Abstract
A new ellagic acid derivative 3,3′-di-O-methylellagic acid 4′-α-
Euscaphis konishii Hayata (E. konishii), belonging to the Staphyleaceae family and being widely distributed in Fujian, Jiangxi, Guangdong, and Hunan provinces of China, has been used in traditional Chinese medicine for the treatment of stomach pain, testicular pain, and prolapse of the anus. 1,2 Previous chemical studies on the Euscaphis genus have led to the isolation of triterpenes, 3 flavonoids, 4 and lignans. 5 However, E. konishii, which is the most abundant species of the Euscaphis genus with more than 300 hectares of man-made forest in Fujian Province of China, has been underemphasized and overlooked for its chemical constituents and activities. 6,7 Significant bioactive constituents of E. konishii, as well as anti-inflammatory and fungistatic compounds, have been reported in the previous literature. 8,9 This paper reports continuing research, which resulted in finding a tumor-inhibiting extract, which led to the isolation of 10 constituents described in this study, among them a new ellagic acid with 9 known compounds (Figure 1). This study describes the isolation, and the identification of the structures and the biological activities of these compounds from the twigs of E. konishi
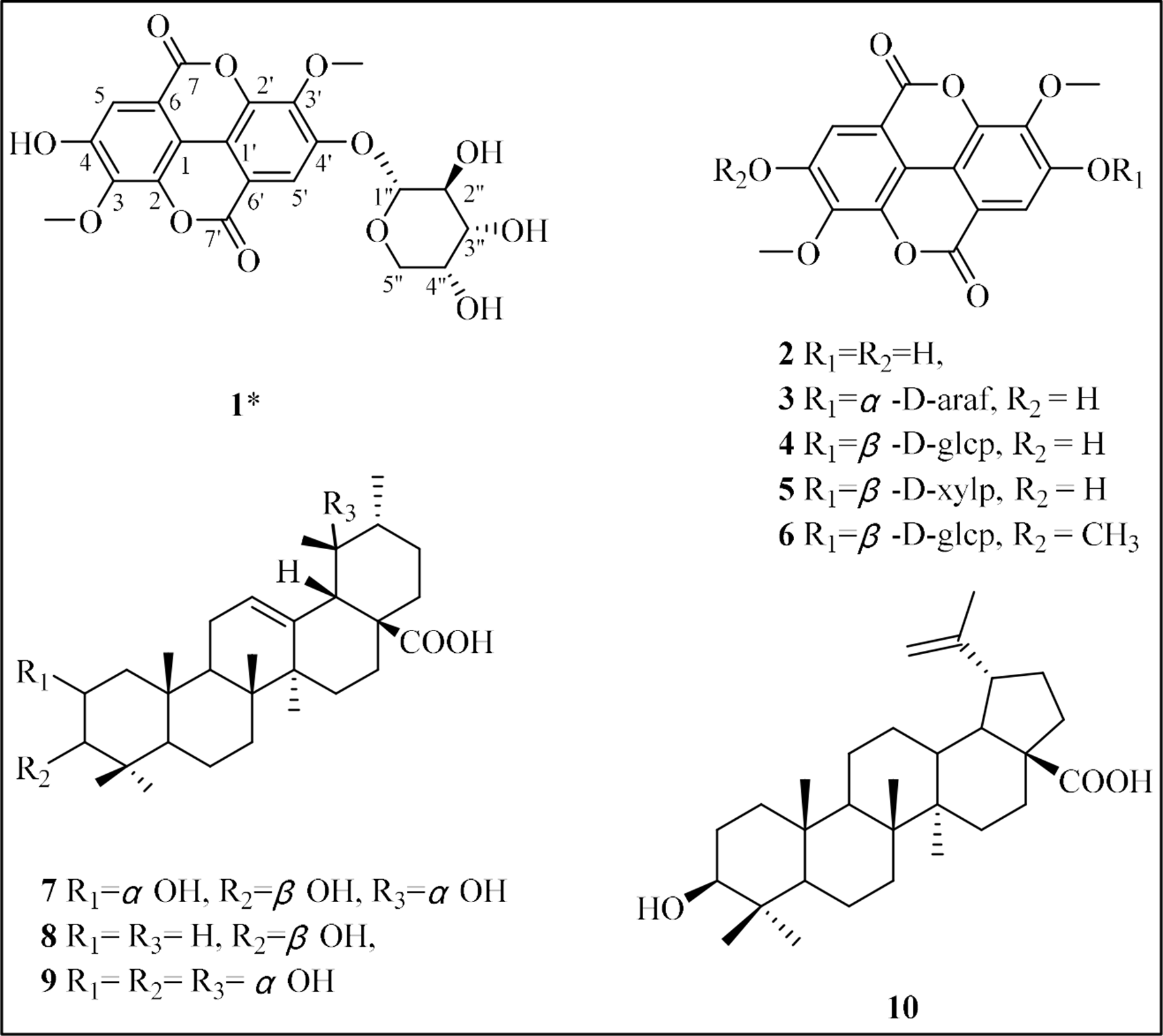
The structure of compounds 1-
Results and Discussion
3,3′-Di-O-methylellagic acid 4′-α-
1H- and 13C-NMR Spectroscopic Data for Compound 1.

aIn DMSO-d6 (100 MHz).
bIn DMSO-d6 (400 MHz).
Its structure was further confirmed by 2D NMR, which indicated the HMBC correlations from H-5 to C-1 (δ C 111.9), C-7 (δ C 158.5), and H-5′ to C-1′ (δ C 114.2), C-7′ (δ C 158.6), and H-1″ to C-4′ (δ C 152.8). These data fully demonstrate that the compound is a glycoside derivative of ellagic acid. 11 The 2 methoxyl groups were located at C-3 and C-3′ on the basis of their 13C-NMR chemical shifts at (δ C 61.0) and (δ C 61.7). 12
The sugar was hydrolyzed and compared with α-

The key HMBC correlations of compound 1.
Except for the 3,3′-di-O-methylellagic acid 4′-α-
Their cytotoxicities were determined against the Human Hepatocarcinoma cell line (HepG2 cells). The results in Table 2 indicate that ellagic derivative compounds showed moderate cytotoxic activities against HepG2 cells lines. Compounds
Cytotoxicity Values of Compounds 1

Experimental
General Experimental Procedures
Waters W2695-QDA High Performance Liquid Chromatograph-Mass Spectrometer was purchased from Waters Corporation, United States; CPA225D electronic analytical balance from Sedolis Scientific Instrument Beijing Co., Ltd.; high-speed multifunction pulverizer from Yongkang Tianhao Shengshi Industry and Trade Co., Ltd.; BS-100A automatic partial collector from Shanghai Huxi Analytical Instrument Factory Co., Ltd.; WRR melting point instrument from Zhengzhou Nanbei Instrument Equipment Co., Ltd.; BS-214D electronic balance from Sedolis Scientific Instrument Beijing Co., Ltd.; LC-20AP preparative high performance liquid chromatography (HPLC) from Shimadzu Corporation, Japan; CO2 constant temperature cell incubator from USA Shellan company; Bruker AV400 (400MHz) nuclear magnetic resonance instrument from German Bruker; Agilent 6520 high-resolution mass spectrometer from USA Agilent Technologies; Nicolet impact 5700 Fourier transform infrared spectrometer from USA Thermoelectric Corporation; silic agel from Qingdao Ocean Chemical Co., Ltd.; D101 macroporous adsorption resin, Sephadex LH-20 from General Electric Company, United States; PRP-512A resin from Beijing Jufu Resin Factory; YMC-Pack ODS-A reversed phase chromatography column (20 × 250 mm, 5 µm and 10 × 250 mm, 5 µm) from YMC, Japan; Diamonsil C18 analytical reversed-phase chromatography column (C18 250 × 4.6 mm, 5 µm) from Beijing Dima Technology Co., Ltd.; methanol, dichloromethane, petroleum ether, and ethyl acetate from Xiyu Chemical Co., Ltd.; methanol and acetonitrile from Merck Company; 5-FU and MTT from Sigma company.
Plant Material
The twigs of E. konishii were collected from San’ming District, Fuzhou, Fujian Province, the People’s Republic of China (PRC) with 26°34′39″N and 117°41′11″E. The plant was identified by Professor Shuangquan Zou from Fujian Agriculture and Forestry University (FAFU), PRC. A voucher specimen (No. 20161105) was preserved in the Department of Pharmaceutical Engineering, College of Plant Protection, FAFU (PRC). The twigs were oven-dried at 65 °C for 24 hours and crushed before extraction.
Extraction and Isolation
The air-dried and powdered twigs of E. konishii (10 kg) were extracted with 95% EtOH (3 × 80 L) by reflux. The crude extract (670 g) was mixed with the diatomite (670 g) and successively extracted with petroleum ether (PE), ethyl acetate (EtOAc), and ethanol (EtOH). The solvents were evaporated to obtain PE (106 g), EtOAc (150 g), and EtOH (150 g). The EtOAc fraction subjected to chromatography on silica gel (200-300 mesh) and eluted with a CH2Cl2-MeOH gradient (100:0-0:100) to yield 15 fractions (Fr.1-Fr.15). Fraction 12 (4.46 g) was subjected to a Sephadex LH-20 column and eluted with methanol to obtain 5 fractions (Fr.12.1-Fr.12.5). Fraction 12.2 (214 mg) by preparative HPLC [CH3CN-H2O (55:45), v/v, 8.0 mL/min, 210 nm] resulted in the isolation of compounds
Acid hydrolysis of compound
Cytotoxicity Assay
The compounds and different fractions of the extractions were tested for their cytotoxicity against HepG2 cells by the MTT method, 23 with 5-FU being used as a positive control. The cells were plated in a 96-well plate at a density of 5 × 103 cells per well in 180 µL and incubated for 24 hours. After different concentrations of the compounds were added to the plates, the cells were cultivated for another 48 hours, and subsequently 20 µL of MTT (5 mg/mL) were added to each well. After 4 hours, the culture medium was removed, and the cells were dissolved with 150 µL DMSO in each well by vigorously shaking the plate. Finally, the absorbance was assessed by a microplate reader at 570 nm. The IC50 values were calculated for the inhibition rateas follows: (OD value of control group – experiment group)/(OD value of control group – OD value of blank group) × 100%
Supplemental Material
Supplementary Material 1 - Supplemental material for Chemical Constituents and Their Activities From the Twigs of Euscaphis konishii Hayata
Supplemental material, Supplementary Material 1, for Chemical Constituents and Their Activities From the Twigs of Euscaphis konishii Hayata by Jingxin Chen, Lin Ni, Yao Zhang, Yingsa Zhu, Wei Huang and Shuangquan Zou in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 31700292) and Special Funds for Science and Technology Commissioners of Fujian Province (103/KTP19108A and 103/K1517070A).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
