Abstract
Background:
Multidrug-Resistant Tuberculosis (MDR-TB) is fast becoming a major public health concern, with 80% of the reported global MDR-TB deaths occurring in high burden countries including Namibia where drug susceptibility testing is not routinely performed. Previous studies on TB in Namibia have primarily focused on TB and HIV co-infection and MDR-TB development. However, no study to date has specifically examined the epidemiology of MDR-TB mortality or its associated risk factors at a national level. Thus, this study aimed at examining the variation of mortality among MDR-TB patients in Namibia and identifying its risk factors.
Design and methods:
The study adopted a retrospective cohort study design using the 2014–2017 MDR-TB records, and a Gompertz PH model with Gamma (shared) frailty for the frailty modelling of the MDR-TB mortality and its associated risk factors.
Results:
There were more MDR-TB deaths among females, HIV positive patients with pulmonary TB in the Khomas region. MDR-TB mortality was more likely to occur for patients who were aged 55 and above (HR = 3.57, p < 0.001, 95% CI: 2.18–5.91), HIV positive (HR = 2.07, p < 0.001, 95% CI: 1.39–3.08), and from the Khomas (HR = 3.68, p = 0.001, 95% CI: 1.72–7.87), Kunene (HR = 4.45, p = 0.022, 95% CI: 1.24–15.91), Omusati (HR = 2.70, p = 0.022, 95% CI: 1.15–6.31), and Oshana (HR = 2.51, p = 0.021, 95% CI: 1.15–5.48) regions.
Conclusions:
It is therefore recommended that the Namibian government and policy makers consider conducting outreach sessions to increase awareness on MDR-TB including early detection and screening programmes, and patient’s adherence, especially among female patients aged 55 and above, with HIV and those living in these highlighted regions.
Introduction
Tuberculosis (TB) is a chronic respiratory infectious disease caused by the pathogen Mycobacterium tuberculosis and spreads through air droplets by sneezing and coughing of an infected person. 1 It is treatable using drugs such as Isoniazid and Rifampin, two of the most potent TB drugs. 2 However, the bacteria that cause TB can develop resistance to the antimicrobial drugs used to cure the disease. Such TB bacteria is referred to as the Multidrug-Resistant TB (MDR-TB) and is associated with a higher fatality rate, especially among Human Immunodeficiency Virus (HIV) infected patients. 3 MDR-TB is much more difficult and becoming costly to treat than a drug susceptible TB. It has become a major public health problem and an obstacle to global TB control.4,5 Ou et al. 6 reported that globally MDR-TB occurred in an estimated 460,000 cases and resulted in 230,000 deaths in 2017 and accounted for 3.6% of all new cases and 17% of treated cases. In 2015, MDR-TB cases accounted for 16% of new TB cases (about 1 in 6) and 48% of previously treated TB cases (nearly 1 in 2). Of the estimated 74 000 cases of MDR-TB in 2015, only 60% were diagnosed (due to limited access to rapid and quality diagnosis) and treated. Over 2000 cases of Extensively Drug Resistant-Tuberculosis (XDR-TB) in the European region were detected in MDR-TB patients in 2015, meaning that 1 in 4 MDR-TB patients had XDR-TB, with most XDR-TB cases occurring in countries with a high burden of MDR-TB. 7 In Africa, the true burden of Drug resistant TB is poorly described with only 51% of countries having a formal survey completed on it. In the absence of such, modelled estimates were used and a total of 92,629 drug resistant tuberculosis cases were estimated with 42% of these occurring in Nigeria and South Africa. 8
Despite TB being a long-standing and widespread disease, the global burden attributable to TB continues to be a major public health concern. In 2013 alone the estimated new cases of TB worldwide were 9.0 million of which 1.5 million resulted in deaths. 9 According to WHO, 3 80% of these deaths occurred in 30 high-burden countries such as Congo, Kenya, Tanzania, South Africa and Ethiopia, and Namibia is not excluded from such. WHO 2 estimated that about 30% of patients with TB in Namibia go undiagnosed, untreated, or unreported. Additionally, drug resistant TB is one of the greatest threats to ending TB in Namibia. 3 According to the Ministry of Health and Social Services [MoHSS], 10 the number of reported cases of MDR-TB in Namibia increased from 137 in 2014 to 190 in 2015 and to 387 in 2016. This was partly due to the scaling up of laboratory testing for TB drug resistance during the anti-TB drug resistance survey organized by the MoHSS National TB and Leprosy Programme, in 2015. Furthermore, looking at the trend in reported cases of drug resistance in Namibia from 2010 to 2020 inclusive of MDR-TB cases, it was observed that the number of people developing DR-TB and those on treatment has been decreasing from 2017 whilst the number of people with DR-TB who were successfully treated started increasing from 2014. 10 Similarly, looking at the reported cases of MDR-TB from 2007 to 2015, it was revealed that there was an increase in the number of MDR-TB cases between 2007 and 2009 (from 116 to 201) and then in 2014 to 2015 (from 137 to 190), while across the age and sex distribution for 2015, more males between the ages of 25–44 years were resistant to the anti-TB drugs than their female counterparts. 10 MoHSS 10 further states that the treatment outcome in Namibia for MDR-TB patients in 2015 showed that 35% of the MDR-TB patients were cured, 29% had completed their treatment, 21% died, and 10% were lost to follow-up, while 5% were still on treatment, failed or transferred to other facilities.
Although, MDR-TB treatment requires a course of second line drugs for at least 9 months up to 20 months, supported by counselling and monitoring for adverse events, drug susceptibility testing is not routinely performed in Namibia, making the reported prevalence of MDR-TB in the country an underestimate of the true burden. Previous studies on TB in Namibia have primarily focused on TB and HIV co-infection and MDR-TB development. Studies like Ricks et al. 11 identified risk factors for MDR-TB development, such as previous TB treatment and hospitalization, while Shipanga 12 explored spatial clustering of TB mortality in the Erongo region of Namibia. However, no study to date has specifically examined the epidemiology of MDR-TB mortality or its associated risk factors at a national level. Thus, this study aimed at filling this gap by focusing on regional variability, providing insights to inform evidence-based policies and interventions to reduce MDR-TB mortality rates. The identified risk factors associated with deaths among MDR-TB patients from this study can further be considered as useful insights for evidence-based health policies and programmes on TB and MDR-TB in the prevention of high MDR-TB mortality rates in the country, while adding value to the body of scientific knowledge on MDR-TB mortality in Namibia and globally.
Material and methods
The study adopted a retrospective cohort study design using the 2014–2017 MDR-TB records from the MoHSS’s electronic Tuberculosis (eTB) database. The eTB database is a system that was created by MoHSS to provide the monthly summaries information of TB patients across regions in Namibia since 2010. The records of all MDR-TB patients registered on the eTB database from 2014 to 2017 were considered as the sample of the study. The study only used records of patients who were diagnosed with MDR-TB and classified as MDR-TB patients in the eTB database, and not all forms of DR-TB. Figure 1 shows how the sample of the study was selected. A total of 1514 DR-TB patients were recorded from 2014 to 2017 in the eTB database, with 1432 classified as MDR-TB and 82 as XDR in the eTB database. Since this study’s focus was on MDR-TB patients, all patients classified as XDR were excluded from this study. Of the 1432 MDR-TB patients, 1208 were recorded as alive, while 224 were recorded as dead in the eTB database. Likewise, since this study was focusing on MDR-TB mortality, the 1208 patients were classified as censored patients in this study, while the 224 patients were classified as the patients who experienced the event, with the event being death in this study.

Selection of the sample of the study.
Furthermore, Microsoft Excel (2013) was used to clean the data, prior to performing the data analysis of this study, while STATA (version 16) was used to perform the descriptive analysis, survival parametric modelling, and frailty modelling. Data cleaning included the exclusion of all patients classified as XDR from the data since the study’s focus was on MDR-TB patients. Variables within the eTB databases included the time to event variable which was the length of treatment (i.e. the difference between the date the MDR-TB patient started the treatment and the date the patient experienced the event, discharged, lost to follow-up or finished the treatment), as well as the patients’ socio-demographic characteristics such as their sex, age category, and region, and clinical characteristics such as HIV status, treatment type, site of TB disease, number of previous TB treatments, and patient status. Table 1 gives a brief description of the study variables. STROBE checklists were followed in this study (see Supplemental file).
List of study variables.

Throughout the centuries, the survival analysis technique has been popularly used in the biomedical research fields and solely linked to the investigation of mortality rates. With its development dated far back as to the 17th century. 13 In the last few decades, the applications of survival analysis in biomedical researches have been widely used in evidence-based medicine to examine the time-to-event series as well as to predict survival/death events, with the time-to-event series used in illustrating the occurrence time to any dichotomous event. 14 As the year progressed and with more innovative statistical methods being developed and used in the health sciences fields, so were the modifications of the survival analysis technique with the implementation of these innovative statistical methods within its framework. In 1972, David Cox developed a proportional hazard model, which derives robust, consistent, and efficient estimates of covariate effects using the proportional hazards assumption with the baseline hazard rate unspecified.13,15 To date, the Cox Proportional Hazard (CPH) model is the most widely used semi-parametric survival data modelling in the health sciences and medical research fields for investigating the association between the survival time of patients and one or more predictor variables 16 while taking into account the effect of censored observations. However, now and again, the hazard for all the predictor variables can be proportional and there can be more than a single event types under consideration with each individual understudy undergoing exactly one type of event as can be the case for TB patient treatment outcomes (dead, stopped, transferred out, defaulted and interrupted). As a result, more advanced model modification of the CPH model such as the stratified CPH and extended CPH models will be applicable to use for handling the non-proportional hazard while techniques such as the competing risk survival models can be applied to situations with more than one single event type under consideration. Qi 17 suggested techniques such as the Accelerated Failure Time (AFT) model, Weibull and Gompertz models, in addition to the general CPH model, for the analysis of survival time data. Although not commonly used for the analysis of clinical trial data and medicinal researches, the AFT model measures the direct effect of the predictor variables on the survival time instead of the hazard as done in the general CPH model framework.
One of the CPH model assumptions is that the survival times be independent. However, there are situations when these times are not independent due to each individual understudy having some common features or some study characteristics that are shared by more than one observed event time. Thus, the frailty and shared frailty survival models are applicable to these situations. 17 Frailty models are the survival data analogue to regression models, which account for heterogeneity and random effects. A frailty is a latent multiplicative effect on the hazard function and is assumed to have unit mean and variance θ, which is estimated along with the other model parameters, while a frailty model is a heterogeneity model where the frailties are assumed to be individual- or spell-specific. 18 Similarly, Chen et al. 19 discussed that a frailty is an unobservable random effect. For multivariate time-to-event data, it represents the unobserved covariates shared by correlated event times. On the other hand, a shared frailty model is a random effects model where the frailties are common (or shared) among groups of individuals or spells and are randomly distributed across groups. 18 The shared frailty model assumes that the common random effect (frailty) has a multiplicative effect on the individual hazard. Conditional on the frailty, the event times are independent and thus have hazards that are similar to the univariate model. 20
Survival analysis
Survival analysis is a collection of statistical procedures for data analysis for which the outcome variable of interest is time until an event occurs, with time defined in terms of years, months, weeks, or days from the beginning of a follow-up of an individual until an event occurs. Also, time can refer to the age of an individual when an event occurs, with event defined in terms of death, disease incidence, relapse from remission, recovery (e.g. return to work) or any designated experience of interest that may happen to an individual.
21
A survival function

where

accordingly.
22
Theoretically, at the start of the study, since no one has experienced the event yet, the probability of surviving past time 0 will be one, and if the study period increased without limit, eventually nobody would survive, so the survivor curve will eventually fall to zero. The Kaplan-Meier (KM) is the most widely used non-parametric method of estimating equation (1) and utilizes information from both subjects who have experienced an event as well as right-censored subjects. The KM estimator at time

where
Log-rank test
The log-rank test, a nonparametric procedure for comparing two or more survival functions, is a test of the null hypothesis that all the survival functions are the same, versus the alternative that at least one survival function differs from the rest. 24 Kleinbaum and Klein 21 suggested that this (log-rank) statistic, like many other statistics used in other kinds of chi-square tests, makes use of observed versus expected cell counts over categories of outcomes, with the categories for the log-rank statistic defined by each of the ordered failure times for the entire set of data being analysed. The log-rank statistic tests the null hypothesis that at all time points the survival functions for all groups are equal, against the alternative hypothesis that at least one survival function is different from the others for some time periods, with the (general) log-rank statistic given by:

where
Frailty and non-frailty model fittings description
Frailty is a random component designed to account for variability due to unobserved individual-level factors that are otherwise unaccounted for by the other covariates/predictors in the model. Frailty effect, on the other hand, can be defined as when the population level hazard eventually decreases over time because the “at risk group” has an increasing proportion of less frail individuals.
21
Let
Exponential accelerated failure time (AFT) model
Exponential AFT model is used when the researcher assumes a constant failure rate.
25
This model assumes that the hazard rate is constant. In other words, the risk of the event of interest occurring remains the same throughout the period of observation.
21
With scale

for all

for
Weibull proportional hazard (PH) model
The Weibull PH model is a two-parameter model with flexibility to characterize both increasing and decreasing hazard over time. It is commonly used to assess product reliability, analyse life data and model failure times.
26
It has the property that if the AFT assumption holds then the PH assumption also holds (and vice versa). This property is unique to the Weibull model
27
and holds if

for all

for
Log-normal AFT model
Log-normal AFT model is used when the variable is the product of a large number of independent, identically distributed variables in the same way that a normal distribution results when the variable is the sum of a large number of independent, identically distributed variables. 21 This model is not a PH model as the hazard increases to a maximum and then monotonically decreases over time, resulting in a log-normal distribution of the event time. The hazard function of a log-normal AFT model can be defined as:

where

for
Log-logistic AFT model
Log-logistic AFT model is applicable in cases where the logarithmized outcome variable follows a logistic distribution. In survival analysis, it is used to model hazard rates that are initially increasing and finally decreasing.
28
To be precise, it allows the hazard to increase for a shape parameter

for all

for
Gompertz PH model
The Gompertz model is a two-parameter PH function and is similar to the CPH model but has a defined baseline hazard function with a shape parameter
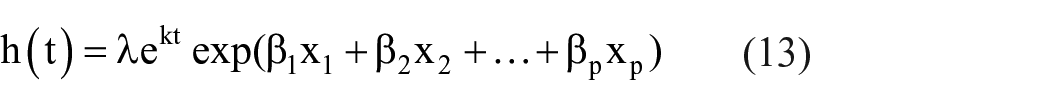
for all

for
Model comparison and diagnostics
In this study, the Akaike Information Criterion (AIC) and Bayesian Information Criteria (BIC) techniques were used to choose the best fit model to use in modelling the MDR-TB mortality among the 10 fitted models (5 with frailty & 5 without frailty) described above. The model with the lowest AIC & BIC values was considered as the better fit and, thus, further used in the identification of the risk factors associated with MDR-TB mortality. In this study, the event of interest was the death of MDR-TB patients during treatment and was coded as 1 for death occurring by TB and 0 for censor (no death). The time to event variable in this study was the length of treatment of MDR-TB patients (calculated as the difference between the date the patient started the treatment and the date the patient experienced the event of interest (death) or when the study ended), while the spatial variation variable was the organization units (i.e. the different hospitals/clinics where the patients were enrolled for TB care, categorized by regions). The censoring variable was the patient status - whether the patient completed the treatment and got discharged, lost to follow-up or died, while the frailty variable was derived from the age variable considering elderly patients (aged 55+). The covariates were the patients’ socio-demographic characteristics (such as sex, age category and region), and clinical characteristics (such as HIV status, site of TB disease, treatment type and number of previous TB treatments).
Ethics considerations
This study was reviewed and approved by the University of Namibia Research Ethics Committee with approval number: SOS-0040, dated 4 March 2022. Permission to use the secondary data from the MoHSS’ eTB database was obtained from MoHSS with approval number: 17/3/3 PMS, dated 30 July 2020 and the collected data was treated with confidentiality. In addition, the data was used for nothing else besides the purpose of the study and the information was not to be revealed to any other parties. The collected data did not contain patient’s names or information that could identify or reveal the patients’ identity. Also, this study followed all ethical standards for research without direct contact with human or animal subjects as there were no names of persons or household addresses recorded in the 2014–2017 MDR-TB data collected for this study.
Results
Descriptive analysis
Out of the 1432 MDR-TB patients considered in this study, a total of 224 (15.6%) events (deaths) and 1208 (84.4%) censored cases were recorded from 2014 to 2017 as shown in Table 2. Of the 1432 MDR-TB patients, 801(55.9%) were males while 631(44.1%) were females, 610 (42.6%) were in the 18–34 age group, 618 (43.2%) were HIV negative, 796 (55.6%) had more than 3 previous TB treatment, 672 (46.9%) were on new TB treatment, and 1331 (93.0%) had pulmonary TB. However, out of the 224 MDR-TB deaths, the majority were among patients aged 35–54 years (n = 106), tested positive for HIV (n = 128) and had pulmonary TB (n = 203). Similarly, 107 deaths were among patients who had more than 3 previous TB treatments, while 92 were among patients who defaulted.
Distribution of MDR-TB patients’ by socio-demographic and clinical characteristics (2014–2017).

Furthermore, it can be concluded that the average duration of deaths among the MDR-TB patients was approximately 5 months and 3 days in 2014–2017, with a lower and upper bound of 5–6 months. Looking at the life table (Table 3), it can be observed that about 92% of the MDR-TB patients survived the first month of treatment, while about 53% survived the fourth and ninth months. From Figure 2, it can be observed that, overall, all MDR-TB patients had high survival chances at the beginning of their treatment, however, their survival rate started decreasing as the duration of their stay (in months) on the TB treatment increases. In other words, the longer a MDR-TB patient stayed on treatment the lower their chances of survival. In addition, the curve showed that mortality hazard was high in the first 3 months, after which the survival rate had reduced to 0.55 (55%) till the end of the study period. In other words, no MDR-TB deaths were recorded after month 4 as the patients had either survived or were lost to follow-up or transferred to other facilities.
Life table of MDR-TB patients.

Beginning total = Number of patients at the beginning of the study.
Time interval is measured in months since diagnosis, that is, length of treatment.
There were no MDR-TB patient cases recorded for 4–7 months duration in 2014–2017.

KM graph of survival time estimate from 2014 to 2017.
Moreover, from the fitted log-rank test for equality of survivor functions, it can be concluded that the survival distributions across the MDR-TB patients’ sex (p = 0.050), number of previous TB treatments done (p = 0.022), HIV status (p < 0.001), treatment type (p < 0.001), region (p = 0.004) and age category (p < 0.001) characteristics differ significantly at a 5% level of significance. However, the survival distributions did not differ significantly across the patients’ site of TB disease (p = 0.169). These significant characteristics were later used in the MDR-TB mortality modelling.
Modelling the risk factors of MDR-TB mortality
Since the observations in this current study were clustered into groups such as regions, in order to account for the unobserved heterogeneity in individual risk to diseases and death, the shared (Gamma) frailty modelling technique was considered when modelling the MDR-TB mortality. To determine the best fit model to use in modelling the MDR-TB mortality, two sets of models were considered – one set was the models with frailty, while the other one was without frailty. As a result, 10 models were fitted, namely: exponential accelerated failure time (AFT) model, Weibull proportional hazard (PH) model, log-normal AFT model, log-logistic AFT model, Gompertz PH model, exponential AFT model with a Gamma frailty, Weibull PH model with a Gamma frailty, log-normal AFT model with a Gamma frailty, log-logistic AFT model with a Gamma frailty and Gompertz PH model with a Gamma frailty, and their respective AIC and BIC values shown in Table 4. Among the models fitted without (Gamma) frailty, it can be seen that the fitted Gompertz PH model (AIC = 1452.83, BIC = 1589.63) had the lowest AIC and BIC values. This meant that a Gompertz PH model was the best fit model to use when modelling the MDR-TB mortality without frailty consideration. Likewise, among the models fitted with frailty, it can be seen that the fitted Gompertz PH model (AIC = 1451.84, BIC = 1604.41) had the lowest AIC and BIC values (Table 4). This meant that a Gompertz PH model with Gamma frailty was the best fit model to use when modelling the MDR-TB mortality with frailty consideration. Thus, it can be concluded that the Gompertz PH model is the best fit model to use for modelling the MDR-TB mortality, with the Gompertz PH with Gamma frailty model the best fit for the frailty modelling of the MDR-TB mortality. Table 5 shows the outputs obtained from the fitted Gompertz PH with and without Gamma frailty models.
AIC and BIC values of the fitted 10 models.
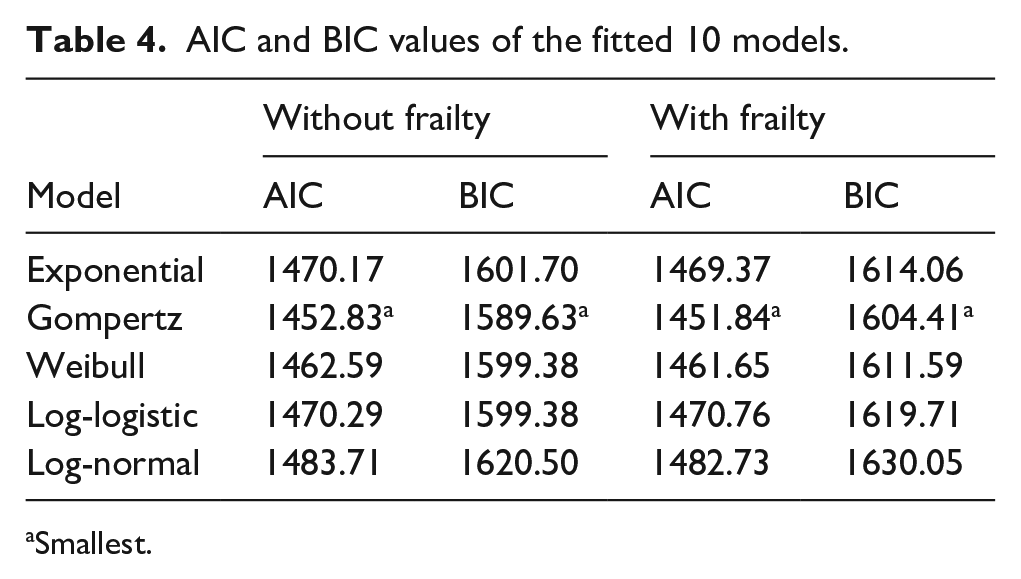
Smallest.
Output from the best fit models.

5% level of significance; (ref) = Reference group.
Gompertz without frailty
From Table 5 at a 5% level of significance, it can be concluded that the MDR-TB patients who were males (HR = 0.70, p = 0.012, 95% CI: 0.53–0.92) had 0.7 times lower risk of mortality compared to the patients who were females. Looking at the age category characteristic, the MDR-TB patients who were aged 35–54 (HR = 1.54, p = 0.009, 95% CI: 1.11–2.13) and 55+ (HR = 3.29, p < 0.001, 95% CI: 2.19–4.95) years had between 1.5 and 3.3 times higher risk of mortality compared to those who were aged 18–34 years. Additionally, MDR-TB patients who were from the Khomas (HR = 3.37, p = 0.001, 95% CI: 1.69–6.75), Kunene (HR = 3.55, p = 0.018, 95% CI: 1.25–10.11), Omusati (HR = 2.46, p = 0.022, 95% CI: 1.14–5.32), and Oshana (HR = 2.43, p = 0.018, 95% CI: 1.17–5.08) regions had between 2.4 and 3.6 times higher risk of mortality compared to the patients from the Erongo region. Similarly, MDR-TB patients who had failure re-treatment (HR = 2.04, p = 0.021, 95% CI: 1.11–3.72) had 2.0 times higher risk of mortality compared to those who had come in after defaulting on their treatment, while those who were new to treatment (HR = 0.66, p = 0.031, 95% CI: 0.45–0.96) had 0.7 times lower risk of mortality. However, MDR-TB patients who were HIV positive (HR = 1.86, p < 0.001, 95% CI: 1.35–2.57) had 1.9 times higher risk of mortality compared to those who were HIV negative.
Gompertz with gamma frailty
From Table 5 at a 5% level of significance, it can be concluded that the MDR-TB patients who were males (HR = 0.68, p = 0.018, 95% CI: 0.50–0.94) had 0.7 times lower risk of mortality compared to the patients who were females. Looking at the age category characteristic, the MDR-TB patients who were aged 35–54 (HR = 1.48, p = 0.029, 95% CI: 1.04–2.11) and 55+ (HR = 3.59, p < 0.001, 95% CI: 2.18–5.91) years had between 1.5 and 3.6 times higher risk of mortality compared to those who were aged 18–34 years. Additionally, MDR-TB patients who were from the Khomas (HR = 3.68, p = 0.001, 95% CI: 1.72–7.87), Kunene (HR = 4.45, p = 0.022, 95% CI: 1.24–15.91), Omusati (HR = 2.70, p = 0.022, 95% CI: 1.15–6.31), and Oshana (HR = 2.51, p = 0.021, 95% CI: 1.15–5.48) regions had between 2.5 and 4.5 times higher risk of mortality compared to those from the Erongo region. Similarly, MDR-TB patients who had failure re-treatment (HR = 2.24, p = 0.023, 95% CI: 1.12–4.50) had 2.2 times higher risk of mortality compared to those who had come in after defaulting on their treatment. However, MDR-TB patients who were new to treatment (HR = 0.65, p = 0.042, 95% CI: 0.42–0.98) had 0.7 times lower risk of mortality. Furthermore, MDR-TB patients who were HIV positive (HR = 2.07, p < 0.001, 95% CI: 1.39–3.08) had 2.1 times higher risk of mortality compared to those who were HIV negative.
Spatial pattern of deaths among MDR-TB patients
Duration of stay
The spatial map in Figure 3 shows the duration (in months) of stay of the MDR-TB patients at hospitals across the different regions in Namibia while on TB treatment. It can be observed from the map that MDR-TB patients tended to stay longer (2–2.7 months) at hospitals in the Otjozondjupa and Kavango regions (dark blue colour), long (1.7–2 months) in hospitals at the Erongo, Omusati, Oshikoto, and Zambezi regions (sky blue colour), but tended to stay for a short period (1–1.7 months) in hospitals at the //Karas, Omaheke and Ohangwena regions (light blue colour) and for a shorter period (0.8 of 1 month) in hospitals at the Khomas, Hardap, Oshana and Kunene regions (very light blue colour). This map appears to be clustered, which suggests that there are spill over effects between regions. Overall, the shortest duration of stay among the MDR-TB patients was 0.8 month whilst the longest duration of stay was 2.7 months.

A clustered map of the MDR-TB patients’ duration of stay variation in Namibia.
Sex and region
Figure 4 shows that there was no difference between the MDR-TB mortality dispersion between males and females. However, there was less female mortality as well as male mortality in the Omusati, Kavango, Zambezi, Erongo, Omaheke and //Karas regions. Figure 5 shows the MDR-TB mortality dispersion across the 14 regions of Namibia. It can be observed from the map that higher MDR-TB mortality cases were recorded in the Hardap, Omaheke and Omusati regions (dark blue colour), while a high number of MDR-TB deaths cases were recorded in the Kavango, Erongo, Zambezi, and Ohangwena regions (sky blue colour). However, a low number of MDR-TB deaths cases were recorded in the Kunene and Khomas regions (light blue colour), while a lower number of MDR-TB mortality cases were recorded in the //Karas, Otjozondjupa, Oshikoto and Oshana regions (very light blue colour).
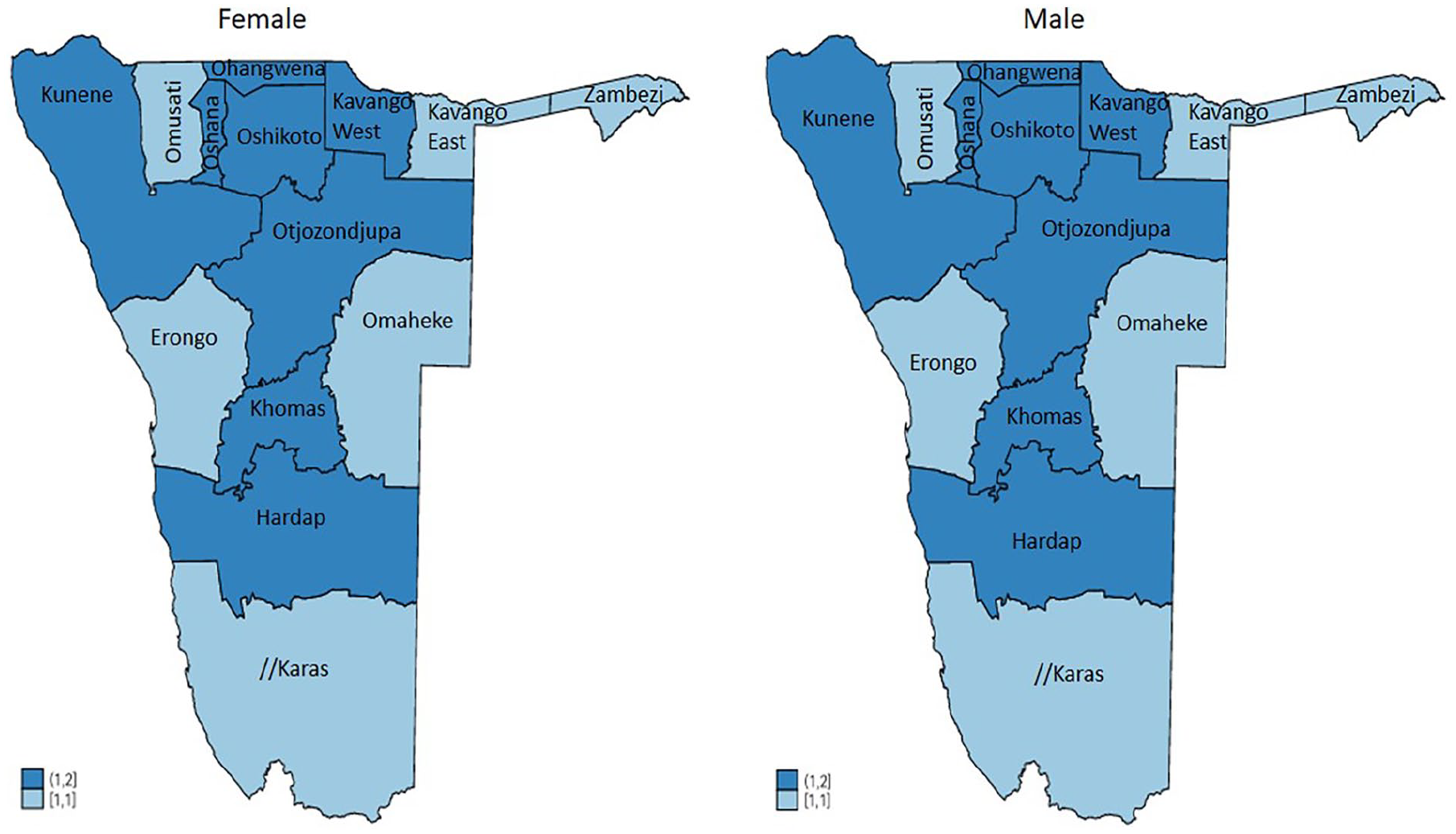
A spatial map for the MDR-TB patients’ mortality variation per sex in Namibia.

A spatial map of MDR-TB patients’ mortality variation per region in Namibia.
Discussion
The main aim of this study was to examine the variation of mortality among MDR-TB patients in Namibia and identify its risk factors. Of the 10 different models fitted to determine the best fit model to use in modelling the MDR-TB mortality, the Gompertz PH model was identified as the best fit model to use, while the Gompertz PH model with Gamma (shared) frailty was identified as the best fit model to use for the frailty modelling of the MDR-TB mortality, due to their minimum AIC and BIC values. Moreover, MDR-TB patient’s characteristics such as sex, age category, HIV status, region, treatment type and number of previous TB treatments had a significant effect on MDR-TB mortality. However, the MDR-TB patient’s site of TB disease did not have a significant effect.
From this study, it was revealed that out of the 1432 MDR-TB patients between 2014 and 2017, the majority were males. This can be due to the high tendencies towards alcohol and drug abuse among men, and the interruption of their medications and treatments due to a number of behavioural and psychosocial factors such as peer pressure, anxiety, and rejection of diagnosis, stigma and cultural beliefs. This finding is similar to the findings found by Limenih and Workie 30 in Ghana who reported that there were more MDR-TB patients among the males than females and that male MDR-TB patients were associated with a high likelihood of experiencing unsuccessful treatment outcomes. This study further revealed that the majority of the MDR-TB deaths recorded were among patients who were aged 35–54 years old, although MDR-TB patients who were at least 55 years old had a higher risk of mortality compared to those who were younger. In other words, as the age of the patient increases, their survival probability decreases. This may be due to the fact that older patients might have underlying conditions such as high blood pressure which lead to weak immune systems, which then lead to poor response to medication. Additionally, the older the patients gets, the more prone they are to contracting other adult-related health illnesses or diseases such as diabetes, arthritis, osteoporosis, stroke and hypertension (to mention a few) thereby affecting their immune system that is already weakened by TB. 31 Similar findings have been observed in a study done by An et al. 32 in China where MDR-TB patients aged 40–60 and 60+ years were identified to be at a significant risk of death.
Furthermore, out of the MDR-TB deaths, the majority were recorded among patients who were diagnosed with HIV, with such patients having higher risk of mortality. This finding is not surprising as HIV weakens the immune system which is supposed to protect the body from any attacks, thereby increasing the risk of mortality 31 in people with TB as they have weaker immune systems than HIV negative patients and the co-infections of TB with HIV can be very fatal on their immune system. Also, HIV positive patients in general have a higher rate of adverse reactions. This study findings concurs with Shipanga 12 and Woya et al. 33 where it was revealed that TB patients co-infected with HIV had shorter survival duration than HIV negative patients, with Woya et al. 33 concluding the same for MDR-TB patients. Likewise, the majority of the MDR-TB deaths recorded were among patients who were from the Khomas and Kavango (East & West) regions. This is not surprising as the Khomas region has the largest population in the country and receives the highest number of MDR-TB and non-MDR-TB patients countrywide (including referrals from other regions) due to it being the capital city with two of the largest state hospitals and the main referral centres in the country with operational implications such as high patient load, huge patient turn over, extensive surgical operation and exhaustive waiting list. While for the Kavango regions, the hospitals are often understaffed, under equipped and lack sufficient medical supplies to adequately treat MDR-TB patients. Additionally, MDR-TB patients’ from the Khomas, Kunene, Omusati, and Oshana regions had a higher risk of mortality compared to those from the Erongo region. This can be attributed to the fact that majority of the hospitals in these regions are referral centres for all patients from clinics and health centres in the regions and from neighbouring regions, regardless of the type of illnesses they are experiencing, which bring about operational challenges in terms of high patient load, manpower, treatment materials and equipment, huge patient turn over and extensive surgical operation among others. However, hospitals in the Omusati, and Oshana regions can be attributed to poor medical facilities operation and shortage of highly skilled personnel.
Moreover, the study revealed that there was a uniform dispersal of high MDR-TB mortality cases among MDR-TB patients who had come in after defaulting on their treatment and those new to treatment. This can be due to the patients’ immune system which might already be compromised at the time of reactivation of their (prescribed) TB treatment while for patients new to treatment, it can be due to the late diagnosis and commencement of their TB treatment. This also concurs with the findings made by Woya et al. 33 where it was concluded that newly enrolled MDR-TB patients were at higher risk of dying than those who were previously enrolled. However, this current study found that there were more MDR-TB mortality cases among MDR-TB patients’ who had more than three previous TB treatments. This can be attributed to weak immune systems, where the body becomes resistant to certain (repeated) drugs.
Limitations
This study made use of secondary data obtained from the MoHSS’ eTB database for the period of 2014–2017 and was limited to available variables within the dataset. As a result, the findings about the geographical differences may have changed and thus interpretations from these must be made with wariness. Furthermore, there was no further information about the 1208 MDR-TB patients recorded as alive within the dataset to identify whether or not these patients included cured or recovered patients. Likewise, since treatment regimen is a key contributor to treatment outcomes and knowing whether or not a patient receives care in an area other than where they live, unfortunately, there were no information captured within the dataset on the treatment regimen used by the patients on treatment during the study period and on their (current) location of residence. Similarly, there were no records of co-morbidities captured within the dataset for the TB patients, including the MDR-TB patients.
Conclusion
MDR-TB mortality is a complex issue that has no simple solution. With its mortality less likely to occur in patients who were males and new to treatment compared to patients who came in after defaulting on their treatment, and more likely to occur in HIV positive patients who were at least 55 years old, from the Khomas, Kunene, Omusati, and Oshana regions compared to patients from the Erongo region, MDR-TB mortality requires intervention from all government and non-governmental organizations. It can be prevented at all costs by implementing an effective directly observed treatment short course strategy. It is therefore recommended that good control measures in hospitals and other congregate settings be implemented to protect HIV positive patients who are at least 55 years old, from the Khomas, Kunene, Omusati, and Oshana regions, as well as other patients and health care workers. All efforts have to be made to provide MDR-TB patients with tailored services and by ensuring family members and treatment supporters are fully involved in the care and support of the patients. Moreover, health care workers should ensure that MDR-TB patients fully understand why their (TB/MDR-TB) treatment should not be interrupted and the consequences of poor adherence (such as poorer prognosis, higher risk of failure, putting family members and contacts at risk, and having to receive more complicated and longer treatments).
In addition, from this research study’s findings, it is strongly recommended that the Namibian government and policy makers consider conducting outreach sessions/courses to increase awareness on MDR-TB including early detection and screening programmes, patient’s adherence and mortality, most especially among female patients aged 55 years and above, with HIV and those living in the Khomas, Kunene, Omusati, and Oshana regions. Also, this research study recommends that more health facilities be built in these regions to take off pressure from already-existing facilities, while at the same time the government should employ more skilled medical personnel to cover backlogs and ensure that sufficient medical supplies are provided to health facilities. Further studies on this topic need to consider other factors that might have an effect on MDR-TB mortality such as COVID-19 status, body weight, pregnancy, lactation, anaemia, nutrition status, smoking, alcohol/drug abuse, and place of work. Likewise, future research can make use of latest TB datasets and use a longitudinal study design which can enable the researcher to focus on a longer study period. Moreover, since this study revealed a high MDR-TB deaths among HIV positive patients with pulmonary TB who have had more than three previous TB treatments, a further epidemiological study is recommended for the possible identification of associated risk factors of these deaths among these specific patient types.
Supplemental Material
sj-pdf-1-phj-10.1177_22799036251336969 – Supplemental material for Frailty modelling for multidrug-resistant tuberculosis mortality in Namibia
Supplemental material, sj-pdf-1-phj-10.1177_22799036251336969 for Frailty modelling for multidrug-resistant tuberculosis mortality in Namibia by Opeoluwa Oyedele and Paulina Mweshitya Shikongo in Journal of Public Health Research
Footnotes
Acknowledgements
The Ministry of Health and Social Services (in Namibia) is thanked for availing the 2014-2017 MDR-TB data.
Ethical considerations
This study was reviewed and approved by the University of Namibia Research Ethics Committee with approval number: SOS-0040, dated 4 March 2022. Permission to use the secondary data from the MoHSS’ eTB database was obtained from MoHSS with approval number: 17/3/3 PMS, dated 30 July 2020 and the collected data was treated with confidentiality. In addition, the data was used for nothing else besides the purpose of the study and the information was not to be revealed to any other parties. The collected data did not contain patient’s names or information that could identify or reveal the patients’ identity. Also, this study followed all ethical standards for research without direct contact with human or animal subjects as there were no names of persons or household addresses recorded in the 2014-2017 MDR-TB data collected for this study.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Conceptualization: PMS and OO; Study design: PMS and OO; Literature search: OO and PMS; Data extraction & verification: PMS; Formal analysis: OO and PMS; Writing – original draft: OO; Writing – review & editing: OO and PMS. All authors reviewed and edited the manuscript and approved the final version of the manuscript for publication.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
