Abstract
Objective:
The aim of this study was to examine the prevalence of morbidity and mortality in patients with seropositive rheumatoid arthritis (RA).
Methods:
Data were obtained from national population-based registries in the period 1998–2009. Prior to the seropositive RA diagnosis (International Classification of Disorders 10th revision M05), we identified a total of 21,558 patients and 87,384 age- and sex-matched control subjects. Odds for morbidity were calculated before and after the RA diagnosis. We estimated the overall survival based on the Kaplan–Meier method.
Results:
Patients with RA had statistically significantly higher odds for a number of co-morbidities prior to the onset of RA including diseases of the musculoskeletal system (odds ratio (OR) 3.10, 95% confidence interval (CI) 3.00–3.21), diseases involving the immune system (OR 1.45, 95% CI 1.29–1.64), endocrine diseases (OR 1.09, 95% CI 1.01–1.17), diseases of the circulatory system (OR 1.08, 95% CI 1.03–1.14) and diseases of the respiratory system (OR 1.30, 95% CI 1.22–1.38), compared with age- and sex-matched control subjects. After the RA diagnosis, the same trend was seen with higher odds for the same co-morbidities. We found a 5-year survival of 80% (95% CI 78–81%) for patients with RA, while for control subjects it was 88% (95% CI 88–89%).
Conclusion:
Compared with age- and sex-matched controls, patients with seropositive RA have higher odds for several co-morbidities prior to and, particularly, after the diagnosis of RA. Furthermore, patients with RA have a lower overall survival compared with age- and sex-matched controls.
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by inflammation, primarily of the small joints of the hands and feet. RA affects approximately 0.3–1% of the adult population worldwide 1 with a peak onset of the disease between 40 years and 70 years of age 2 and the prevalence rises with age. 3 The long-term prognosis of RA has improved over the last decades primarily attributed to early diagnosis and effective pharmacological treatments such as methotrexate and biological agents 4 and has resulted in stable clinical remission or at least low disease activity achievable in most patients. 5 However, the increased life expectancy of the background population has also resulted in patients with RA being older and therefore having longer disease duration. 6 The risk of co-morbidities thus becomes a central issue in RA, particularly as co-morbidity can be a threat to the improvement in the long-term prognosis in patients with RA. A review from 2008 has reported that patients with RA may have more serious co-morbid conditions, for example, ischaemic heart diseases, heart failure, hypertension and infections compared with persons without RA and with significant worse outcomes. 7 Furthermore, other factors of importance for the development of co-morbidity include treatments of glucocorticoids and lifestyle of patients, such as smoking and physical inactivity. 8 –10 It is documented that co-morbidity is associated with more active RA, poorer physical function and lower health-related quality of life 11 and can shorten the life expectancy of patients with RA. 7 The shorter life expectancy in RA has primarily been attributed to the increased incidence of cardiovascular diseases 12 in addition to an increased incidence of infections, osteoporosis, leading to bone fracture and the development of malignancies. 13 The importance of identifying significant predictors for the prognosis in RA has previously been pointed out. 14 The Danish population-based registers provide unique opportunities to gather knowledge about morbidity before and after a diagnosis of RA. Thus, the aim of the present study was to examine morbidity before and after a diagnosis in a national sample of patients with seropositive RA and to estimate the overall survival compared with an age- and sex-matched control population.
Methods and materials
Population
This population-based study includes data obtained from nationwide registries for the period 1998–2009. We followed up patients and control subjects throughout the entire period or until emigration, death or end of follow-up (31 December 2009), whichever comes first. The registries made it possible to trace the information of the patients retrospectively, for example, 3 years before the RA diagnosis and prospectively, for example, 3 years after the diagnosis of RA. In Denmark, all residents since 1968 have been recorded in the civil registration system and unique personal identification numbers ensure accurate linkage of information among the registries. The linkage to the civil registration system makes it possible to trace information of the vital status of patients and control subjects.
In Denmark, all hospital contacts of patients are recorded in the Danish National Patient Registry (NPR) by type and date of contact. NPR was established in 1977 and records administrative information, primary and secondary diagnoses, diagnostic procedures and treatment procedures using the International Classification of Disorders 10th revision (ICD-10) system. Diagnoses from private and public hospitals are recorded in NPR. In the present study, all patients registered in the NPR from 1998 to 2009 with a diagnosis of ICD-10 M05 (seropositive RA) were included. Thus, the study population is representative for all patients in Denmark with a diagnosis of seropositive RA (ICD-10 M05). In accordance with the World Health Organization criteria for ICD-10, we extracted information of morbidity as primary and secondary diagnoses and subdivided into main disease groups. The design for morbidity/mortality evaluation have formerly been described. 15,16
Matched controls
We used data from the civil registration system to randomly select suitable control subjects that could match the patients at the time of diagnosis (ICD-10 M05), with respect to age and sex and geographic location. The controls were matched on the exact year of diagnosis provided in NPR. The controls were required to have no M05 diagnosis before or at the time of matching, and the age matching was done on the exact year. Based on the selected criteria for matching, the ratio between patients and controls was 1:4. The matching was successful for more than 99% of the observations.
Ethical approval
The study was reported to the Danish Data Protection Agency. Data handling did not involve revealing the identity of any patients or control subjects; hence, no ethical approval was required.
Statistical analysis
In the analysis of data, we developed a conditional logit model as a simple logistic regression may overestimate the odds ratio (OR) in a matched case–control design. The explanatory variable was the patient–control group and the dependent variables were the 21 major ICD-10 diagnosis groups. Furthermore, we performed a second analysis with ICD-10 diagnosis occurring in more than 1% of either patients or control subjects. OR and 95% confidence intervals (CIs) were calculated for the main diagnosis. Furthermore, we used the Kaplan–Meier method to estimate the overall survival of patients and control subjects. Finally, based on the Cox proportional hazard model we calculated hazard ratios. A significance level of 0.05 was assumed for all tests. We performed all analyses using SAS 9.1.3 (SAS, Inc., Cary, North Carolina, USA).
Results
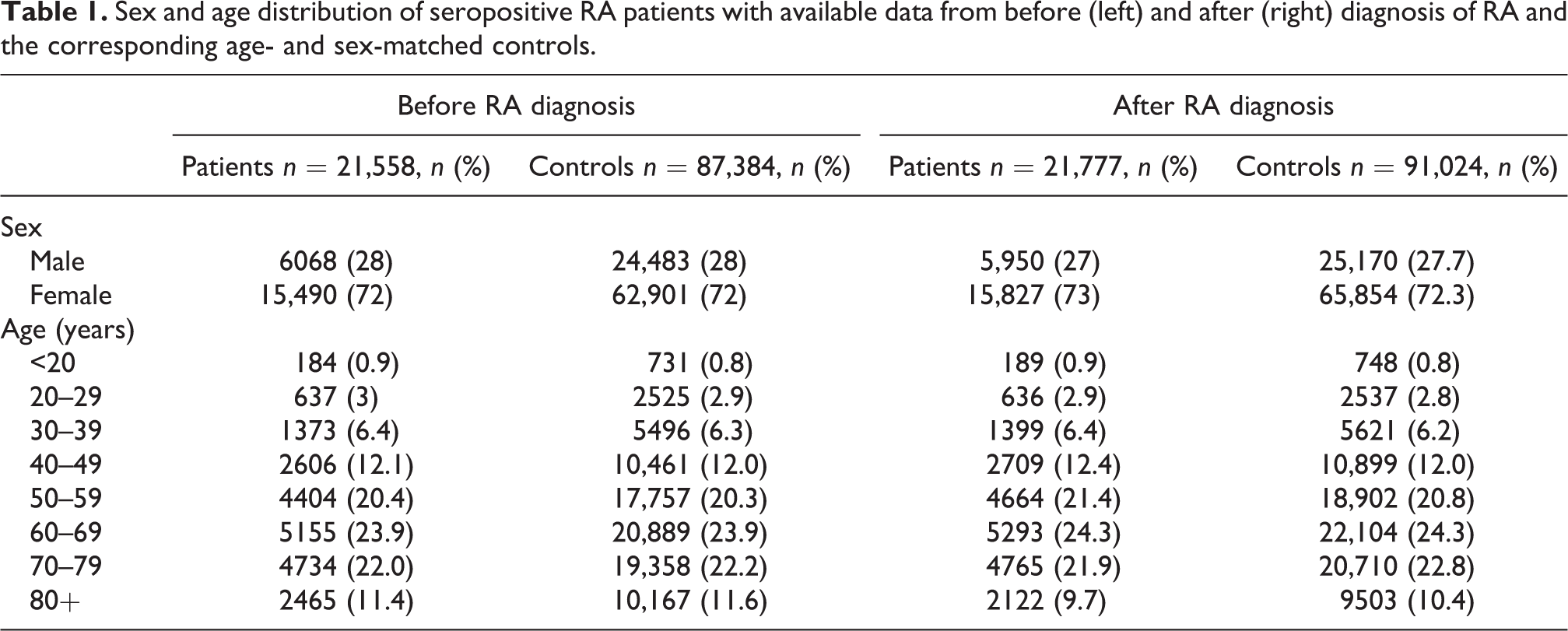
In the present study, we identified 21,558 patients and 87,384 age- and sex-matched control subjects before the diagnosis of seropositive RA, 21,777 patients with RA and 91,024 age- and sex-matched control subjects after the diagnosis of seropositive RA. Table 1 illustrates the sex and age distribution of seropositive RA patients with available data from before and after diagnosis of RA and the corresponding age- and sex-matched controls.
Sex and age distribution of seropositive RA patients with available data from before (left) and after (right) diagnosis of RA and the corresponding age- and sex-matched controls.
Morbidity before and after diagnosis
The morbidities identified in this study population are presented in Table 2. We found higher odds for diseases involving the immune system, for example, primary immunodeficiencies and autoinflammatory disorders (OR 1.45, 95% CI 1.29–1.64) and infectious and parasitic diseases, for example, intestinal infectious diseases, viral infections characterized by skin and mucous membrane lesions and human immunodeficiency virus disease (OR 1.32, 95% CI 1.20–1.46) prior to the RA diagnosis. After an established RA diagnosis, we found even higher odds (OR 1.55, 95% CI 1.41–1.71) and (OR 1.41, 95% CI 1.31–1.53), respectively.
Co-morbidities, divided by ICD-10 classification groups, in patients with seropositive RA and age- and sex-matched controls, and odds ratios for a contact with the health care system due to each ICD-10 classification group.

ICD-10: International Classification of Disorders 10th revision; OR: odds ratio; CI: confidence interval.
aObservations of a diagnosis from the ICD-10 classification group.
For endocrine diseases, primarily consisting of type-2 diabetes mellitus, we found higher odds prior to the RA diagnosis compared with the control population (OR 1.09, 95% CI 1.01–1.17). Furthermore, we found high odds for diseases of the circulatory system, consisting of, for example, hypertension, arterial fibrillation and angina pectoris prior to the diagnosis of RA (OR 1.08, 95% CI 1.03–1.14) and higher odds of diseases of the respiratory system (OR 1.30, 95% CI 1.22–1.38), which mainly consisted of chronic obstructive pulmonary disease and pneumonia. After the RA diagnosis, and compared with control subjects, the same trend was seen with even higher odds for the same co-morbidities as seen prior to the diagnosis of RA (Table 2).
Overall survival of patients with RA and matched control subjects
The overall survival was lower in patients with RA than in control subjects (Figure 1). We found a 5-year survival rate of 80% (95% CI 78–81%) for patients with RA, while for control subjects it was 88% (95% CI 88–89%). The overall survival was persistently about 10% lower for patients with RA compared with control subjects (Figure 1). After 12 years, the probability of survival in patients with RA was 61% (95% CI 61–63%) and for the control subjects 74% (95% CI 74–75%). Furthermore, we found a hazard ratio of 0.59, showing that controls had 41% lower risk of dying compared to patients with RA.

Overall survival of patients with rheumatoid arthritis and age- and sex-matched control subjects.
Discussion
The study population included in this study is based on national population-based registries that have made it possible to identify all existing hospital-registered diagnoses of each patient before and after the established seropositive RA diagnosis. We have shown that patients with RA have higher odds of several co-morbidities a number of years before the diagnosis of RA and documented that the odds of the same co-morbidities are even more increased after the diagnosis, compared with a control population. The large population of patients with RA included in this study confirms a lower overall survival in patients with RA compared with an age- and sex-matched control population.
We found higher odds of diseases of the musculoskeletal system and connective tissue several years before an established RA diagnosis. This suggests that RA may have a long-term preclinical course presenting with musculoskeletal symptoms that are not yet manifested as measurable parameters for a clinical diagnosis of RA, according to the American College of Rheumatology/European League Against Rheumatism classification criteria for RA. 17 In support, increased levels of anti-citrullinated peptide antibodies have been found 2.5 years prior to the clinical diagnosis of patients with RA. 18 Furthermore, in some patients, radiographic joint destruction was present at the time of diagnosis, indicating the presence of joint inflammation in a significant period before the diagnosis. 19
Furthermore, the present study has documented higher odds of diseases involving the immune system and infectious diseases, both before and after the diagnosis of RA. Previously studies have shown that the immune pathology in patients with RA may exist for more than 10 years prior to the onset of the clinical disease. 18,20 Likewise, it is well known that RA increases the odds of infections compared with the healthy controls. 21 So, while treatment with immunosuppressive therapy or corticosteroids may explain the increased odds of infection after the RA diagnosis, the explanation for the increased odds of infection prior to the diagnosis may be found in the presence of immune dysregulation. 22
Our study shows higher odds of diseases of the circulatory system with 12.7% versus 9.4% in patients with RA compared with controls, endocrine disorders 4.3% versus 3.1% and respiratory diseases 6.5% versus 3.8% of patients with RA compared to controls, respectively. In addition, other studies that have compared the prevalence of co-morbidities among RA patients with those in the general population have shown a higher prevalence of cardiovascular events, 23,24 type-2 diabetes mellitus 25 and chronic obstructive pulmonary disease. 26 However, it is remarkable that these diseases were also more frequent prior to the diagnosis of RA. This has not previously been described, although studies have documented co-morbidities in patients with early RA. 27,28 One explanation for this finding might be that a prediagnostic inflammatory process has an impact on the development of RA, type-2 diabetes mellitus and cardiovascular diseases, 27 while another explanation may be that some co-morbidities are attributed to an environment and/or lifestyle behaviour that exist prior to the RA diagnosis. Overall, we showed that the prevalence of co-morbidities were highest among patients after the RA diagnosis. This may be explained by patients with RA have a higher inflammatory state making them at higher risk for both benign and malignant co-morbidities. More studies may examine this association.
In this study, we showed a protective effect of RA diagnosis concerning pregnancy, childbirth and the puerperium. One possible explanation for this finding may be that women with symptoms warranting medical attention are more unlikely to become pregnant.
The lower overall survival in patients with RA, documented in this study, is consistent with previous studies showing a lower survival in patients with RA compared with healthy populations. 29,30 In addition, population-based studies, including a meta-analysis, have documented a lack of improvement in survival over the last four to five decades in patients with RA 31,32 and contradict the significant decline in the expected mortality documented in the general population in the last decades. 33 This suggests that the earlier diagnosis and improvement in therapeutic strategies for patients with RA have not yet had a major impact on the survival for the patients. There are several possible interpretations of the increased mortality in RA. However, co-morbidities are a key explanation. 34 In the present study, the multi-co-morbidity documented in the RA population may contribute with important explanations of the increased mortality. Thus, future studies aiming to reduce multi-co-morbidities in the early onset of the RA may have important implications for the survival rate in the RA population. There is a need for therapeutic as well as lifestyle strategies to prevent development of several morbidities in patients with RA. Furthermore, increased knowledge of determinants contributing to the mortality gap between patients with RA and the general population is urgently needed.
Strengths of this study include the large number of subjects, the long observations period of patients with RA and age- and sex-matched controls as well as the presentation of how co-morbidities are distributed in RA patients in a real-life setting. Data were obtained from national databases, which provide a unique opportunity for sufficient follow-up both retrospectively and prospectively. In Denmark, all patients with a hospital contact are registered in the NPR and the diagnoses included in the database are reported by clinicians working throughout the entire Danish hospital system. However, the study has also some limitations. Although it has been shown that NPR is suitable for epidemiological studies based on records of the RA diagnosis, 35 the validity of the specific diagnoses in this study could not be tested. Furthermore, the database only includes diagnoses made in hospitals, which may have underestimated the number of co-morbidities in our study as the RA diagnosis has been shown to be included in NPR for patients registered as inpatients and for patients with more than one hospital registration with RA. On the other hand, patients with RA may be offered more frequent screening for recognized co-morbidities than the background population, which may increase the observed rate of co-morbidities in our study. It cannot be ruled out that the higher odds documented for co-morbidities in patients with RA after the diagnosis may be, at least partly, due to surveillance bias. Furthermore, we found that 10.9% of RA patients had a RA diagnosis before the ICD-10 M05 arthritis diagnosis. Thus, the inclusion criteria of a diagnosis of M05 may have limited the amount of RA patients in our population with the exclusion of patients diagnosed with M06, that is, rheumatoid factor (RF)-negative RA. Hence, our results may only be generalizable to population with RF-positive RA patients. In this study, we identified patients with RA together with the different co-morbidities from NPR. This restricted the included conditions to patients registered at private or public hospitals and consequently the generalizability may be limited to hospital-based populations. On this background, the number of co-morbidities may possible be even higher than shown in this study. Further, we developed a conditional logistic regression model and matched controls on age, gender and geographic location, thus it is possible that there exists unobserved heterogeneity in factors not included in the available data for this study. We have included a wide range of co-morbidities, and it is important to note that some of the co-morbidities are not chronic conditions. Thus, the severity of the different co-morbidities is necessary to take into account, when interpreting these results. Finally, although the co-morbidities included in this study may explain some or all of the relationship between RA diagnosis and mortality, we did not adjust for pre-RA diagnosis co-morbidities. Thus, this study does not make it possible to determine the independent association between RA and mortality.
Our results confirm a high prevalence of co-morbidities in patients with RA, highlighting the importance of health professionals treating patients with RA have an increased focus on treatments of co-morbidities but also on prevention of these, which may have a positive impact on the survival of patients with RA. Implementation of existing recommendations, for example, concerning cardiovascular diseases 36 and development of recommendations for other morbidities need to be implemented in clinical practice.
In conclusion, patients with seropositive RA have higher odds of a variety of co-morbidities compared with controls, and the odds of these co-morbidities is present several years prior to the diagnosis of RA. Patients with seropositive RA have a lower overall survival than an age- and sex-matched control population. Results from this study support the importance of preventing co-morbidities in patients with RA and adds further evidence that efforts to prevent co-morbidities should be initiated early for people who have high odds of developing RA and for the general health information.
Footnotes
Author contributions
PJ and JK created, initiated and managed the project. KL drafted the manuscript. JK and RI performed the statistical analyses and commented on the manuscript. BAE and MØ commented on the methods and critically revised the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
