Abstract
Background:
Approximately 50% of infertility cases are attributed to male factors. Acupuncture has long been employed as a complementary therapy to enhance male infertility treatment outcomes. This study aimed to assess the impact of electroacupuncture (EA) therapy on sperm motility and TMSC in male infertility patients.
Design and methods:
This randomized clinical trial involved 30 male infertility patients divided into 2 groups. Consecutive sampling was utilized among men diagnosed with infertility at the Fertility Clinic Sekar, Dr. Moewardi General Hospital, Surakarta. Both groups underwent assessments of sperm motility and TMSC before and after the intervention. The first group received Coenzyme Q, while the second group received Coenzyme Q + EA.
Results:
The Qoenzyme Q + EA group exhibited no significant difference in motility levels before treatment, with an average motility of 41.40% ± 13.33 and a TMSC level of 33.59 × 106 ± 27.91. Post-treatment, motility remarkably increased by 56.40% ± 11.78, and the TMSC level rose by 78.63 × 106 ± 58.38 in the Qoenzyme Q + EA group. Conversely, the Qoenzyme Q pre-treatment group had an average motility of 48.07% ± 15.77 and a TMSC level of 30.20 × 106 ± 34.82. After Coenzyme Q treatment, a significant decrease in motility by 42.80% ± 18.03 and TMSC level by 28.22 × 106 ± 15.16 was observed.
Conclusion:
Combining Coenzyme Q + EA had a more significant impact on sperm motility and TMSC levels than Coenzyme Q alone. These findings underscore the differential effects of Coenzyme Q + EA and Coenzyme Q on sperm motility and TMSC levels, suggesting potential therapeutic implications for male reproductive health. Future studies with larger sample sizes are warranted to validate and expand upon these results.
Introduction
Infertility is a disease characterized by the failure of a partner to clinically initiate pregnancy after 12 months of regular unprotected sexual intercourse. 1 Globally, infertility affects 15% of couples, and male infertility accounts for half of all cases. 2 In addition, male infertility can result in psychological trauma, emotional instability, and mental stress, which can negatively impact the couple. 3
History taking, physical exams, laboratory test findings, and semen analysis are used to diagnose infertility. The Total Motile Sperm Count (TMSC) is calculated by multiplying the motility parameter’s value by the volume and concentration of the sperm and dividing the result by 100%. TMSC, which combines the three sperm parameters of motility, volume, and concentration, can predict spontaneous pregnancy more accurately than the WHO categorization system for sperm analysis. 4
Although commonly utilized, conventional medical therapy does not ultimately improve the semen parameters in male infertility. 5 Therefore, the effectiveness of male infertility treatments has traditionally been increased using complementary therapies, such as acupuncture. 6 According to prior studies, acupuncture has been proven beneficial in raising sperm concentration and motility when used alone or in conjunction with other treatments.7 –9 However, few studies have been found yet that discuss how acupuncture can help patients with male infertility by increasing the quality of their sperm. In addition, differences in sperm motility and TMSC in male infertility patients undergoing electroacupuncture therapy have not been studied.
Methods
Study design
This study follows an experimental design and employs a randomized clinical trial research approach. The study was conducted at the Sekar Fertility Clinic, Dr. Moewardi General Hospital, Surakarta, from March to October 2022.
Participants
The research subjects were men who came to the Sekar Fertility Clinic, Dr. Moewardi General Hospital, who had a complete examination with a diagnosis of infertility according to the inclusion and exclusion criteria. Sperm sampling was done by masturbating in a room close to the laboratory. Time since the last ejaculation was recorded and sperm samples were examined according to the 2021 WHO semen analysis criteria for volume, sperm concentration, total sperm count, percent motile spermatozoa, and percent morphology of normal spermatozoa. 10
Inclusion criteria: Men aged 25–50 years, semen samples from male patients examined at the Fertility Clinic of Dr. Moewardi General Hospital, willing to provide semen samples for analysis before and after intervention, abstinence of 2–7 days, minimum ejaculate volume of 1 ml, no abnormalities found in reproductive organ examinations, no alcohol consumption in the last 12 months. Exclusion criteria: men with azoospermia in semen analysis, testosterone consumption within the previous 3 months, and men who have recently undergone genital surgery within the last 6 months.
Subjects who met the eligibility criteria were randomly divided into two groups with a 1:1 ratio between control and intervention. Randomization was performed using an online randomization generator on the https://www.randomizer.org website. The control group (Coenzyme Q group) consisted of 15 male infertility patients who received standard medical therapy for infertility, including the administration of clomiphene citrate and antioxidants as determined by physician judgment. The intervention group (Coenzyme Q + EA group) also consisted of 15 patients who received standard medical care for infertility in addition to EA.
The size of the subject is calculated according to the formula:
N = sample size in each group
Zα = level of significance or type I error
Zβ = type II error
SD = standard deviation
X1 = mean of the control group, obtained from the literature study
X2 = mean of the treatment group, determined by the researcher
F = estimated attrition rate for the study
Interventions
The sperm sample was subsequently subjected to sperm analysis to assess parameters, including motility and TMSC. Sperm motility refers to the ability of sperm to move effectively. TMSC is calculated by multiplying sperm concentration by the total semen volume and then multiplying by the motility percentage. Both groups were evaluated based on factors such as age, body mass index (BMI), duration of infertility, smoking status, sperm motility, and TMSC values.
Insertion of the needle into the acupuncture points selected based on the meridian system to points CV3 (Zhongji), CV6 (Qihai), ST29 (Guilai), SP6 (Sanyinjiao) bilateral, LI4 (Hegu) bilateral, and ST36 (Zusanli) bilateral, and KI3 (Taixi) bilaterally (Table 1).11,12
Statistical analysis
Age and BMI were analyzed using the Independent t-test for normally distributed data or Mann-Whitney for abnormally distributed data. In addition, the length of infertility, smoking, and fertility rate were analyzed using Chi-Square. If the numerical ratio data is normally distributed, the dependent t-test was performed. In cases where the numerical data for the ratio was not normally distributed, the non-parametric Wilcoxon test was performed. Data were analyzed using the IBM-SPSS version 23 statistical application.
Results
In Table 2, variables age, BMI, length of fertility, smoking, and fertility rate had p-values >0.05.
Baseline characteristics.

EA: Electroacupuncture; SD: Standard deviation.
Based on Table 3, the average motility and TMSC levels in the pre-treatment Coenzyme Q + EA group were 41.40 ± 13.33% and 33.59 ± 27.91 × 106, respectively. Post-treatment Coenzyme Q + EA, the average motility, and TMSC levels increased to 56.40 ± 11.78% and 78.63 ± 58.38 × 106, respectively. In contrast, the pre-treatment Coenzyme Q group had average motility and TMSC levels of 48.07 ± 15.77% and 30.20 ± 34.82 × 106, respectively, which decreased to 42.80 ± 18.03% and 28.22 ± 15.16 × 106, respectively, post-treatment Coenzyme Q (see Figures 1 and 2).
Differences in motility and TMSC values of each group before and after treatment.

TMSC: Total motile sperm count.
Dependent t-test (motility) and Wilcoxon (TMSC).
Significant in p < 0.05.

Differences in Motility Values of the Two Groups Pre-treatment and Post-treatment.
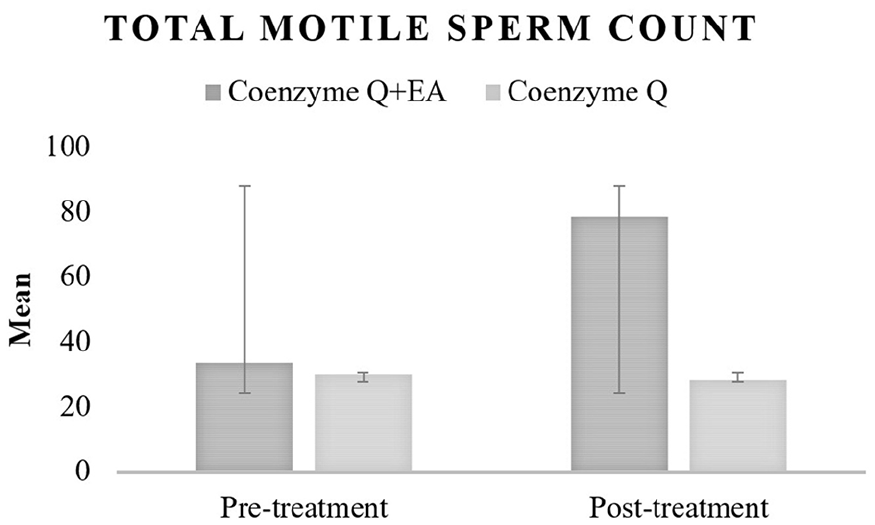
Differences in TMSC Values of the Two Groups, Pre-treatment and Post-treatment.
Significant differences were observed in motility (p < 0.001) and TMSC (p = 0.001) levels between the pre-treatment and post-treatment Coenzyme Q + EA group, indicating a significant improvement in motility and TMSC values among patients receiving Coenzyme Q + EA. In contrast, there were no significant differences in motility (p = 0.203) and TMSC (p = 0.650) levels within the Coenzyme Q group before and after treatment. This suggests that patients receiving Coenzyme Q alone did not experience significant changes in motility and TMSC values (Table 3).
Discussion
There are numerous causes of male infertility. However, the two primary mechanisms underlying infertility will result from these varied circumstances. The first mechanism is the stimulation of cell hypoxia and an increase in the creation of excess ROS by factors such as obesity, immature sperm, radiation, leukocytosis, alcohol, and cigarettes. 13 The second process, conversely, is heat exposure to the scrotum, which might result in heat stress and increased ROS production. 14 These two mechanisms both cause an overabundance of ROS to be produced. The accumulation of ROS leads to two types of processes. The first process is activating the hypothalamic-pituitary-adrenal (HPA) axis and stimulating the hypothalamus to release corticotropin-releasing hormone (CRH). CRH stimulates the production of adrenocorticotropic hormone (ACTH) in the anterior lobe of the pituitary gland. ACTH will affect the adrenal glands to release the hormone cortisol in response to oxidative stress. This hormone negatively affects the anterior pituitary gland, resulting in decreased secretion of LH and FSH. As a result, spermatogenesis will be reduced. 15 The second process, however, is excessive lipid membrane peroxidation that results in defects in cell membranes and mitochondrial malfunction, which inhibits spermatogenesis and sperm motility. 13 Besides the two mechanisms above, there are also theories regarding Ca2+ signaling, which plays an essential role in capacitation, hyperactivation, chemotaxis, and acrosomal reactions. Sperm cells respond to changes in Ca2+ levels through the EF-hand protein, which belongs to the calcium and integrin-binding protein 1 (CIB1) family of proteins. Disruption of Ca2+ signaling by CIB1 and cell cycle regulator cyclin-dependent kinase 1 (CDK1) is associated with idiopathic oligoasthenozoospermia. Dysregulation of these proteins interferes with Sertoli cell proliferation, thereby increasing germ cell apoptosis and causing defects in spermatogenesis. 16
Significant differences in motility and TMSC levels between coenzyme Q + EA pre-treatment and post-treatment suggest that acupuncture treatment after pharmacological therapy for male infertility improves sperm motility and TMSC levels compared to pharmacological treatment alone. This study’s findings align with other studies on acupuncture in improving male infertility. A meta-analysis of seven randomized clinical trials found that acupuncture as an adjuvant therapy could increase the effect of pharmacological treatment, marked by improving sperm motility (SMD = 4.10, 95% CI: 1.09–1.72) and sperm concentration (SMD = 1.07, 95% CI: 0.739−1.40). 8 Another systematic review based on twelve randomized clinical trials reported that acupuncture as a sole or adjuvant therapy had been shown to improve sperm quality. 17 Another meta-analysis examining four randomized clinical trials showed a significantly increased percentage of sperm concentration (p < 0.001, CI 4.91–7.92). 9 A case report by Nareswari et al. 7 reported an increase in sperm motility in one patient from 25% to 67%, changing the patient’s diagnosis from severe oligoasthenoteratozoospermia to severe oligo-teratozoospermia.
Various hypotheses underlie the mechanism of acupuncture in helping improve men’s infertility. The first mechanism is the modulation of the autonomic nervous system at the segmental (spinal) level, which causes activation of the parasympathetic nervous system, resulting in a vasodilating effect that improves circulation and prevents heat stress.11,18 The second mechanism is an increase in the production of antioxidant enzymes, which are superoxide dismutase (SOD) and glutathione peroxidase (GPx) as antioxidant enzymes that inhibit the production of reactive oxygen species (ROS). 7 The third mechanism, however, involves the main level and is characterized by modulation of the hypothalamus-pituitary-adrenal (HPA) axis, which results in an increase in brain endorphins and a decrease in the expression of adrenal Neuropeptide Y. (NPY). Both processes boost the HPG axis’s activity, increasing FSH and LH secretion.15,19 It improved spermatogenesis results from these three pathways. However, the exact method by which acupuncture enhances Ca2+ signaling by CIB1 and CDK1, boosting proliferation, is still unknown. 7
Limitations
This study has significant limitations despite supporting earlier research on the use of acupuncture in male infertility. The study employed a relatively small sample size, albeit meeting the minimum sample size calculation described in the research methodology. This limitation may impact the study’s generalization of its findings to a larger population. Furthermore, confounding factors such as variations in ejaculation timing were not included in the analysis, potentially affecting the observed results. Therefore, future research with a larger sample size and a more comprehensive consideration of confounding factors is warranted.
Conclusion
Combining Coenzyme Q + EA had a more significant impact on sperm motility and TMSC levels than Coenzyme Q alone. These findings emphasize the differential effects of Coenzyme Q + EA and Coenzyme Q on sperm motility and TMSC levels, suggesting potential therapeutic implications for male reproductive health. Future studies with larger sample sizes are needed to further validate and expand on these results.
Footnotes
Acknowledgements
The authors thank you, Universitas Sebelas Maret and Dr. Moewardi General Hospital, for supporting this study.
Author contributions
U.R.B: conception and design, drafting and submission of the manuscript, review and editing, obtained funding, B.M: acquisition of data, review and editing, obtained funding, T.P: review and editing, obtained funding, I.N: intervention, review and editing, obtained funding, A.L: acquisition of data, material support, E.M: acquisition of data, material support, C.H: statistical analysis, material support, L.S: intervention, material support, M.S: intervention, analysis and Interpretation of data, H.P: intervention, analysis and Interpretation of data, A.S.W: recruiting patients, statistical analysis, M.F.A: recruiting patients, statistical analysis. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding is obtained from fund Non-Anggaran Pendapatan dan Belanja Negara (APBN) number 254/UN27.22/PT.01.03/2022.
Ethics approval
This study has been approved by the Research Ethics Committee of Dr. Moewardi Hospital, Surakarta, Indonesia, No. 743/V/HREC/2022. Informed consent was obtained from all participants at enrollment.


