Abstract
Background:
Surface-enhanced Raman scattering (SERS) has become a useful spectroscopic tool for studying biomolecule structures. The main types of plasmonic substrates used in biological systems are Au nanoparticles (AuNPs), whose surface plasmon resonance depends on the nanoparticle size, morphology, particle interspace, and so on.
Methods:
In this study, AuNP colloids with different sizes were synthesized and used as the sensors to probe SERS signals of different biomarkers and biomolecules.
Results:
The results showed that an AuNP colloid of ~50 nm had excellent SERS effects in probing various molecules, and could be preserved for about 3 months with excellent repeatability and reproducibility (RSD <5%) in terms of the probed signal intensity (rhodamine 6G and crystal violet). Meanwhile, the fabricated AuNPs were applied to study the SERS signals and structural information of bovine serum albumin (BSA) in aqueous solution. It was found that SERS could rapidly provide the structural information and vibration characteristics of BSA.
Conclusion:
It was concluded that biocompatible AuNP colloid may be a promising biosensor in the rapid and label-free detection of biological systems.
Introduction
Raman spectroscopy is a vibrational spectroscopic technique based on the inelastic scattering to reflect the molecular structures and changes of samples, and can be considered as the molecular “fingerprint” of specific molecules.1-3 However, the original Raman signals are generally very weak and only about 10-10 times of the intensity of the incident laser. 4 Fortunately, the Raman effects can be amplified by approximately 1014 times when molecules are attached to the surfaces of noble metal nanoparticles; that is, the so-called surface-enhanced Raman scattering (SERS). 5 In view of its good sensitivity, SERS has been widely used in physics, chemistry, material science, medicine, and other fields. The enhancement effect of SERS depends on the enhancement substrate, and various types of substrates have been reported, including solid substrate, liquid colloid (Au, Ag, Cu, etc.), immuno-tags, and so on. 4 Among these, the nano-sized gold colloid has been applied extensively for its merits of excellent enhancement effects, convenient synthesis, biocompatibility, easy operation, and desirable economy. At present, it is widely accepted that electromagnetic enhancement and chemical enhancement are responsible for the SERS effects. It has been reported that the size, shape, morphology, and interspacing of nanoparticles that play key roles in local surface plasmon resonance (LSPR) contribute a lot to the electromagnetic enhancement. 6 In this study, Au nanoparticle (AuNP) colloids of different sizes were synthesized and their SERS effects were compared by probing different biomarkers.
As the basic components and functional units of organisms, proteins are involved in all metabolic processes of living cells and tissues. It has been confirmed that SERS has great potential in high-throughput biomolecule detection, especially in related studies of proteins. 7 Currently, SERS-based protein exploration methods mainly include label-free direct detection and tag indirect detection. The label-free method can obtain the internal amino acid skeleton information of proteins more directly by simply attaching them to the enhanced substrates. By contrast, tag indirect detection mainly depends on the functionalized decoration of nanoparticles to provide the SERS signals of the interaction between the probed protein and its ligands. 8 Although tag-labeled detection has developed more rapidly in recent years, it is unavoidably complex for extensive applications. To date, the label-free method still plays a dominant role in SERS tests due to its simple operation, good sensitivity, and excellent robustness. The most popular noble metal nanoparticles used in label-free detection include AuNPs, Ag nanoparticles, and Au/Ag particles. However, it had been certificated that Ag nanoparticles have a significant impact on the protein structure and metabolic processes of purine. 9 Hence, in this study, AuNPs were fabricated and applied to explore the structural information of bovine serum albumin (BSA) by the SERS method. Our results indicate that the appropriate AuNPs may be a promising biosensor for the label-free SERS detection in biological and biomedical systems.
Experimental section
Materials
Reagents used in this study include HAuCl4•4H2O (AR, Sinopharm Co. Ltd., Beijing, China), Na3C6H5O7 (Alfa Aesar Co. Ltd., US), crystal violet (Amresco Co. Ltd., US), and rhodamine 6G (Amresco Co. Ltd., US). All the above reagents were analytically pure. BSA was purchased from Dingguo Co. Ltd., Beijing, China. Ultrapure water (18.0 MΩ cm-1) was used in all the experiments.
Preparation of gold colloid
In accordance with the procedure published by Lee and Meisel, 10 50 ml chloroauric acid (24.28 mM)was heated to boiling, and then 1 ml sodium citrate solution was added to heat for another 30 min. After that, the reacted solution was cooled to room temperature to obtain AuNP colloids. Here, three different concentrations of sodium citrate solution (23.5 nM, 58.4 nM, 87.1 nM) were selected for reaction with chloroauric acid in order to obtain AuNP colloids of different sizes.
Characterization of gold colloids
UV-visible absorption spectra of the synthesized gold colloids were detected by UV-Vis spectrometer (UH4150, HITACHI Co. Ltd., Japan). The scanning range was set as 200–1000 nm with a scanning speed of 600 nm/min. Particle size analysis was performed with dynamic light scattering measurements (Malvern Instruments Ltd., Malvern, UK). The morphology and size of the AuNPs were observed using a transmission electron microscope (TEM) (JEM-1200EX, JEOL, Japan), with an accelerating voltage of 75 kV and an electric current of 40 mA. The average particle sizes were calculated using the Nano Measurer software.
SERS detection and data analysis
The Raman signals were collected by a portable Raman spectrometer (Laser Raman spectrometer JW-1, Beijing JieWei Technology Co. Ltd., China) equipped with a 785 nm, 200 mW diode laser. The wave number range was 400–1800 cm-1, the resolution was 3.5 cm-1, and the laser bandwidth was 0.2 nm.
To compare the enhancement effects of AuNPs with different sizes, a 20 μl sample (R6G or CV) was mixed with 500 μl of gold colloids of different sizes in the sample vial, and then the SERS signals were collected immediately. The concentrations of R6G and CV solutions were 20 nM and 24.5 nM, respectively. The integration time was set to 10 s and the final spectrum was the average of three repeated scans.
To obtain the optimal SERS spectra of BSA, several types of combination modes of BSA solution with AuNPs were studied. First, 20 μl BSA (7.53 nM) was mixed with 500 μl of gold colloids of different sizes in the sample vial to collect the SERS signals. Second, different volume ratios of gold colloids to BSA solution (5:1, 10:1, 25:1, 50:1) were explored to obtain the most optimized signals. In each test, the integration time was 20 s and the final spectrum was the average of three repeated scans. All samples were measured 10 times to obtain the mean spectra and standard deviations. The original data were sequentially preprocessed by removal of cosmic rays, baseline correction, and normalization. The fingerprint region was cut and the spectra data were analyzed using Origin 8.0 software.
Results and discussion
Characterization of AuNPs
In this study, the sizes of AuNP colloids were adjusted by changing the amount of sodium citrate to reduce chloroauric acid. The UV absorption properties of AuNPs can be characterized by the peak position, half bandwidth, and peak intensity. As shown in Table 1 and Figure 1(a), with decreasing concentrations of sodium citrate, λmax of Au colloids is broadened and the peak position shifts from 524 to 540 nm. The plasma resonance absorption of Au particles is mainly caused by the dipole oscillation of conductive electrons in the electromagnetic field. Different dipole oscillations correspond to different sizes of the particles. 11 The broadened and red-shifted maximum absorption peak indicated that AuNPs with larger sizes were produced in the solution. The particle size distribution range of different gold colloids was measured by a particle size analyzer (see Table 1), and the results were coincided with those of UV absorption. Thus, it can be concluded that the concentration of sodium citrate had a great influence on the particle size range of gold colloids. To be specific, the lower concentration of sodium citrate could lead to lower nucleation rate of the agglomeration process, so that a lower number of particles could grow into crystals. As a result, the particles usually had larger sizes. From TEM images, it can be seen that the synthesized AuNPs are smooth and uniform, and mostly present a round or oval shape (see Figure 1(b)). According to the Nano Measurer software results, the average particle sizes were 100 ± 18 nm, 50 ± 12 nm, and 20 ± 8 nm for different concentrations of sodium citrate solution. Therefore, it can be seen that the size of gold nanoparticles can be easily controlled by changing the amount of sodium citrate to reduce chloroauric acid.
Characterization results of AuNPs.
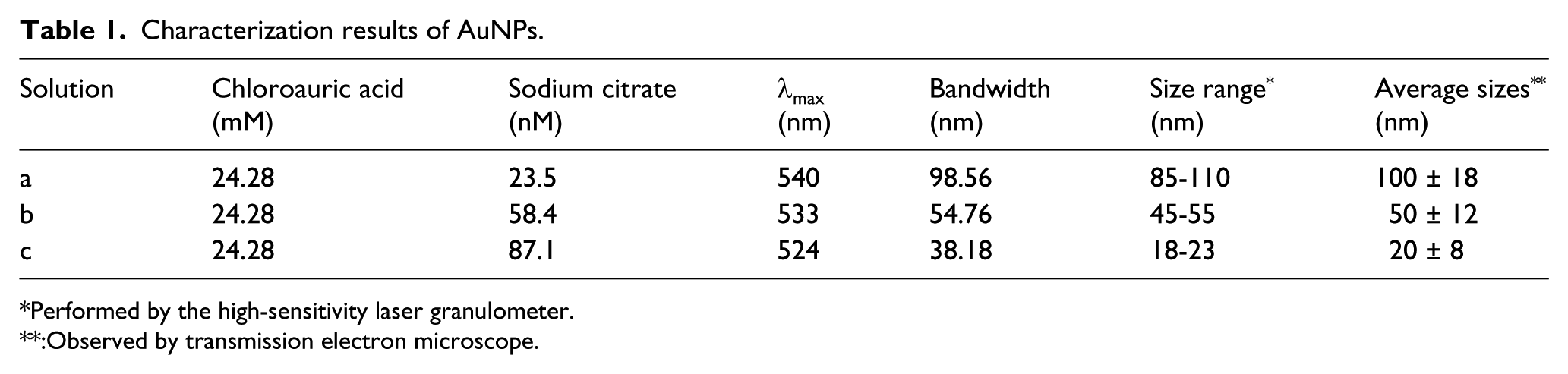
Performed by the high-sensitivity laser granulometer.
:Observed by transmission electron microscope.
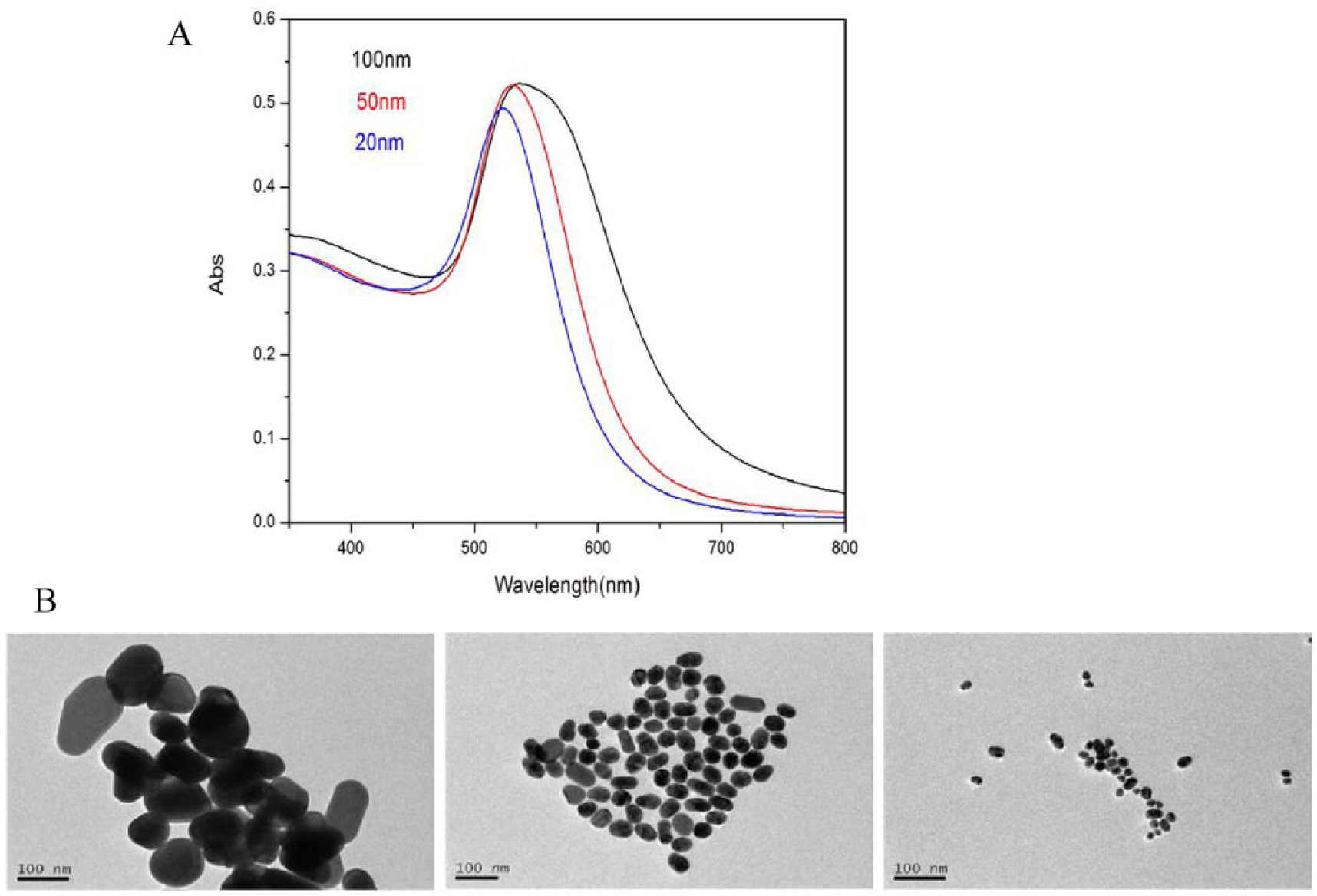
TEM images (A) and UV-Visible absorption spectra (B) of different Au nanoparticles.
Enhancement effects of AuNPs of different sizes
R6G and CV were chosen as markers to evaluate the enhancement effects of the synthesized gold colloids. The normal Raman signals of R6G were weak, but the SERS peaks of R6G clearly showed up in spite of strong fluorescence when AuNPs were applied. The fabricated AuNPs exhibited high sensitivity, and the detecting concentration could be down to nanomolar or even picomolar concentration for R6G and CV. Different sizes of AuNPs showed different enhancement effects (see Figure 2(a)). This might be because different sizes of AuNPs can produce different LSPR. 12 It was easy to observe that the analytes could be absorbed onto the surfaces of Au particles rapidly to cause particle aggregation and produce the SERS signals. The gold colloid of 50 ± 12 nm had the strongest enhancement for R6G and CV (see Figure 2(a) and (d)).
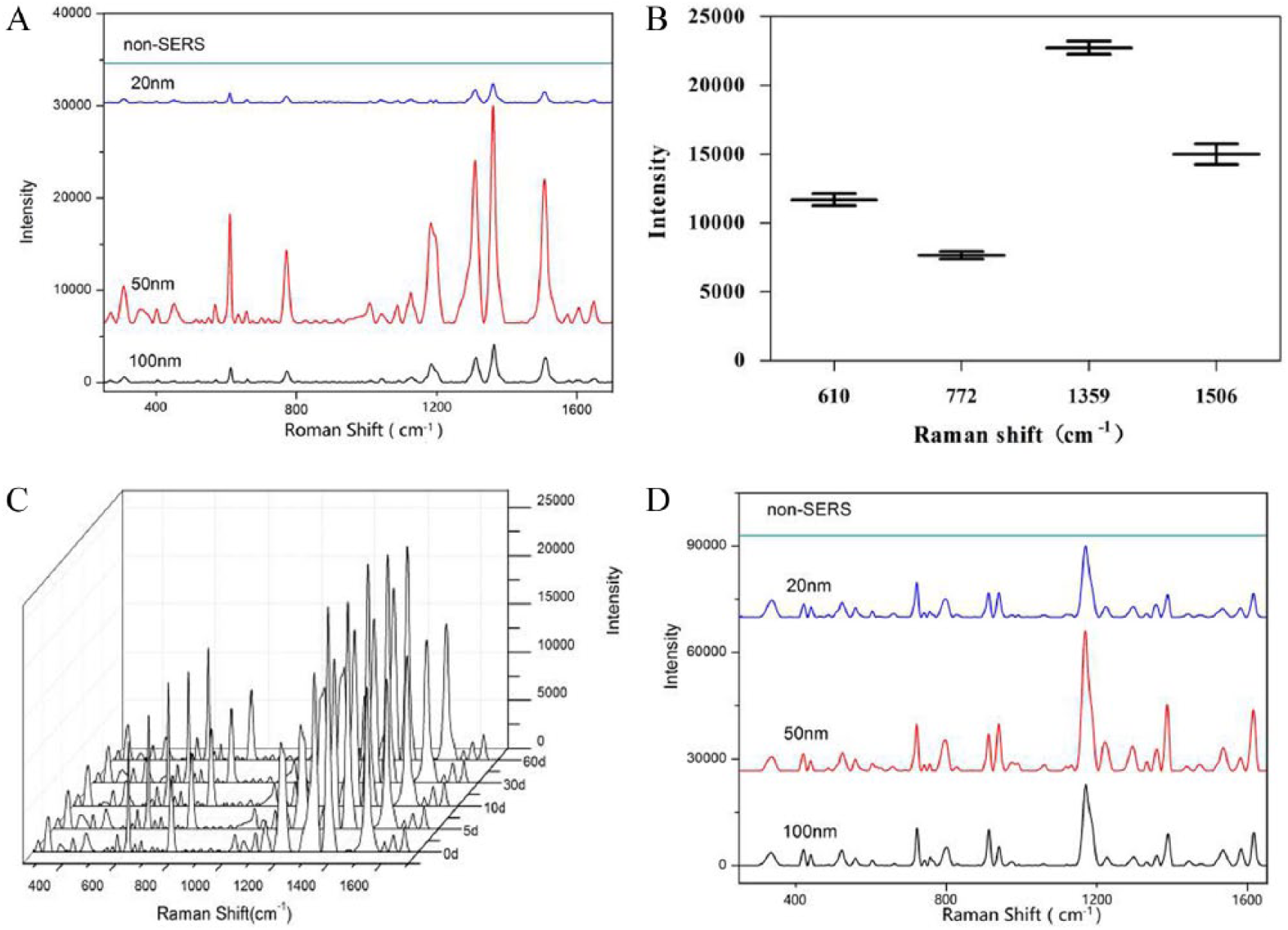
Enhancement effects of AuNPs with different sizes: SERS spectra (A), signal repeatability and reproducibility (B), and colloid stability (C) of R6G; SERS spectra of CV (D).
In Figure 2(a), the typical Raman peaks of R6G are at 615cm-1 (in plane ring C-C-C deformation), 774cm-1 (out of plane C-H deformation), 1360cm-1 (C=C stretching vibration), and 1513cm-1 (C=O stretching vibration). 13 The relative standard deviations (RSDs) of the typical peak intensities are calculated to be less than 10% when the gold colloid of 50 ± 12 nm is used as the enhanced substrate (see Figure 2(b)), and the SERS signals of R6G are stable for the storage period not more than 3 months (see Figure 2(c)). This indicated that the synthesized gold colloids had great stability, repeatability, and reproducibility. As for the other biomarker CV, the results were consistent with those of R6G (see Figure 2(d)).
SERS exploration of BSA
Proteins are complex biomolecules consisting of different amino acids. Proteins with different structures possess different biological functions. Rapid detection and analysis of their structural information is still a challenging issue for biological and biochemical scientists. 14 In this study, the SERS of BSA was explored by applying gold nanoparticles with different sizes. As shown in Figure 3(a), the Raman signals of BSA can be easily observed by SERS with AuNPs of about 100 nm, 50 nm, and 20 nm. Comparatively, the AuNPs of 50 ± 12 nm show the strongest Raman peaks at 1100–1200 cm-1 and 1400–1600 cm-1; meanwhile, other typical peaks at 955 cm-1, 1159 cm-1, and 1550 cm-1 correspond, respectively, to the symmetric stretching vibration of carboxylic acid groups, the stretching vibration of the C-N bond in the phenylalanine residues and the tyrosine residues, and the C-N stretching vibration of phenylalanine and acid amides.15,16 Although the SERS signals of BSA with AuNPs of 20 ± 8 nm are weak, typical peaks at 955 cm-1, 1159 cm-1, and 1550 cm-1 can still be observed. As for the AuNPs of about 100 nm, characteristic peaks at 955 cm-1, 1007 cm-1, 1159 cm-1, 1205 cm-1, and 1550 cm-1 can be observed. Taken together, the AuNPs of ~50 nm in size showed the strongest enhancement effects for most vibrations. This result was consistent with other works. 17 This strongest enhancement effects may be due to the controlling synthesis in which the particle size was more than 50 nm and the particle interspace was less than 10 nm. Under this condition, the LSPR frequency of the substrate was matched with the incident laser frequency and the effective coupling between nanoparticles could be induced to maximize the enhancement. 18 In addition, considering the production of SERS hotspots suitable for protein detection requires proper aggregates, it is necessary to explore the appropriate ratio of gold colloid to BSA. As shown in Figure 3(b), the best enhancement effects can be achieved when the volume ratio of gold nanoparticles to BSA is 25:1.
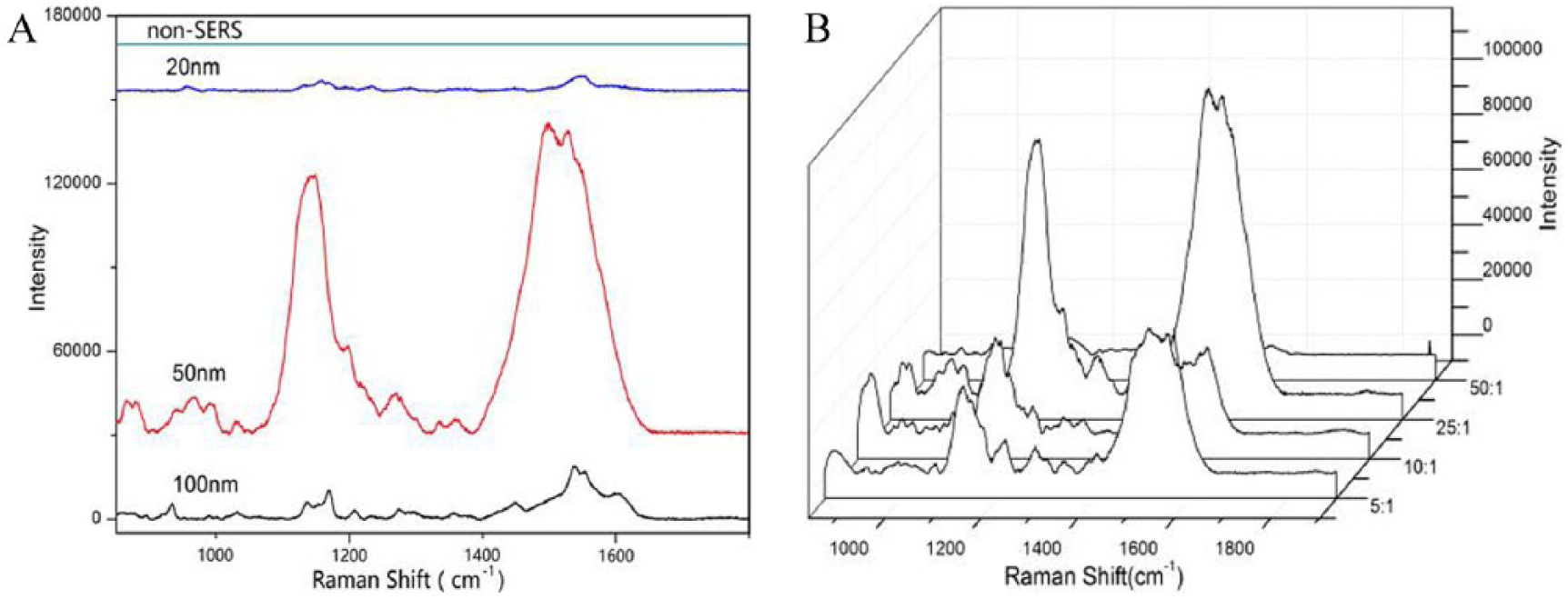
SERS spectra of BSA: SERS spectra of BSA by Au nanoparticles with different sizes (A); SERS spectra of BSA for different volume ratios of gold nanoparticles to BSA (B).
Conclusion
In this study, AuNP colloids with different sizes were synthesized and used as the sensors to probe the SERS signals of different biomarkers and biomolecules. The fabricated AuNP colloids had numerous merits of outstanding enhancement effects, convenient synthesis, biocompatibility, easy operation, and desirable economy. It was found that the AuNP colloid of 50 nm had excellent SERS effects in probing various molecules (rhodamine 6G, crystal violet, and BSA), and could be preserved for about 3 months with good repeatability and reproducibility (RSD <5%) in terms of the probed signal intensity. Meanwhile, the fabricated AuNPs were applied to study the SERS signals and structural information of BSA in aqueous solution, and it was proved that SERS could rapidly provide the structural information and vibration characteristics of BSA. Our work indicates that biocompatible Au nanoparticle colloids may be a promising biosensor in the rapid and label-free detection of biological systems.
Footnotes
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research work is supported by the General project of Science and Technology of Beijing Municipal Commission of Education (Grant No.201810005031).
