Abstract
Background and objective:
Angiotensin-converting enzyme inhibitors are first-line therapies for hypertension and related cardiovascular and renal conditions. The study aimed to characterize the adverse event profile of ACE inhibitors using the U.S. Food and Drug Administration Adverse Event Reporting System.
Methods:
This retrospective pharmacovigilance study analyzed FAERS reports submitted from January 1, 1979, through March 31, 2025, for nine ACE inhibitors (benazepril, captopril, enalapril, fosinopril, lisinopril, moexipril, quinapril, ramipril, and trandolapril) identified as primary suspect drugs. Extracted variables included patient age, sex, reporter type, and reported adverse events. Descriptive statistics were used to summarize reporting frequencies. Comparisons between subgroups (male vs female, <18 vs ≥18 years, and healthcare professional vs consumer reporters) were conducted using reporting proportions. Reporting proportion ratios (RPRs) were calculated to compare the proportion of specific adverse events between predefined reference categories. As FAERS does not provide denominator data, findings reflect reporting patterns rather than incidence or risk estimates.
Results:
A total of 124,638 adverse event reports were identified. ADEs were disproportionately higher in adults and elderly patients compared to those <18 years. Males generally reported more ADEs than females, though some drugs showed the opposite trend. Healthcare professionals submitted the majority of reports (pooled RR = 2.22, 95% CI: 2.20–2.25). The most commonly reported ADEs were angioedema, cough, hypotension, and acute kidney injury.
Conclusion:
ACE inhibitor adverse event profiles vary by drug, patient age, and sex. Adults and elderly patients carry the highest burden of ADEs, though pediatric cases remain clinically relevant for specific agents. These findings support tailored monitoring and risk mitigation strategies in clinical practice.
Introduction
Hypertension, defined as chronically elevated blood pressure, is a major modifiable risk factor for cardiovascular disease, chronic kidney disease, and dementia, and remains the leading contributor to global morbidity and mortality.1–3 Despite being largely preventable, it continues to drive the global burden of ischemic heart disease, stroke, and heart failure.1–3
Among available therapies, angiotensin-converting enzyme (ACE) inhibitors are first-line agents owing to their dual cardioprotective and renoprotective benefits. By blocking the conversion of angiotensin I to angiotensin II, these agents promote vasodilation and reduce sodium retention, thereby lowering blood pressure and limiting cardiac remodeling.4–7 Their clinical utility extends across hypertension, coronary artery disease, myocardial infarction, chronic kidney disease, and heart failure with reduced ejection fraction.5,6
However, ACE inhibitor use is associated with a spectrum of adverse drug events (ADEs). These range from common and bothersome effects, such as dry cough and hyperkalemia, to rare but life-threatening complications like angioedema.5,8–10 While clinical trials established their efficacy and common side effects, they are often underpowered to fully characterize the incidence, severity, and real-world clinical presentation of these rarer ADEs.
Pharmacovigilance systems like the FDA Adverse Event Reporting System (FAERS) are therefore critical for identifying and quantifying these post-marketing safety risks. Containing over 28 million reports from healthcare providers, consumers, and manufacturers, FAERS is a cornerstone resource for drug safety surveillance.11–13 To address this gap in real-world safety data, this study leverages the FAERS database to systematically evaluate and characterize ACE inhibitor–associated adverse events, with a specific focus on severe reactions like angioedema. The study aimed to quantify the reporting rates, identify demographic or clinical risk factors, and ultimately inform safer prescribing practices.
Methods
Data source
This retrospective descriptive study analyzed adverse event reports for angiotensin-converting enzyme (ACE) inhibitors using the U.S. Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS), a publicly available post-marketing pharmacovigilance database. FAERS is a spontaneous reporting system that supports the FDA’s ongoing safety surveillance of approved drugs and therapeutic biologics. It is structured as a relational database composed of multiple data files, including patient demographic and administrative information (age, sex, reporting country, report type, and dates), drug information (suspect, concomitant, and interacting products; role codes; dosage and therapy dates), adverse event terms coded using the Medical Dictionary for Regulatory Activities (MedDRA), indications for use, report source (healthcare professional, consumer, or manufacturer), and patient outcomes such as death, life-threatening events, hospitalization, disability, and congenital anomaly. Each case is assigned a unique identifier and may include follow-up submissions that update the original report.
Adverse event reports are submitted voluntarily by healthcare professionals and consumers, while manufacturers are legally required to report serious and unexpected adverse events to the FDA. Reports are transmitted electronically, standardized, and incorporated into publicly available quarterly data files. As a spontaneous reporting system, FAERS is subject to inherent limitations, including underreporting, variable data completeness, duplicate reporting, and reporting bias, and it does not allow estimation of true incidence rates or confirmation of causality. Nevertheless, FAERS remains a critical tool for signal detection, hypothesis generation, and regulatory decision-making.
Inclusion and exclusion criteria
All reports submitted to FAERS, in which an ACE inhibitor (benazepril, captopril, enalapril, fosinopril, lisinopril, moexipril, quinapril, ramipril, or trandolapril) was listed as the primary suspect drug, were included. This retrospective pharmacovigilance study analyzed FAERS reports submitted from January 1, 1979, through March 31, 2025, for the nine ACE inhibitors identified as primary suspect drugs. FAERS reports were screened to identify cases involving the nine ACE inhibitors of interest. Only reports in which the ACE inhibitor was designated as a Primary Suspect (PS) drug were included.
The following reports were excluded:
(1) reports listing ACE inhibitors solely as Secondary Suspect (SS), Concomitant (C), or Interacting (I) medications to minimize confounding.
(2) duplicate reports, for which only the most recent version of each case ID was retained according to FDA recommendations.
(3) reports with missing or invalid case identifiers.
(4) reports submitted outside the predefined study period (January 1, 1979, through March 31, 2025).
Duplicate detection was further supported by cross-checking patient demographics, event dates, and clinical outcomes to eliminate additional redundancies. For example, reports in which metformin was coded as the primary suspect for hypoglycemia while lisinopril was recorded as a concomitant medication were excluded.
Data collection and analysis
Extracted variables included patient demographics (age and sex), reporter type (healthcare professional vs non-healthcare professional), and adverse events categorized by Medical Dictionary for Regulatory Activities (MedDRA) Preferred Terms Version 28.1. Although the FDA Adverse Event Reporting System includes a race/ethnicity variable, this field is non-mandatory and frequently incomplete. In our dataset, a substantial proportion of reports lacked information on race, which limited the reliability of subgroup analyses. Therefore, race/ethnicity was not included in the present analysis. Descriptive statistics (absolute counts and percentages) were used to summarize the distribution of adverse events across individual ACE inhibitors. Because FAERS is a spontaneous reporting database and does not provide denominator data (i.e. the total number of exposed ACE inhibitor users without adverse events), true incidence or risk estimates cannot be calculated. Therefore, comparisons between subgroups (sex, age category, reporter type) were conducted using reporting proportions rather than risk ratios. The reporting proportion ratio (RPR) was calculated by dividing the proportion of a specific adverse event among reports in one subgroup by the corresponding proportion in the comparison subgroup. About 95% confidence intervals were computed using the Wald method. Heterogeneity across drugs was assessed using the I2 statistic.
Ethical considerations
The study used publicly available, de-identified data from FAERS. In accordance with U.S. federal regulations (45 CFR 46), this analysis does not constitute human subjects research and therefore did not require Institutional Review Board (IRB) approval or informed consent.
Results
A total of 124,638 adverse event reports were identified from FAERS submissions through March 31, 2025, spanning nine ACE inhibitors. Across all nine ACE inhibitors, a total of 126,190 reports were included in the analysis. Age information was available for 117,169 cases (92.85%), while 9021 reports (7.15%) had missing age data. Sex information was available for 112,272 cases (88.97%), with 13,918 reports (11.03%) lacking sex specification. Reports with missing demographic data were excluded from age- and sex-stratified analyses but retained in the overall adverse event frequency analysis.
Year-wise stratification of adverse event reports for ACE inhibitors
Year-wise stratification of adverse event reports for ACE inhibitors as a class demonstrated a clear temporal trend in the FAERS database. Reporting remained relatively low and stable during the late 1980s and early 1990s, followed by a gradual increase beginning in the mid-1990s. A more pronounced upward trajectory was observed after 2010, with reports rising sharply and reaching a peak between 2018 and 2020. This period represented the highest reporting burden for the ACE inhibitor class. After 2020, a modest decline and subsequent stabilization were observed; however, reporting levels remained substantially higher than those recorded in earlier decades. Overall, the findings indicate a significant long-term increase in reported adverse events for ACE inhibitors, likely reflecting expanded utilization, improved pharmacovigilance systems, and enhanced reporting practices over time (Figure 1).

Year-wise trend of adverse event reports for ACE inhibitors as a class in the FAERS database.
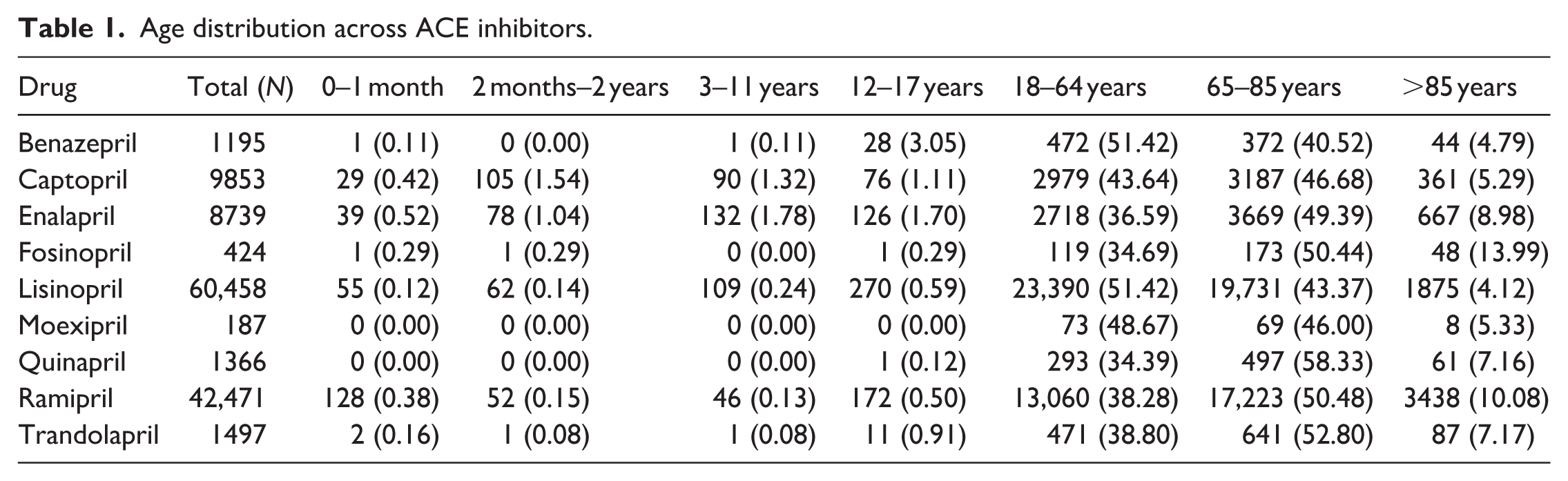
Age distribution across ACE inhibitors
The age distribution of adverse event reports varied across angiotensin-converting enzyme (ACE) inhibitors, with the majority of reports occurring in adults and older adults for all drugs. For lisinopril (n = 60,458), most reports were among individuals aged 18–64 years (51.42%) and 65–85 years (43.37%), with minimal pediatric involvement (<1%). A similar pattern was observed for ramipril (n = 42,471), where 50.48% of reports involved patients aged 65–85 years and 38.28% aged 18–64 years. For enalapril (n = 8739) and captopril (n = 9853), reports were also concentrated among adults aged ≥65 years (49.39% and 46.68%, respectively), followed by those aged 18–64 years. Pediatric reports were uncommon across all agents. Less frequently reported ACE inhibitors, such as fosinopril, quinapril, and trandolapril, demonstrated a predominance of reports in the 65–85-year age group (50.44%, 58.33%, and 52.80%, respectively). Benazepril reports were primarily among adults aged 18–64 years (51.42%) and 65–85 years (40.52%). Overall, adverse event reporting was heavily concentrated among middle-aged and elderly populations (Table 1).
Age distribution across ACE inhibitors.
Gender distribution across ACE inhibitors
Gender distribution was generally balanced across most ACE inhibitors. For lisinopril, males accounted for 52.49% of reports and females for 47.51%. Similarly, ramipril, captopril, and benazepril demonstrated near-equal gender representation, with slight male predominance. In contrast, fosinopril showed a higher proportion of male reports (62.80%), whereas quinapril had a greater proportion of female reports (59.28%). Moexipril reports were slightly more common among females (53.80%). Overall, the data suggests no major gender disparity in adverse event reporting for most ACE inhibitors, though minor variations exist among specific agents (Table 2).
Gender distribution across ACE inhibitors.

Reporters type across ACE inhibitors
Reporter type varied considerably between ACE inhibitors. Reports for enalapril (87.48%), fosinopril (86.17%), and ramipril (80.97%) were predominantly submitted by healthcare professionals (HCPs). Benazepril and trandolapril also showed a majority of HCP-reported cases (78.02% and 75.44%, respectively). In contrast, captopril reports were primarily submitted by consumers (64.32%), representing the highest consumer-reported proportion among the included agents. Lisinopril had a mixed reporting pattern, with 63.24% from healthcare professionals and 36.76% from consumers. Quinapril and moexipril demonstrated relatively balanced distributions between HCP and consumer reports. These findings indicate variability in reporting sources depending on the specific ACE inhibitor (Table 3).
Reporters type across ACE inhibitors.

Most frequently reported adverse events across ACE inhibitors
The most frequently reported adverse events differed across ACE inhibitors. For Lisinopril, angioedema was the most common event (14.64%), followed by cough (8.06%) and drug hypersensitivity (6.97%). Ramipril reports were led by acute kidney injury (11.10%), hypotension (7.40%), and dyspnea (6.78%). Acute kidney injury was also the leading event for enalapril (13.63%) and fosinopril (21.93%), highlighting a potential class-related renal safety concern. Captopril was most frequently associated with cough (6.88%), dermatitis (4.32%), and hypotension (4.23%). For Benazepril, the most commonly reported events were completed suicide (12.22%), angioedema (11.97%), and hypotension (7.53%). Quinapril reports were dominated by recalled product administered (14.79%), followed by fall (10.61%) and ineffective drug (9.30%). Trandolapril was most frequently associated with dyspnea (8.08%), cough (7.28%), and acute kidney injury (6.61%), while moexipril most commonly involved cough (8.56%), angioedema (7.49%), and ineffective drug (6.95%). Overall, while some adverse events such as acute kidney injury, hypotension, and angioedema appeared across multiple ACE inhibitors, the relative frequency of specific events varied substantially between individual agents, supporting heterogeneity in safety reporting profiles (Table 4).
Most frequently reported adverse events across ACE inhibitors (top 3 per drug).

Disproportionality analyses were performed to detect pharmacovigilance signals. For each adverse event, a 2 × 2 contingency table was constructed using ACE inhibitor reports and all other drugs in FAERS as an internal reference. Three measures were calculated: the Proportional Reporting Ratio (PRR), calculated as [a/(a + b)] ÷ [c/(c + d)]; the Reporting Odds Ratio (ROR), calculated as (a × d)/(b × c); and the Information Component (IC), a Bayesian measure calculated as log2[a/N ÷ ((a + b)/N × (a + c)/N)]. A signal was considered significant when PRR ≥ 2 with χ2 ≥ 4, ROR lower 95% CI > 1, and IC025 > 0.
Angioedema
Angioedema exhibited marked heterogeneity in proportional reporting across ACE inhibitors. Lisinopril demonstrated the highest reporting proportion (14.64%), followed by benazepril (11.97%) and fosinopril (11.79%), whereas captopril (4.16%) and trandolapril (4.14%) showed comparatively lower proportions. Enalapril (7.22%) and moexipril (7.49%) occupied an intermediate range. Notably, angioedema did not rank among the leading reported events for quinapril or ramipril. These findings indicate substantial variability in reporting patterns within FAERS. Given the known class effect of bradykinin-mediated angioedema, the observed differences likely reflect variations in prescribing volume, population susceptibility, duration of exposure, and stimulated reporting rather than definitive differences in intrinsic drug-specific risk. Angioedema demonstrated a strong and statistically significant disproportionality signal among ACE inhibitor reports in FAERS (PRR = 85.94, χ2 = 489,203, ROR = 96.22, IC = 6.00, IC025 = 5.97), meeting all signal detection thresholds. These findings are consistent with the well-established bradykinin-mediated mechanism of ACE inhibitor-induced angioedema and provide quantitative pharmacovigilance evidence supporting its clinical significance.
Acute kidney injury
For acute kidney injury (AKI), fosinopril showed the highest reporting proportion (21.93%), followed by enalapril (13.63%) and ramipril (11.10%). Benazepril and trandolapril had moderate proportions, while lisinopril, moexipril, and captopril showed lower frequencies. The higher reporting with fosinopril may relate to an older patient population and greater baseline renal comorbidity rather than a direct causal effect. The FAERS disproportionality analysis revealed a robust pharmacovigilance signal for AKI associated with ACE inhibitors across all three detection methods (PRR = 16.04, χ2 = 1702; ROR = 17.60, 95% CI: 17.27–17.94; IC025 = 3.89), indicating that AKI is reported approximately 16–18 times more frequently with this drug class than with all other drugs in the database.
Hypotension
Hypotension was reported across all ACE inhibitors, with notable variability in magnitude. The highest proportional reporting was observed with fosinopril (9.67%) and quinapril (9.22%), followed by enalapril (7.52%), benazepril (7.53%), and ramipril (7.40%). Lower proportions were documented with trandolapril (6.15%), lisinopril (4.78%), moexipril (4.81%), and captopril (4.23%). The clustering of higher proportions among certain agents may reflect differences in clinical indication, baseline cardiovascular status, or titration practices. ACE inhibitors show a strong, consistent pharmacovigilance signal for hypotension across all four detection methods (PRR = 7.75, χ2 = 52,433, ROR = 8.25, IC = 2.92), with hypotension reported nearly eight times more frequently than with other drugs. This is statistically robust and aligns with the drug class’s known blood pressure-lowering mechanism.
The rate of adverse events in patients <18 versus ≥18 years
The forest plot compares the rate of adverse events (AEs) between patients younger than 18 years and those aged 18 years or older for different ACE inhibitors. Reporting proportion ratios (RPRs) comparing patients <18 years with those ≥18 years demonstrated consistently lower reporting proportions in the pediatric group across all ACE inhibitors. All RPR estimates were below 1.0, with 95% confidence intervals not crossing unity, indicating statistically significant differences in reporting proportions between age groups. These findings reflect differences in reporting patterns rather than incidence or risk. Among the most reported drugs, lisinopril and ramipril accounted for the majority of AE cases, particularly in adults and the elderly. In contrast, pediatric reports were relatively rare but notable for captopril and enalapril, especially in infants and children. This suggests that while ACE inhibitors are associated with adverse events across all age groups, the burden is disproportionately higher in adult and elderly populations. In contrast, pediatric events are limited in number but still clinically relevant for certain drugs (Figure 2).

The rate of adverse events in patients <18 versus ≥18 years.
Adverse event rates in male and female patients
Figure 3 illustrates the comparison of adverse event (AE) rates between male and female patients receiving different ACE inhibitors. The pooled reporting proportion ratio (RPR = 0.94, 95% CI: 0.93–0.95) indicates a statistically lower reporting proportion among males compared with females overall. However, substantial heterogeneity (I2 = 97%) suggests variability across individual drugs, indicating that sex-related differences in reporting are not consistent.

Adverse event rates in male and female patients.
The rate of adverse events reported by healthcare professionals and consumers
The analysis of adverse event reporting by reporter specialty shows a marked predominance of healthcare professionals compared to consumers across all ACE inhibitors, with notable variations by individual drug. For example, healthcare professionals accounted for the majority of reports for lisinopril (36,190 vs 21,039 consumer reports) and ramipril (33,081 vs 7774), reflecting a stronger contribution from clinicians in documenting adverse reactions for these widely prescribed agents. In contrast, drugs such as quinapril and moexipril demonstrated a more balanced distribution of reports between healthcare professionals and consumers, suggesting that patient-driven reporting played a relatively larger role for these agents. The pooled reporting proportion ratio (RPR = 2.22, 95% CI: 2.20–2.25) indicates that adverse events were reported more frequently by healthcare professionals compared with consumers. This reflects differences in reporting behavior within FAERS rather than differences in true event occurrence (Figure 4).
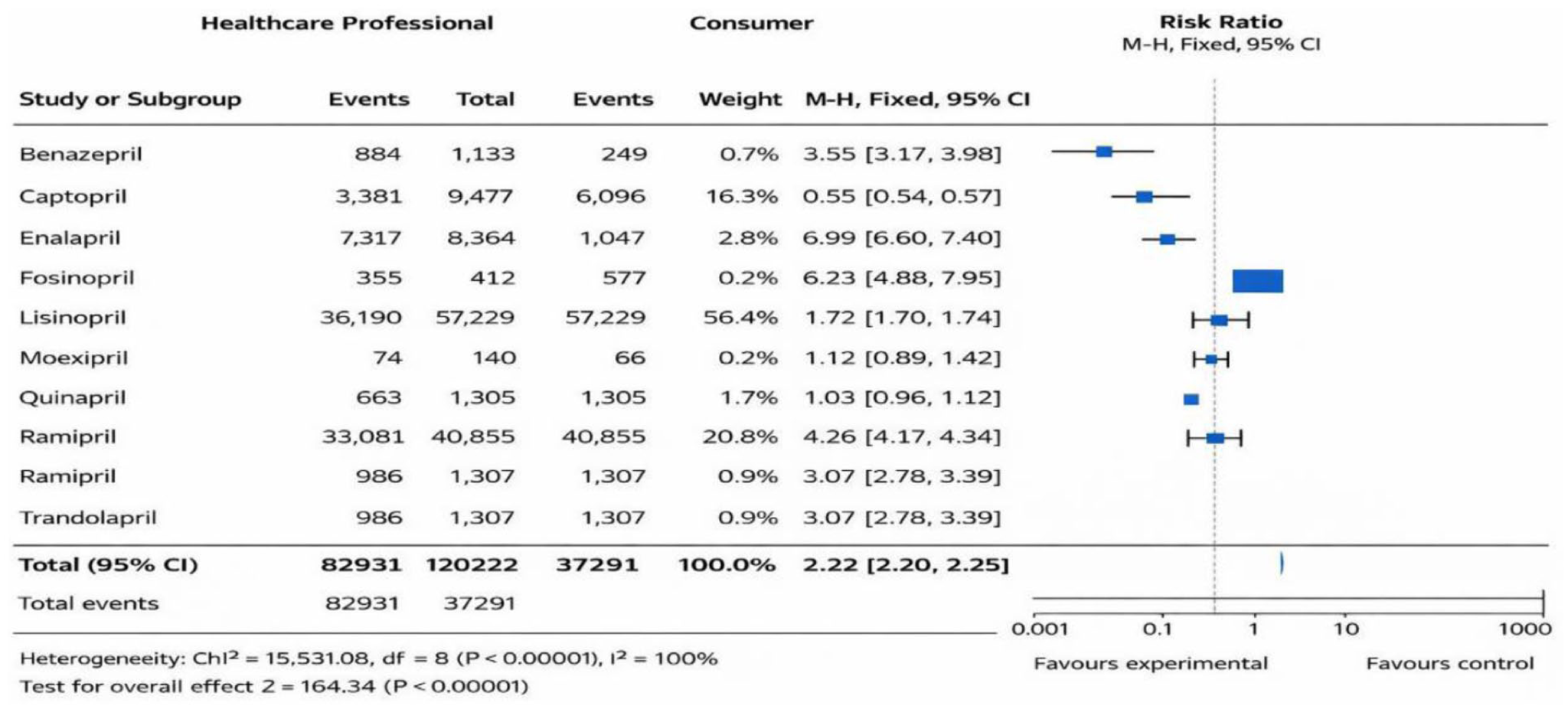
The rate of adverse events reported by healthcare professionals and consumers.
Discussion
This study offers a thorough examination of actual adverse event (AE) reports for nine distinct ACE inhibitors, providing important information about the safety profiles of these medications in a range of patient demographics. A significant burden of adverse events (AEs) in the adult and elderly populations, unique and occasionally surprising AE profiles for specific medications, a slight but irregular gender gap in AE reporting, and a significant role for healthcare professionals in pharmacovigilance reporting are among the main conclusions of the analysis.
The most remarkable observation is the distinct demographic distribution of AEs. The forest plot analysis shows that pediatric patients (<18 years) have a significantly lower reporting frequency of reported adverse events (AEs) than adults (≥18 years) for all ACE inhibitors tested. This is consistent with the primary usage of these drugs for illnesses such as hypertension and heart failure, which are common among older persons. According to Ruksakulpiwat et al. 14 hypertension raises the risk of stroke and heart disease, which are the major causes of death and disability worldwide, especially among older persons. According to WHO, older age is a risk factor for high blood pressure, and a projected 1.4 billion persons aged 30–79 years had hypertension in 2024, accounting for 33% of the population in this age group. 15 The number of adverse events (AEs) in the 18–64 and 65–85 age groups across all medicines demonstrates that the patient population being treated determines the observed AE pattern. However, the availability of pediatric reports, particularly for captopril and enalapril, is clinically significant. This most likely reflects the allowed use of these specific ACE inhibitors in pediatric populations for illnesses like proteinuric renal disease or heart failure, indicating that while the absolute number of reports is minimal, attention is still essential when prescribing these medications to children.
Despite the fact that ACE inhibitors work by blocking the renin-angiotensin system, our research shows significant differences in their adverse event (AE) profiles, which could influence clinical judgment. Its function as a significant class-wide risk was confirmed by the frequent reporting of class-related adverse events (AEs) such as cough, angioedema, and hypotension. Angioedema was more common for benazepril (11.97%), lisinopril (14.64%), and fosinopril (11.79%). There were also clear medication-specific differences: reports of completed suicide were surprisingly high for benazepril and lisinopril (12.22% and 5.05%, respectively). This finding may be due to confounding variables rather than direct drug effects, but it still merits more research. Strong associations between enalapril and ramipril (13.63% and 11.10%) and acute kidney injury as well as drug interactions (particularly 10.50% for enalapril), highlight the importance of renal monitoring and cautious medication revision. With a high incidence of AKI (21.93%), reports of rhabdomyolysis (8.49%), and metabolic acidosis (6.13%), fosinopril showed a unique profile. In contrast, quinapril’s adverse event reports were primarily dominated by falls (10.61%) and recalled products (14.79%), which may indicate safety concerns in elderly, fragile patients. According to Na Takuathung et al., 257 RCTs were included in the meta-analysis out of 378 RCTs that matched the eligibility requirements. The reporting of dry cough, hypotension, dizziness, and hyperkalemia was significantly higher when ACE inhibitors were compared to a placebo. 16 According to Messerli et al., ACE inhibitor users were far more likely than ARB users to experience angioedema, cough, pancreatitis, and gastrointestinal bleeding. ACE inhibitor-associated angioedema was more than three times as common (hazard ratio: 3.31 [95% CI: 2.55, 4.51]). 17 According to Borghi et al., among the wide range of cardiovascular medications, ACEI has a very good safety and tolerability profile. Patients with more serious medical conditions or those receiving treatment from many drug classes are more likely to experience side effects such as dry cough, hypotension, hyperkalemia, headache, dizziness, and renal impairment. 18 These variations demonstrate the importance of taking into account each agent’s distinct adverse event pattern in addition to pharmacokinetics and efficacy when choosing an ACE inhibitor.
Research limitations
Several limitations specific to suicide-related adverse events warrant acknowledgment. FAERS data are subject to underreporting bias, which is likely to be particularly pronounced for suicide-related outcomes due to social stigma, medicolegal considerations, and the inherent difficulty in ascertaining causality in fatal or near-fatal events. Reports may originate from healthcare providers, patients, or manufacturers with varying levels of clinical detail, and the cause-death or intent (e.g. suicidal ideation vs completed suicide vs accidental overdose) may be inaccurately classified or incompletely documented. Furthermore, FAERS does not capture baseline psychiatric history, concomitant psychotropic medications, or underlying risk factors—all of which are critical confounders when evaluating suicide-related signals. The presence of a disproportionality signal should therefore not be interpreted as evidence of a causal relationship between ACE inhibitors and suicidal behavior, but rather as a hypothesis-generating finding requiring confirmation through controlled epidemiological studies.
Conclusion
This large-scale analysis of ACE inhibitor adverse events demonstrates that while these drugs are a cornerstone of cardiovascular therapy, their safety profiles are not uniform. The reporting proportion of AEs is overwhelmingly concentrated in adult and elderly populations, but pediatric exposure requires monitoring. Significant differences exist between individual drugs, with some associated with severe, unexpected events like suicide or rhabdomyolysis that merit further study. Gender appears to influence AE reporting proportion in a drug-specific manner, and healthcare professionals remain the backbone of pharmacovigilance, though consumer reports provide critical supplemental data. These findings can inform clinical practice by highlighting drug-specific hazards and underscoring the need for tailored patient monitoring and continued post-marketing surveillance.
In comparative analysis across agents, clinically meaningful variability in reporting patterns was observed for key adverse events. Angioedema was disproportionately reported with lisinopril, benazepril, and fosinopril, whereas acute kidney injury was most prominent with fosinopril, enalapril, and ramipril. Hypotension clustered more frequently with fosinopril and quinapril, while hyperkalemia showed higher proportional reporting with enalapril. Cough, a recognized class effect, demonstrated greater reporting proportions with moexipril, lisinopril, and trandolapril. Although these findings reflect differences in reporting behavior within FAERS rather than comparative incidence or causal risk, they suggest that the safety profile of ACE inhibitors is not uniform at the reporting level. Awareness of these differential reporting patterns may assist clinicians in individualized agent selection, particularly in patients at increased risk for renal dysfunction, electrolyte imbalance, hypotension, or bradykinin-mediated reactions
Footnotes
Acknowledgements
The authors extend their appreciation to Prince Sattam bin Abdulaziz University for funding this research work through the project number (PSAU/2025/03/32516).
Ethical considerations
This analysis utilized publicly available, fully de-identified data from the FDA Adverse Event Reporting System (FAERS); as such, the study was deemed not to constitute human subjects research, and no Institutional Review Board approval or individual patient consent was required.
Consent to participate
As this study involved the analysis of a fully de-identified, public database with no direct patient contact, individual patient consent was neither required nor obtained. The FAERS data collection process itself operates under federal regulations where the primary reporters (healthcare professionals or consumers) submit the reports, and patient consent for this submission is governed by the FDA’s regulatory framework for pharmacovigilance.
Author contributions
The author was responsible for the conception, design, data collection, analysis, interpretation, and writing of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to Prince Sattam bin Abdulaziz University for funding this research work through the project number (PSAU/2025/03/32516).
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
