Abstract
Background:
Herbal medicines are widely used globally; however, public awareness of their appropriate use and potential adverse effects remains limited. This study aimed to assess the knowledge and awareness of herb–drug interactions among the general population in Saudi Arabia, a context where research in this area is scarce.
Methods:
A cross-sectional survey was conducted using an electronic questionnaire disseminated through social media platforms. Data were analyzed with SAS® version 9.4, and multiple linear regression analyses were performed to identify factors associated with participants’ knowledge scores.
Results:
A total of 834 respondents participated in the study. The majority were female (64.4%) and married (48.7%). More than half of the participants (57%) demonstrated moderate knowledge of herb–drug interactions. Age and region were negatively associated with knowledge levels, whereas female gender and prior use of herbal products were positively associated.
Conclusions:
The findings reveal a moderate level of public knowledge regarding herb–drug interactions in Saudi Arabia. Targeted educational initiatives and public health interventions are urgently needed to improve awareness and promote safe practices.
Introduction
Herbal medicine, a core component of complementary and alternative medicine (CAM), is widely practiced across diverse cultures.1,2 Over the past few decades, the global use of herbal medicines and natural products has increased substantially. 3 These products are frequently marketed as dietary supplements and are readily available as over-the-counter remedies. 4 It is estimated that more than 70% of the global population relies on traditional herbs for the treatment of various illnesses. 5 In Saudi Arabia, the use of herbal remedies alongside conventional drug therapy is particularly common.6 –8 This growing interest has attracted the attention of regulatory agencies, researchers, and healthcare practitioners, given its implications for patient safety and healthcare delivery.
Herbal products contain a wide range of bioactive compounds derived from stems, seeds, leaves, roots, and flowers. 3 Although often promoted as “natural” and perceived as harmless, these products can pose risks due to their complex chemical composition, possible contaminants, and potential interactions with pharmaceutical drugs. 3 Reports of adverse effects highlight the importance of evaluating their safety profiles systematically. Consequently, there is a growing need for rigorous research and regulatory oversight to ensure their safe integration into healthcare practices.
However, unlike conventional pharmaceuticals, dietary supplements are not routinely subjected to pre-marketing safety evaluations by the U.S. Food and Drug Administration (FDA), nor are all marketed supplements registered. 9 In addition, phytochemical variability and content inconsistency between different preparations and batches further complicate safety evaluations.10,11 These regulatory and quality-control gaps are particularly concerning because clinical evidence demonstrates that herbal medicines can interact with conventional drugs. 12 Such herb–drug interactions are generally classified as pharmacokinetic or pharmacodynamic. 12 Pharmacokinetic interactions occur when herbal supplements influence drug absorption, distribution, metabolism, or elimination, potentially altering therapeutic efficacy or increasing adverse effects. 13 Pharmacodynamic interactions, though less frequent, occur when an herbal supplement modifies the pharmacological action of a co-administered drug, leading to synergistic or antagonistic outcomes. 14 Both interaction types illustrate why awareness of herbal medicine use is essential in clinical practice.
Despite this, public awareness remains limited. In Saudi Arabia, previous studies have found inadequate knowledge of herbal medicine use 7 and insufficient labeling information on natural health products. 15 Yet, no study has specifically investigated public understanding of herb–drug interactions. Identifying knowledge gaps in this area is critical for healthcare providers to support safe practices and reduce risks. Therefore, this study aimed to evaluate the knowledge and awareness of herb–drug interactions among the general population in Saudi Arabia.
Methods
Study design, population, and sample
This study employed a cross-sectional descriptive design and was conducted between March and August 2021. The target population comprised residents of Saudi Arabia, with the aim of assessing their knowledge and awareness of herb–drug interactions. Eligibility criteria included adults aged 18 years or older without formal medical education. Exclusion criteria were children, healthcare professionals, and non-residents temporarily visiting Saudi Arabia. The required sample size was calculated using Raosoft® software, which indicated a minimum of 385 participants based on a 5% margin of error, a 95% confidence level, and a 50% response distribution. Ethical approval for this study was granted by the Ethical Committee of Prince Sattam bin Abdulaziz University (Reference Number: SCBR-475/2025). Informed consent is typically obtained digitally by presenting a detailed consent statement on the first page of the online survey. Participant confidentiality was strictly maintained, as no personal identifiers such as names or national identification numbers were collected.
Survey development and administration
The study questionnaire was originally developed in English, as most relevant literature, frameworks, and validated tools in this domain were available in English. It was subsequently translated into Arabic by two language experts fluent in the local dialect. A back-translation into English was then performed by three bilingual specialists to ensure semantic and conceptual equivalence. Since existing Arabic tools did not adequately capture the domains of interest, a new questionnaire was designed, adapted, and refined through forward–backward translation and expert review to ensure both content validity and linguistic accuracy. The final instrument consisted of two sections. The first section collected demographic information and data on prior use of herbal products. The second section assessed knowledge of herb–drug interactions through eleven items. Each correct answer was scored as 1, while incorrect answers were scored as 0, yielding a total knowledge score ranging from 0 to 11. Knowledge levels were categorized as follows: <5, inadequate; 5–8, moderate; and >8, high. The questionnaire underwent a multi-step validation process.
Content validity was evaluated by six healthcare professionals (three family physicians and three pharmacists) who reviewed each item for clinical relevance, comprehensiveness, and alignment with the study objectives. They assessed whether the questions adequately captured knowledge and awareness of herb–drug interactions. Face validity was then assessed by two laypersons who regularly use herbal supplements. They evaluated the questionnaire for clarity, readability, and ease of understanding to ensure it was appropriate for the general public. Based on feedback from both groups, minor modifications were made to wording and structure to improve clarity. Finally, internal consistency reliability was assessed using Cronbach’s alpha, which exceeded 0.7, confirming acceptable reliability. The questionnaire was distributed electronically via social media platforms to reach the general public across Saudi Arabia. Eligible participants were invited to complete the survey after providing informed consent.
Statistical analysis
Data were entered and analyzed using Statistical Analysis Software (SAS®), version 9.4. Descriptive statistics were used to summarize the frequency distributions of questionnaire items. Categorical variables were compared using the chi-square test, while relationships between independent variables (risk factors) and the dependent variable (knowledge score) were assessed using bivariate analyses, including the independent t-test and chi-square test, as appropriate. A p-value of ≤0.05 was considered statistically significant. Multiple linear regression analyses were performed to identify variables associated with participants’ knowledge of the potential adverse effects of herbal products and common herb–drug interactions.
Results
Characteristics of the study sample
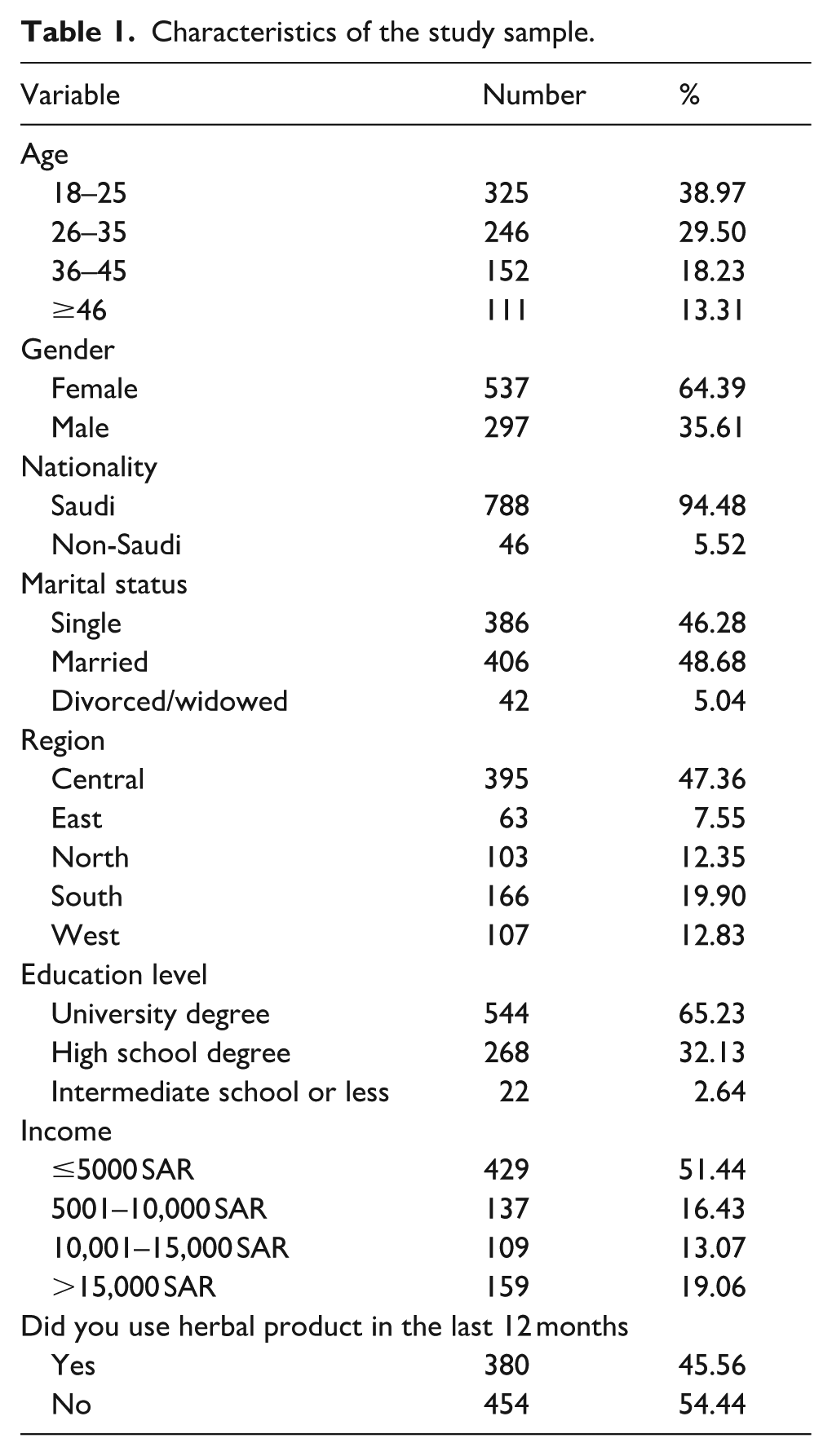
The study was conducted over a 6-month period, during which 834 individuals completed the questionnaire. The majority of respondents were female (64.4%) and nearly half were married (48.7%). Most participants were Saudi nationals (94.5%), and approximately 69% were between 18 and 35 years of age. More than two-thirds (65.2%) held a university degree. In the preceding 12 months, 45.6% of respondents reported using herbal products. The demographic characteristics of the study sample are presented in Table 1.
Characteristics of the study sample.
The knowledge of herb-drug interactions among the public
Table 2 presents the distribution of participants’ responses to the knowledge items. Approximately 63.7% of respondents agreed that herbal products can affect the absorption of medicines, while more than two-thirds (68.0%) acknowledged that herbal products may cause side effects. In contrast, over half of the participants (58.1%) did not agree that consuming grapefruit juice with certain medications could alter their effectiveness, and 55.2% disagreed that taking St. John’s wort concurrently with medications could influence drug efficacy. Conversely, more than half of the respondents (55.0%) agreed that patients receiving anticoagulants should avoid excessive intake of specific foods such as garlic, ginger, broccoli, and spinach.
The knowledge of herb-drug interactions among the public.

Each participant’s knowledge score was calculated as the sum of correct responses across the 11 knowledge items, with possible scores ranging from 0 (lowest knowledge) to 11 (highest knowledge). As shown in Table 3, the majority of respondents (86.0%) scored between 3 and 8 points. Knowledge levels were further categorized, as presented in Table 4. Most participants (59.1%) demonstrated a moderate level of knowledge, while 30.5% had low knowledge. Only 11.6% of respondents achieved a high level of knowledge.
Knowledge calculated score (range 0–11).

Knowledge level classification (low = score 0–4, medium 5–8, high score 9–11).

Participant’s attitude toward drug-herb interactions
In assessing participants’ attitudes toward herb–drug interactions, more than 90% agreed that it is important to be aware of potential interactions between medications and herbal products. Just over half of the respondents (55.4%) reported having previously heard about such interactions, while 46.3% indicated that they had discussed the issue with their physician (Table 5).
Participant’s attitude toward drug-herb interactions.

The potential predictors for drug-herbal interaction knowledge
Table 6 summarizes the predictors of knowledge regarding herb–drug interactions. A negative correlation was observed between age and knowledge levels. Participants aged 18–25 years (p = 0.0488) and 36–45 years (p = 0.0420) demonstrated significantly lower knowledge compared with those aged ≥45 years. Regional differences were also identified, with participants from the western region exhibiting lower knowledge scores than those from the central region (p = 0.0095). In contrast, gender was positively associated with knowledge, as female participants reported significantly higher scores than males (p < 0.0001). Prior use of herbal products was also associated with knowledge scores, with respondents who had used herbal products in the past 12 months demonstrating greater knowledge than non-users (p = 0.0077).
Potential predictors for herbal products knowledge.

Discussion
Herbal remedies are commonly utilized in many nations to prevent and treat health problems. In Saudi Arabia, their use is widespread and influenced by cultural and religious beliefs.6,16 As observed by Alghamdi et al. and Ullah et al., while the Saudi healthcare system is primarily focused on modern medicine, traditional, locally manufactured herbal medicines remain highly popular among the general population.17,18 In our study, approximately 46% of participants reported using herbal products within the last 12 months. This prevalence is consistent with previous research from Jeddah, Saudi Arabia, which reported a usage rate of 42%. 16 However, the safe integration of these products into modern healthcare is challenged by a significant lack of research on potential herb-drug interactions (HDIs). A primary concern is the general population's limited awareness of these interactions. This study evaluated such knowledge and found that a majority (57%) of the Saudi public possessed a medium level of understanding.
A common assumption among patients is that herbal supplements are inherently safe and harmless due to their natural origin. However, clinical evidence demonstrates that not all herbal supplements are safe and that they can produce adverse effects, particularly when co-administered with chronic prescription medications. 19 This risk is compounded by the fact that herbal products do not undergo the same rigorous regulatory scrutiny for safety and efficacy as conventional pharmaceuticals. Al Akeel et al. have emphasized that herbal medications should be approved for use only after a thorough risk-benefit analysis and should be promoted as regulated herbal drugs. 6 Despite these known risks and the existence of governing laws, herbal medications continue to be widely used in Saudi Arabia.
The current study assessed knowledge of HDIs among the general population in Saudi Arabia. Most participants (57.91%) demonstrated a medium level of knowledge, followed by low (30.45%) and high (11.36%) knowledge levels. General awareness of HDIs was relatively high; 63.67% of participants agreed that herbal products could influence the absorption of prescribed drugs, and 68% acknowledged that these products might cause adverse effects. However, knowledge of specific drug-herb interactions was medium to low. For instance, a significant proportion of participants incorrectly believed that grapefruit juice (58%) and St. John's wort (55%) do not interact with prescription drugs. Interestingly, over 55% of participants were aware of potential interactions between anticoagulant drugs and herbal products such as garlic, ginseng, and spinach. These findings align with a study from Hail, Saudi Arabia, which revealed a significant deficit in understanding, with 58.24% of participants exhibiting insufficient knowledge of HDIs. 20 A systematic review further supports this observation, indicating that a substantial proportion of patients concurrently use herbal supplements and prescription medications despite a poor understanding of potential interactions. 21 These results underscore an urgent need for improved patient education and greater clinical vigilance regarding herbal medicine use.
Participants in this study exhibited a positive attitude toward learning about HDIs. The vast majority (90%) believed it is important to be aware of possible interactions between herbal products and prescription drugs, and 55% reported having heard about HDIs. Nonetheless, 53.72% had never inquired with their physician about such interactions. This communication gap is a critical finding, consistent with other research in Saudi Arabia showing that 70% to 91% of patients fail to disclose their herbal supplement use to healthcare providers.7,19,22 This lack of disclosure increases the risk of adverse HDIs. Therefore, healthcare providers must proactively inquire about patients’ use of herbal supplements and educate them on the potential benefits and risks.
Our analysis revealed several demographic predictors of knowledge. Gender was positively associated with knowledge level, with females demonstrating a higher understanding than males. This finding agrees with studies from Saudi Arabia, Nigeria, and Indonesia, which identified gender as a significant factor in herbal medicine knowledge, with women generally being more knowledgeable.16,23,24 Conversely, some studies have found no association between gender and knowledge of herbal therapies. 13 A significant association was also found between knowledge and prior use of herbal products; surprisingly, respondents who had not used herbal products in the previous year demonstrated higher knowledge levels. Furthermore, knowledge of HDIs was negatively correlated with age, with participants aged 45 and below showing lower knowledge than older participants. This may be attributed to older adults' greater experience with and awareness of herbal supplements, as noted in previous research.15,25,26 Geographical location was also a significant factor; participants from the western region exhibited lower knowledge than those from the central region, potentially reflecting the considerably lower usage of and interest in herbal supplements in the west (42%) compared to the central region (over 90%). 27 These predictors highlight the need for targeted educational campaigns to enhance knowledge of herbal products and ensure their safe and effective use.
Our findings highlight significant gaps in public knowledge and awareness regarding HDIs. In the current era of digital health, these gaps could be addressed through innovative digital platforms and online resources. For instance, mobile applications and clinical decision-support tools have been developed to help patients identify and avoid clinically significant HDIs. 28 Similarly, web-based resources can serve as accessible educational tools, though the variability in their quality and reliability must be addressed. 29 The growing role of telemedicine and digital consultations in complementary and alternative medicine also presents an opportunity for healthcare professionals to integrate digital tools into patient education strategies. 30 Together, these digital approaches could complement traditional health education campaigns and significantly strengthen public awareness of HDIs, particularly in a setting like Saudi Arabia where digital health adoption is rapidly advancing.
Strengths and limitations
This cross-sectional descriptive study provides valuable insights into public knowledge and awareness of herb–drug interactions in Saudi Arabia and establishes a foundation for future research and targeted interventions. However, several limitations must be acknowledged. First, the cross-sectional design captures data at a single point in time and therefore cannot establish causality or assess changes in knowledge and awareness over time. Second, the use of social media platforms for questionnaire distribution introduced a sampling bias. Third, although the sample size was relatively large (n = 834), the demographic profile was skewed, with a predominance of female respondents (over 64%) and participants holding a university degree (over 65%). This does not fully represent the general Saudi population and, therefore, limits the generalizability of the findings.
Conclusion
This cross-sectional study conducted among the general public in Saudi Arabia revealed that knowledge regarding herb-drug interactions (HDIs) is predominantly low to medium. Despite the growing popularity of herbal remedies and their frequent concomitant use with prescription medications, public awareness of potential interactions remains inadequate. Our findings underscore an urgent need for targeted educational initiatives and public health campaigns designed to improve awareness of HDIs and promote the safe co-administration of herbal and conventional medicines. To mitigate associated risks, healthcare providers should proactively counsel patients on this topic. Furthermore, we recommend that policymakers and regulatory authorities consider implementing stricter labeling requirements for herbal products to warn consumers of potential interactions. Enhancing public access to credible, easily understandable information on HDIs is also essential to safeguard patient health and support the informed use of herbal supplements within the population.
Supplemental Material
sj-docx-1-phj-10.1177_22799036251390939 – Supplemental material for Public knowledge and awareness of drug-herb interactions: A cross-sectional study in Saudi Arabia
Supplemental material, sj-docx-1-phj-10.1177_22799036251390939 for Public knowledge and awareness of drug-herb interactions: A cross-sectional study in Saudi Arabia by Ahmed A. Albassam, Nehad Jaser Ahmed and Abdullah K. Alahmari in Journal of Public Health Research
Footnotes
Acknowledgements
We would like to extend my heartfelt thanks to Mona Ali Alsubaie and Wateen Mubarak Alshahrani who assisted in collecting data for this research. Their hard work and commitment were crucial to the success of this project, and I am truly grateful for their contributions.
Ethical considerations
Ethical approval for this study was granted by the Ethical Committee of Prince Sattam bin Abdulaziz University (Reference Number: SCBR-475/2025). Informed consent is typically obtained digitally by presenting a detailed consent statement on the first page of the online survey.
Author contributions
A. Albassam: Writing– review & editing, Project administration, Supervision, Methodology, Investigation. N. Ahmed: Writing– original draft, Software, Formal analysis, Conceptualization. A. Alahmari: Validation, Data curation, Resources, Visualization.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to Prince Sattam bin Abdulaziz University for funding this research work through the project number (PSAU/2024/03/31977).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All information and data for this research will be made available upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
