Abstract
Introduction:
Paper is the most important material of the printing industry and is being improved due to the increasing needs of industry. The most important process to improve the optical and physical properties of paper is the surface coating. Paper has a smoother and opaquer surface with surface coating. In addition, brightness, whiteness, and yellowness values are improved with surface coating. Ultraviolet (UV) light in sunlight causes changes in the structure of the paper and coating chemicals and accordingly causes yellowing. Para-amino benzoic acid (PABA), due to its chemical structure, is a UV-blocking agent used in sunscreen creams.
Methods:
The effect of PABA on paper gloss, yellowness, and lightfastness values was investigated by adding it into the surface coating formulation. For this purpose, coating formulations were prepared with cationic starch and 3%, 5%, and 7% PABA; coatings were applied to the paper according to laboratory condition standards. Coated papers were printed with an IGT-C1 lab-type offset printability device with magenta ink. The printed papers were then exposed to a light fastness test with Solarbox. The color, brightness, and yellowness values of all samples before and after lightfastness were measured with an X-Rite spectrophotometer. The gloss of coated and printed paper sample values was measured with a BYK-Gardner glossmeter.
Results:
The gloss values of paper to which PABA was added, increased; however, it was determined that these gloss values decreased after the lightfastness test.
Conclusions:
PABA-added cationic starch coatings are more affected by UV light than cationic starch coatings.
Introduction
The surface coating process, applied to paper, the most commonly used material in the printing sector, is undertaken to meet higher expectations. The basis of the surface-coating process is the coating, which uses a formula prepared with pigment, binding agent, and a number of additive agents. At the end of the coating process, the paper becomes more effective in terms of its optical and physical properties and shows better printability characteristics. 1 These properties are usually printed or unprinted brightness, surface smoothness, whiteness, and opacity.2,3
Light fastness or resistance is also important for the quality of the printed sample, and the conservation and maintenance of its function. The main reason for paper discoloration is UV light from daylight. This is especially the case for outdoor prints. Light fastness refers to the resistance of a printed product or ink to discoloration. Light source is not the only reason for discoloration: light intensity, exposure time, angle, the surface properties of paper, humidity, climatic conditions, pigment properties, etc. also cause discoloration. Accelerated light-aging tests are available in accordance with certain standards to determine light fastness. By this means, it is possible to determine in advance whether an ink is resistant or not.4–6
Nowadays, the paper industry uses starch in very large amounts and in different ways either in paper production or as a surface coating material. The starch generally functions as a binding agent in the formula. It is the most commonly used binder type when compared with the alternatives such as natural or synthetic polymers and latexes.7,8 One of the reasons for its widespread use is that it is easily accessible and low-priced. 9 The purpose of using starch in paper or in a surface coating includes to provide resistance to the penetration of liquids, to obtain better surface properties, and to improve specific physical properties such as stretch, strain, and tear resistance.10,11 Ground, primed starch, enzyme-converted starch, ammonium persulfate-oxidized starch, starch ether, and cationic starch are some of the starch types used in the paper industry.12,13 In recent years, the use of cationic starch has become more prevalent than other types. Cationic starch has a number of environmental advantages. Studies have shown that the use of cationic starch reduces biochemical oxygen levels, or the waste load, by half.14–16 Paper producers and even corrugated cardboard producers use starch in their processes to increase physical resistance, and due to the acceleration of pigment deterioration in the recycling process.17–18 It may be possible to increase resistance to light by adding chemicals into the coating formula during its preparation. Here, UV filters come into play. Para-amino benzoic acid (PABA) is a type of solar filter commonly used in sunscreens (Figure 1). The material is in the form of a white-yellow solid crystal powder. Despite realizing only 1% dissolution in water, it is very soluble in alcohol and ether: 4-Amino benzoic acid is used as a UVB (280–320 nm) filter in sunscreen products. It distributes UV light from the sun to its form by means of four conjugated double bonds, oxygen, and non-bonding double electrons in nitrogen in its chemical structure and, thus, is used as a UV protector (Figure 1).
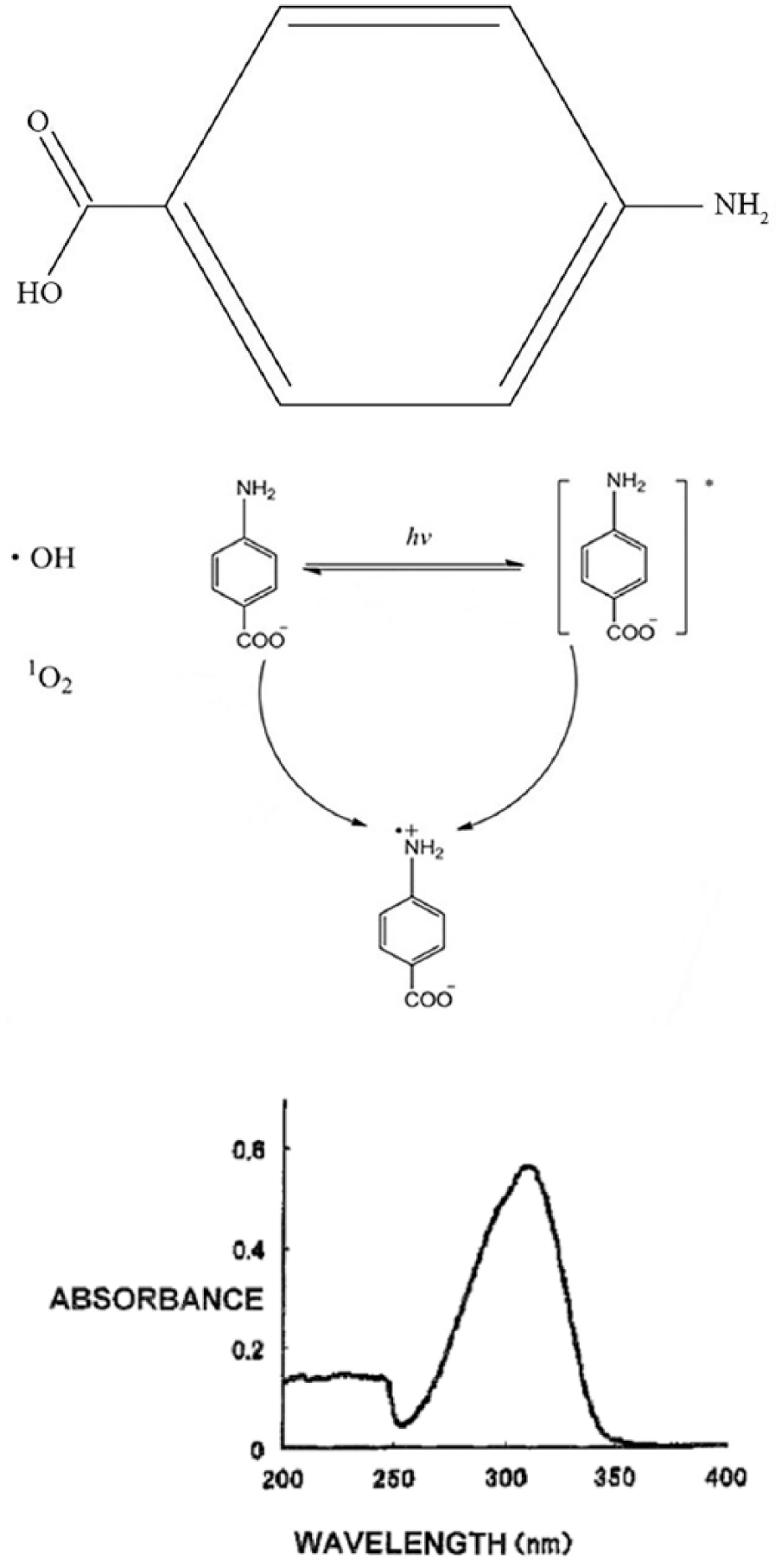
The chemical formula of para-amino benzoic acid (PABA), and its circuit with UV and the UV spectrum.
Methods
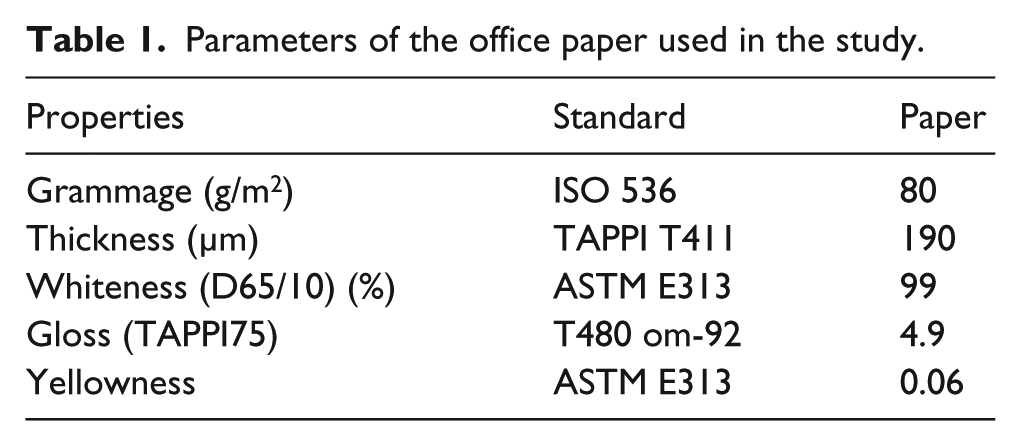
In the study conducted, cationic starch, one of the most frequently used binders, and white office paper were used in paper coatings. The properties of the office paper used in the study are given in Table 1. The formulations were prepared by adding 4-Amino benzoic acid (PABA) at concentrations of 3% (F1), 5% (F2), and 7% (F3) into cationic starch to understand its effect on the optical and physical properties of the paper. Coatings were applied under laboratory conditions with a laboratory-type automatic coating machine.
Parameters of the office paper used in the study.
Unprinted papers were measured for yellowness according to standard ASTM E313. Printing was performed using DYOPAN LR-5600 Magenta commercial offset printing inks with an IGT C1 Laboratory-Type Offset Printability Tester under the pressure conditions of 400 N/m2 at a standard density value on prepared coatings. Print results were measured with CIE L*a*b* values using an X-Rite eXact handheld spectrophotometer according to ISO 12647-2:2013. Color difference ΔE values were measured according to CIE ΔE* 1976 ISO 13655 (spectral range between 400 nm and 700 nm, D50 light source, 2° observer, polarization filter and 0:45-degree geometry). The color change formula is given below. Calculations were made by taking the average of three measurements 19 :

Gloss measurements were performed using a BYK-Gardner GmbH glossmeter according to the ISO 2813: 2014 standard. Afterwards, printed and coated papers were subjected to a lightfastness test in accordance with BS 4321:1969. 20 After the lightfastness test, CIE L*a*b*, gloss, and yellowness measurements were again performed on the samples.
First of all, for the lightfastness process, the CIE L*a*b* color and gloss values of the samples were measured. The printed and unprinted samples were then subjected to lightfastness tests in a sealed cabin—a device named Solarbox 1500—under constant UV light for 192 hours according to BS 4321-1969. The CIE L*a*b* color and gloss values of the printed and unprinted samples that were subjected to discoloration were measured again, and the difference between the initial and final readings was calculated. Color changes were evaluated according to the Blue Wool scale.

Results
1. After coatings were applied to the white paper, the color measurement, ΔE, did not change much when cationic starch coated paper color didn’t change remarkable, whereas, in the PABA-added coatings, an increase in the ΔE color difference in the direction of yellowing was observed as the concentration of the PABA ratio increased (Figure 2).
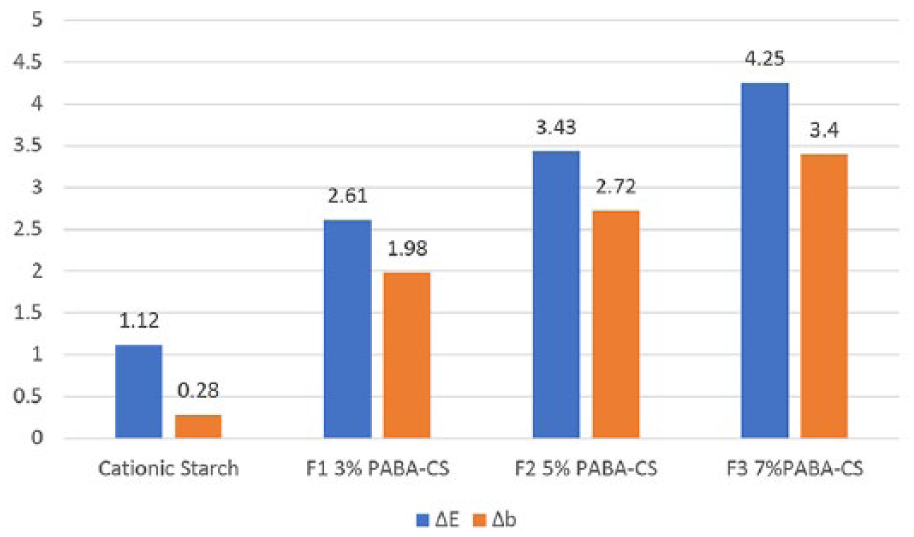
Color measurements of coatings on unprinted white paper (ΔE, Δb).
2. The cationic starch coating was not very effective in lightfastness (ΔEwhitepaper: 8,68; ΔEcationicstarch: 8,8).
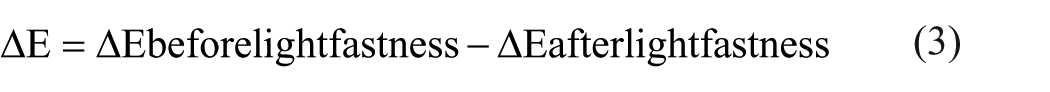
3. As the concentration of 4-Amino benzoic acid (PABA) increased in coatings, the color difference, ΔE, on unprinted paper increased (Figure 3).
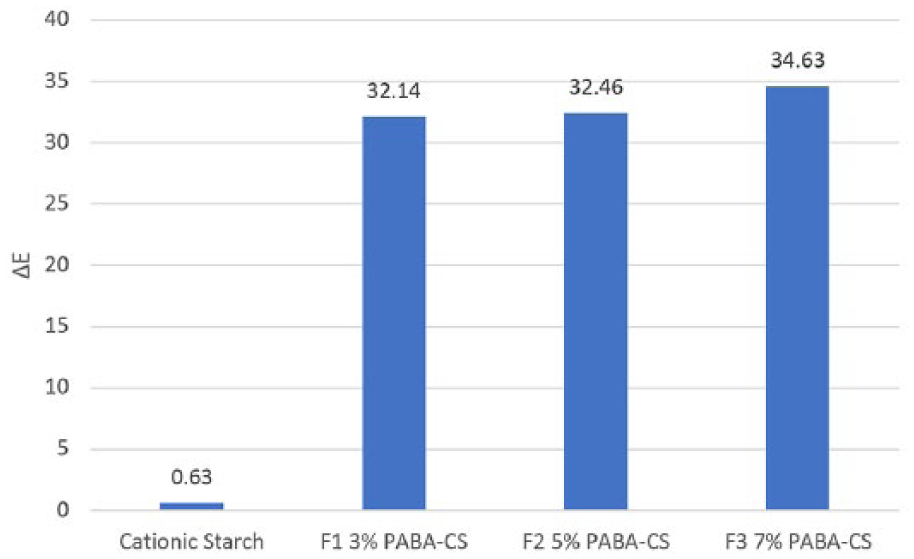
ΔE color difference after lightfastness for unprinted papers.
4. It was determined that the color difference, ΔE, decreased by approximately 2 after the lightfastness test on the prints made on the cationic starch-coated paper. In other words, less discoloration occurred on cationic starch-coated paper. This is due to the fact that the pigment remains on the surface of the cationic starch-coated paper because it cannot penetrate into the paper fibers, therefore the effect of the paper’s own discoloration on the total ΔE change is small (ΔEuncoatedpaper: 5,29; ΔEcationicstarch: 3,40).
5. After printing on paper that is coated with 4-amino benzoic acid (PABA)-added starch, the ΔE color difference increased as the PABA concentration increased as a result of lightfastness (Figure 4).
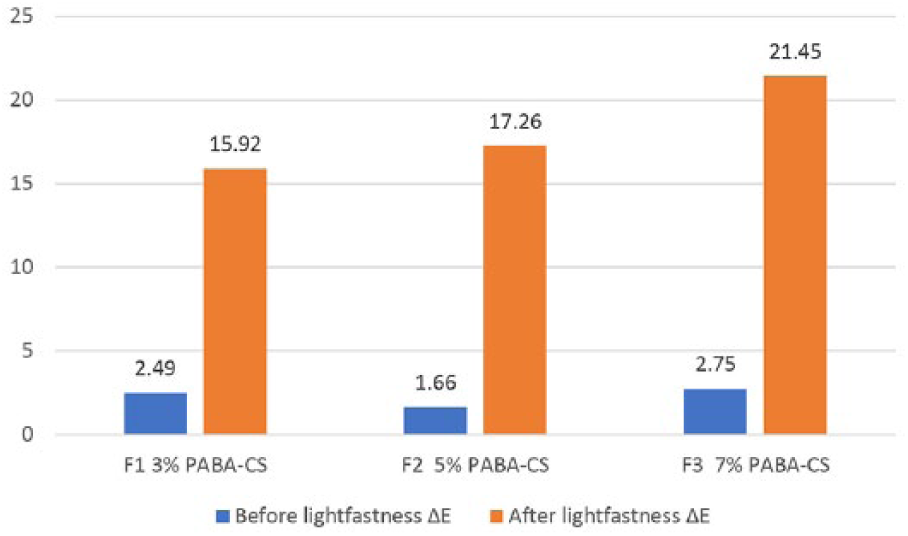
ΔY yellowness difference according to the lightfastness test after printing on paper coated with 4-amino benzoic acid (PABA)-added starch.
6. Yellowness increased on paper coated with 4-amino benzoic acid (PABA)-added starch after the lightfastness test according to the measurements of unprinted paper (Figure 5).
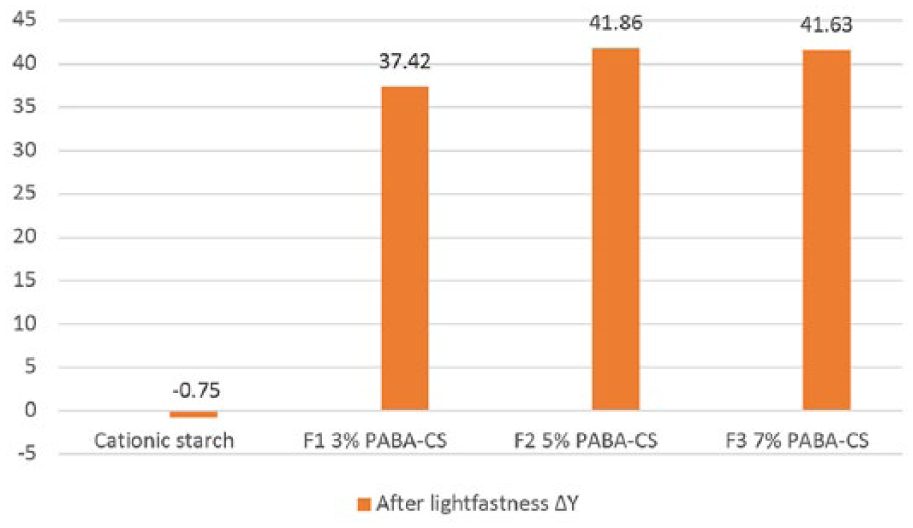
ΔE color difference before and after the lightfastness test after printing on paper that is coated with 4-amino benzoic acid (PABA)-added starch.
7. When printed on paper with PABA, the color difference, ΔE, was approximately three-fold. This was due to the fact that PABA added its own color to the ink.
8. The color differentiation and yellowing on the paper with the PABA-added coating is due to the cross-linking of PABA with its paired electrons to the cationic starch polymer. As a result, the paired electrons in PABA are affected by UV light in lightfastness and become chromophore group, and therefore, color change increases and color turns to yellow in PABA with the most paired e-.
In the brightness measurements made with unprinted samples, it was observed that the cationic starch coating increased the brightness, and maintained this increase when 3% and 5% PABA was added; however, the brightness decreased when 7% PABA was added (Figure 6). After the lightfastness test, brightness levels differed. Brightness tended to increase again in the sample with cationic starch, while it decreased in the PABA-added samples (Figure 7). In the brightness measurements taken on the printed samples, it was determined that the samples with cationic starch and PABA had high brightness, but the 7% PABA sample had lower brightness than the others. A similar tendency was found after the lightfastness test (Figure 7).
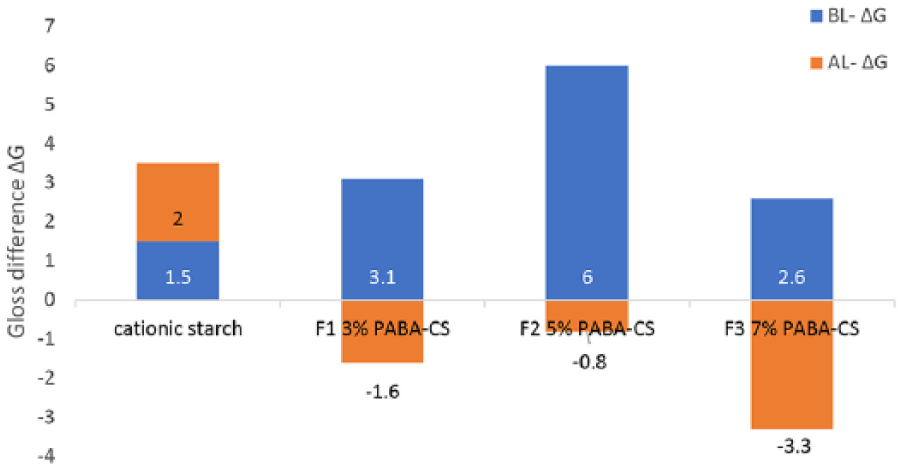
Gloss change ΔG before and after the lightfastness test on printed samples.
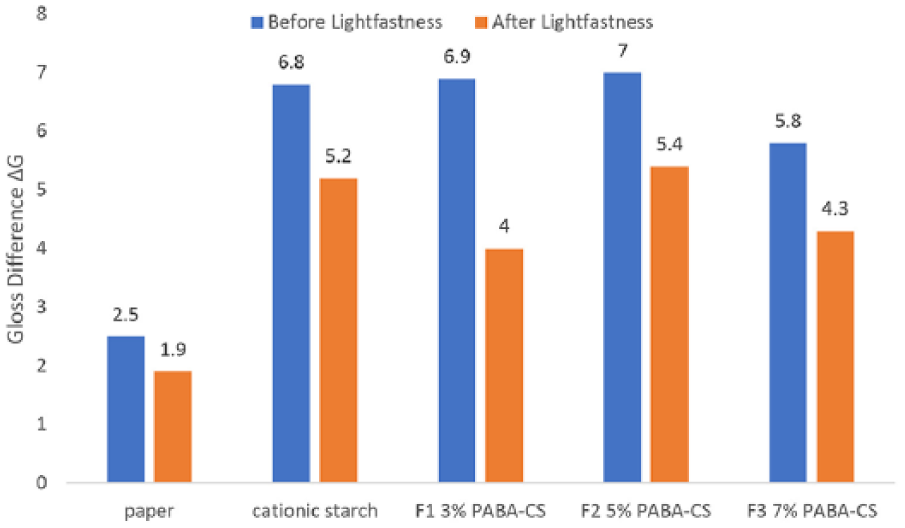
Gloss change ΔG before and after the lightfastness test on printed samples.
Conclusions
Yellowing occurs due to the fact that the paired electrons are greatly affected by UV because of the chemical structure of the 4-amino benzoic acid (PABA), which is planned to be used for the inhibition of lightfastness. Therefore, it is not suitable for the use on paper to be printed.
When the brightness results were examined, it was seen that the brightness of paper with 5% PABA among unprinted papers increased 2-fold, but as a result of lightfastness, this ratio decreased to 1.5-fold.
The highest brightness value in unprinted papers was seen in 3-5% PABA-added papers.
Brightness decreases in PABA-added paper as a result of lightfastness.
The highest brightness value was also seen in 3-5% PABA-added papers among printed papers.
The brightness value decreases after lightfastness test in all printed papers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
