposure times of the specimens to the solutions, and bacterial invasion were not evaluated and should be assessed in future studies. Second, the surfaces of the specimens used in this study were flat and did not completely mimic the surface structure of actual prostheses inserted in the oral cavity. In addition, the effects of saliva were not taken into consideration. Finally, further studies on the PEKK polymer are necessary to obtain more data and facilitate comparisons.
Conclusions
Within the limitations of the study, the findings suggest that denture cleansers significantly increase the surface roughness of PMMA, polyamide, and PEKK denture base resins and significantly decrease the surface hardness of polyamide and PMMA; the decrease in the hardness of PEKK is not significant. Compared with CU, effervescent cleansers significantly alter the roughness and hardness of polyamide. Thus, denture cleansers should be carefully used depending on the denture base material.
Footnotes
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
