Abstract
Introduction:
Besides mechanical cleaning, mouthwashes, and denture cleansers are typical supplements for oral care. Many chemical mouthwashes and denture cleansers provide positive effects, but finding safe and natural alternatives should be the priority.
Aim of study:
This study aims to investigate the effectiveness of natural materials as mouthwash and denture cleansers.
Material and method:
In examining the antimicrobial efficacy of mouthwashes, the minimum inhibitory concentration values were determined for five groups: distilled water (negative control), 0.2% chlorhexidine, chitosan solution, Saussurea costus solution, and chitosan-Saussurea solution. As for denture cleansers, the efficacy was evaluated after immersing polymethylmethacrylate (PMMA) disks in the previous solutions. The cell viability of Human Gingival Fibroblast (hGFs) after treatment with tested solutions was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays. Data was analyzed using ANOVA with Bonferroni post hoc tests.
Result:
The prepared solutions showed comparable antimicrobial efficacy and cell viability as chlorohexidine. The chitosan-Saussurea costus solution demonstrated a higher antimicrobial effect and better prevention of microorganisms’ adherence to PMMA disks and cell viability. It also did not affect the roughness of PMMA in the short term.
Conclusion:
The new denture cleaning and mouthwash agent, which contains the natural constituents of chitosan and Saussurea costus, demonstrates significant antibacterial effectiveness and serves as a viable eco-friendly alternative to commercial mouthwash, as well as exerting minimal influence on denture roughness.
Clinical implications:
Chitosan and Saussurea costus have antibacterial and anti-inflammatory properties that can potentially enhance oral health. These materials present a viable path toward the production of safe, efficient, and sustainable oral care products due to their natural and biocompatible substitutes.
Introduction
Denture-base materials and artificial teeth accumulate deposits similar to natural teeth. The oral cavity, whether natural or synthetic, is coated with a layer of salivary glycoprotein and immunoglobulin called “pellicle” within 30 min. This layer is about 0.5–1.5 μm thick. The pellicle serves as a surface on which oral residues, such as mucus, food particles, desquamated epithelial cells, and microorganisms (bacteria and fungus), easily attach.1–3
Inadequate oral hygiene is the primary cause of gingivitis, which is caused by the accumulation of microbial biofilms on the surfaces of teeth. 4 The first stage of periodontal disease is plaque-induced gingivitis. If left without treatment, gingivitis would spread and penetrate the soft and bone tissue that supports teeth, ultimately leading to tooth loss. 5 Treatment of plaque-induced gingivitis includes mechanical plaque control (brushing, flossing, and interdental aids) and chemical plaque control as an adjunct to mechanical plaque control. 6 Mouthwashes are frequently used to complement other tools for maintaining oral hygiene. These products contain synthetic and/or natural elements that act on microorganisms by preventing some of their enzymatic reactions and growth inhibition, thus affecting biofilm. 7
However, for edentulous patients, a complete denture (CD) remains as the main choice of treatment. Polymethyl methacrylate (PMMA) resin has been effectively utilized in the production of CDs and is still acknowledged as the most esteemed benchmark. The benefits of utilizing common PMMA resins include their convenient handling and manipulation.3,8
Biofilm contains various types of microbial cells that attach to surfaces. These microbial cells create a micro-ecosystem where they release byproducts (such as acids) that might compromise the integrity of the surface. The microorganisms involved in dental biofilm include Streptococcus mutans, Candida albicans, Staphylococcus aureus, Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, and others.9,10 Biofilm on tissues and dentures causes denture stomatitis, a multifactorial oral pathological condition along with erythema, bleeding, and halitosis. 11
Maintenance of denture hygiene involves mechanical, chemical, or combined approaches. Mechanical cleaning by brushing can reduce denture biofilm in a quick and cost-effective manner, but inadequate brushing can erode the denture base material and generate surface flaws that facilitate bacterial colonization and pigmentation. Meanwhile, the chemical procedure entails the immersion of the denture in denture cleansers or mouthwash for a certain duration of time to eliminate food particles and bacteria without affecting the mechanical and physical properties of the denture.12,13
Mouthwashes are typically used for cleaning dentures. This is a beneficial practice for achieving and maintaining optimal oral hygiene. 14 Mouthwash has a higher usage frequency compared to denture tablets. 15
Chemically, cellulose-based biopolymers created by deacetylating chitin make up the abundant cationic polymer known as chitosan. It has a number of appealing qualities, including hydrophilicity, biocompatibility, non-toxicity, and a wide range of antimicrobial activities for gram-positive and gram-negative bacteria as well as fungi.16,17 Chitosan can be used to prevent and treat denture stomatitis due to its antifungal properties. 18 Chitosan has been identified as a promising denture cleanser due to its superior antifungal properties compared to commercial cleansers, and that it effectively controls plaque and gingival inflammation when used as a mouthwash; therefore, chitosan may serve as a substitute for mouthwash. 5
In the subalpine region of Jammu and Kashmir, Saussurea costus (Costaceae family) is utilized for treating rheumatism, various ailments, headaches, ulcers, cough, colds, and throat infections, while in Korea, it is commonly incorporated in traditional prescriptions for inflammatory diseases. 19 Its anticancer, antioxidant, anti-inflammatory, antifungal, and antibacterial properties have attracted the scientific community’s attention to investigate the underlying mechanisms of these properties.20,21 Researchers have demonstrated S. costus’s potential as a natural addition or alternative to traditional antibacterial medicines. 22
Chlorhexidine is widely used to prevent and eliminate plaque. A 0.2% concentration of chlorhexidine-based mouthwashes are typically prescribed as oral rinses for oncology patients rehabilitated with oral prostheses. 23 Substantial antibacterial action has been demonstrated at concentrations ranging from 0.2% to 4%. 24 In comparison to mechanical brushing and effervescent cleaning tablets, the 4% solution exhibits enhanced antimicrobial efficacy against Candida albicans and Streptococcus mutans on acrylic dentures and soft silicone linings. Chlorhexidine has been shown to have effective antibacterial properties and biofilm removal efficacy when used as a denture cleanser. 25 Although chlorhexidine is the most effective chemical agent for plaque management, its extended use can cause tooth discoloration and impair tongue functions like taste perception and sensitivity.26,27 Recent findings have led to an increased focus on naturally derived antimicrobial products as an alternate strategy for targeting cariogenic bacteria. These products are typically believed to have less adverse effects.
This study aims to investigate and compare the antimicrobial effect of chlorhexidine, chitosan, Saussurea costus, and chitosan-Saussurea costus as mouthwash and denture cleansers against the main resistant microorganisms responsible for oral diseases namely streptococcus mutanus, Escherichia. Coli, and Candida albicans and the effect of testing solutions on the roughness of the acrylic denture base material. In addition, the study also aims to examine the effect of these solutions on the viability of human gingival cells (hGFs). The null hypothesis proposes that the prepared solutions (1) would not substantially affect the antimicrobial and biocompatibility properties of mouthwash, and (2) would not affect microbial adherence and alter the surface roughness of the denture material as a denture cleanser.
Material and methods
Groups of the study
The study focuses on the antimicrobial effect and cell viability of five groups, which include distilled water (d.w as a negative control, Iraq), chlorhexidine (CHX as positive control, 0.2% chlorhexidine Gluconate mouthwash, Ecolab; UK), chitosan (CS) solution, S. costus (SC) solution, and chitosan-S. costus (CS + SC) solution. The G-Power software was used to determine the sample size with an effect size of 0.5, power of 0.80, and an alpha level of 0.05. It was determined that each group would contain 10 samples. The tests were performed at the Basic Sciences/ College of Dentistry/Mustansiriyah University. The operator was kept uninformed of the treatment solutions during the evaluation process to avoid conscious or unconscious bias.
Solution preparation
Chitosan solution
The solution was prepared by dissolving 2 gm of chitosan (Poly D-glucosamine) derived from shrimp shells with a particle size of 30–80 nm ⩾75%, (C3646, USA) in a 1% (v/v) solution of glacial acetic acid (Thomas Baker Chemicals Pvt. Ltd. India). The mixture was stirred using a magnetic stirrer at 50ºC for 12 h to dissolve the chitosan completely. The pH of the solution was maintained in the range of (5.6–5.8) by adding 10 M of sodium hydroxide NaOH (Hainan Yanghang Industrial Co., Ltd, China). The prepared solution was then stored at a cold temperature until use.18,28
Saussurea costus solution
Saussurea costus leaves were purchased from an herbal store in the local market in Baghdad, Iraq. These leaves were grounded into fine powder after being rinsed with distilled water and air-dried. The ground material was kept in sealed plastic boxes at room temperature of 25ºC until further examination. For the S. costus solution preparation, 2 gm of the prepared powder was mixed in 100 mL of distilled water. The solution was kept in the refrigerator until usage after heating and filtering.21,29
Chitosan-Saussurea costus solution
The Chitosan-Saussurea costus solution was prepared by mixing 10 mL of the previously prepared chitosan solution with 10 mL of the Saussurea costus solution. The mixture was left on the stirrer for about 30 min to ensure the homogenous mixture’s preparation.
Characterization of chitosan and Saussurea costus powder
The S. costus and chitosan powder were analyzed for particle size and morphology using the Field Emission Scanning Electron Microscope (FE-SEM, inspect f50 FE-SEM, The Netherlands). Moreover, the chemical bands were examined using Fourier Transform Infrared Spectroscopy (FTIR) analysis. An infrared absorption spectrum was produced using a Bruker Tensor-27 IR (Germany). The spectra created a profile of the sample, which is similar to a unique molecular fingerprint.
Polymethyl methacrylate disks preparation
150 polymethyl methacrylate (PMMA; Veracril, Colombia) disks with 10 mm diameter and 3 mm thickness were manufactured using a lost-wax technique following the manufacturer’s instructions. To achieve a consistent initial roughness, all the disks were sequentially finished with silicon carbide papers up to grit size 1000. 18 Next, the ruge wheel was used to polish the samples using pumice and water at 1500 rpm to prevent the samples from being distorted by excessive heat. The process continued until a glossy surface was attained (Figure 1). 30 Subsequently, all the disks were washed with distilled water for 1 min and stored in the same medium for 48 h to reduce residual monomer. The surface roughness was measured using a profilometer (TR220, Shandong Ipre Inspection Technology Co., Ltd., China) (Figure 2). The samples were then sterilized using ultraviolet radiation (UV KW-4AC (Chemat Technology Inc., USA). A surface profilometer quantified the test samples’ surfaces before immersion in denture cleaner solutions, and the values were used as pre-immersion control measurements. After immersion in the testing solutions and incubation, digital calipers were used to measure the PMMA disks’ inhibitory zones in millimeters, indicating the relative antimicrobial efficacy of the testing solutions against the tested microorganisms. A profilometer was used to evaluate the surface roughness after immersion. This method quantifies how cleansers affect denture surface topography.

PMMA disks after finishing and polishing process.

Profilometer apparatus to test the Ra value (μm) of the prepared disks after polishing.
In vitro MTT assay
Human gingival fibroblast cells (hGFs; PCS-201-018, ATCC, USA) were cultivated at 37°C in a humidified environment with 5% CO2 in Dulbecco’s Modified Eagle Medium (DMEM; Sigma-Aldrich, USA) enriched with 10% fetal bovine serum (FBS; 30-2020, ATCC, USA). hGFs were received after two passages and cultured until 70% confluence in 100-mm diameter culture dishes. Only cells with passage numbers <10 were used in this study. The cells were conducted on 96-well plates (Santa Cruze, Biotechnology, USA) and planted at 7000 cells/well. After 24 h, the merging monolayer was achieved. The cells were treated with the testing solutions at 300, 200, 100, 50, and 25 μg/mL concentrations. The cells’ viability was calculated 24 h after exposure by getting rid of the media, followed by adding 28 μL of the MTT solution at a concentration of around 2 mg/mL, and then incubating the cells at 37°C for 2.5 h. Following the removal of the MTT solution, the residual precipitates in the wells were solved using Dimethyl Sulphoxide (DMSO) at a quantity of about 130 μL and then incubated with shaking at 37°C for 15 min. 31
Antimicrobial test
Two types of bacterial and fungus strains were used in this study. These include E. coli (ATCC® 25922), S. mutans (ATCC® 35668), and Candida albicans (ATCC®11006). The microbial cultures were sub-cultured in Mueller Hinton agar (for Candida albicans and bacterial strains cultures). Using sterile swabs, 0.2 mL of the bacterial strain (1.5 × 108 CFU/mL) was evenly swabbed on agar plates. To evaluate the antimicrobial efficacy of the testing solutions as mouthwash, a sterile metal cork borer was used to make five uniformly spaced wells (about 4 mm in diameter) per plate on the surface of the culture agar. Each well was filled with 0.2 mL of solution of each group under aseptic conditions and kept at room temperature for 1 h. For the antimicrobial test of the solutions as denture cleanser, acrylic disks were immersed in the solutions for 20 min, 22 and the soaked disks were placed in the prepared wells in the culture agar and stored at room temperature for 1 h. The plates were incubated for 24 h at 37°C. The clear area around the wells was identified as inhibition zones, which were then evaluated.
Measurement of surface roughness
The PMMA disks were subjected to daily soaking in disinfection solutions and distilled water for 20 min at room temperature. After immersion, the samples were meticulously rinsed with water and preserved in water at ambient temperature. The immersion process was reiterated for 10 days. The surface roughness was evaluated following the 10-day immersion period utilizing a surface profilometer. The roughness parameter, Ra, was measured and reported as mean values accompanied by standard deviations.
Ethical approval
This study was reviewed and approved by the Mustansiriyah University College of Dentistry Research Ethics Committee (MUCOD, No. REC127) on 1 May 2023.
Statistical analysis
Statistical Package for Social Sciences (SPSS) version 25 and Microsoft Excel were used for results analysis. Descriptive and Inferential statistics (one-way ANOVA) were used to accept or reject the statistical hypotheses. The Bonferroni test was used to compare the significance difference between the means of the testing groups.
Results
The FE-SEM micrographs for the nano-chitosan and S. costus powder are presented in Figure 3(a) and (b). The images show that the nano-chitosan particles primarily have a spherical appearance, whereas the S. costus particles have an irregular morphology.

FE-SEM images of: (a) nano-chitosan powder, (a*) particle size distribution chart of chitosan, (b) S. costus powder, and (b*) particle size distribution chart of S. costus.
Characteristic bands were observed in the nano-CS powder. The wide peak at 3450 cm−1 is assigned to the stretching vibration of the O–H group; the peak at 3299 cm−1 is associated with the stretching vibration of the N–H group, while the peak at 2898 cm−1 is attributed to the stretching of the C–H group. The peaks at 1655, 1560, and 1310 cm−1 represent the amide I band, amide II band, and amide III band, respectively. The peak at 1410 cm−1 corresponds to CH2 bending. Moreover, the peak at 1150 cm−1 is related to the vibration stretching of the C–O–C bond. As seen in Figure 4(a), the spectra produced by dissolving chitosan in 1% acetic acid did not exhibit variations in the distinctive peaks. 32 The FTIR spectrum of S. costus, as shown in Figure 4(b) exhibited absorption peaks at 3408, 2359, 1602, and 896 cm−1. The prominent peak at 3408 cm−1 corresponds to the O–H bond of phenolic functional groups; the peak at 2359 cm−1 arises from the presence of C=C, and the peak at 1602 cm−1 is attributed to (C=O). 33
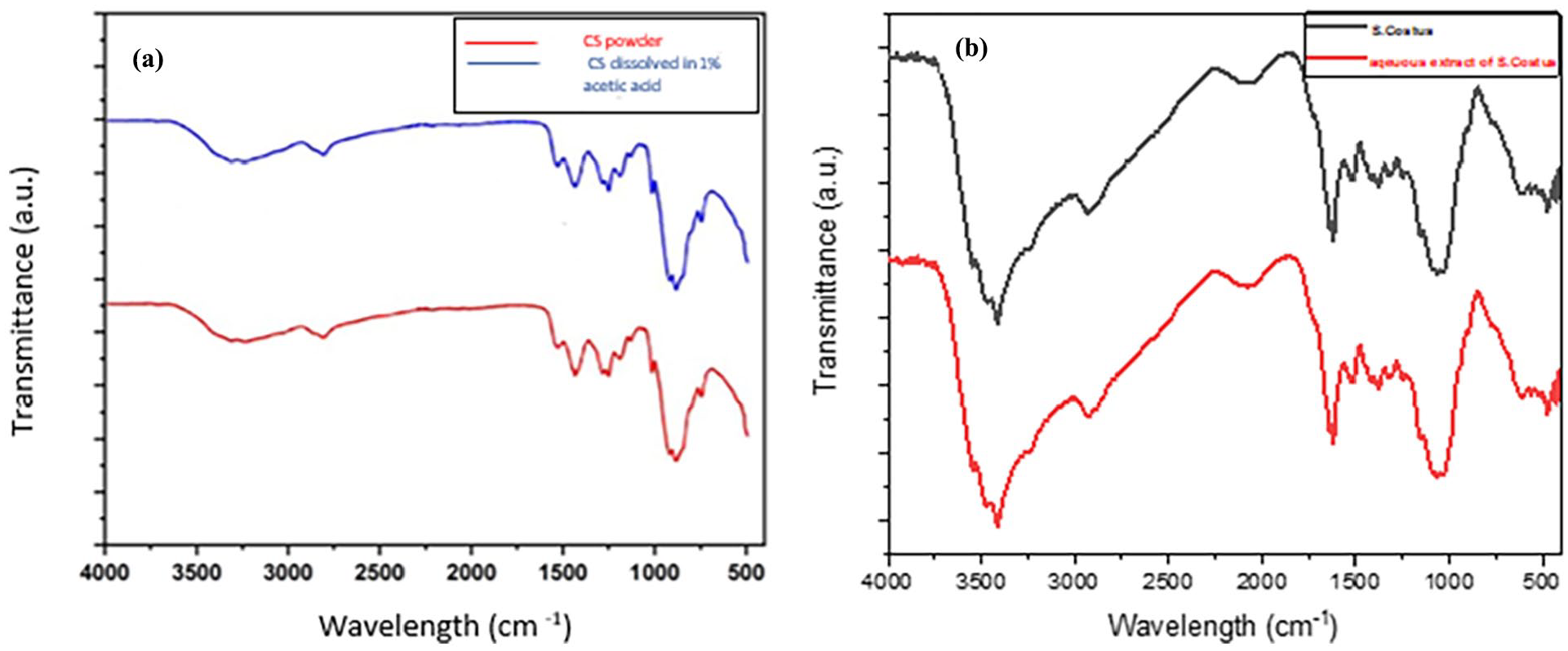
FTIR spectrum of: (a) nanochitosan powder and chitosan solution and (b) S. costus and S. costus solution.
Cell viability was assessed by measuring the formation of formazan crystals resulting from the reduction of MTT tetrazolium by living cells. The data in Figures 5 and 6 illustrate the dose-response viability of the hGFs when exposed to various concentrations of CS, SC, CS + S. costus mixture solutions, distilled water (the negative control), and chlorhexidine (the positive control). This experiment aimed to determine the IC50, which represents the concentration at which 50% of cell growth is inhibited, using the MTT assay. The data presented in the Tables 1 and 2 are expressed as means and standard deviations. The herbal solutions with concentrations of 300, 200, 100, 50, and 25 exhibited reduced cytotoxicity towards the cells. An increase in the concentration of the herbal-prepared solutions resulted in a higher level of cytotoxicity towards the hGF cells. Furthermore, it was observed that the cell survival of both the test and control groups is higher at concentrations of 100, 50, and 25 µg/mL compared to concentrations of 300 and 200 µg/mL. This finding suggests that the cytotoxic effect of the substance depends on the dose. Furthermore, the IC value of the assay conducted on the herbal solutions (including chitosan, S. costus, and a mixture of chitosan-S. costus solutions) exhibited higher values compared to a 0.2% chlorhexidine solution (190.9, 178.2, 225.7, and 97.6 μg/mL, respectively).

The effect of different concentrations of −ve, +ve control, and prepared solutions on cell viability, according to MTT assay. The box plot reports the cell viability of different solutions at: (a) 300 µg/mL, (b) 200 µg/mL, (c) 100 µg/mL, (d) 50 µg/mL, and (e) 25 µg/mL.

Comparison of the effect of different solutions with different concentrations on cell viability.
Descriptive statistics of cell viability of solution at different concentration by MTT assay.

One way ANOVA of cell viability of all concentrations.

The negative control group (distilled water) shows no inhibition zone, which means that the water has no antibacterial activity. The CS, SC, and CS + SC mixture solutions showed antibacterial activity comparable to and higher than chlorohexidine for both solutions and PMMA immersed in the solutions. According to the ANOVA results, a significant difference in the antibacterial activity of the solutions against the S. mutans, E. coli, and Candida albicans microbial cultures was found between the various groups, as shown in Tables 3 and 4 and Figures 7 and 8.
Descriptive statistics of inhibition zones against microbial strains; ANOVA summery for −ve control, +ve control, and experimental groups.

Descriptive statistics of inhibition zones around the PMMA disks immersed in different solutions against microbial strains.


Inhibition zones formed around mouthwashes and denture cleansers against: (a) S. mutanus and (b) E. coli, and (c) Candida albicans.

The box plot reports the effect of −ve, +ve, and prepared solutions on inhibition zone (mm) against S. mutanus bacteria: (a1) inhibition zone formed around solutions used as mouthwash and (a2) inhibition zone formed around PMMA disks after immersion in the testing solutions); E. coli (b1) inhibition zone formed around solutions used as mouthwash and (b2) inhibition zone formed around PMMA disks immersed in the testing solutions) and Candida (c1) inhibition zone formed around solutions used as mouthwash and (c2) inhibition zone formed around PMMA disks immersed in the testing solutions).
The surface roughness assessments indicated no substantial changes in the denture materials following immersion in the cleansing agents. All the groups, including the control, demonstrated comparable roughness values ranging from 0.275 to 0.289 microns, as displayed in Table 5 and Figure 9.
Descriptive statistics of roughness values of the PMMA disks immersed in different solutions compared to the control group (before immersing).


The box plot reports the roughness (µm) values of PMMA after immersing in cleansing solutions compared to control (before immersing).
Discussion
This study examined the formulation of herbal products that would have the same impact as common products. The cell viability of the prepared solutions was investigated and compared with chlorhexidine; their effect against microorganisms when used as mouthwash was examined. The suitability of these solutions as a denture cleaner was also evaluated.
Denture stomatitis, gingivitis, and caries are recognized to have multiple factors, one of which is inadequate denture maintenance and poor oral hygiene. 34
The MTT assay is commonly employed as a standard method for assessing the cytotoxicity of different compounds across a range of conditions or concentrations. In this work, human gingival fibroblasts (hGFs) were considered essential due to their significant role in the periodontal connective tissue and their contribution to wound healing. 35 The determination of the IC50 value (50% half inhibitory concentration) of relevant treatments can be achieved by comparing the rate of cell viability of the control group and the other solutions. Determination of the half-maximal (50%) inhibitory concentration (IC50) is essential for understanding the pharmacological and biological characteristics of the solutions, drugs, and materials. 36
As a result, the present study conducted an in vitro experiment to evaluate the prepared solutions’ cytotoxicity at different concentrations compared to a 0.2% chlorhexidine gluconate solution. The study observed a dose-dependent relationship between the prepared solutions and their impact on the human gingival fibroblast (hGF) cells after 24 h. This indicates that lower doses of herbal-prepared solutions have a limited effect on the viability of these cells. Conversely, exposure to herbal solutions at the IC50 dosage resulted in a significant level of cell viability, exceeding that of the 0.2% chlorhexidine gluconate. The results indicate that increased concentrations of herbal solutions may lead to toxicity when exceeding a concentration of 300 µg/mL. This finding agrees with a previous study that investigated the cytotoxicity of Annona squamosa pulp extract as a mouthwash through in vitro investigation. 37
There has been significant research interest on plant extracts with high quality antibacterial activities. Extensive studies have been conducted on numerous phytochemicals, focusing particularly on the antimicrobial effects exhibited by several plant species. Various herbs, including cinnamon, garlic, basil, curry, ginger, sage, curcumin, thyme, mustard, and others, have been found to possess antibacterial properties, as demonstrated by their crude extracts. These plants also possess a bactericidal effect against many bacteria. 38 The findings of this investigation demonstrate that the zone inhibition of E. coli, S. mutans, and Candida albicans exhibited an increase when exposed to a mixture of chitosan and S. coutus, as compared to the individual solutions of each material. These results suggest the presence of a synergistic impact between the materials (see Tables 3 and 4). Numerous studies have been conducted on plant extracts to investigate their antibacterial properties and to discover new antimicrobial compounds, all of which have yielded positive outcomes. In addition, we conducted a comparison between the antimicrobial properties of CS and SC and a 0.2% chlorhexidine solution, which is widely utilized as an antiseptic mouthwash. Chlorhexidine’s antibacterial activity stems from its capacity to adhere to the negatively charged microbial cell wall, leading to the liberation of cellular components. Chlorhexidine is recommended as a denture cleansing solution, although excessive use may cause stains on acrylic resins. 39 It is also worth noting that patients may develop an allergic reaction to chlorhexidine, a phenomenon that has been occurring more frequently, and can result in a variety of symptoms ranging from moderate to life-threatening.18,40,41
Chitosan has attracted significant research interest in different fields. Chitosan possesses several qualities, including antimicrobial, antioxidant, biocompatible wound healing, and mucoadhesive characteristics.42,43 It exhibits anti-adherence properties that result in modifications to bacterial surfaces, changes in the expression levels of bacterial surface ligands, and adsorption to hydroxyapatite crystals on the tooth surface. The aforementioned qualities are accountable for chitosan’s bactericidal and bacteriostatic properties. 5
The antibacterial efficacy of chitosan is dependent upon its degree of deacetylation (DD), molecular weight (MW), and acidity level (pH). The utilization of high-DD chitosan implies that this particular chitosan derivative possesses a substantial quantity of cationic charges, enabling effective interaction with the anionic components of lipopolysaccharides found on the membranes of bacterial cells. The cellular membrane is disrupted through electrostatic interaction between chitosan and lipopolysaccharide. The inhibitory effect of high-molecular-weight chitosan on the cellular internalization of chitosan molecules is particularly pronounced in Gram-negative bacteria, characterized by their double-membrane structure. 44 Chitosan has been reported to disrupt mRNA synthesis and protein incorporation potentiality. Furthermore, it possesses anti-inflammatory characteristics due to its impact on the levels of prostaglandin E2. 45 Hasriati et al., 46 demonstrated that chitosan possesses sufficient antibacterial activity to decrease the total bacteria count peri-miniscrew and its efficacy does not substantially differ from that of chlorhexidine.
Our study showed that all the solutions except distilled water formed an inhibition zone when used as a mouthwash or denture cleanser. The mean inhibition zone in the chitosan group was higher than that of the chlorhexidine group when the inhibition zones of the experimental groups were compared. This suggests that the antimicrobial activity of chitosan was superior to that of the chlorhexidine solution. This result is consistent with the findings of Manvi et al., 47 which demonstrated that the chitosan solution was more effective than commercial denture cleanser solutions. The findings of Srimaneepong et al. 18 proposed that chitosan solutions could be used as an inexpensive and safe denture cleaning solution. Our findings are also consistent with that of Costa et al., 48 who asserted that chitosan is effective against the preponderance of bacteria and can be used as an alternative to conventional mouthwashes.
The phytochemical analysis of an extract from the Saussurea costus identified 48 chemical substances, each demonstrating distinct mass spectral fragmentation characteristics. Fatty acids represent the largest proportion of the phytochemicals at 73.8%. Tannins at 8.2%, followed by carbohydrates at 6.9%, terpenoids at approximately 4.3%, carboxylic acids at 2.5%, hydrocarbons at 2.4%, and sterols at about 1.5%. Phenolic compounds represent the smallest proportion of the phytochemicals at 0.2%. 49 Research indicates that tannins, terpenoids, and phenolic compounds possess antimicrobial capability. 50 Reports have also indicated that fatty acids serve as self-defense agents in organisms and exhibit a range of biological actions. 51 Carbohydrates have not been shown to possess therapeutic effects independently; however, they may enhance the effectiveness of essential therapeutic components. 52 Sterol molecules have been demonstrated as cancer inhibitors, anti-inflammatory agents, and anti-viral agents. 53 Our findings align with that of Idriss et al. 49 which indicated that the antimicrobial effect of the aqueous extract of Saussurea costus demonstrated no activity in an inhibitory zone of less than 7.0 mm.
Previous studies have documented the antibacterial properties exhibited by S. costus. 21 In a study conducted by Al-Zawawi et al., 54 a randomized controlled trial (RCT) was conducted to examine the effectiveness of herbal-based oral rinses in reducing post-operative inflammation following non-surgical instrumentation. The study specifically focused on patients with a known allergy to chlorhexidine, aiming to explore alternative therapeutic options for periodontal inflammation. The findings indicated that herbal-based mouth rinses can be considered viable alternatives to chlorhexidine for post-operative prescriptions after periodontal therapy. According to Al-Zawawi et al., 54 mouthwashes derived from S. costus have been proposed as a potential alternative to chlorhexidine for treating periodontal inflammatory diseases. In this study, the S. costus solution demonstrated the formation of an inhibition zone in denture cleanser and mouthwash groups comparable to chlorhexidine. This finding aligns with the results of Deabes et al., 55 which indicated that S. costus extract may significantly contribute to defense mechanisms against human multi-resistant bacteria and could serve as an alternative to antibiotics for treating specific diseases. Additionally, it aligns with the findings of Soliman et al. 56 which highlighted the efficacy of S. costus phytochemicals in managing various C. albicans species.
The surface roughness of dentures greatly affects microbial colonization. Microbial accumulation is facilitated by the surface imperfections of the denture, even on a clean prosthetic. A roughness threshold of 0.2 µm has been proposed to prevent biofilm formation on hard and artificial dental surfaces. 57 In this study, the roughness values of the PMMA material groups before and after immersion in cleansing solutions were higher than the threshold point. The PMMA material groups displayed a non-significant increase in roughness following immersion. According to Alfouzan et al., 2 Gajwani-Jain et al., 58 and Carvalho et al. 59 reported that, when compared to various chemical cleansers, PMMA samples submerged in chlorhexidine caused the least increase in surface roughness. On the other hand, in chlorhexidine-immersed specimens, Jariyanurut et al. 60 demonstrated that the chlorhexidine with higher ionic concentration resulted in a greater release of soluble components, such as plasticizers, that interact with the cross-linking agent (ethylene glycol dimethacrylate) in the acrylic resin, thus increasing surface roughness.
A meta-analysis showed that natural components have no significant impact on the surface roughness of acrylic resin. 61 Jariyanurut et al. 60 stated that denture cleansers containing chitosan caused fewer surface roughness changes than other groups, which was attributed to the formation of stronger bonds between the −NH2 groups of chitosan and the substances in PMMA which enhance the formation of more stable polymer matrices.
The utilization of medicinal plants for treating and preventing oral problems can offer occasional benefits to rural populations or those in poor socioeconomic conditions owing to the affordability and accessibility of herbs in various parts of the country. Consequently, medicinal herbs may serve as a feasible substitute for traditional antimicrobials, in addition to avoiding the side effects of chemical agents found in some current medications. 62 However, the current study was conducted in vitro. The study’s limitations include the short period for assessing the roughness value of PMMA. Additionally, this investigation does not highlight the effect of various denture immersion durations. The antibacterial properties of saliva may play a role in reducing microbial adherence, a factor not addressed in the current investigation. Consequently, the results do not accurately reflect the clinical conditions of various biofilm microorganisms. Future studies can be conducted by studying the effect of time and the effect of these prepared natural solutions on the color of natural and artificial teeth as well as the mechanical and physical properties of dentures fabricated through conventional methods or 3D printing and CAD/CAM milling. The effect of an acquired salivary pellicle on microbial adhesion is another direction for future studies. The assessed parameters must be examined in an in vivo model. This is in addition to exploring the effect of the prepared solutions on the bacteria and microbes isolated from the mouths of patients of various ages, genders, and medical health.
Conclusion
A new denture cleaning and mouthwash agent incorporating the natural ingredients of chitosan and Saussurea costus showed effective antibacterial and antibiofilm actions without toxicity. Its efficacy is similar to that of commercial mouthwash, taking into account the limitations. Compared to commercially available herbal mouthwash and denture cleaning, this combination is more affordable and widely available.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
