Abstract
Background:
Patients with inflammatory rheumatic conditions such as rheumatoid arthritis, polymyalgia rheumatica and ankylosing spondylitis are at increased risk of common comorbidities such as cardiovascular disease, osteoporosis and anxiety and depression which lead to increased morbidity and mortality. These associated morbidities are often un-recognized and under-treated. While patients with other long-term conditions such as diabetes are invited for routine reviews in primary care, which may include identification and management of co-morbidities, at present this does not occur for patients with inflammatory conditions, and thus, opportunities to diagnose and optimally manage these comorbidities are missed.
Objective:
To evaluate the feasibility and acceptability of a nurse-led integrated care review (the INtegrating and improving Care for patients with infLammatory rheUmatological DisordErs in the community (INCLUDE) review) for people with inflammatory rheumatological conditions in primary care.
Design:
A pilot cluster randomized controlled trial will be undertaken to test the feasibility and acceptability of a nurse-led integrated primary care review for identification, assessment and initial management of common comorbidities including cardiovascular disease, osteoporosis and anxiety and depression. A process evaluation will be undertaken using a mixed methods approach including participant self-reported questionnaires, a medical record review, an INCLUDE EMIS template, intervention fidelity checking using audio-recordings of the INCLUDE review consultation and qualitative interviews with patient participants, study nurses and study general practitioners (GPs)
Discussion:
Success of the pilot study will be measured against the engagement, recruitment and study retention rates of both general practices and participants. Acceptability of the INCLUDE review to patients and practitioners and treatment fidelity will be explored using a parallel process evaluation.
Trial Registration:
ISRCTN12765345.
Background
The impact of inflammatory rheumatic conditions including rheumatoid arthritis (RA), psoriatic arthritis (PsA), ankylosing spondylitis (AS), polymyalgia rheumatica (PMR) and giant cell arteritis (GCA) for patients extends far beyond the musculoskeletal system. Patients with these conditions are at an increased risk of common comorbidities such as cardiovascular disease (CVD), osteoporosis and depression, 1 –6 leading to increased morbidity and mortality, which is likely to be due to pre-existing lifestyle factors (e.g. smoking) plus the disease physiology and its treatment. Multimorbidity, the co-occurrence of two or more long-term conditions (LTCs), is increasingly common. 7 People with multimorbidity have reduced quality of life and poorer clinical outcomes, despite using significant health resources and as such effective management of these patients is a priority area for future research. For patients with inflammatory rheumatological conditions, such as RA, these morbidities (particularly CVD, osteoporosis and depression) are more common and more severe, 8 but crucially often less well managed than in the general population. 9 Despite advances in the treatment of RA, excess mortality remains, highlighting the need for aggressive management of comorbidities. 10
There is a progressive move towards providing integrated care in primary care settings, rather than specialist care. Such community-delivered care is frequently nurse-led and aims to provide not only assessment and treatment for the ‘index’ LTC but also a holistic assessment of other comorbid conditions. In UK primary care, management of many LTCs (including diabetes and CVD) is incentivized by the quality and outcomes framework (QOF) component of the GP contract, leading to an improved process of care for those selected QOF conditions, 11 although inflammatory rheumatological conditions are not included within QOF. Hence, routine primary care monitoring of these conditions is not routine, with the exception of RA, where an annual review is a target within QOF although the content of the review is not specified. At present, NICE guidance advocates people with RA should have an ‘annual review’ with a holistic assessment of their condition plus assessment for associated comorbidities (e.g. CVD and osteoporosis). 12 However, this is often fragmented between primary and secondary care, resulting in duplication of some screening, for example, CVD (with cost implications), while other morbidities remain unrecognized and hence untreated. We have previously shown that primary care RA annual reviews focus on CVD and osteoporosis screening (as previous QOF domains) while other key holistic elements, such as case finding for depression, are lacking 13 meaning opportunities for intervention are missed. 14
Nurse-led care is increasingly advocated for management of chronic inflammatory arthritis to increase the number of interventions taken to treat related morbidities. 15,16 To date much of this has been delivered in specialist rheumatology services rather than primary care, but given the multimorbidity expertise in primary care, 17 we hypothesize that delivering nurse-led integrated care reviews for patients with inflammatory rheumatological conditions may provide both patient and cost benefits while enabling earlier identification, intervention and management of multimorbidities. While RA is one of the commonest inflammatory rheumatic disorders consulted for in primary care, 18 and to date has been studied most frequently with regard to impact and assessment of comorbidities, 1,3,5,14 –16 given that similar comorbidities occur across other inflammatory conditions 1–6 we aimed to assess whether such an approach would be feasible across a broader range of inflammatory rheumatological conditions.
Aims and objectives
The overall aim of this pilot study is to evaluate the feasibility and acceptability of a nurse-led integrated care review for people with inflammatory rheumatological conditions in primary care. The primary objective for this pilot study will be to assess the feasibility of conducting a larger scale randomized controlled trial (RCT) by evaluating: – the overall engagement, recruitment and study retention rates of both general practices and participants; – the feasibility and acceptability of delivering the INtegrating and improving Care for patients with infLammatory rheUmatological DisordErs in the community (INCLUDE) review for both patients and practitioners.
Secondary objectives are to estimate the parameters needed for a realistic sample size calculation for a definitive RCT. Decision-making about progression to a full trial will also be informed by the process evaluation, which will examine the acceptability of the intervention as well as assess the fidelity of the intervention delivery. The development of the study protocol followed the Standard Protocol Items: Recommendations for Interventional Trials guidelines 19 and was developed in accordance with published definitions of pilot and feasibility studies. 20
Methods
Patient and practitioner involvement and engagement
The study was developed with input from the Haywood User Group and Research User Groups at Keele. A specific patient and practitioner involvement and engagement (PPIE) group has met to discuss the purpose and content of the proposed intervention to identify patient priorities (which were agreed as CVD, osteoporosis and mood) and to develop and review the questionnaire content. Key health professional stakeholders (primary care, rheumatology and nursing) have participated in an expert group to discuss practicalities of implementing the review and ensure that relevant QOF metrics (e.g. CVD) are captured. Following these, an evidence-based template (INCLUDE computer template based in EMIS GP software) has been produced to support the integrated care review.
Design and setting
The INCLUDE study is a multicentre pilot cluster feasibility study in UK primary care with parallel process evaluation. General practices will be recruited from West Midlands North via the National Institute for Health Research (NIHR) Clinical Research Network (CRN). The units of randomization will be the general practices and the units of observation will be adults aged 18 years and older who have a Read code for one of the five inflammatory rheumatic conditions of interest (RA, PsA, AS, PMR and/or GCA).
Participants
GP practices
GP practices will be recruited through NIHR CRN West Midlands North. Practices are eligible for inclusion if they use the clinical operating system EMIS Web and are willing to take part and provide clinic facilities for the recruitment period.
The balance between scientific considerations and the need for consent is a recognized issue in cluster trials. 21 –24 Following discussion with their practice team, the senior GP partner in each practice will provide informed consent for the practice to participate, acting as ‘guardian’ for patients and their care. GP practice consent to participate will be formalized through written agreements. To avoid selection bias and differential consent, the inclusion and exclusion criteria will be agreed and applied with all participating practices prior to practice sign up and randomization in the study.
Randomization
Randomization to the intervention or control group will be performed at GP practice level using stratified block randomization; stratification will be by practice size (splitting by order of highest/lowest practice sizes) and using block sizes of 2 and/or 4 within each stratum to ensure balanced clusters and individual patient numbers across study arms.
Patients within control practices will continue to receive usual care from their GP practice. Eligible patients within intervention practices will be invited to attend a nurse-led integrated care review (the INCLUDE review) which includes case finding and assessment for common comorbidities such as CVD, osteoporosis and anxiety and depression. The groups will be compared to assess the potential effect of the intervention on a series of outcome measures assessed by questionnaire. Measurements will take place at baseline, 3 and 6 months.
Patient eligibility criteria
Patients will be eligible if they are adults aged 18 years and older registered with a participating general practice who have a Read code in their electronic record for one of the five inflammatory rheumatic conditions of interest (RA, PsA, AS, PMR or GCA) and are capable of giving written informed consent in English. Practice lists will be screened by the lead GP for each practice, prior to randomization of the practice, and vulnerable patients (e.g. patients on the practice register for severe enduring mental health problems or significant cognitive impairment (such as dementia) and/or in the palliative phase of their illness) will be excluded, as will people who reside in a nursing home as alternative arrangements will be in place for their long-term care.
Study procedures
The study flow chart is summarized in Figure 1. Eligible patients at both control and intervention practices will be sent a study baseline pack which includes an invitation letter, Patient Information Sheet, Baseline Questionnaire with consent form and prepaid return envelope in the post from their GP practice. They will be asked to complete and return the questionnaire and consent form. To maximize response rates, reminder postcards will be sent after 2 weeks and a reminder invitation pack will be sent after 4 weeks. The same procedure will be followed for both intervention and control practices. Patients who do not respond or consent at baseline will be considered non-responders or decliners, respectively, and will be not be contacted again. Participants who consent and self-report that they have one of the above named conditions will remain in the study unless they request to be withdrawn. Participants who consent at baseline but self-report that they do not have one of the conditions of interest will be deemed ineligible for the study and excluded. All responders who provide their contact details and consent to the trial at baseline will be sent a postal questionnaire at 3 and 6 months, with reminder mailings following the same procedure as at baseline. Participants will be asked to consent to medical record review. Full general practice medical records of consenting participants will be accessed and securely downloaded to obtain information on consultations, prescriptions, investigations and referrals in the medical records at 12 months.

Study flow chart.
Usual care
All participants (in both intervention and control arms) will continue to receive usual GP care from their general practice (and specialist care if appropriate) for the duration of the study.
Intervention practices
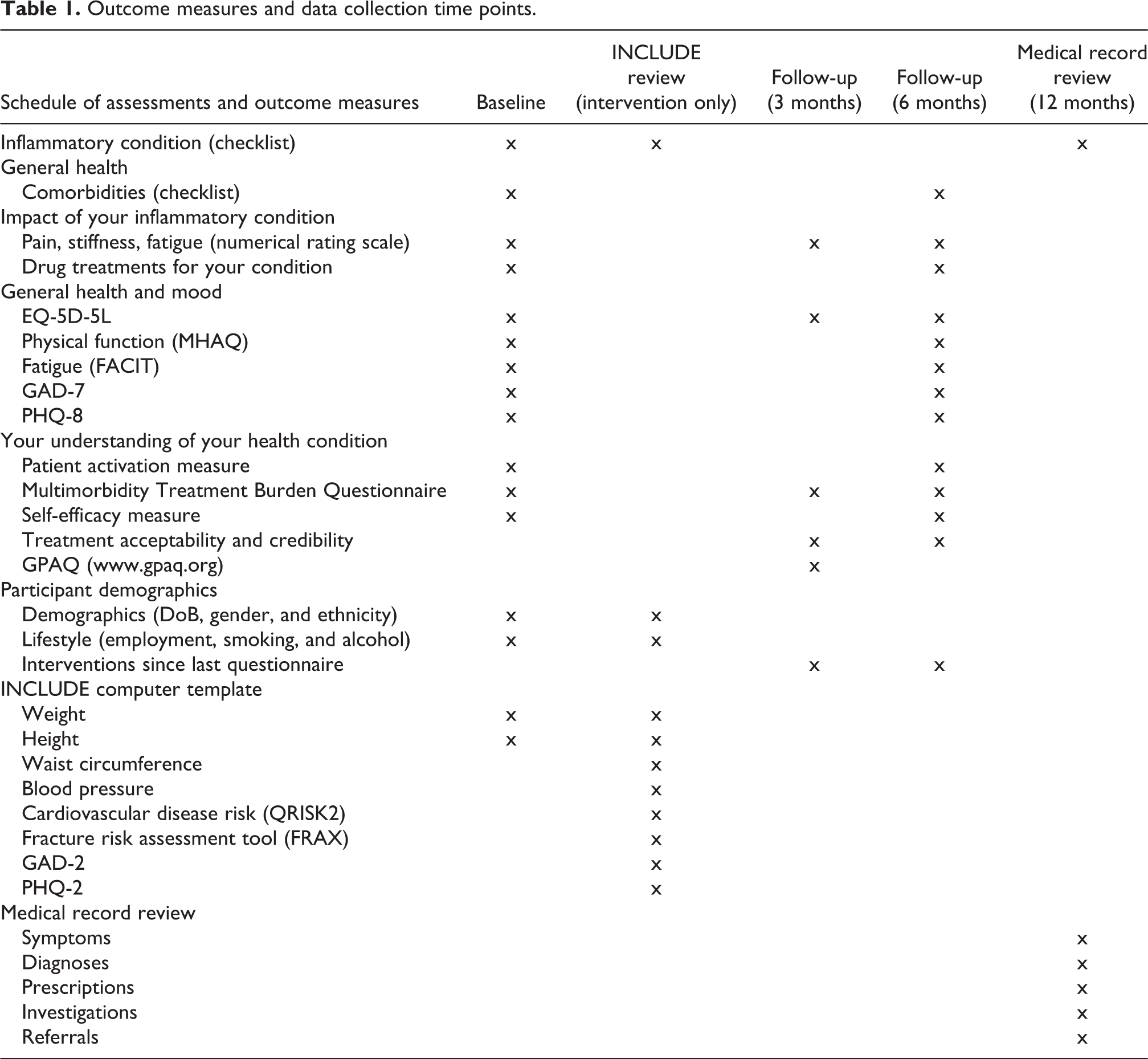
In addition to the questionnaires mailed to all participants, patients from intervention practices who have consented to participate in the study will also be invited to attend an INCLUDE review appointment (the intervention) to take place at their GP practice. This will be a patient-centred consultation, delivered by an INCLUDE study nurse trained to include case finding, identification and assessment of CVD, anxiety and depression and osteoporosis. Outcome measures used in the questionnaire and review are summarized in Table 1. The review will be recorded using an EMIS template specifically developed for the study. Patients will agree an individualized management plan, which could include provision of written advice, self-management support and signposting/referral to appropriate services (e.g. blood pressure monitoring within practice) and be provided with a summary card sheet at the end of the consultation for information.
Outcome measures and data collection time points.
Data collection
Questionnaire outcome measures
Table 1 illustrates the outcome measures to be collected at each time point. In order to retain patients’ anonymity, all data will be non-identifiable and stored in password-protected, encrypted files, separate from signed consent forms. Key domains will include health-related quality of life (EQ-5D-5L), 25,26 pain intensity (numerical rating scale 0–10), physical function Modified Health Assessment Questionnaire (MHAQ), 27 fatigue Functional Assessment of Chronic Illness Therapy (FACIT), 28 anxiety General Anxiety Disorder 7 (GAD7) 29 and depression Patient Health Questionnaire 8 (PHQ8), 30,31 Patient Activation Measure, 32 Multimorbidity Treatment Burden Questionnaire 33 and self-efficacy for Managing Chronic Disease. 34 Items measured at follow-up only will include treatment acceptability and credibility, 35 patient satisfaction General Practice Assessment Questionnaire (GPAQ) 36 and healthcare utilization.
INCLUDE review data
At the INCLUDE review, the nurse will conduct a holistic consultation to include case finding, identification and assessment of CVD (QRISK2), 37 obesity (assessing body mass index), osteoporosis Fracture risk assessment tool (FRAX) 38 and anxiety and depression (GAD2 and PHQ2 with full measures used as appropriate). 29–31 Outcomes in terms of an individualized management plan will be recorded with the patient being provided with a summary sheet at the end of the consultation for information. The review will be recorded using a study specific EMIS computer template (the INCLUDE template) which has been developed with primary care clinicians and health informatics specialists. This INCLUDE template will be saved as part of the patient’s clinical care record within EMIS at the GP practice. The data will be de-identified, downloaded and securely transferred to Keele Clinical Trials Unit (CTU) for analysis.
Medical record review
Entire set of general practice medical records of consenting participants will be accessed and securely downloaded at 12 months after baseline, to obtain information on consultations, prescriptions, investigations and referrals.
Process evaluation
A sample of participants attending the INCLUDE review will be asked to provide informed consent to the intervention being audio-recorded (to assess fidelity of delivery of the intervention). Face-to-face written consent will be obtained by a researcher in the practice waiting room prior to the review. On consent, the participant will be provided with an audio recording card (confirming consent given) to hand to the study nurse. To assess perceptions of acceptability of the review, semi-structured interviews (either face-to-face or by telephone) will be undertaken with a purposive sample of participants following their attendance at the INCLUDE review. Where an INCLUDE review consultation has been recorded, the method of tape-assisted recall (TAR) 39 will be used to prompt discussion in these interviews. All the study nurses and GPs will also be invited to participate in semi-structured interviews. The study nurses will be interviewed twice – once after training to explore their views of the training delivered prior to the start of the study, and again at the end of the intervention period to focus on perceptions of acceptability and feasibility of the INCLUDE review. In these interviews, TAR will be used to stimulate discussion. The topic guide for GP interviews will explore the acceptability, feasibility and impact on routine practice of the INCLUDE review.
Sample size
As this is a pilot trial, a formal sample size calculation is not required. Informed by previous work examining consultation rates for inflammatory disorders in primary care, 18 we aim to recruit approximately 100 participating individuals per arm to ensure that the range of conditions are represented. To achieve this desired sample size, we estimate that 200 patients will need to be invited per arm, allowing for approximately 50% (n = 100) consenting to participate. These assumptions are based on trials with similar recruitment methodology conducted at our research centre previously and will be tested in this pilot trial. We estimate that between two and six GP practices per arm (depending on practice size and demographics) will be required.
Data analysis plan
Quantitative analysis
As a pilot study, analysis will be exploratory and focus upon process outcomes (e.g. recruitment and retention and use of the INCLUDE template). Descriptive statistics will be used to describe the questionnaire response rates between the groups. Analysis will focus on process outcomes (recruitment, retention, intervention fidelity and change in practice through medical record review).
We will assess the number of potentially eligible patients, number of eligible patients, consent rate, retention rate and follow-up rates which will be presented in total and stratified by study arm. The success of the pilot trial will be assessed using criteria adapted from Avery et al.
40
: Recruitment rates: Uptake of eligible patients (red: <25%, amber: uptake 25–50% and Green: uptake >50% of eligible patients); Retention rates: follow-up rates across 3 and 6 months (red: <50%, amber: 50–70% and green: follow-up rate > 70%); Intervention uptake rates (i.e. patient uptake from invitation to INCLUDE review; red: <30%, amber: 30–50% and green: >50%).
We will summarize baseline patient characteristics by study arm and will calculate means and confidence intervals of outcome measures by study arm, and determine which outcome is most sensitive to change over time, in order to inform the sample size calculation for the main trial. No formal statistical testing of superiority of clinical outcome measures between treatment groups will be undertaken: The study will retain a specific focus on assessing/testing the feasibility of the design and methods towards carrying out a main trial and of descriptive evaluation of clinical measures.
Qualitative analysis
Researchers will listen to the audio-recordings of the INCLUDE consultations and use a predefined fidelity checklist specifically developed for the study to assess intervention fidelity. A descriptive analysis will be produced.
The qualitative interviews will be transcribed, and the interview transcripts will form the data which will be analysed by members of the study team adopting a constant comparison approach. 41,42 Themes will be generated through initial coding of text segments, followed by re-coding with meetings between the team to enable discussion and agreement of overarching themes. Triangulation will allow us to compare qualitative and quantitative data in order to give a greater understanding of how the intervention was implemented. Data will also be mapped to the domains of normalization process theory 43 in order to understand the context within which the INCLUDE intervention was implemented in the pilot study.
Ethics approval and dissemination
The study was approved by the Wales REC 5 Research Ethics Committee (REC reference 17/WA/0427). Health Research Authority approval for the study was obtained on 11 January 2018. All participants provided written informed consent. Patient recruitment commenced on 21 March 2018 and is ongoing. The research team intend to disseminate outcomes from the study in peer-reviewed journals and at relevant conferences. The main findings from the study will be available on the Keele University website and displayed on posters in participating GP practices. Participants will have access to study results on request.
Study monitoring
An independent Trial Steering Committee (TSC) has been appointed. Study monitoring will be conducted according to a Trial Monitoring Plan developed by the Trial Management Group based on the trial risk assessment and in accordance with Keele CTU and Sponsor Standard Operating Procedures, and agreed by the TSC. Monitoring will also be undertaken by the approving Research Ethics Committee in the format of annual progress reports and the NIHR Collaborations for Leadership in Applied Health Research and Care West Midlands in the format of quarterly progress reports. The study is deemed a low-risk study since it uses a signposting intervention already common in primary care for other patient groups. As such, the outcomes being signposted from the INCLUDE review are already used in routine clinical practice, and adverse events are uncommon and generally minor. Study clinicians including GPs will be asked to report unexpected and related adverse events and serious adverse events they become aware of during the trial, with the exception of planned hospitalizations, for a period of 3 months following the INCLUDE review only. Reporting procedures will be made clear during the protocol study training and will be contained in site files for all clinicians involved in the study.
Discussion
Inflammatory rheumatological conditions such as RA, PsA, AS, PMR or GCA are associated with a number of morbidities such as CVD, osteoporosis, anxiety and depression which impact negatively on quality of life, yet often these comorbidities are under-recognized and treated.
This article describes the protocol for a pilot RCT with parallel embedded process evaluation aiming to assess the feasibility (eligibility, enrolment and retention rates, barriers to recruitment, questionnaire completion rate and intervention participation) and acceptability of a nurse-led integrated care review (the INCLUDE review) for patients with inflammatory rheumatological conditions in primary care. PPIE has been integral to the development of the study. Results from this study will inform decisions to progress to a full-scale randomized control trial.
Footnotes
Authors’ note
This project was undertaken with the support of Keele CTU Trials Unit, Keele University, UK and NIHR Clinical Research Network West Midlands. The views expressed are those of the author(s) and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health and Social Care.
Acknowledgments
The authors would like to thank all of the patients and staff at the participating general practices, members of the patient and practitioner involvement and engagement groups and members of the Trial Steering Committee for their valuable input in study design and conduct.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article presents independent research jointly funded by the National Institute for Health Research (NIHR) Collaborations for Leadership in Applied Health Research and Care West Midlands (CLAHRC WM) and the Haywood Foundation. CJ, ELH and CCG are part funded by the NIHR CLAHRC WM. CDM is funded by the NIHR CLAHRC WM, the NIHR School for Primary Care Research and an NIHR Research Professorship in General Practice (NIHR-RP-2014-04-026).
