Abstract
Introduction
The Cancer and Aging Research Group (CARG) model predicts chemotherapy-related toxicities in older patients; however, its applicability has not been validated in Taiwanese patients. This study aims to validate the CARG model in older Taiwanese patients with solid tumors.
Methods
Patients (N = 258) aged ≥65 years with solid tumors from a single medical center, slated for first-line chemotherapy, were recruited between 2018 and 2021, with follow-up until December 31, 2022. Patients were categorized into low- (N = 85), medium- (N = 117), and high- (N = 56) risk based on CARG. Validation of CARG involved receiver operating characteristic (ROC) curves. Individual CARG variables were analyzed using univariate analysis for their impact on toxicities and survival.
Results
Toxicities of grades ≥3 were 38.8%, 44.4%, and 67.9% (P = .001) in the three ascending risk groups, and there were significant differences in both hematological (P = .002) and non-hematological (P < .001) toxicities. ROC was 0.631 (95% CI: 0.562-0.700), indicating satisfactory discrimination. One-year overall survival rates were 88.7%, 79.7%, and 63.8%, respectively, in ascending-risk groups, with high-risk groups showing decreased survival (P = .002). In the multivariate analysis, decreased hemoglobin, history of falls, and inability to walk one block remained significantly associated with toxicity. For overall survival, the inability to take medications was the only independent predictor.
Conclusion
This prognostic study validated the CARG model in a heterogeneous solid tumor cohort in Taiwan. In addition to predicting both hematological and non-hematological toxicities, CARG could offer insights into patient survival among older individuals with cancer.
Introduction
Advancements in medicine have led to increased life expectancy, resulting in a growing annual diagnosis of cancer among older patients. In 2022, individuals aged ≥65 years constituted over 9.8% of the global population. 1 By 2030, approximately 70% of cancer diagnoses are projected in individuals aged ≥65 years. 1 Despite the challenges associated with advanced age, effective treatment to enhance meaningful survival should not be discouraged.
Although chemotherapy remains integral to cancer treatment, its efficacy is associated with a substantial incidence of treatment-related adverse events. Older adults, due to poor physical reserve, concomitant comorbidities, and a lack of social support, may become particularly susceptible to chemotherapy-related toxicity. 2 Furthermore, measures to reduce toxicity, such as prescribing reduced dosages, may sometimes compromise treatment efficacy. 3 Therefore, administering standard chemotherapy dosages while minimizing the risk of treatment-related toxicity is crucial.
Geriatric assessment (GA), incorporating dimensions such as physical activity, nutrition, mood, cognition, and social function, is widely employed to identify vulnerable older patients undergoing cancer treatment. 4 Indeed, European Society of Medical Oncology (ESMO) has recommended the implementation of GA and management (GAM) in the care of older cancer patients. 5 The Cancer and Aging Research Group (CARG) previously constructed a model predicting chemotherapy toxicity in older adults with cancer. 6 The CARG model consisted of 11 parameters based on patient characteristics, cancer type, number of chemotherapy drugs, chemotherapeutic dosing, laboratory values, and a GA component. Notably, the CARG demonstrated improved toxicity prediction over the Karnofsky Performance Scale, a finding further validated by the same study group. 7 In a recent prospective study, the CARG was compared to G8 and VES-13 questionnaires, and was found to be more practical in daily use than G8. 8
In the original CARG study, all patients aged ≥65 years with solid tumors scheduled to receive a new chemotherapy regimen were eligible for inclusion. However, Asians constituted less than 5% of the original cohort. 6 This is the first study to the authors’ knowledge to validate the CARG model in a population of older Taiwanese patients with solid tumors.
Materials and Methods
Patients
A total of 258 consecutive patients diagnosed with solid tumors were enrolled between 2018 and 2021 at a medical center in Taiwan. The eligibility criteria included age ≥65 years, a scheduled first-line chemotherapy treatment, the ability to complete GA measures, and informed consent. This study was approved by the Linkou Chang Gung Memorial Hospital institutional review board (201600916B0).
The CARG Model
The original CARG model comprised 11 variables: age, tumor site, number of chemotherapy drugs, dosing, laboratory values, and GA questions. 6 Patients were categorized into three groups based on their total risk score: low- (0-5 points), medium- (6-9 points), and high- (10-19 points) risk, aiming to distribute patients into tertiles.
The GA component included in the CARG model consists of specific questions assessing patient functionality and overall health status. These questions evaluate several key domains: (1) Functional status: Patient-reported ability to perform activities of daily living (ADL), instrumental activities of daily living (IADL), Karnofsky performance status (KPS), incidence of falls, and ability to walk one block; (2) Comorbidities: The total number of medical conditions requiring ongoing treatment or monitoring; (3) Nutritional status: Recent history of unintentional weight loss; (4) Cognitive function: Assessed by the Blessed Orientation-Memory-Concentration test; (5)Psychological state: Evaluated using the Hospital Anxiety and Depression Scale (HADS); and (6) Social support and activity: Patient-reported ability to take medications independently and maintain social activities.
Data Collection
The study procedures mirrored those employed in the original CARG study. All variables in the model were collected prior to planned chemotherapy. Chemotherapy-related toxicities were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (version 3.0) and documented through a chart review. All included patients were followed up until death or December 31, 2022. The same physicians screened the patients as well as provided care for cancer. The reporting of this study conforms to the TRIPOD guidelines for transparent reporting of a multivariable prediction model. 9
Statistical Analysis
Basic patient demographic data are summarized as proportions (%) for categorical variables and medians with interquartile ranges (IQR) for continuous variables. Differences in patient categorical variables between our study cohort and the original CARG cohort were compared using chi-square tests or Fisher’s exact tests if any cell had fewer than five observations.
Observed grades 3-5 toxicity rates among the three predefined groups were compared using a chi-squared test of proportions. The performance of the model was evaluated using the area under the curve (AUC) of the receiver operating characteristic curves. Additionally, the overall survival (OS) of all patients was compared using Kaplan-Meier curves. Log-rank tests were used to determine the differences between survival curves.
All 11 variables from the CARG model were evaluated using univariate logistic or Cox regression analyses to determine the influence of each variable on chemotherapy-related toxicities and OS. All variables in the univariate analysis with P-values of < .05 were further analyzed and included in the multivariate analysis. All statistical analyses were performed using SPSS software (version 23.0; IBM Corp., Armonk, NY, USA). Statistical significance was set at P < .05.
Results
Patient Characteristics
Patient Characteristics.

Several significant differences were observed in patient characteristics between the two cohorts. Our CARG cohort included more patients with gastrointestinal and breast cancers, stage I-III disease, education below the high school level, Asian race, and receiving chemotherapy in a first-line setting.
Geriatric Assessment and Laboratory Values.

ADL, activities of daily living; IADL, independent activities of daily living; KPS, Karnofsky performance status; SD, standard deviation; BMI, body mass index; WBC, white blood cell.
Chemotherapy Toxicity
Grade 3-5 Chemotherapy Toxicity.

ANC, absolute neutrophil count; WBC, white blood cell.
Model Validation
Under CARG risk scoring, the patients were stratified into low-(N = 85, 32.9%), medium-(N = 117, 45.3%), and high-risk (N = 56, 21.7%) groups. The risks of grade 3-5 toxicity were 38.8%, 44.4%, and 67.9% in patients with low-, medium-, and high-risk groups, respectively (P = .001; Figure 1A). The AUC for the mCARG model was 0.631 (95% confidence interval: 0.562-0.700; Supplemental Figure 1). The Relationship Between Risk and Grade 3 or Higher Chemotherapy-Related Toxicities. (A) Incidence of These Toxicities Were Significantly Increased in Higher Risk Groups in Both the Original CARG Cohort (P < .001) and Our Patients (P = .001). (B) Both Hematological (P = .002) and Non-hematological (P < .001) Toxicities had Significantly Increased Incidence by CARG Risk Groups.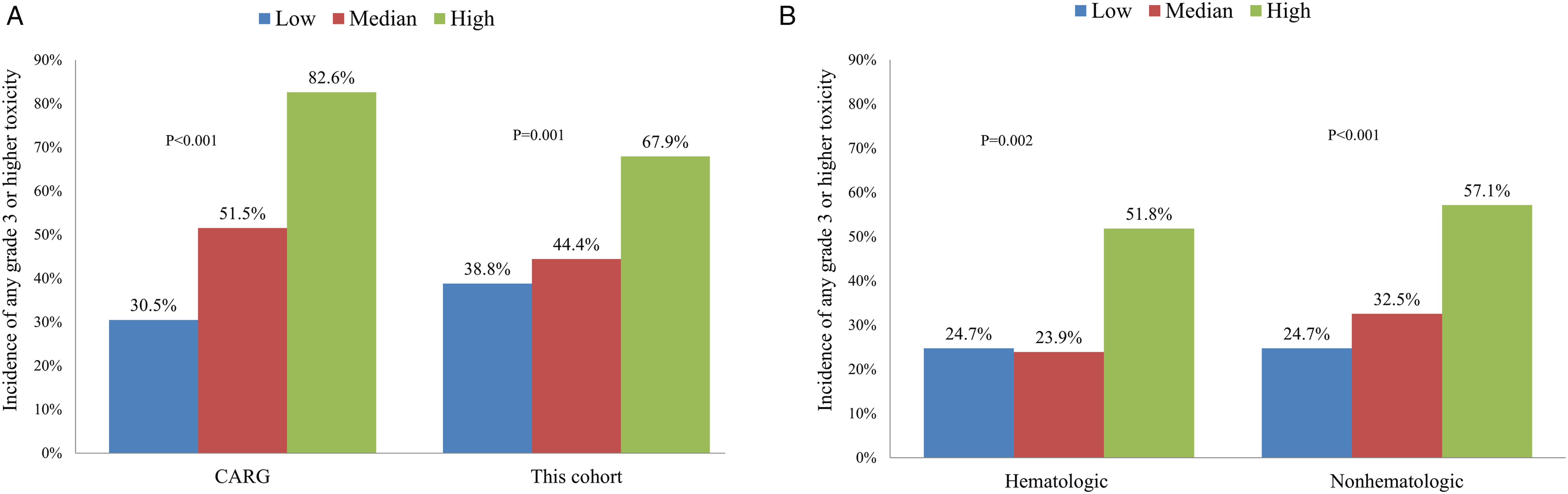
When examining hematological toxicities, our patients showed significantly increased toxicities in ascending risk groups (24.7%, 23.9%, and 51.8%, P = .002; Figure 1B). Similarly, in terms of non-hematological toxicities, patients also demonstrated significantly increased toxicities in the ascending risk groups (24.7%, 32.5%, and 57.1%, P < .001; Figure 1B).
With a median follow-up time of 33 months (range 2-47 months), 67 patients (26.0%) had died by the end of the study. The overall mortality rates were 14.1%, 27.4%, and 41.1% for patients in the low-, medium-, and high-risk groups, respectively (P = .002; Figure 2). Survival of Older Patients Undergoing Chemotherapy After 12, 24, and 36 Months. Patients of Higher Risk Groups had Decreased Survival Compared to the Low-Risk Group.
The hazard ratios were 1.97 (95% CI 1.01-3.82, P = .05) and 3.34 (95% CI 1.66-6.72, P = .001) for medium- and high-risk patients, respectively, compared with low-risk patients.
Univariate and Multivariate Analysis of Toxicity and OS
Univariate and Multivariate Analysis of Grade 3-5 Chemotherapy Toxicity and Overall Survival.

GI, gastrointestinal; GU, genitourinary.
To further explore the independent predictors of toxicity and overall survival, we conducted multivariate analyses including variables that reached statistical significance in univariate models (P < .05). In the multivariate analysis, decreased hemoglobin, history of falls, and inability to walk one block remained significantly associated with toxicity. For overall survival, the inability to take medications was the only independent predictor.
Discussion
This study revealed that chemotherapy toxicity is prevalent in older patients, with nearly half experiencing grade ≥3 adverse events, and 1% succumbing to treatment-related complications. The original CARG study predicted chemotherapy-related toxicity in older patients. Our Taiwanese CARG study similarly demonstrated effective risk discrimination. The model also successfully predicted toxicity by hematological and non-hematological adverse event categories. Additionally, the CARG model successfully predicted overall survival trends in our patients.
Numerous studies have sought to validate the CARG model in diverse populations, often in comparison with other evaluation methods. Suto et al 10 applied CARG to a Japanese population, where the model significantly demonstrated validity in predicting chemotherapy toxicity. 10 Additionally, they observed that Eastern Cooperation Oncology Group (ECOG) performance status did not serve as a reliable predictor. 10 Similarly, in India, a large prospective observational study found CARG to be successful in predicting toxicity, whereas ECOG performance status failed to do so. 11 Another prospective study in Germany, involving 104 patients, compared CARG with Geriatric-8 (G8) and concluded that CARG strongly predicted toxicity, whereas G8 did not. 12 In Canada, CARG was validated in a large population of patients with metastatic castration-resistant prostate cancer. 13 Recently, Pang et al also identified the valid application of CARG in a multiethnic Asian population in Singapore. 14
However, not all studies have corroborated these findings. In a French ELCAPA cohort of patients aged >70 years, Frelaut et al 15 discovered that CARG was a poor predictor of chemotherapy toxicity. 15 Similarly, Chan et al 16 evaluated CARG and G8 in a Hong Kong population 16 and found that both tools were ineffective predictors of toxicities. However, they observed higher CARG scores were associated with unexpected hospitalization. 16 Other factors, such as pain, may be equally crucial in addressing chemotherapy toxicity. 17 These discrepant findings may be explained by the differences in study methodology, patient population, and type of cancer intervention provided.
Similar to the original CARG study, our Taiwanese cohort demonstrated adequate risk discrimination ability. Although the CARG study identified 11 variables significant to toxicity prediction, our study only found 6 of them to be associated with higher toxicities, including age ≥72 years, GU or GI cancer type, polychemotherapy, decreased hemoglobin, falls, and the inability to walk one block. Several factors may have contributed to the disparity in toxicity evaluation between the original CARG cohort and our study. The model, partially relying on patient subjective input, may have been affected by differences in education and cultural variance between the two cohorts, potential masking the ability of the model to predict treatment toxicity in Asian populations. Our cohort primarily possessed educational backgrounds below the high school level, whereas the CARG cohort predominantly held bachelor’s degree or higher. Furthermore, side effects encompass partly subjective parameters such as fatigue, syncope, and neuropathy, serving as vulnerability admissions. Interestingly, fatigue, being the parameter with the greatest subjective influence, exhibited the most prominent distinction between the two groups. This discrepancy may mirror the resilience and cultural attitudes within the population, thereby influencing variations in toxicity.
There were also other observable differences between the original CARG cohort and ours. Notably, our patients had lower stage disease, and were exclusively receiving first-line chemotherapy. In comparison, the original cohort patients had mostly stage IV disease, many of whom were receiving further line treatments. Previous studies have shown that further-line therapies may incur increasing toxicities with diminishing returns on survival. 18 These disparities may have contributed to the numerically lower frequencies of chemotherapy-related toxicities across all risk groups in our study patients.
Interestingly, this study found that the CARG model exhibited significant correlation with OS. However, literature on this phenomenon is currently lacking. Incidentally, a small prospective observational study of non-colorectal gastrointestinal cancer observed that CARG could effectively predict OS. 19 The CARG model incorporated age, sociodemographic factors, tumor and treatment characteristics, laboratory data, and GA variables for toxicity prediction, these same parameters are also applicable for survival prediction in the same population.20,21 Anemia, kidney failure, fall-related injuries—integral components of CARG—may contribute to higher morbidity and mortality.22-24 ECOG performance status of ≥2 would also serve as an indicator of poorer survival. 25
Several of these parameters serve as indicators of frailty. In Shamliyan’s systematic review of 24 studies, weight loss, fatigue/exhaustion, weakness, low physical activity/slowness, and mobility impairment were identified as key parameters for frailty. 26 The review highlighted the prevalence of frailty in geriatric syndromes and its correlation with reduced survival. 26 Handforth et al 27 found that over half of older patients with cancer experienced frailty, leading to increased treatment complications and all-cause mortality. 27 In the CARG model, frailty is represented by factors such as the ability to take medication, walking one block, experiencing falls, and reduced social activity. 6 In this study, the ability to take medication and walking one block were found to be associated with survival. Further research exploring the relationship between CARG scores and survival is warranted.
Several limitations exist within this study. Patient side effects were assessed with CTCAE version 3 instead of version 4 due to the time period of data collection. The role of dose reduction in regards to toxicity is unclear and its data collection incomplete, although monochemotherapy, polychemotherapy, lines of chemotherapy use are recorded for consideration. Additionally, hematological cancers were not included in this study; however, this inclusion may provide more insights into CARG applicability to a wider range of patients with cancer. Future research may include larger numbers of patients with hematological malignancies for broader generalizability of the CARG.
Our CARG investigation focused on a Taiwanese population with mixed solid tumors. In addition to successful validation across all chemotherapy-related toxicities as well as in hematological and non-hematological toxicity subgroups, CARG demonstrated good survival prediction. Therefore, our study suggests that CARG serves as a reliable tool for assessing the adverse effects of chemotherapy on older adults. To further enhance the broader applicability of CARG in patients with universal cancers, additional cohort studies are warranted.
Supplemental Material
Supplemental Material - Validation of a Chemotherapy Toxicity Prediction Model in Older Adults With Cancer in Taiwan
Supplemental Material for Validation of a Chemotherapy Toxicity Prediction Model in Older Adults With Cancer in Taiwan by Chieh-Ying Chang, Yu-Shin Hung, Ming-Chung Kuo, and Wen-Chi Chou in Cancer Control
Footnotes
Authors’ Note
The abstract of this paper was presented at the ASCO Breakthrough Conference as a poster presentation with interim findings. The poster’s abstract was published in ‘Poster Abstracts’ in JCO 42, 223-223 (2024) with DOI:10.1200/JCO.2024.42.23_suppl.223.
Acknowledgements
The authors gratefully acknowledge the assistance of the patients who participated in this study.
Ethical Statement
Author Contributions
Conception and design of study: CYC, YSH, MCK, WCC; Acquisition of data: YSH, MCK; Analysis and interpretation of data: CYC, YSH, MCK, WCC; Drafting of the manuscript: CYC, YSH, MCK, WCC.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grants from the Chang Gung Memorial Hospital at Linkou, Taoyuan, Taiwan, R.O.C. (CMRPG3L1611 and CORPG3N0151); the Ministry of Health and Welfare, Taiwan, R.O.C. (MOHW112-TDU-B-222-124011); and the National Science and Technology Council, Taiwan, R.O.C. (NSTC 111-2314-B-182A-162 and NSTC 112-2314-B-182A-152).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed in the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
