Abstract
Punicalagin, the predominant ellagitannin of pomegranate (Punica granatum L.), shows strong therapeutic potential in chronic diseases, yet medical implementation and therapeutic development remain limited due to poor bioavailability and inconsistent responses in gut-derived metabolites. This review consolidates recent findings (2000–2025) on punicalagin’s biochemical structure, metabolism, therapeutic mechanisms, and delivery strategies. Distinct α/β anomeric forms, together with redox-active hexahydroxydiphenoyl (HHDP) groups linked to a glucose core, underpin punicalagin’s antioxidant and anti-inflammatory activities, with the isomers differing in stability and receptor interactions. Preclinical studies on anticancer demonstrate their effects through NF-κB and STAT3 inhibition, apoptosis induction, and synergy with conventional therapies. Clinical responses vary according to urolithin metabotype, with urolithin A most validated, while urolithin B, C, and D show more limited evidence, highlighting the need for personalized nutrition approaches. Bioavailability has been improved using chitosan nanoparticles and lipid-based self-microemulsifying drug delivery systems (SMEDD), a lipid-based nanoemulsion that enhances lymphatic transport and bypasses first-pass metabolism. Key research areas requiring further study, including standardized preparations, large-scale human trials, clarifying microbiome-dependent variability in urolithin production and sustainable production from pomegranate waste. Punicalagin thus represents a promising nutraceutical candidate, requiring optimized delivery and precision nutrition strategies to achieve clinical adoption.
Introduction
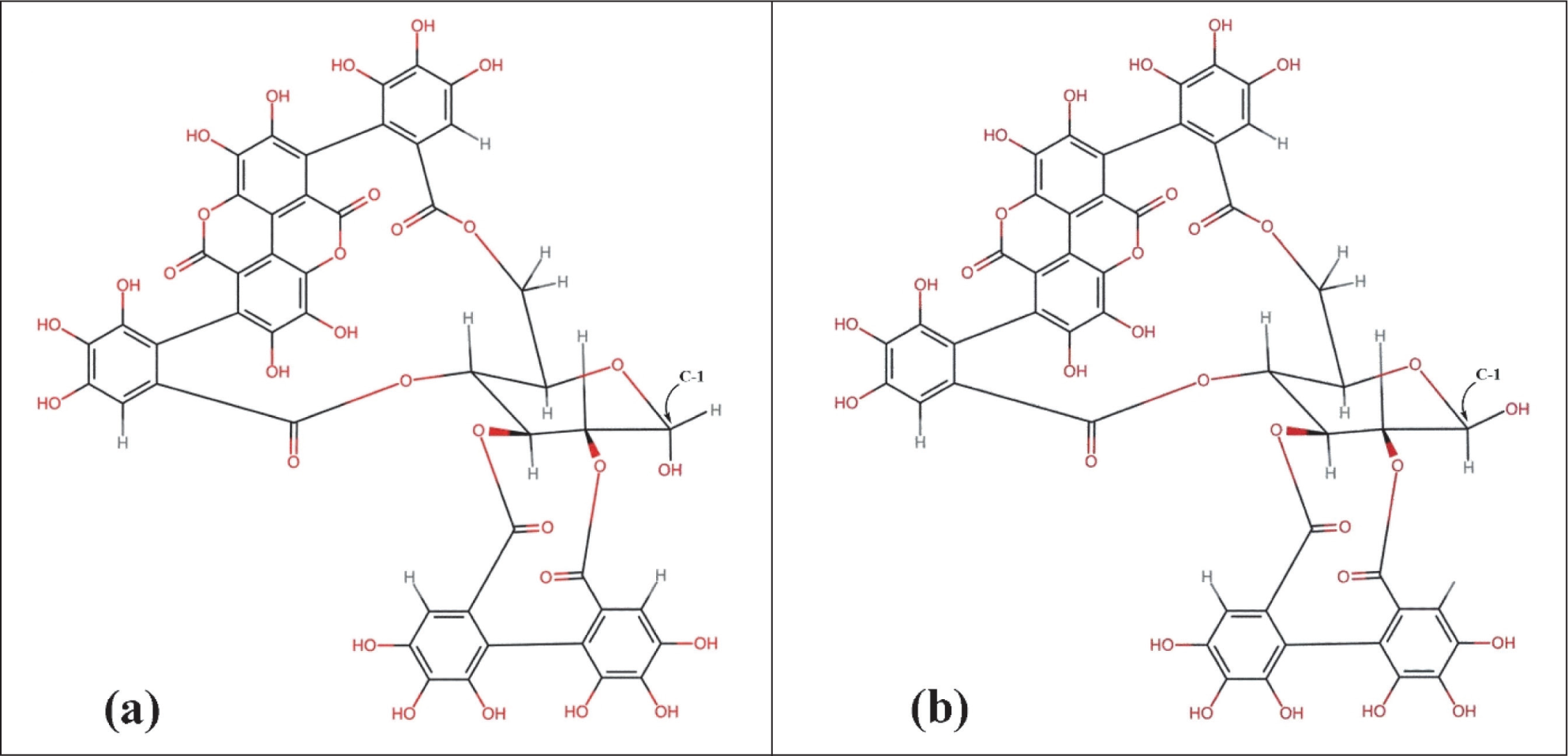
Punicalagin (2,3-(S)-Hexahydroxydiphenoyl-4,6-(S,S)-gallagylglucose, with molecular formula: (C48H28O30) is the most prevalent ellagitannin in pomegranate (Punica granatum L.), mainly robust in the peel and juice. It is the most abundant water-soluble polyphenol with a molecular weight of 1084.7 and is characterized mainly by its structure containing ellagic acid (EA) and HHDP groups (Figure 1). Upon hydrolysis, each HHDP moiety cyclizes to yield EA. Punicalagin represents a dietary ellagitannin, together with its conversion to EA and subsequent microbial metabolites (urolithins). Punicalagin possesses a distinctive structure among ellagitannins, characterized by α and β anomeric isomers that are found due to the open-chain glucose core in its structure. These isomers have slightly different chemical and biological properties that affect the stability, bioavailability, and health effects. 1 Figure1 illustrates α- and β-isomers, which differ in the orientation of their HHDP groups on the glucose core, 2 which influences their chemical stability and susceptibility to hydrolysis. α-punicalagin generally exhibits greater aqueous stability, while β-punicalagin is more prone to hydrolytic cleavage. 3 These structural differences affect their bioavailability, since the rate at which EA is released varies between the two forms. 4 Biologically, this translates into subtle differences in antioxidant capacity, microbial metabolism, and downstream urolithin production,3,5,6 which in turn modulate their health effects.
Structural Comparison of α and β Punicalagin. Each Isomer Features a Glucose Core Esterified with HHDP Groups. The Key Distinction Lies at the Anomeric Carbon (C-1): (a) in α-Punicalagin, the Ester Linkage at C-1 Adopts an Axial (Downward) Orientation; (b) in β-punicalagin, the Linkage Is Equatorial (Upward). This Stereochemical Variation Influences Hydrolytic Stability and Downstream Metabolic Behavior. https://www.ebi.ac.uk/chebi/search?query=%CE%B1-punicalaginChEBI . Modified.
Punicalagin contributes to the antioxidant, anti-inflammatory, and anticancer activities of the pomegranate. Furthermore, it has been shown to have more effective free radical scavenging actions than many other polyphenols. 7 Punicalagin is actively being studied for its influence on gut microbiota, mostly due to its breakdown by intestinal bacteria into urolithins, which have crucial biological activities. 8 There has been widespread investigation of punicalagin in the prevention of chronic diseases such as cardiovascular disorders, diabetes, and cancer, with studies supporting its therapeutic potential. 9 Preclinical evidence supports punicalagin’s role in cardiovascular, neurodegenerative, metabolic, and oncologic contexts, largely through modulation of oxidative stress and inflammatory signaling. 10 According to the latest research, a peculiar isomerization property characterizing punicalagin, where the equilibrium between α- and β-punicalagin determines its biological activity. HPLC-MS and NMR have been used for characterization and tracing of the isomers, and investigation of their metabolic destiny. 11 Although it has a low oral bioavailability, encapsulation and nano-formulation techniques are now being designed to improve its delivery and efficacy. 12 This review aims to thoroughly analyze and describe the current scientific knowledge of the therapeutic potential of punicalagin by studying its structural features, metabolic transformation, and mechanisms of action in chronic diseases. We attempt to explain the relationship between the molecular properties of punicalagin and the observed biological activities, elaborating critically on both the challenges and opportunities for clinical translation. In particular, this work will orient in designing a translational trajectory for reconciling demonstrated preclinical efficacy against pharmacokinetic constraints, while exploring newer methods for bioavailability improvement and therapeutic application.
Methodology
This review synthesizes evidence from a vast array of scientific literature on punicalagin via scanning key databases such as PubMed, Web of Science, Scopus, and Google Scholar. The review considered studies between the years 2000 and 2025 in order to better understand the transformation of views regarding the therapeutic potential of punicalagin. Although it does not strictly conform to all elements of a systematic review, greater priority was given to original research articles exploring punicalagin’s mechanisms of action, bioavailability challenges, and clinical applications. The literature was searched using keywords such as “punicalagin,” “pomegranate ellagitannins,” and “urolithins,” along with words describing their “therapeutic effects,” “bioavailability,” and disease conditions. Priority was given to studies that had a strong experimental design, especially characterization of compounds using HPLC-MS or NMR, well-established in vitro/in vivo models, and clinical trials with clearly defined measures as outcomes, being selected from an initial list of relevant publications. Research was emphasized regarding punicalagin’s interactions with biological pathways (e.g., NF-κB, PI3K/AKT) and its strategies to overcome the pharmacokinetic limitations of this molecule (e.g., nano-formulations, microbiota modulation). Findings were then categorized thematically by disease area (cancer, metabolism, neurodegeneration) and mechanistic insight on these was backed by multiple experimental approaches. Statistical results from the original works were kept to draw interpretive conclusions, including effect size and significance levels. This narrative format enabled an integrative take on punicalagin’s multitargeted bioactivities, with an eye on the translational challenges faced by the compound.
Bioactivity Driven by Metabolites Despite Low Oral Bioavailability
Despite punicalagin having potent biological activities, it presents very little systemic bioavailability in its unaltered form through oral ingestion. However, punicalagin undergoes extensive metabolism in the gut, with the microorganisms there generating EA via hydrolysis, which gets further processed into urolithins (like urolithin A and urolithin B). The metabolites, particularly urolithins, have improved bioavailability and can stay in circulation for a longer time when compared to the parent compound, contributing to the health benefits observed for punicalagin. 13
The explanation for the low oral bioavailability of punicalagin remains its high molecular weight and poor GI absorption. Its microbially produced metabolites have demonstrated potent anti-inflammatory, antioxidative, and anticancer effects. Urolithins prevent chronic diseases by modulating cellular pathways that include NF-κB and Nrf2, cancer, cardiovascular disorders, and neurodegenerative disorders. 14 Newly provided evidence partly explains why interindividual differences in gut microbiota composition could largely contribute to differential urolithin production and ultimately to the differing effectiveness of punicalagin in individuals. 15 The efforts to enhance punicalagin bioavailability have been via encapsulation (nanoparticles, liposomes, etc.) or the simultaneous administration of probiotics to enhance their conversion. 16 Punicalagin continues to be considered a nutraceutical of great potential, even with the barrier of pharmacokinetics, its metabolites being the actual efficacy-bearing components.
Bridging Structural Biochemistry and Therapeutic Applications in Chronic Diseases
At the molecular level, punicalagin and its metabolites (EA and urolithins) modulate key cellular mechanisms implicated in chronic diseases. For example, in cancer models, punicalagin has been reported to inhibit pro-survival signaling pathways, including PI3K/AKT and NF-κB, while promoting apoptosis through activation of p53. These effects are cancer-specific and should not be extrapolated to other diseases without mechanistic evidence.6,17 It has beneficial effects on cardiovascular diseases by improving endothelial function through enhancing nitric oxide (NO) bioavailability and reducing oxidative stress in vascular tissues. 18 Although punicalagin does not cross the blood–brain barrier (BBB), it has been shown to strengthen BBB integrity by reducing oxidative stress and inflammation, thereby exerting indirect neuroprotective effects. Punicalagin may hold potential in managing Alzheimer’s and Parkinson’s diseases by reducing neuroinflammation through microglial modulation. 19
Structural biology and molecular docking studies have shown that punicalagin’s polyphenolic scaffold can bind amyloid-beta fibrils, stabilizing non-toxic conformations and reducing aggregation. 19 Similarly, docking and biochemical assays demonstrate that punicalagin interacts with lipid peroxidation by-products and inflammatory mediators, attenuating oxidative damage in atherosclerosis.9,19 On the other hand, it is this poor bioavailability that has limited its therapeutic application, and consequently, such a problem has had to be circumvented by developing new delivery systems that increase its efficacy, such as nanoparticle encapsulation or complementing it with probiotics.20,21 Thus, integrating the structural biochemistry of punicalagin with the mechanistic understanding of the implicated disease pathways as a multifunctional agent for chronic disease prevention and supportive therapy.
Structure-activity Relationship of Punicalagin
One of the remarkable bioactivities of punicalagin is an inherent property resulting from the distinctive structure of ellagitannins in its structure and subsequent binding to reactive oxygen species (ROS), cellular targets, and metabolic enzymes. Punicalagin’s Structure-Activity Relationship depicts some key structural motifs implicated in its antioxidant, anti-inflammatory, and therapeutic properties.
Molecular Determinants of Antioxidant Capacity
The HHDP moiety of punicalagin contains multiple ortho-dihydroxyl (-OH) groups that serve as the primary structural determinants of its potent antioxidant activity. 22 These ortho-substituted phenolic groups enable efficient electron delocalization through resonance stabilization, significantly enhancing punicalagin’s capacity to neutralize ROS. 23 By having ortho-dihydroxyl groups, punicalagin depends on two oxidative mechanisms, hydrogen atom transfer (HAT) and single electron transfer (SET), to neutralize free radicals like hydroxyl (•OH), superoxide (O₂•⁻), and peroxyl (ROO•) radicals. 24 The ortho-dihydroxyl groups themselves allow the molecule to act as a strong metal chelator, especially for transition metals such as Fe²⁺ and Cu²⁺, preventing the formation of hydroxyl radicals through metal-catalyzed Fenton reactions. 25 Thus, both the scavenging of radicals and the chelation of metals by the HHDP group are crucial functions for punicalagin in reducing oxidative stress in biological systems. 26 Studies with structurally fewer complex analogs have confirmed the importance of the ortho-dihydroxyl groups in punicalagin’s redox activity: the elimination or methylation of these groups clearly diminished its antioxidant potency. 27
The glycosidic linkage of punicalagin’s EA units to a glucose core, together with extensive HHDP esterification, confers greater antioxidant capacity than free EA. 9 This structure-induced improvement in punicalagin solubility and stability in aqueous systems is critical for its bioavailability and biological activity. 20 The bulky glucose moiety creates steric hindrance for the phenoxyl radical intermediate formed in antioxidant reactions, thereby prolonging the radical scavenging activity of punicalagin. 14
In oxygen radical absorbance capacity (ORAC) assays, punicalagin exhibits substantially greater activity (~37,000 μmol TE/g) than free EA (~8,000 μmol TE/g) due to the presence of multiple redox-active sites in punicalagin’s structure. 15 The glucose bond also affects the metabolic fate of punicalagin because it has to be hydrolyzed enzymatically by gut microbiota to liberate EA, which is then metabolized to urolithins. 18 Thus, this controlled release mechanism offers sustained antioxidant activities in vivo, in contrast to the rapid absorption and metabolism of free EA. 19
Moreover, the glucose moiety may interact with cellular membranes and proteins, thereby helping punicalagin modulate certain signaling pathways in addition to its direct antioxidant effects. 20 These structural advantages carry punicalagin forward as a potentially better candidate than its aglycone counterpart for therapeutic intervention in oxidative stress-related ailments. 28
Anti-inflammatory Interaction of Structure-activity Relationship in Punicalagin
The anti-inflammatory action of punicalagin has been demonstrated in defined experimental contexts. This activity occurs due to properties imparted by punicalagin interacting with some of the chief inflammatory pathways (Table 1), such as TLR4 and NF-κB signaling, or oxidative stress induced by metal ions. In particular, the glycosidic bond anomeric configuration (α or β) can strongly influence punicalagin interactions with inflammation receptors and transcription factors. In RAW 264.7 macrophages, punicalagin (10–50 μM, pure compound) suppressed LPS-induced activation of TLR4 and downstream NF-κB/MAPK signaling. 29 Due to its optimal spatial fitting with the TLR4/MD-2 complex, the β-anomeric form of punicalagin binds to TLR4 with a higher affinity than does the α-anomer; 25 consequently, it interferes with the LPS-induced dimerization of TLR4, thereby preventing the downstream activation of NF-κB and MAPK signaling. 30 In vivo, oral administration of punicalagin (50 mg/kg/day) reduced NF-κB activation in a collagen-induced arthritis mouse model. 31 These findings highlight that punicalagin’s anti-inflammatory effects are context-dependent and supported by primary experimental evidence.
Experimental Evidence for Anti-inflammatory Mechanisms of Punicalagin.
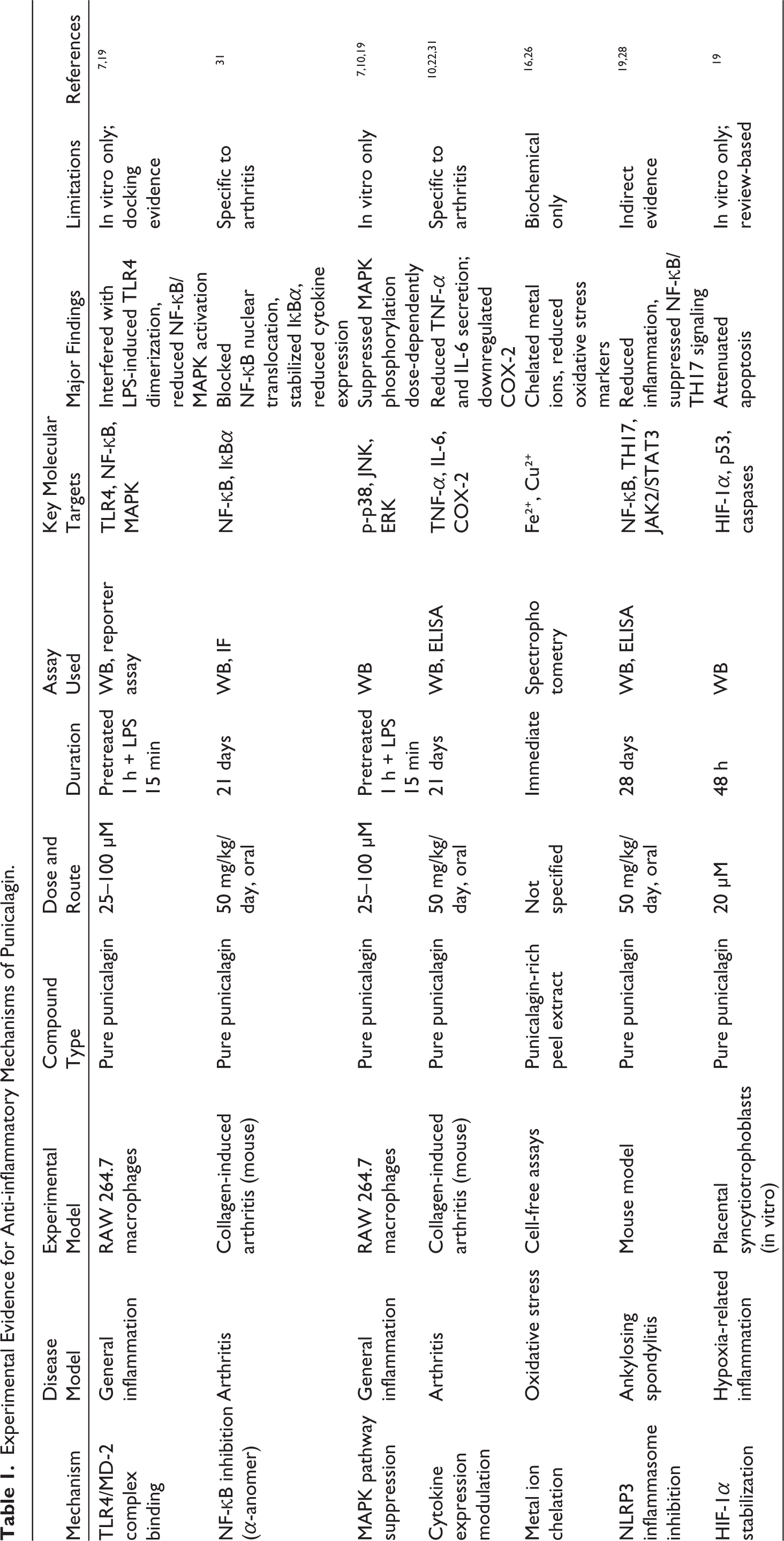
It has been worked out that in its α-anomeric form, the molecule is a more direct inhibitor of NF-κB via nuclear translocation, with this effect presumably in part due to increased hydrogen bonding with IκB kinase (IKK) and, in turn, preventing the degradation of IκBα. 31 In a comparative study, it was observed that β-punicalagin mostly inhibits pro-inflammatory cytokines such as TNF-α and IL-6, whereas its α-anomer predominantly inhibits COX-2 expression. 32
The ortho-dihydroxyl groups present in punicalagin’s HHDP moiety confer upon it the mighty capability to chelate redox-active metal ions implicated in the onset of inflammatory cascades. In adverse cases, free Fe²⁺ can catalyze the Fenton reaction, driving hydroxyl radical (•OH) formation that leads to excessive tissue damage during chronic inflammation. 16 Chelation of Fe²⁺ by punicalagin, however, reduces the availability of free iron, thereby limiting Fenton chemistry and attenuating hydroxyl radical generation. This iron-binding activity also contributes to stabilization of HIF_1α, thereby regulating inflammatory responses under hypoxic conditions. 18 On the other hand, chelation of Cu²⁺ interferes with the assembly of NLRP3 inflammasome, leading to diminished activation of caspase-1 and release of IL-1β. 20 For instance, in atherosclerosis and neurodegenerative disorders, copper dysregulation favors inflammation. 18
Hydrolysis: Stomach and EA (pH-dependent Release)
Punicalagin undergoes an initial phase of biotransformation in the stomach in an acidic environment, wherein the low pH (1.5–3.5) allows for its partial hydrolysis into EA and smaller ellagitannin derivatives. 33 This mechanism is pH-dependent and occurs rapidly. Even under artificial gastric conditions, approximately 40% of punicalagin is converted into EA in just 60 minutes. 34 Types of hydrolysis exist, for example, one induced by stomach acid alone and the second by enzymes found within gastric juice, and both are operating in this instance, with the rate of hydrolysis increasing as the pH decreases.20,35 This step is very important because of the release of EA, which is the main contributor to the microbial metabolism in the intestine. The gastric release of EA from punicalagin occurs more efficiently than from other ellagitannins due to punicalagin’s α/β anomeric configuration, which allows for a more facile acid-catalyzed cleavage. 25 Conversely, EA is more stable in gastric juice than punicalagin, but both EA and punicalagin are scarcely absorbed in the stomach, with the greater portion (~90%) just passing into the small intestine to be further metabolized. 36
Gut Microbiota Transformation
Conjugated or unconjugated EA is released during gastric hydrolysis and subjected to immense biotransformations by the colonic intestinal microbiota. This is mediated by certain bacterial taxa such as Gordonibacter urolithinfaciens and Gordonibacter pamelaeae, which perform sequential conversion of EA through tetrahydroxylation, trihydroxylation, and finally dihydroxylation reactions into different urolithin metabolites. 15 Among microbial transformation steps, EA is transformed into urolithin D, a tetrahydroxy derivative. The intermediate serves as a divergent point toward either urolithin C or urolithin A upon further enzymatic activities of specific microbes. Urolithin C is trihydroxy, whereas urolithin A is a dihydroxy derivative with greater activity in favoring mitochondrial health. Then urolithin A possibly can get converted to urolithin B, which is the least hydroxylated and last product of the pathway37–39 (Figure 2).
Metabolism of Punicalagin. Hydrolysis in the Stomach Liberates Ellagic Acid, Which Undergoes Microbial Transformation to Urolithins (A, B, C, D, Iso Urolithin A) with Interindividual Variation. Urolithin D Acts as an Intermediate in This Pathway. Gallic Acid Is Produced in Parallel via Chemical/Enzymatic Cleavage but Does Not Contribute to Urolithin Formation, Instead Undergoing Absorption and Conjugation with Modest Systemic Antioxidant Activity. https://foodb.ca/compounds/FDB016425
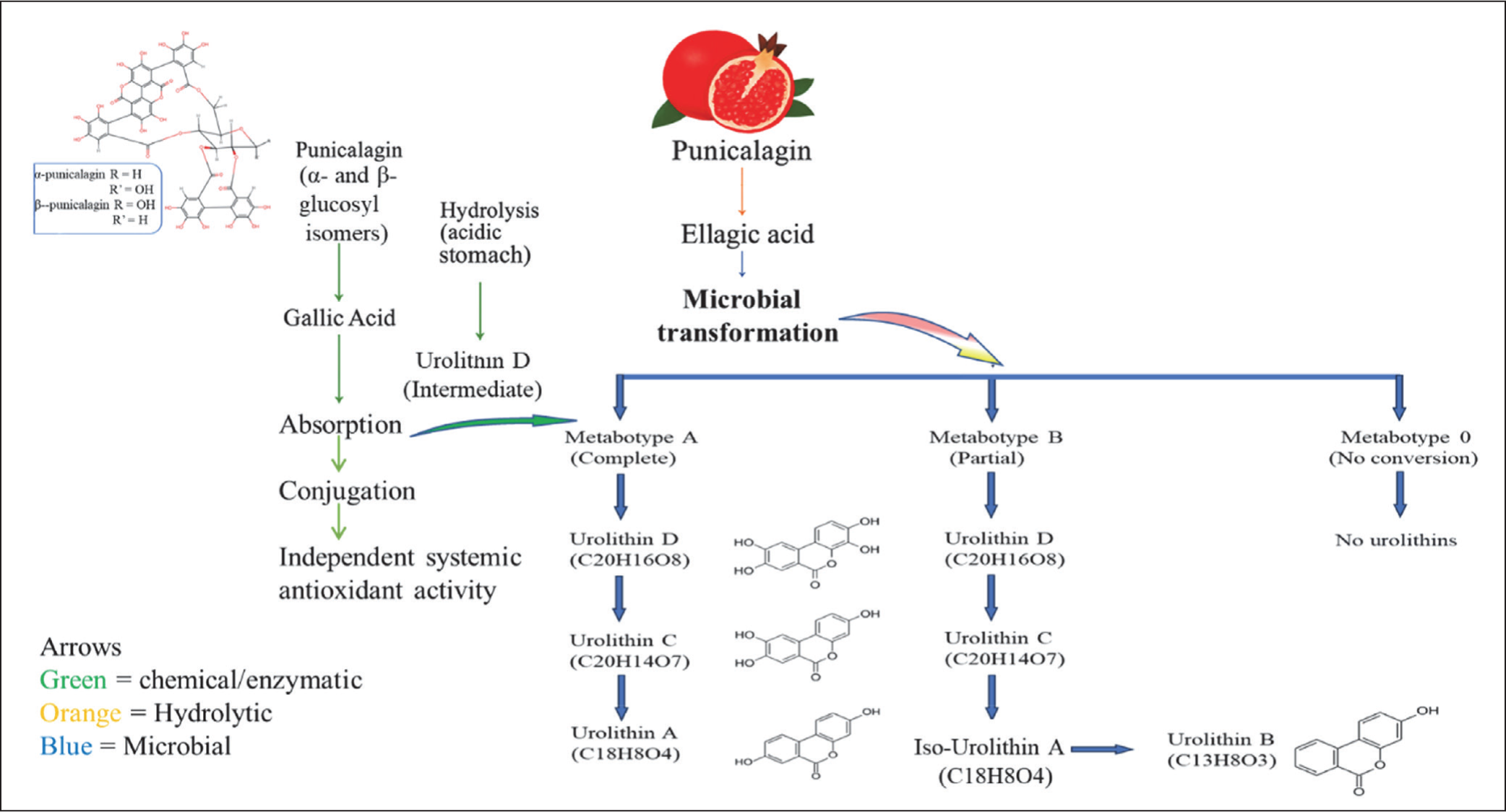
The human population can thus be stratified into three distinct metabotypes of urolithins according to their ability for microbial conversion: Metabotype-A, individuals efficiently produce urolithin A (UA), which is the most bioactive metabolite; Metabotype-B, individuals form urolithin B (UB) as an ultimate product of their conversion; and Metabotype-0 individuals do not possess the necessary microbiota and consequently excrete primarily unmetabolized EA. 40 Though urolithins C and D are thought to be intermediate metabolism products, how they are detected and what they do within human physiology are still being investigated. The absence or presence of these intermediates may be reflections of certain nuanced enzymatic capacities of an individual’s gut microbial consortia.41,42 This population subdivision has clinical ramifications, as evidence suggests that Metabotype_A individuals may derive greater health benefits from punicalagin metabolism. However, current clinical findings are not limited to pure punicalagin and often involve pomegranate extracts or urolithin metabolites, so the conclusion should be interpreted with caution. The composition of an individual’s gut microbiota, and thus urolithin metabotype, is shaped by a number of factors, including long-term diet (especially intake of fiber and polyphenols), antibiotic use, age, and host genetics. 43 According to recent scientific evidence, regular consumption of pomegranate may be capable of modifying gut microbial ecology, potentially toward more favorable communities capable of producing urolithins over time. 32
Therapeutic Divergence Between Punicalagin and Urolithins
The distinct therapeutic effects of punicalagin and urolithins result from their different molecular structures, and their active compounds work at different body locations. The gastrointestinal tract serves as punicalagin’s primary location, where it acts as a strong antioxidant, anti-inflammatory and antimicrobial substance which supports intestinal barrier function and controls gut microbiota.34,44–46 Urolithins exist as small lipophilic compounds which the body absorbs through the bloodstream to distribute throughout muscular, cerebral and vascular body regions.47–52 Urolithin A serves as the most researched urolithin, which currently represents the only metabolite that scientists are testing in human clinical studies about muscle strength and aging and metabolic health.34,48,49 The comparative Table 2 shows a comparison for punicalagin and urolithin A because this combination has been proven to have the most effective therapeutic effects. Urolithin B shows anti-inflammatory effects, while urolithin C and D act as temporary converters, which scientists use to trace microbial conversion efficiency. 53 Urolithin A serves as the main focus of research because it shows practical value for medical use, but the complete range of urolithins demonstrates the different ways that microorganisms metabolize substances, which leads to distinct metabolic patterns in different people. Therapeutic differences depend on the microbial abilities of people because of the differences between individual patients. Metabotype-A individuals produce urolithin A through their metabolic pathway, which starts from EA and progresses to urolithin D, urolithin C and lastly to urolithin A. The iso-urolithin A and urolithin B pathways in Metabotype-B individuals function through the accumulation of intermediates. The microbial abilities of Metabotype 0 individuals prevent them from starting the conversion process, which leads to the absence of urolithins in their samples. The punicalagin and its metabolites show different therapeutic properties, which require scientists to create methods for measuring their effects.
Comparative Therapeutic Properties of Punicalagin and Urolithin A.

Pharmacokinetics
Punicalagin and its metabolites show striking differences as far as their pharmacokinetic profiles are concerned in terms of bioavailability and persistence in circulation. The most notable being native punicalagin that shows extremely poor systemic absorption, recording undetectable plasma concentrations after 4 hours and reaching concentrations less than 0.1 μM in the plasma between 30 and 60 minutes after ingestion (with the elimination half-life being less than 1 hour). 43 This is in stark contrast to microbial urolithins, especially urolithin A glucuronide, which are stabilized in circulation for long periods, with the appearance of peak plasma concentration (Cmax 5–20 μM) from 6 to 24 hours post-consumption and sustain detectable levels for around 48 hours (elimination half-life 12–48 hours). 13 The original purified compound undergoes an alternative pathway of absorption. Above all, it is delivered to a very diverse population of gut microbes in the colon to metabolize and detoxify into bioactive derivatives. This microbial treatment results in more or less a delayed and sustained release profile, with slow uptake and long physiological activity of the drug. 54 Tissue-specific cycles of conjugation and deconjugation and active transport mechanisms lead to preferential accumulation of urolithin A in target organs like the brain, prostate, or colon mucosa, with concentrations usually being 5–10 times higher than in plasma. 14 The lengthy half-life of urolithins and their distribution among body tissues are also affected by their extensive Phase II metabolism (glucuronidation and sulfation) in enterocytes and hepatocytes. This also reduces their direct antioxidant capacity, but may in turn potentiate receptor-mediated signaling effects. 55
Therapeutic Mechanisms and Evidence
Anticancer Effects of Punicalagin
Punicalagin, with its multimodal anticancer action, directly interferes with oncogenic signaling pathways (validated in vitro in breast cancer cells at 50 μM, 24–48 h, pure compound; see Table 3) and synergistic enhancement of conventional chemotherapy. Chiefly, it suppresses NF-κB and STAT3 transcription factors, with downstream results such as the anti-apoptotic Bcl-2 being downregulated by 40%–60% (50 μM) and cell cycle regulator Cyclin D1 55%–75% (50 μM) in a variety of cancer cells (MCF-7, MDA-MB-231, HepG2; Western blot quantification). 61 Molecular studies demonstrate that punicalagin directly binds to IKKβ (Kd = 2.3 μM) and STAT3 SH2 domains (Kd = 3.1 μM), confirmed by molecular docking and SPR; functional inhibition validated by Western blot of phosphorylated STAT3, to inhibit their phosphorylation and nuclear translocation thereafter. 62 Punicalagin is seen to synergize with doxorubicin in breast cancer models (combination index = 0.32–0.45) to sensitize resistant cells by upregulating drug influx transporters (e.g., SLC22A16 by 4.2-fold) and suppressing P-glycoprotein efflux activity (63% reduction at 25 μM). 63 It further shows selective toxicity against cancer stem cells (CSCs) by decreasing CD44+/CD24- populations by 78% in triple-negative breast cancer through inhibition of the Wnt/β-catenin pathway (TCF/LEF reporter activity down by 82%). 64 In vivo, oral administration (50 mg/kg/day) reduces tumor volume by 65%–80% in mouse xenograft models (daily gavage, 21 days; histology and PSA assays) while showing negligible hepatotoxicity (ALT and AST levels unchanged vs. controls). 65
Anticancer Effects of Punicalagin.
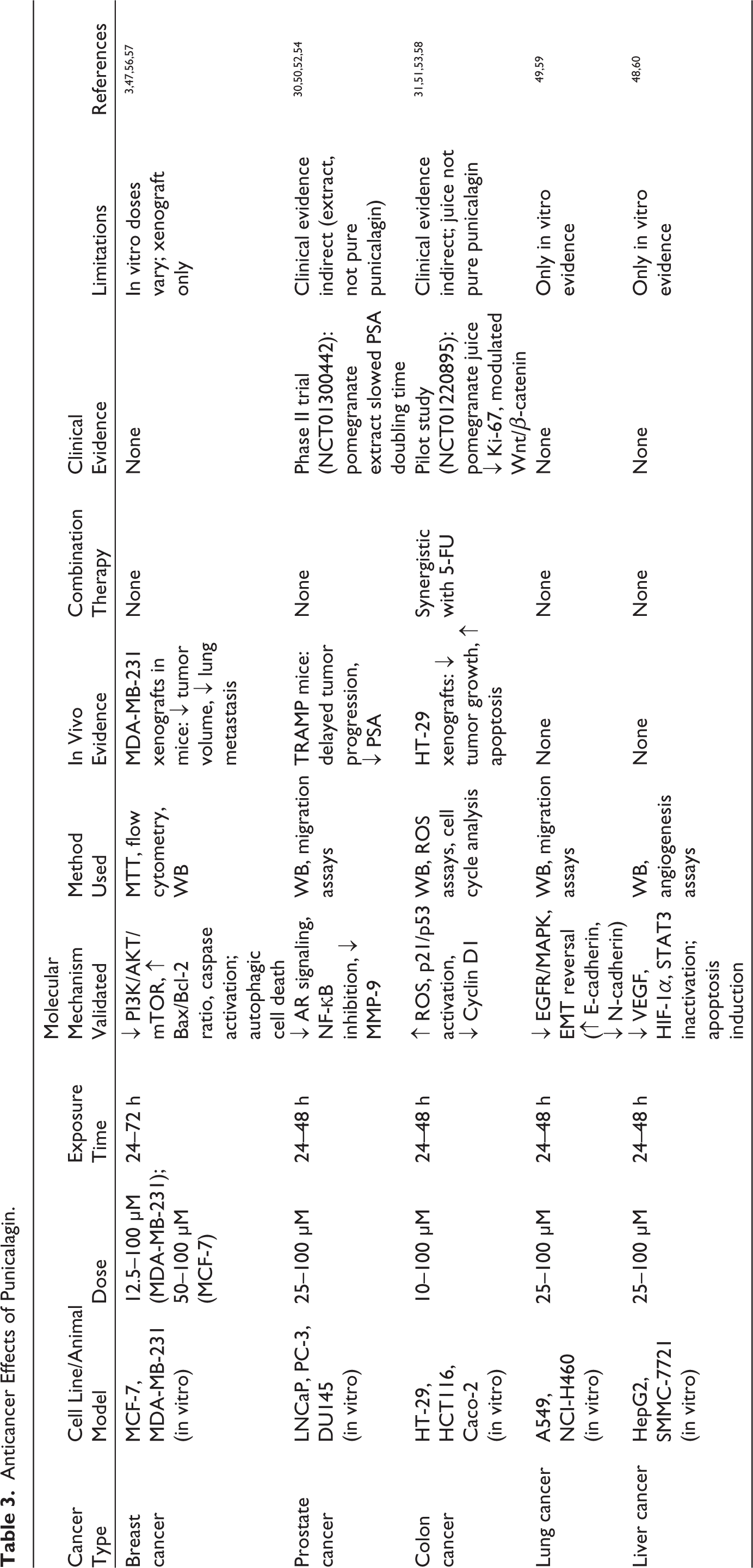
Due to different experimental models and applications (Table 3), punicalagin has displayed anticancer effects in various types of cancers. In vitro tests demonstrate that punicalagin reduces cell growth in breast cancer cell lines (MCF-7 and MDA-MB-231) by modulating PI3K/AKT/mTOR signaling and inducing apoptosis (Western blot of p-AKT and p-mTOR; apoptosis confirmed by caspase activity assays). These findings are specific to breast cancer models and do not imply broader disease specificity. 3 It further suppresses the growth of prostate cancers (LNCaP, PC-3, DU145 cells) through androgen receptor and NF-κB inhibitions (NF-κB nuclear translocation assessed by immunofluorescence; AR signaling by reporter assay) 66 ; whereas, in colon cancer cells (HT-29, HCT116), it induces cell cycle arrest and apoptosis through ROS generation and activation of p53 (ROS quantified by DCFH-DA fluorescence; p53 activation confirmed by Western blot). 67 The other in vitro findings indicate that punicalagin reduces the viability of lung cancer cells (A549, NCI-H460) through EGFR/MAPK pathways, 65 while it inhibits angiogenesis of liver cancer (HepG2, SMMC-7721) via VEGF suppression. 64 In vivo studies have demonstrated that punicalagin is capable of slowing down prostate tumor progression in TRAMP mice by reducing PSA and inflammation, 68 enhancing the efficacy of 5-FU in colon cancer xenografts, 69 and reducing breast cancer metastasis in mouse models through MMP inhibition. 63 Clinical trials involving pomegranate extracts containing punicalagin appear promising, as a Phase II study for prostate cancer (NCT01300442) reported a prolonged PSA doubling time, 70 and a pilot study for colon cancer (NCT01220895) indicated reduced proliferation markers. 32 Taken together, these results spotlight punicalagin’s multimodal anticancer actions, including induction of apoptosis, inhibition of metastases, and synergy with conventional therapeutic agents.
Antidiabetic Actions of Punicalagin
Punicalagin simultaneously targets several pathological processes in diabetes by inhibiting key enzymes and inducing metabolic reprogramming. It is a competitive inhibitor of α-glucosidase with an IC₅₀ of 12.8 μM (vs. 320 μM for acarbose), delaying the intestinal absorption of glucose by 58%–72% in situ. 71 Taking the mechanism into consideration, punicalagin’s HHDP moiety shows the greater capacity of forming hydrogen bonds with catalytic residues (Asp352 and Arg442) of α-glucosidase than acarbose (binding energy of –9.2 kcal/mol vs. –6.4 kcal/mol). 72 In adipocytes, punicalagin-induced browning of white adipose tissue was observed between 10 and 25 μM via PGC-1α activation (3.5 fold), UCP1 expression (4.8 fold), and oxygen consumption rate (an increase of 140%). 73 This activated AMPK phosphorylation of Thr172, followed by SIRT1 deacetylation of PGC-1α (which increased K63-linked ubiquitination by 2.1 fold). 74 In db/db mice, an 8-week treatment (at 100 mg/kg/day) was able to reduce fasting glucose by 42%, improve insulin sensitivity (HOMA-IR decreased by 53%), and increase thermogenic adipocytes in subcutaneous fat by 37% (rather than 8% in controls). 20 It is remarkable that the modulation of gut microbiota by punicalagin (5.7-fold increase in Akkermansia muciniphila) contributed largely to its antidiabetic effect, independent of direct mechanisms. 75
Neuroprotective Mechanisms of Punicalagin
Being multifaceted, punicalagin exerts neuroprotective effects with anti-inflammatory and anti-aggregation mechanisms seen in the neurodegenerative diseases. In BV2 microglial cells, LPS-induced secretion of IL-1β is reduced by 85%, and LPS-induced expression of COX-2 is also reduced by 72% at 20 μM via blockade of the TLR4/MyD88/NF-κB pathway (IκBα degradation inhibited by 90%) _direct evidence by Western blot of IκBα; cytokine reduction by ELISA_.76,77 Molecular docking suggests that punicalagin binds the TLR4/MD2 complex, computational prediction; not experimentally validated, (ΔG = –10.3 kcal/mol) better than the native ligand, LPS (ΔG = –6.7 kcal/mol). 78 For Alzheimer’s pathology, punicalagin can inhibit tau aggregation, suppressing PHF6 filament formation by 68% at 50 μM through direct binding with tau at the VQIVYK motif (Kd = 8.2 μM), as was evidenced by surface plasmon resonance. 79 Immunoreactivity to phosphorylation of tau at Ser202/Thr205 in the hippocampus was decreased by 55%, amyloid-β plaques by 62%, and long-term potentiation was restored to 89% of wild-type levels following 6 months of treatment at 25 mg/kg/day in 3×Tg mouse AD models. 19 The compound strengthens BBB (brain/plasma ratio: 0.18) and preferentially localizes in hippocampal neurons, eliciting antioxidant defenses through activation of Nrf2 (3.2-fold increase in HO-1). 32 Urolithin A, a major punicalagin metabolite, exceed neuroprotective effects (synergistic index = 1.38 with punicalagin), suggesting the gut-brain axis plays a crucial role in its therapeutic outcomes. 76
Bioavailability Challenges & Solutions of Punicalagin
Limitations
Punicalagin confronts bioavailability challenges that limit its therapeutic effects. Being a large molecule (1084.7 g/mol) and an extreme hydrophile, it possesses an extremely limited absorption in the intestine (<5% of the administered dose orally) since passive diffusion is the usual mechanism by which molecules enter across the intestinal epithelium. 25 Any absorbed punicalagin is quickly eliminated through the kidneys, with a plasma half-life in the order of less than an hour, so in other words, it cannot be retained in therapeutic concentrations for long. 36 More than 90% of orally consumed punicalagin reaches the colon unmetabolized, where microbial metabolism to urolithins takes place, causing great interindividual variability based on gut microbiota composition. 15 This predicament is aggravated by factors such as antibiotic use, diet, and genetically inherited polymorphisms of drug-metabolizing enzymes, resulting in uneven distribution of urolithin production in the population. Furthermore, punicalagin is also degraded extensively in the acidic environment of the stomach due to its pH-dependent stability, thus further hampering its bioavailability. 25 These pharmacokinetic drawbacks simply call for more innovative delivery strategies to outwit the poor absorption and rapid clearance of punicalagin while maximizing its conversion to bioactive urolithins.15,36
Delivery Strategies
Advanced delivery systems have been employed to improve punicalagin bioavailability and direct its release in the colon, where microbial conversion to urolithins takes place. Especially promising are chitosan-based nanoparticles sized between 150 and 200 nm, with pH-sensitive coatings that shield punicalagin from gastric degradation and permit controlled release at the neutral pH of the intestines. 32 These nanocarriers also enhance punicalagin’s mucosal adhesion and permeability so that it can remain longer in the gut lumen to be metabolized by microbes optimally. 55 Urolithin production has reportedly been enhanced by as much as threefold by co-administration of punicalagin with certain probiotics, chiefly, Lactobacillus plantarum and Lactobacillus rhamnosus, through the modulation of microbiota. 30 These strains also promote the growth of endogenous Gordonibacter species, the primary bacteria that produce urolithins. 30 Another emerging approach involves the hybrid delivery systems associating punicalagin with prebiotics like fructooligosaccharides (FOS), which selectively enrich urolithin-producing microbiota while delivering the parent compound concurrently. 78
The lipid-based self-microemulsifying drug delivery systems (SMEDD) formulations spontaneously form fine oil-in-water microemulsions upon contact with gastrointestinal fluids. For polyphenols such as punicalagin, SMEDD enhances solubility, promotes lymphatic transport, and reduces first-pass metabolism, thereby improving systemic bioavailability. Recent pharmaceutical studies highlight SMEDD as a promising vehicle for poorly water- soluble nutraceuticals, complementing nanoparticle approaches and expanding the toolkit for punicalagin delivery.12,80,81 For systemic targeting, lipid-based nanoemulsions (e.g., SMEDDS) have demonstrated five to eight-fold increases in punicalagin absorption by facilitating lymphatic transport and bypassing first-pass metabolism.9,11 These strategies collectively address the key pharmacokinetic limitations while leveraging punicalagin’s metabolic pathway for optimized therapeutic outcomes.30,78
Future Perspectives
Precision Nutrition: Metabotype-based Dosing for Personalized Therapy
Pomegranate extract supplementation in the future could be made possible by precision nutrition strategies that consider individual variations in the gut microbiota composition and individual metabolic capability. Approximately 40% are “urolithin non-producers” (Metabotype 0), injecting significant variability in the clinical response to pomegranate interventions. 40 Thus, new strategies are focusing on microbiome profiling aided by 16S rRNA sequencing with the goal to identify Gordonibacter-enriched individuals who will mostly be able to benefit from punicalagin supplementation, 15 coordinated with precision prebiotic/probiotic cocktails capable of a 3.2-fold enhancement of urolithin A production in Metabotype-B individuals, especially with punicalagin when coadministered with specific Lactobacillus strains. 32 On a pharmacometabolomic level, baseline urine urolithin levels are being used to tailor and optimize punicalagin dosing in the range of 50–200 mg/day, 55 whereas recent metabotype-adapted dosing was shown to increase the beneficial effects by 57% over standard dose administration for the reduction of cardiovascular risks. 25
Clinical Trials
Unfortunately, for now, standard experimental reviews have greatly impeded clinical translation of punicalagin research, for trials carry highly variable extracts purported to contain anywhere from 15% to 85% punicalagin. 82 There is, therefore, an urgent need for GMP-grade punicalagin isolates (>95% purity) which allow dose-response studies to be done, 83 combined with validated biomarker panels assessing both urolithin metabolites and NF-κB activity in peripheral blood mononuclear cells. 14 Designs of future Phase II/III trials should allow colon-targeted delivery by means of delayed-release capsules 84 and include microbiome stratification arms, 85 with unified clinical endpoints of high-sensitivity C-reactive protein (hs-CRP) reduction of ≥30%. 86 These are some of the measures being put into effect in the NIH groundbreaking trials that are evaluating punicalagin for prostate cancer prevention. 87
Sustainable Sourcing
Pomegranate processing industry on a global scale contributes to the disposal of more than 3 million tons of peel wastes per annum, and this is an environmental problem, yet an opportunity for a sustainable punicalagin production.88–90 Advanced green extraction techniques have been shown to enhance the recovery of punicalagin from pomegranate peel while simultaneously reducing solvent consumption, offering a more sustainable alternative to conventional methods.91,92 Circular economy paradigms are emerging where pomegranate peel extracts are repurposed for active food packaging films that can extend meat shelf life by several days. 93 They are also being explored as agricultural biostimulants, with studies showing improved germination and growth in crops such as Brassica nigra when fruit peel extracts are applied. 94 Blockchain-enabled supply chain models are being piloted to enhance transparency and traceability, while simultaneously improving farmer income through smart contracts and reduced intermediaries. 95 Life cycle assessment frameworks further demonstrate that integrating such valorization strategies can reduce the overall carbon footprint of pomegranate-derived products and create new avenues for sustainable economic value. 96
Conclusion
Punicalagin acts as a multi-nutraceutical with therapeutic utilities in chronic ailments due to its distinctive structure among ellagitannins, characterized by α/β anomeric forms and extensive HHDP esterification, making it the predominant ellagitannin in pomegranate. chemical structures and metabolisms affected by microbiota. Anticancer effects are documented on the front of apoptosis induction, inhibiting metastasis, and chemosensitization in preclinical models, while antidiabetic and neuroprotective actions further support its pleiotropic potential. Challenges, however, exist in poor bioavailability coupled with the genetic polymorphism influencing urolithin production. Nanotechnology-based delivery (nanoparticles and SMEDDS) and microbiome engineering (probiotics and prebiotics) present themselves as suitable strategies to boost efficacy. Moving ahead, the focus should embrace: (a) impaired precision nutrition based on urolithin metabotyping, (b) high-quality clinical trials underpinned with purified punicalagin, and (c) sustainable harvesting from the pomegranate waste. Structural-based innovation and delivery system integration for supply, along with customized approaches to the patient, ultimately help to transform punicalagin from a dietary polyphenol into a clinically recognized therapeutic agent, hence straddling the realms of folk medicine and modern pharmacotherapy.
Footnotes
Acknowledgements
We thank Dr. Muayad Masoud, the College of Chemistry Department at Arab American University-Palestine, for assistance regarding the structural elucidation of punicalagin isomers.
Author Contribution
The sole author made conception and design, acquisition of data, analysis and interpretation of data; drafted the article and revised it critically for important intellectual content; submitted to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. The author is eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
Consent to Participate
Not applicable.
Consent to Publication
Not applicable.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics Approvals
This study does not involve experiments on animals or human subjects.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable.
Publisher’s Note
All claims expressed in this article are solely those of the author and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Tools
The author declares that they have not used artificial intelligence (AI) tools for writing and editing of the manuscript, and no images were manipulated using AI.
