Abstract
Background:
Cardiopulmonary exercise testing (CPET) is the gold-standard for quantification of peak oxygen uptake (VO2) and cardiorespiratory and muscle responses to exercise. Its application to Duchenne muscular dystrophy (DMD) has been scarce due to the notion that muscle weakness inherent to disease restricts the cardiorespiratory system from reaching maximal capacity.
Objective:
To investigate the utility of CPET in DMD by 1) establishing whether patients can perform maximal-effort exercise for valid VO2 peak assessment; 2) quantifying VO2 peak repeatability; 3) characterizing muscle and cardiorespiratory responses; 4) comparing VO2 peak to 6-min walk distance (6MWD).
Methods:
Twenty-seven DMD and eight healthy boys (6 years and older) underwent CPET using an incremental work-rate protocol for leg (ambulatory) or arm (non-ambulatory) cycling with measurement of heart rate (HR) and gas-exchange variables from rest to maximal-effort. The oxygen cost of work (ΔVO2/Δwork-rate) was calculated, and peak exercise parameters (VO2, HR, O2 pulse, ventilation (VE) and ventilatory threshold (VT)) were considered valid if the respiratory exchange ratio ≥1.01.
Results:
VO2 peak was valid (81.5% of patients), repeatable (intraclass correlation coefficient = 0.998) and low in ambulatory and non-ambulatory DMD compared to controls (19.0 ± 6.0; 10.7 ± 2; 35.2 ± 4.5 mL/kg/min respectively). VT was low (30.8 ± 10.7; 19.4 ± 3.0; 61.2 ± 6.9% VO2 peak) reflecting significant muscle metabolic impairment. Peak HR in ambulatory-DMD (172 ± 14 bpm) was similar to controls (183 ± 8.3 bpm), but O2 pulse was low (3.4 ± 1.0; 6.5 ± 1.1 mL/beat). Peak VE/VO2 (ambulatory = 42.1 ± 6.8; non-ambulatory = 42.2 ± 7.8; controls = 34.3 ± 4.6) and ΔVO2/Δwork-rate were elevated (ambulatory = 12.4 ± 4.9; non-ambulatory = 19.0 ± 9.7; controls = 10.1 ± 0.8) revealing ventilatory and mechanical inefficiency. Despite strong correlation between VO2 peak and 6MWD, severity of impairment was discordant.
Conclusion:
Valid CPET is feasible in DMD, revealing low VO2 peak due to abnormal muscle metabolic and cardiorespiratory responses during dynamic exercise. CPET reveals cardiorespiratory limitations in DMD boys with unremarkable 6MWD, and should be considered an integrative approach in clinical care and assessment of emerging therapeutics.
Keywords
Introduction
Duchenne muscular dystrophy (DMD) is a devastating X-linked neuromuscular disease caused by a gene mutation in dystrophin. Loss of this key sarcolemmal protein renders muscle susceptible to contraction-induced damage, causing cycles of muscle degeneration and regeneration and cumulative replacement of muscle by fat and fibrotic tissue. Patients experience progressive weakness and fatigability, early loss of ambulation and death from respiratory muscle or cardiac failure. 1 Disease progression is also associated with reduced daily physical activity 2 which is expected to worsen clinical outcomes by impairing cardiorespiratory fitness and limiting capacity for exercise. This has secondary implications as low levels of physical activity increase the risk of developing comorbidities (obesity, metabolic and cardiovascular disease) and all-cause mortality. 3 Fortunately, promising disease-modifying therapies are on the horizon for both ambulatory and non-ambulatory boys with DMD, with potential to yield a milder muscle phenotype and allow boys to engage in greater daily activity and possibly improve cardiorespiratory fitness and physical ability to exercise. Thus, there is a need for an assessment approach that can be used to quantify these parameters in DMD boys with varying disease severities and help identify physiological systems that may additionally constrain exercise capacity. While skeletal muscle is certainly a dominant factor, the contribution of cardiorespiratory impairments to reduced exercise capacity and daily function in DMD remains unclear.
The gold-standard for exercise capacity determination is cardiopulmonary exercise testing (CPET), a modality that simultaneously assesses cardiac, respiratory and muscle physiological responses to increasing levels of exertion. 4 Through measurement of gas exchange, CPET provides quantification of cardiorespiratory fitness, also known as peak oxygen uptake (VO2 peak), a metric that reflects the balance between maximal capacity of the cardiovascular and respiratory systems to transport oxygen (cardiac output) and ability of contracting muscles to extract and utilize oxygen (arteriovenous oxygen difference) during dynamic exercise. 5 VO2 peak is well-established as an important diagnostic and prognostic health indicator in clinical settings, 3 whereby a higher VO2 peak is inversely associated with adiposity and cardiovascular risk in all ages. 6 CPET is used in a wide spectrum of populations to elucidate underlying pathophysiology and exercise limitations, assess therapeutic efficacy, and individualize exercise prescription. 7
Although CPET is increasingly being used to investigate exercise intolerance in neuromuscular disorders (i.e. primary mitochondrial myopathy, 8 Barth syndrome, 9 Pompe, 10 Charcot Marie Tooth, 11 Friedreich's ataxia, 12 Beckers muscular dystrophy 13 and other dystrophies14–16), its application to DMD has been very limited.13,17,18 The largest study in DMD to date 17 was conducted prior to corticosteroids being the standard of care for these patients, and reported markedly decreased VO2 peak along with a low cardiorespiratory response to maximal effort exercise. Whether or not patients achieved established criteria reflecting maximal-effort exercise was not reported, confounding interpretation of their CPET results. Indeed, the feasibility of obtaining valid measures of maximal effort (VO2 peak and heart rate) in neuromuscular disorders has long been questioned, as muscle weakness and fatigue are thought to prevent the cardiorespiratory system from reaching its maximal capacity.14,19,20
This study seeks to fill gaps in knowledge about CPET in DMD and includes ambulatory and non-ambulatory patients compared to healthy age-matched boys. Our aims are to: 1) establish whether boys with DMD can perform maximal effort CPET and attain valid criteria for VO2 peak assessment; 2) determine VO2 peak test-retest repeatability; 3) quantify VO2 peak and cardiorespiratory responses; and 4) compare the severity of disease classification using CPET to standard functional tests. Collectively, these data will demonstrate the utility of CPET for the DMD population and support its integration into clinical research and ultimately patient care.
Materials and methods
Participants
Boys aged 6 years and older with a genetically-confirmed DMD diagnosis and on a stable glucocorticoid regimen were recruited from the Center for Neuromuscular and Rare Diseases at University of Florida (UF) and ongoing investigations at UF between February 2021 and April 2024. Inclusion criteria for ambulatory boys were ability to independently walk at least 100 meters without an external assistive device, 2 and non-ambulatory boys had to be able to lift a cup to their mouth (Brooke Upper Extremity score of 3). 21 Exclusion criteria included presence of unstable or concomitant medical problems that are contraindicated for CPET as outlined by the American College of Sports Medicine (ACSM), 22 including severe cardiomyopathy or cardiac conduction abnormalities, presence of a secondary condition that impacts muscle function or muscle metabolism, history of rhabdomyolysis, inability to comply with instructions and/or current or prior participation in investigational drug or gene-therapy studies. Age-matched healthy controls were also included, using the same exclusion criteria. The research protocol was approved by Institutional Review Board 1 (Project number: 202101689) at the UF. Informed consent and assent were obtained from the parent and patient respectively. All procedures performed in our human participants were in accordance with the ethical standards of the 1975 Helsinki declaration.
Study design
All procedures were conducted in UF's Clinical Research Center by an experienced team of exercise physiologists, clinicians (research nurse or cardiologist) and physical therapists. Body mass (kg) and height (cm) were obtained from all participants. All boys with DMD underwent CPET, pulmonary function testing (PFT) and an echocardiogram prior to exercise. Functional measures (6 min walk distance (6MWD), 10 meter run/walk (10 m r/w), Northstar Ambulatory Assessment (NSAA)) were assessed in the ambulatory boys with DMD for correlation with VO2 peak. Healthy controls underwent CPET only. A subset of patients and controls were invited back to undergo a second CPET between 1 and 3 weeks after the first CPET for repeatability assessment. A list of current medications was obtained and reviewed by the study clinician prior to start of exercise.
Procedures
Pulmonary function testing (PFT): PFT was performed in all DMD patients in a seated position with a spirometer (Carefusion Microlab Portable spirometer, Micro Medical Limited, Kent, United Kingdom), in accordance with the American Thoracic Society (ATS) recommendations 4 before each CPET. The spirometer was calibrated before each test. Forced vital capacity (FVC) and forced expiratory volume in 1 s (FEV1) were measured and compared to reference values as percent predicted. 23 Maximal inspiratory pressure (MIP) and maximal expiratory pressure (MEP) were measured using the Carefusion MicroRPM device (CareFusion, Kent, United Kingdom) according to ATS recommendations to provide global estimate of inspiratory and expiratory muscle strength respectively. Predicted values for MIP and MEP were calculated. 24
CPET: This non-invasive test was carried out according to guidelines developed by ACSM 22 and the ATS, 4 and tailored for pediatric populations. 6 The equipment was calibrated according to manufacturer instructions before testing. All participants were familiarized to the equipment, cycling protocol and Borg scale (6 to 20) for rating of perceived exertion (RPE). 25 CPET was conducted after at least 2 h of fasting. Each subject was instrumented with a 12-lead ECG (CardioSuite Cardio Card, Nasiff Associates, Inc, New York, USA) for heart rate (HR) monitoring and fitted with a facemask (Hans Rudolph Inc, Kansas City, MO) connected to a calibrated metabolic gas mixing chamber (ParvoMedics, TrueOne, Salt Lake City, Utah, USA) to measure VO2, carbon dioxide production (VCO2) and ventilation (VE). Vital signs (HR, blood pressure, oxygen saturation) and RPE were obtained after 10 min of rest in the seated position, followed by 3 min of metabolic data collection. Ambulatory boys performed CPET on a recumbent stationary electronically braked cycle ergometer (Lode Corrival, Lode BV, Gronigen, The Netherlands). Pedal shoes and straps were used to ensure a stable foot placement. Non-ambulatory patients performed arm ergometry (Angio, Lode BV, Gronigen, The Netherlands) while seated in a wheelchair placed in a locked position.
A graded cycling exercise protocol was developed based on previous experience in patients with primary mitochondrial myopathy. 8 It consisted of a 3-min warm-up at no-load, followed by 2-min incremental stages optimizing achievement of a physiological steady-state. Workload was increased by 5 watts per stage (in boys with a 6MWD < 400 meters) or 10 watts/stage (in boys with 6MWD >400 meters) to peak exertion. In the last 15 s of each stage, the Borg RPE was obtained, and HR and any symptoms (muscle pain, dyspnea, fatigue) were noted. The ECG was continuously monitored for ischemic signs and arrhythmic events. Verbal encouragement was provided throughout the test, and the exercise protocol was terminated when the subject voluntarily stopped because of exhaustion, if they were unable to maintain a cycling cadence above 40 revolutions per minute (rpm) for both arm and leg cycling protocols, or the study team ended the test (based on achieving criteria of peak-effort or any adverse findings as described in ACSM guidelines 22 ). After peak exercise was reached, the patient underwent a cooling down phase of two minutes unloaded pedaling. Blood pressure and oxygen saturation were obtained immediately post-exercise and recovery and the subject remained seated and instrumented for five minutes of recovery. Patients and parents were told to contact the study team if any symptoms occurred within 48 h after CPET.
CPET data analysis: Data were extracted from the Parvomedics and CardioSuite software for analysis. For the resting phase, data were averaged for 2 min. Peak exercise variables (VO2, VCO2, VE, respiratory rate, tidal volume (Vt) and HR) were recorded as the highest average 30-s value within a phase where the respiratory exchange ratio (RER) ≥1.01. VO2 peak was expressed in absolute terms (mL·min−1) and indexed by weight (mL·kg−1·min−1) for intersubject comparisons and relative to normative data for children as percent predicted using published equations for VO2 peak 26 and heart rate. 27 The following parameters were calculated from directly measured outcomes: oxygen cost of work (ΔVO2/Δwork-rate, WR) 28 ; oxygen pulse (VO2/HR); the chronotropic index (CI); ventilatory equivalent for oxygen (VE/VO2); VE/VCO2 slope for the linear exercise period; ventilatory reserve (VR) 6 ; and Vt elevation (Peak Vt/FVC). The ventilatory threshold (VT) was manually calculated by the V-slope method and expressed as a percentage of predicted. The CI was not calculated in the non-ambulatory DMD boys because arm-ergometry and beta-blockers impact peak HR achieved with exercise.4,29 For further clarification on above-mentioned CPET variables, see Appendix 1.
Validity of VO2 peak assessment: Historically, a sustained plateau in VO2 between two final exercise workloads is considered reaching physiological limits and attainment of maximal aerobic capacity. 5 However, this is infrequently observed in clinical populations or children and the highest measured VO2 is used (VO2 peak) 6 as long as the RER criteria is attained. For our study, we determined validity of maximal-effort exercise by achieving RER ≥ 1.01. This value (lower than the cut-off established for adults where peak RER > 1.10) is commonly used in pediatric exercise testing, 26 whereas values < 1.0 are considered to reflect submaximal exercise. 30 The RER is obtained exclusively from ventilatory expired gas analysis and is a highly accurate and reliable gauge of subject effort. 5 Achievement of ≥85% age-predicted maximal heart rate is also commonly used to indicate maximal effort during a CPET, 5 although this can be more variable and affected by beta-blocking agents that blunt HR responses to exercise. 5 Due to the potential use of cardiac medications in boys with DMD, we based maximal effort by ability to achieve RER ≥ 1.01. Age-predicted maximal HR was calculated using the equation = 208 − 0.7 × age. 27 Borg scale RPE was collected throughout exercise to reflect patient symptomatology for myalgia, muscle fatigability and dyspnea. Values ≥17 out of 20 are typically used to reflect maximal effort exercise. 25
Determination of physiological limitations of VO2 peak: The VT usually occurs between 45–65% VO2 peak in healthy untrained subjects. 5 An earlier onset (< 40%) indicates skeletal muscle as a prominent limitation to VO2 peak, either due deconditioning and/or abnormal muscle oxidative capacity. Criteria suggesting cardiac function as limiting to VO2 peak include 1) chronotropic incompetence (<0.80 once RER ≥ 1.01 29 ; and 2) a low peak oxygen pulse (<80% predicted). As respiratory limitation criteria, we assessed whether patients exhausted their ventilatory reserve during exercise (final VR < 20%), 30 and whether they had insufficient tidal volume recruitment (Vt/FVC <50%) at peak exercise.
Functional measures: To characterize functional ability, the 6MWD, 10 m w/r and NSAA were administered after at least a 1-h rest. Validated standardized instructions and methodology were used for all functional measures. 31 The 6MWD was assessed on a 25-meter course using standard guidelines for DMD, 32 and the distance traversed in 6 min was recorded in meters. To account for differences in age and height in the DMD cohort, the % predicted 6MWD was also calculated. 33 The 10 m w/r was performed with participants wearing shoes up to 3 times, with the fastest time used for analysis. 34 The NSAA, which comprises 17-items of physical function yielding a clinician-rated score between 0 to 34 was also conducted. 35
The individual percentage scores of predicted values for VO2 peak %pred and 6MWD%pred were each classified and then compared. For both variables, classifications were made according to previously established guidelines 13 ; normal >82%; mildly reduced (61–81%); moderately reduced (51–60%) and severely reduced (≤50%).
Statistical analysis: All statistics were analyzed using Prism, Version 10.2.2 for macOS. Data were checked for normal distribution using the Qplot. Quantitative descriptive statistics are presented as mean ± SD. One-way ANOVA was used to compare CPET outcomes at rest and peak exercise. The Tukey's multiple comparisons test was used to adjust for P values in multiple comparisons. Repeatability (test-retest reliability) of VO2 peak was determined on two separate days (within a 3-week period), with values compared using a paired-samples t-test and linear regression. SAS macro INTRACC was used to calculate the intraclass correlation coefficient (ICC). A Bland-Altman plot was used to quantify systematic and random error. To determine within-participant reproducibility, the coefficient of variation (CoV), the ratio of standard deviation to the mean, was calculated. Individual levels of VO2 peak were compared to the 6MWD using Pearson correlation, and to the 10 meter walk/run and NSAA using Spearman correlation.
Results
Participant demographics
Twenty-seven boys with DMD completed CPET: 22 (aged 9.4 ± 2.5 years) were ambulatory and performed leg cycling, and 5 (aged 13.4 ± 2.5 years) were non-ambulatory and underwent arm cycling. The Brooke Upper Extremity score ranged from 1 to 3 in the non-ambulatory cohort. One ambulatory boy with DMD had mildly reduced cardiac function (ejection fraction = 45%). All other participants had an ejection fraction >45%. All boys with DMD were on a stable corticosteroid regimen, and many were on cardiac medications (Angiotensin-converting enzyme inhibitors, Mineralocorticoid receptor antagonists) for prophylactic or treatment purposes (see Table 1). Two of these boys with DMD (both non-ambulatory, weight > 30 kg) were on a beta-blocker (12.5 mg carvedilol BID), Table 2. Three patients were on approved exon-skipping therapy, but none had participated in an investigational drug study (i.e. gene-therapy). Eight healthy age-matched controls (9.2 ± 1.0 years) underwent leg cycling CPET. They were not taking any medications and were physically active participating in physical education classes at school.
Demographics and medications of study participants.

Values are mean ± SD. MRAs, mineralocorticoid receptor antagonist; ACE, Angiotensin-converting enzyme. ASignificantly different from Controls and A-DMD, p < 0.05; BSignificantly different from A-DMD; CSignificantly different from Controls.
Peak effort CPET variables in all study participants.
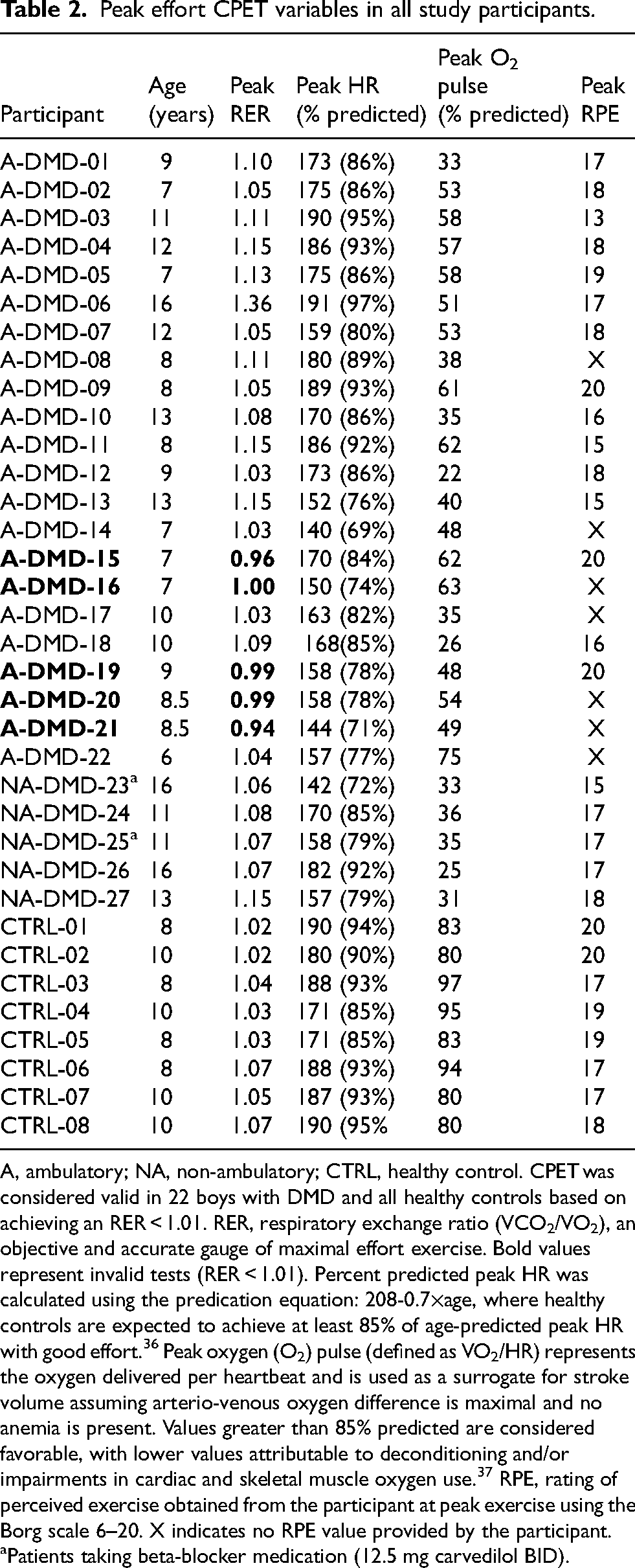
A, ambulatory; NA, non-ambulatory; CTRL, healthy control. CPET was considered valid in 22 boys with DMD and all healthy controls based on achieving an RER < 1.01. RER, respiratory exchange ratio (VCO2/VO2), an objective and accurate gauge of maximal effort exercise. Bold values represent invalid tests (RER < 1.01). Percent predicted peak HR was calculated using the predication equation: 208-0.7×age, where healthy controls are expected to achieve at least 85% of age-predicted peak HR with good effort. 36 Peak oxygen (O2) pulse (defined as VO2/HR) represents the oxygen delivered per heartbeat and is used as a surrogate for stroke volume assuming arterio-venous oxygen difference is maximal and no anemia is present. Values greater than 85% predicted are considered favorable, with lower values attributable to deconditioning and/or impairments in cardiac and skeletal muscle oxygen use. 37 RPE, rating of perceived exercise obtained from the participant at peak exercise using the Borg scale 6–20. X indicates no RPE value provided by the participant. aPatients taking beta-blocker medication (12.5 mg carvedilol BID).
Validity of peak effort CPET in DMD
All patients complied with test instructions. Twenty-two out of 27 (81.5%) boys with DMD, as well as all healthy participants, achieved RER ≥ 1.01 indicating that these tests were valid for analysis of peak physiological comparisons (Figure 1). In the 5 non-valid tests (all in ambulatory participants), RER ranged from 0.94 to 1.00 (Table 2). In valid tests, mean peak RER (controls = 1.04 ± 0.02; ambulatory DMD = 1.10 ± 0.07; non-ambulatory DMD = 1.08 ± 0.03) and HR % predicted (controls = 90.9 ± 4.0; ambulatory DMD = 85.5 ± 7.5; non-ambulatory DMD = 81.5 ± 7.6%) did not differ significantly between the three groups, confirming the peak-effort nature of exercise. Subjective ratings of RPE at peak-exercise were obtained in twenty boys with DMD (74% of patients) and not provided in seven due to a focus on maintaining cadence and completing the final stage of exercise (Table 2). RPE values ranged from 13 to 20 in DMD and were >17 in all healthy controls. In five tests, despite achieving RER ≥ 1.01, DMD boys reported lower than maximal RPE (<17), suggesting peak effort RPE may not be a valid criterion in DMD populations. The main reasons for terminating the exercise test were inability to maintain cycling cadence above 40 revolutions per minute and muscle fatigue. The minimum age of patients performing valid tests (6 years) did not differ from non-valid tests (7 years).
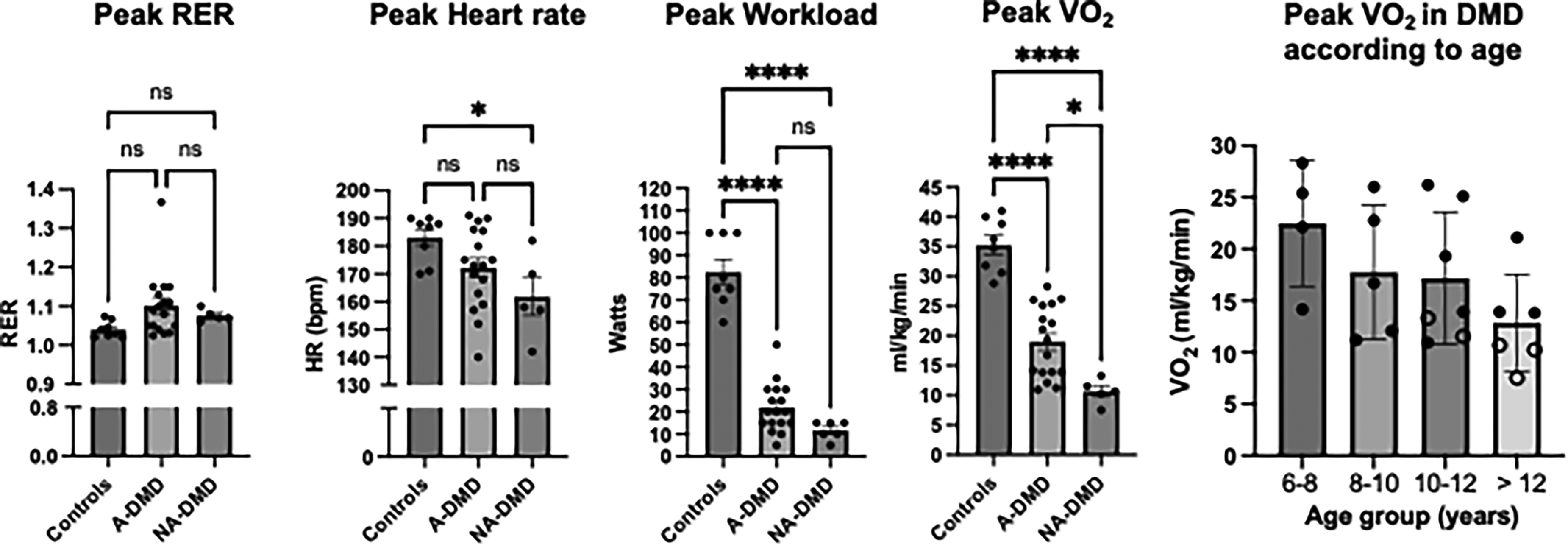
Comparison of peak data in all valid tests of healthy controls, ambulatory (A-DMD), and non-ambulatory (NA-DMD) boys with DMD. Achievement of maximal effort is demonstrated by a respiratory exchange ratio >1.01 across all groups. Peak heart rate in A-DMD was similar to controls, and lower in NA-DMD. Peak workload and VO2 were significantly low in both groups of DMD. VO2 peak varied according to age group in the DMD cohort. Bars indicate mean and standard error of mean for each group. ns, non-significant, * p < 0.05; **** p < 0.0001. Open circles indicate VO2 peak values in non-ambulatory DMD.
Test-retest of VO2 peak
CPET was performed on two separate days (within a 3-week period) in 5 boys with DMD and 5 healthy controls to assess the repeatability of VO2 peak as an outcome measure, Figure 2. In the DMD cohort, the correlation between test 1 and test 2 for VO2 peak was 0.98 (p < 0.0001), with the mean of test 1 (700.2 ± 276.0 mL/min) not significantly different (p = 0.454) from test 2 (692.4 ± 272.1). The average CoV for the DMD cohort was 1.9% (ranging from 0.14 to 2.7%). Similar results were found for the cohort of healthy controls. When analyzed as a collective group (DMD and controls), the correlation between test 1 and test 2 was 0.99 (p < 0.001), with no difference in mean VO2 peak between tests (Test 1: 955.1 ± 384; Test 2: 940.4 ± 368, p = 0.33). The Bland-Altman plots did not show a systematic bias between tests, and the CoV for VO2 peak was <3%. The ICC was 0.998 indicating excellent reliability of VO2 peak.
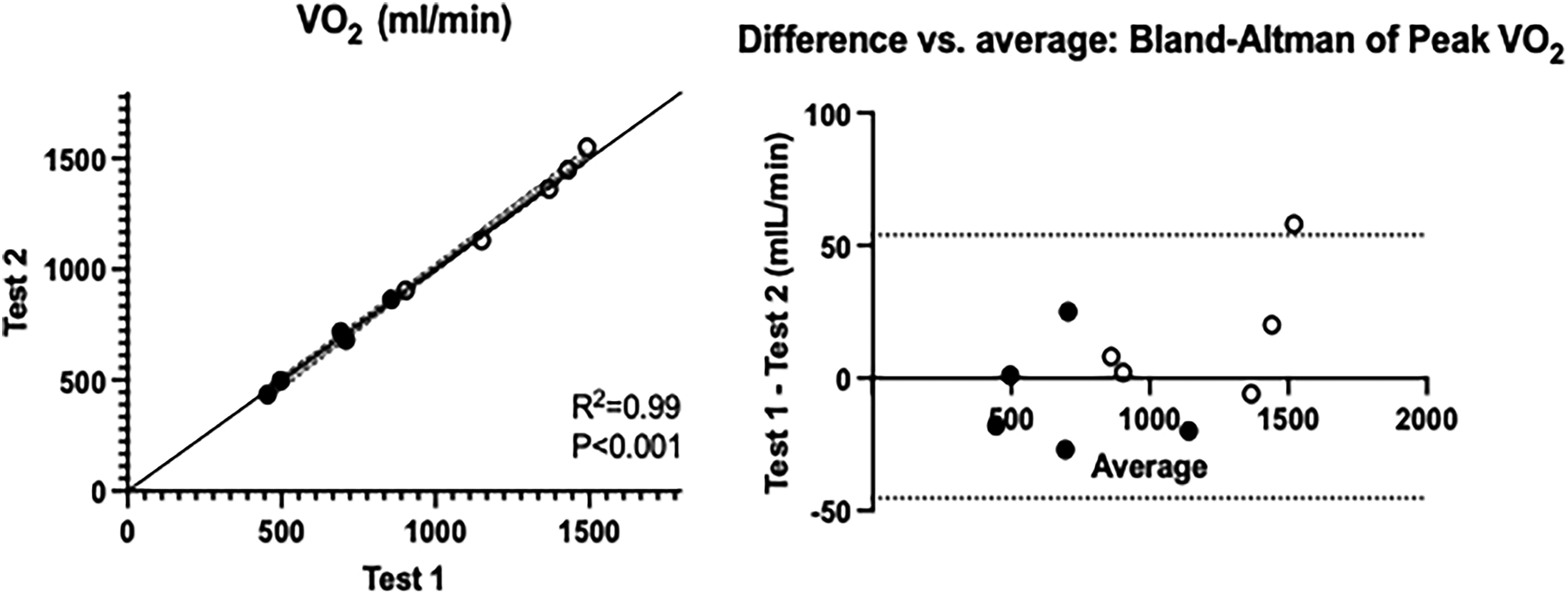
Repeatability of VO2 peak assessed with CPET on two separate occasions within a 3-week period. Test 1 and Test 2 values are shown (left) for DMD (closed circles) and heathy controls (open circles). The Bland-Altman plot (right) demonstrates the average difference between tests.
CPET variables at rest
At rest, all parameters (systolic blood pressure, diastolic blood pressure, VO2 and VE) were similar amongst groups, with the exception of HR which was elevated in ambulatory boys with DMD (108 ± 11 bpm) compared to non-ambulatory (97 ± 6 bpm) and healthy controls (94 ± 7 bpm), Table 3. Resting HR did not differ between non-ambulatory DMD and controls, reflecting clinical management of tachycardia. PFT revealed normal or near-normal function (FVC = 96 ± 15% and 87 ± 11% predicted; and FEV1 to be 96 ± 18 and 80 ± 25% predicted) in ambulatory and non-ambulatory boys, respectively. MIP was 60.6 ± 18 cm H2O (87 ± 25% predicted) in ambulatory and 51.7 ± 7.5 cm H2O (63 ± 13% predicted) in non-ambulatory, and MEP was 68.3 ± 22.0 (77 ± 23% predicted) in ambulatory and 47.7 ± 17.8 mmH20 (46 ± 14% predicted) in non-ambulatory boys with DMD.
Physiological variables obtained at rest and peak cycling exercise for valid tests.

Values are shown for individuals achieving peak effort as reflected by RER ≥ 1.01, and depict means ± SD. Results of the ordinary one-way ANOVA are shown. A = A, ambulatory; NA, non-ambulatory; CTRL, healthy control; SBP, systolic blood pressure; DBP, diastolic blood pressure; FVC, forced vital capacity, FEV1, forced expiratory volume in 1 s; ΔVO2/ΔWR, ratio of oxygen uptake to work rate reflecting the oxygen cost of work; CI index, chronotropic index, defined as change in HR from rest to peak exercise relative to HR reserve (the difference between age-predicated maximal HR and resting HR); VE/VCO2 slope, relationship between ventilation and VCO2; Vt elevation: tidal volume recruitment at exercise relative to forced vital capacity. For further clarification on these CPET variables, see Appendix 1. ND, not done as calculation requires values obtained from PFT; ns, non significant.
Significantly different from Controls based on one-way ANOVA with multiple comparisons;
Significantly different from A-DMD based on one-way ANOVA with multiple comparisons.
Significantly different from other group based on unpaired T-test.
Peak exercise capacity and limiting factors
Absolute VO2 peak was significantly lower in DMD compared to controls (p < 0.0001), reaching 42.4 ± 13.9 and 22.5 ± 4.9% of age-predicted values in ambulatory and non-ambulatory DMD respectively (Table 3). Taking body weight into consideration (Figure 1), relative VO2 peak values after leg-cycling in ambulatory boys were almost half that of age-matched controls (19.0 ± 6.0 versus 35.2 ± 4.5 mL/kg/min). VO2 peak was lowest after arm-cycling in non-ambulatory boys with DMD (10.7 ± 2.1 mL/kg/min). When analyzed according to age group, there were no statistical differences in VO2 peak.
Similarly, peak workload and exercise duration were significantly lower in DMD compared to age-matched controls (Table 3). ΔVO2/ΔWR, which reflects the mechanical efficiency of the musculoskeletal system 28 was elevated in DMD, particularly in non-ambulatory boys. The ventilatory threshold was detected in the majority (85%) of DMD patients, and undetermined in 4 patients (consistent with other studies where due to data scatter, it is not possible to determine in ∼10% of tests 38 ). The average VT was significantly lower in ambulatory (13.9 ± 4.8 mL/kg/min) and non-ambulatory (9.2 ± 1.1 mL/kg/min) DMD boys compared to age-matched controls (27.4 ± 3.1 mL/kg/min). Furthermore, in contrast to controls who reached VT at 61.2 ± 6.9% of VO2 peak, both ambulatory and non-ambulatory DMD boys reached VT at lower levels (30.8 ± 10.7 and 19.4 ± 3.0% respectively, Figure 3). This early onset of VT (<40%) supports skeletal muscle as a limiting factor to VO2 peak.
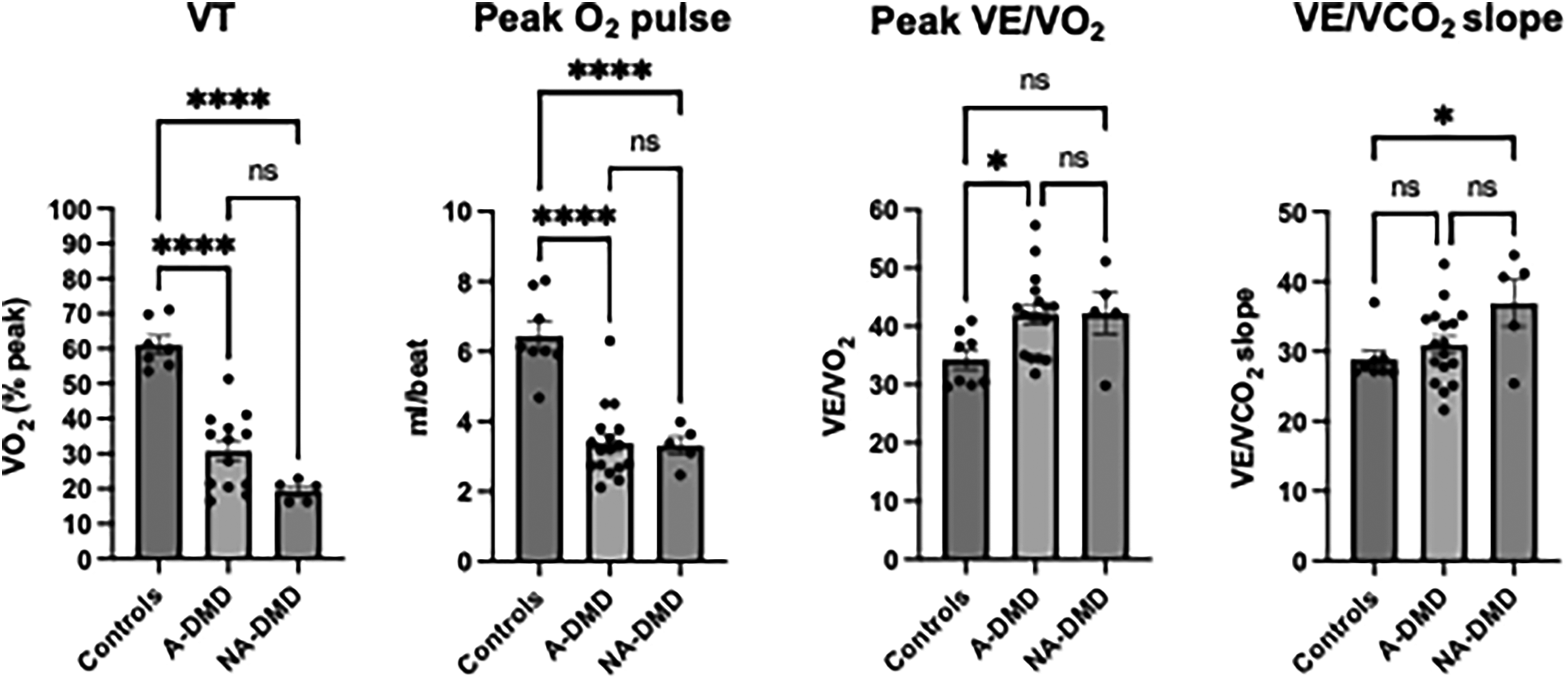
CPET reveals the multi-factorial nature of exercise impairment in DMD. Ambulatory (A-DMD) and non-ambulatory (NA-DMD) boys with DMD had a low ventilatory threshold (VT) and peak oxygen pulse compared to healthy controls. Ventilatory inefficiency (high peak VE/VO2 and VE/VCO2 slope) was also apparent. Bars indicate mean and standard error of mean for each group. ns, non-significant, * p < 0.05; **** p < 0.0001. See Appendix 1 for further description of CPET variables.
Ambulatory boys with DMD were able to reach a peak HR reflective of near-maximal effort exercise (average HR peak of 172 ± 14 bpm) and similar to healthy controls (183 ± 8.3 bpm). Non-ambulatory boys (162 ± 15 bpm) achieved a lower average peak HR (Figure 1), which may be explained by the smaller muscle mass used during arm ergometry exercise 4 as well as use of beta blocker medications in two patients. The mean CI was lower in ambulatory boys with DMD compared to controls (0.70 ± 0.14; 0.83 ± 0.07, p < 0.05). Likewise, peak O2 pulse was low in all ambulatory (mean 3.4 ± 1.0 mL/beat) and non-ambulatory (mean 3.3 ± 0.6 mL/beat) DMD boys compared to controls (6.5 ± 1.1 mL/beat; p < 0.01). There were no abnormal ECG findings and no adverse events reported with CPET.
Peak VE in ambulatory (24.0 ± 6.0 L/min) and non-ambulatory (21.9 ± 6.6 L/min) patients was approximately half that of controls (44.9 ± 7.9 L/min), reflecting the lower overall peak work rate. This difference was due primarily to a lower peak respiratory rate whereas tidal volume did not significantly differ between DMD and controls (Table 3). VR was normal (>20%) in both ambulatory and non-ambulatory patients. However, ventilatory efficiency at peak exercise was abnormal in DMD (Figure 3). Peak VE/VO2 was higher in patients (ambulatory = 42.1 ± 6.8; non-ambulatory = 42.2 ± 7.8) compared to controls (34.3 ± 4.6, p < 0.05). Similarly, the VE/VCO2 slope was elevated (ambulatory = 30.9 ± 5.4; non-ambulatory = 37.0 ± 7.5; controls = 28.9 ± 3.4, p < 0.05). Tidal volume recruitment was insufficient (lower than 50% of vital capacity) in both groups of DMD, with lower values in non-ambulatory compared to ambulatory boys (27.8 ± 3.6 versus 32.7 ± 5.1; p < 0.05).
Relationship of VO2 peak to functional measures
In ambulatory boys with DMD, 6MWD ranged from 150–509 m with an average of 373 ± 97 m, equating to 60 ± 16% predicted. The average time to complete the 10 m w/r was 4.7 ± 1.4 s (ranging from 3.3 to 13.8 s) and the NSAA score was 24.1 ± 6.7 (range 6 to 30); reflecting reduced walking ability and functional abilities respectively. There was a significant correlation between VO2 peak and 6MWD (R2 = 0.61; p < 0.01), NSAA score (r = 0.66, p < 0.01) and 10 m walk/run (r = -0.74, p < 0.01), Figure 4). Despite the strong correlation between VO2 peak and 6MWD, in 60% of cases there was discordance in classification of the severity of individual percentage scores of VO2 peak %pred and 6MWD%pred (Table 4). In all these cases, measured VO2 peak reflected greater impairments in exercise capacity compared to 6MWD. For example, patients were classified as having mild or moderately reduced 6MWD relative to a severely reduced VO2 peak.
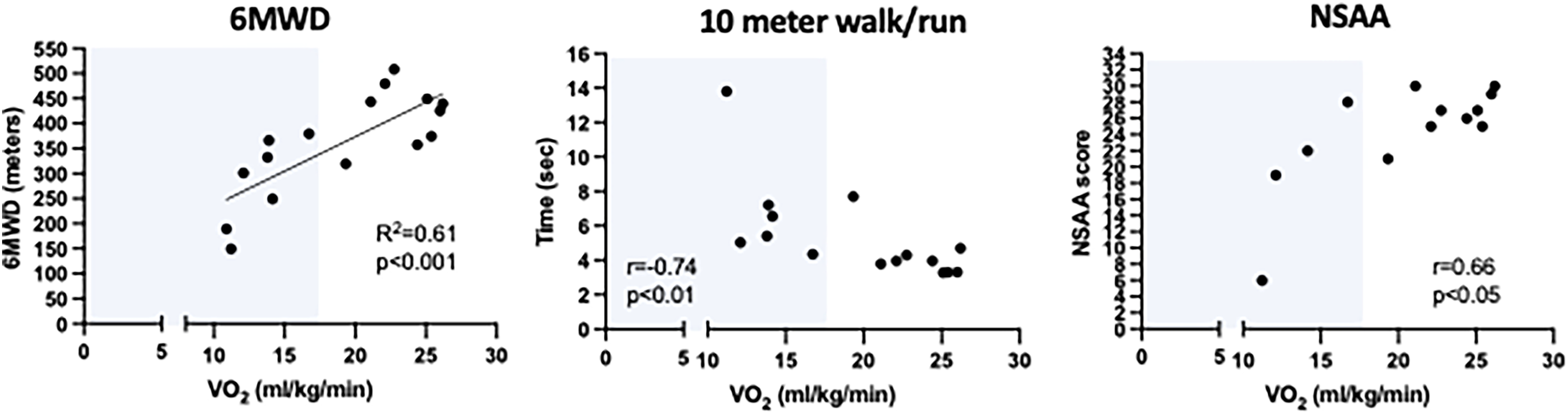
Correlation of VO2 peak with standard measures of walking and functional ability in ambulatory boys with DMD. The Pearson (for 6MWD) and Spearman (for 10-meter walk/run and NSAA) are shown. The shaded area represents VO2 peak values below the disability threshold. 6MMD, six minute walk distance; NSAA, North Star Ambulatory Assessment.
Discussion
CPET is the gold-standard for quantification of VO2 peak, and can be used to identify factors that limit exercise capacity in clinical populations as it provides information regarding the integrated functioning of the cardiovascular, respiratory and skeletal muscle systems during exercise. 39 Despite this, CPET is rarely used in DMD, and it is not known whether this patient population can reach physiological peak oxygen uptake because of significant skeletal muscle weakness preventing the cardiorespiratory system from reaching maximal capacity. In this study, we conducted CPET using ATS recommendations in 27 ambulatory and non-ambulatory DMD boys without adverse events. Our results highlight: 1) the feasibility of performing high-quality CPET, with the majority of patients reaching criteria reflecting maximal or near-maximal cardiorespiratory capacity despite significant muscle weakness; and 2) the multi-factorial nature of exercise limitations with abnormal muscle metabolic and cardiorespiratory responses underlying reduced VO2 peak. Additionally, we demonstrate high reproducibility of VO2 peak and correlations between VO2 peak and clinical measures of ambulatory and physical function. Collectively, these findings support the use of CPET as an integrative approach to quantify physiological function, monitor patient health and evaluate therapeutic efficacy in DMD.
Validity of peak effort CPET in DMD
To accurately interpret VO2 peak, it is essential to determine whether the participant provided maximal or near-maximal effort. 30 Peak RER is one of the most accurate and reliable gauges of subject effort in healthy and clinical populations as it is independent of motivation and reflects the physiological response (greater increase in VCO2 relative to VO2) at higher exercise intensities.5,29 Veneman et al. 14 recently validated the use of peak RER as a criterion for maximal performance in adults with neuromuscular disorders, but it has rarely been reported in DMD. 13 In our study, we discovered that 81.5% of our DMD patients met or exceeded this criterion (RER ≥ 1.01), which is similar to the percentage reported in adults with neuromuscular disorders. 14 These data indicate that valid assessment of VO2 peak is feasible in boys with DMD. Notably, valid tests were conducted in boys as young as 6 years of age (Table 2), boys with severely compromised functional abilities (i.e. NSAA score of 6) and non-ambulatory boys with extensive impairments in upper extremity function (i.e. Brooke Upper Extremity score of 3). Relevant to the potential use of CPET in evaluating therapeutics, measurement of peak RER is essential to ensure consistent patient effort in serial exercise tests. In other words, an improvement in VO2 peak following an intervention can only be ascribed to the intervention if similar maximal effort was provided during the test. In contrast, use of RPE scores on the Borg scale to gauge patient effort may not be reliable, as scores could not be obtained or did not reflect physiological effort in many patients. This is consistent with previous reports in other pediatric populations whereby RPE scores were not always captured due to concerns regarding understanding and validity.6,26
Test reproducibility
VO2 peak is highly reproducible,40,41 with intraindividual day-to-day variation reported to be between 4% to 6% in individuals with no known cardiopulmonary pathology and up to 10% in patients with various diagnoses. 20 To our knowledge, no studies have determined test-retest repeatability of VO2 peak in patients with neuromuscular disorders. 42 Our data show excellent test-retest repeatability of VO2 peak in boys with DMD and age-matched controls, with a coefficient of variation (<3%) in line with previous reports of VO2 peak. 20 High repeatability of any given outcome is important to ascertain therapeutic responsiveness. Further research is needed to establish a standardized response mean and clinically meaningful difference of VO2 peak over time in the DMD population.
VO2 peak is severely reduced in DMD
CPET revealed significantly lower VO2 peak in DMD compared to healthy age-matched boys. Based on predictive values, the majority of boys with DMD in our cohort are classified as having severe exercise intolerance (VO2 peak <50% peak predicted)13,15 Table 4. The clinical significance of quantifying VO2 peak (and improving it) lies in the fact that low VO2 peak is a strong and independent risk factor for cardiovascular, metabolic and other diseases in both adult and pediatric populations. 6 A prognostic exercise capacity of 18.0 mL/kg/min is considered a threshold for functional disability and necessary for independent community living, with values below this associated with increased risk of comorbidities (obesity, cardiometabolic disease) and mortality.43,44 Furthermore, low VO2 peak during childhood or adolescence tracks to adulthood and is associated with a clustering of metabolic risk factors and cardiovascular diseases,45–47 emphasizing the importance of quantifying VO2 peak in earlier years. In our study, almost half of the ambulatory boys (8 out of 17) achieved values below 18.0 mL/kg/min. These low values suggest that a large proportion of individuals with DMD are close to the disability threshold or have low cardiorespiratory reserve and are at increased risk of developing cardiometabolic diseases. Given these findings, therapeutic strategies targeting dystrophic muscle should aim to increase VO2 peak and cardiorespiratory reserve to have even more powerful clinical significance. While it is premature to know whether emerging DMD therapeutics (such as gene-therapy, exon-skipping, givinostat or other novel medications) or exercise training can improve these parameters, our data show that CPET offers an objective and reliable approach to quantify their physiological impact.
Classification of the severity of aerobic (VO2 peak) and ambulatory (6MWD) capacity in ambulatory boys with DMD.
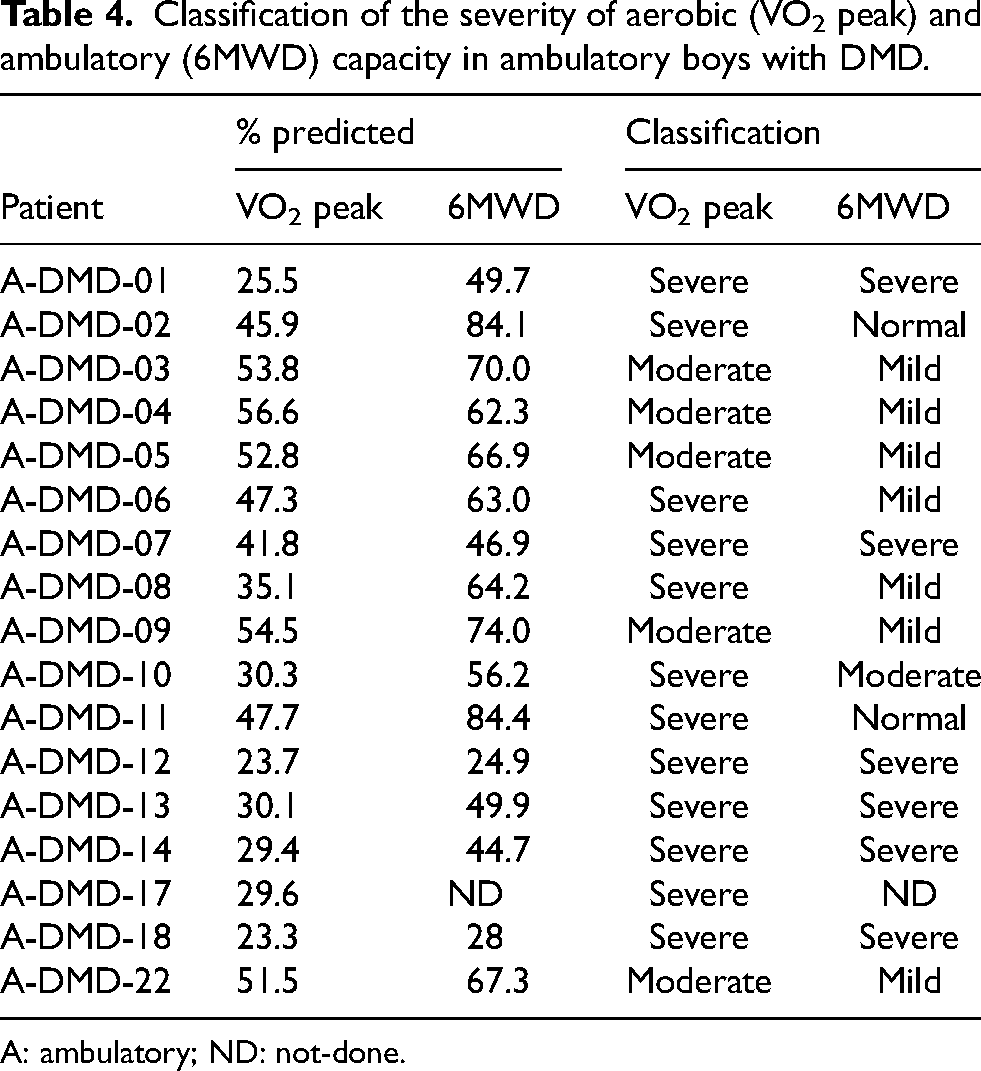
A: ambulatory; ND: not-done.
Physiological limitations to peak exercise in DMD
Muscle limitations: Given the primary pathology in DMD, skeletal muscle is expected to limit exercise capacity. This notion is based on findings that boys with DMD have low muscle and greater fat mass, 48 meaning there is less muscle available to perform aerobic work and extract oxygen. Data from our DMD patient cohort support this, showing both ambulatory and non-ambulatory DMD boys have an early onset of VT (<40%), similar to previous reports in other neuromuscular disorders.11,15 A low VT signifies a reduced capacity for oxidative metabolism within exercising muscles, consistent with prior reports in mdx mice as well as DMD patient muscle biopsy indicating impaired mitochondrial respiration and ATP generation. 49 In addition, CPET revealed a low peak work rate relative to bodyweight (watts/Kg) compared to healthy controls, indicating weakness in leg muscles used for cycling exercise. Although we did not quantify lean mass, a greater muscle fat composition would be expected to contribute to a lower VO2 peak and work relative to body weight (given less metabolically active tissue). Collectively, these results demonstrate that skeletal muscle limitations predominantly constrain peak exercise capacity in DMD. These limitations are likely due to the underlying pathophysiology (i.e. mitochondrial abnormalities relating to calcium influx, fiber atrophy, fibrofatty changes) as well as deconditioning associated with a sedentary lifestyle. Boys with DMD have low daily step counts 2 and spend most of their awake time in sedentary behaviors and low-intensity physical activity. 50 Muscle weakness and fatigue likely precipitate this sedentary behavior and collectively result in a vicious cycle of deconditioning that is quantified during CPET.
Cardiac limitations: The exercise-induced increase in cardiac output (product of stroke volume and HR) is not well-documented in DMD. 17 Prior studies have suggested that patients with neuromuscular disorders cannot attain a peak HR indicative of maximal effort exercise (>85% age-predicted maximal HR).16,19 Two prior CPET studies in DMD reported low mean peak HR (136 ± 13 bpm in 13 boys 17 ; 134 ± 7 bpm in 3 boys 13 ) and suggested that 1) boys with DMD may not have enough functional skeletal mass and strength to perform greater work and increase HR; or 2) the disease process in cardiac muscle affected maximal performance of the heart. 17 It is important to highlight two points regarding the Sockolov study. 17 First, it was performed in 1977, which was prior to corticosteroids being the standard of care for DMD and may have contributed to the lower HR response to exercise. Second, peak RER was not reported, casting uncertainty whether the low HR reflected submaximal versus maximal effort of patients during CPET. Conversely, in the study by Bartels et al., 13 peak effort was achieved in two of the three DMD boys based on the end-exercise RER despite low peak HR. In our study, mean peak HR in valid CPET assessments of ambulatory DMD boys (all on a steady regimen of steroids) was almost 40 bpm higher (172 ± 14 bpm or 85.5 ± 7.5% of age-predicted maximum) compared to these reports. Our findings are consistent with those reported in adults with neuromuscular disease, where the majority of patients achieved >85% of age-predicted maximal HR. 14 However, in our assessment of chronotropic incompetence (a common limitation to VO2 peak in patients with cardiovascular disease), the average CI index was lower (p < 0.05) in ambulatory DMD (0.70 ± 0.14) than in healthy controls (0.83 ± 0.07). While this may be taken to suggest an inadequate HR response to exercise, it may also be explained by the high resting HR in DMD patients. Resting tachycardia is common in DMD and believed to relate to decreased cardiac parasympathetic activity and/or increased sympathetic activity. 51 During incremental exercise, increases in HR are normally due to decreased vagal tone and increased sympathetic outflow. 5 Our findings demonstrate a robust increase in HR from rest to peak exercise in most boys with DMD, suggesting that these autonomic responses are at least partially intact during exercise. Furthermore, these findings reveal that skeletal muscle fatigue and weakness in DMD do not necessarily limit increases in HR.
An attenuated stroke volume can also contribute to low VO2 peak, a finding reported by Sockolov et al. 17 in boys with DMD. In our study, calculated O2 pulse was lower in DMD patients relative to controls. Although this parameter is often used as a surrogate for left ventricular stroke-volume during exercise, this assumption is only valid if arterio-venous O2 extraction is maximal. 5 This assumption may not be valid in DMD muscle as suggested by the blunted a-vO2 difference reported by Sockolov, 17 along with our current data showing a low VT. Without direct cardiac measures, we are not able to distinguish whether low peak O2 pulse reflects involvement of the heart (decreased contractility) and/or impairment in maximal muscle O2 extraction with exercise. Furthermore, a reduced stroke volume response to exercise can reflect either cardiac disease or deconditioning, noting that a few weeks of physical inactivity causes volume unloading of the heart in healthy individuals. 52 Although cardiac disease is unexpected in the ages of our patient cohort, a recent study using exercise cardiac magnetic resonance imaging (cMRI) reported reduced systolic function during in-magnet, submaximal exercise in asymptomatic young boys (mean age = 11.4 years) with DMD 53 . The authors highlight the use of exercise stress and cardiac imaging to unmask early signs of cardiac abnormalities. Collectively, their results and our findings warrant further study using both modalities (cMRI and CPET) to elucidate cardiac function during exercise in DMD.
Ventilatory response to peak exercise in DMD: Ventilation is seldom a limiting factor to peak exercise in healthy individuals. 30 In our cohort of DMD patients, peak ventilation was lower than controls, however ventilatory reserve was sufficient (>20%). This latter finding, along with normal lung volumes suggest there was little to no relative ventilatory limitation to peak exercise in DMD per se. Rather, the level of VE relative to VO2 peak was excessive (>40 liters of air per liter of oxygen uptake) in our DMD patients, reflecting ventilatory inefficiency. Similarly, the VE/VCO2 slope was elevated in DMD, suggesting a greater ventilation is needed to eliminate the same amount of carbon dioxide compared to healthy controls.
The mechanism(s) underlying this hyperventilatory response to exercise may be multi-factorial. Excess CO2 produced by bicarbonate buffering of lactic acid (resulting from increased muscle anaerobic glycolysis) can trigger exaggerated VE. Additionally, as suggested by studies in neuromuscular diseases with primary mitochondrial impairment,8,54 hyperventilation may be regulated by feedback (metabolically-sensitive chemoreceptors and ergoreflexes) directly from skeletal muscle, reflecting limited capacity for oxidative metabolism relative to oxidative demand in working muscle. Our findings of an early VT (described above) certainly support these mechanisms. Another possible underlying cause is the stimulation of mechanoreceptors from respiratory muscle weakness and inability of patients to generate tidal volumes appropriate for the level of work. 16 Insufficient tidal volume recruitment (peak VT < 50% FVC) was detected in both ambulatory and non-ambulatory groups of DMD patients, findings that have previously been reported in other dystrophinopathies 15 and Pompe disease 10 and have been taken to indicate respiratory muscle impairment. The peak inspiratory and expiratory pressures measured in our DMD patients support this notion. 55 The dynamic nature and increased demands associated with CPET may more accurately reveal the degree of weakness and fatigue of respiratory muscles in DMD. In line with this, ΔVO2/ΔWR was elevated in DMD (ambulatory DMD = 12.4 ± 4.9; non-ambulatory = 19.9 ± 9.7; controls = 10.1 ± 0.8 mL/min/watt, p < 0.05). This higher overall oxygen cost to perform work in DMD could reflect mechanical and metabolic inefficiency of muscles (including an increased work of breathing by respiratory muscles).
Comparison of CPET to functional tests
6MWD, timed function tests, and NSAA are currently used as standard assessments in clinical studies of DMD and other neuromuscular disorders. However, none of them quantify cardiorespiratory limitations to exercise or physical activity. Our data show strong correlations between VO2 peak and these standard tests. A lower VO2 peak was associated with a lower 6MWD (absolute and % predicted), a correlation which provides insight into functional capacity of patients who fall below the prognostic exercise capacity of 18.0 mL/kg/min. For example, a 6MWD of 350 meters is often used to stratify functional ability in boys with DMD. Our data reveal that boys who achieve a distance as great as 400 meters may also have underlying impairment in cardiorespiratory fitness. In addition, patients who have a 10-meter walk-run time greater than 4.0 s tended to fall into the disability threshold and may be at increased risk for developing cardiovascular or metabolic disease. While VO2 peak relates to these clinically relevant outcomes, our data suggest that in the majority of patients with DMD, it is more sensitive in detecting the severity of disability. For example, severely reduced VO2 peak was detected in patients in whom the 6MWD % predicted suggested normal or mild-moderately reduced ambulatory capacity. These findings are similar to a previous report in dystrophinopathies, 13 and suggest that CPET can reveal underlying cardiorespiratory limitations in DMD boys who have unremarkable 6MWD or NSAA scores. Furthermore, CPET can objectively distinguish between poor versus peak effort through measurement of RER, unlike the 6MWD or (other standard outcomes) which provides no measure of effort likely contributing to variability in serial testing.
The value of CPET in DMD clinical care and emerging therapeutics
CPET has been integral to the functional evaluation of patients with cardiovascular and pulmonary diseases for over half a century.5,7 It is routinely used as a prognostic indicator, in the assessment of exercise capacity, identification of underlying limitations, personalization of exercise training prescription, and investigation of treatment efficacy. These clinical applications are expected to be the same for neuromuscular disorders. 42 In our DMD cohort, at least half of the patients were either below or close to the disability threshold with low cardiorespiratory reserve. Assessment of VO2 peak in DMD may provide neuromuscular clinicians with a more global health measure to assess patient risk of developing secondary cardiometabolic disease, detect disease progression and reveal quantitatively different roles of cardiorespiratory and skeletal alterations. This has potential to personalize patient management and optimize therapeutic decision-making (i.e. which factors to target for rehabilitation). Exercise training improves cardiometabolic disease risk and increases VO2 peak in clinical populations, 7 including neuromuscular disease.56–58 Although this remains to be demonstrated in DMD, CPET could be used in the prescription of safe exercise parameters and assessment of response. The emergence of disease-modifying therapies is expected to improve skeletal muscle function and exercise capacity. This creates a promising role for CPET as it can characterize this anticipated ‘new’ DMD phenotype from a physiological perspective and document multi-systemic benefits of such therapies. For example, successful microdystrophin gene-transfer may be expected to improve myofiber resilience and resistance to fatigue, which will be quantified as improved VT, ventilatory efficiency, oxygen cost of exercise and VO2 peak.
In addition, CPET provides information complimentary to standard measures of cardiac and pulmonary function in DMD, due to the increased demands on the cardiorespiratory system during exercise, For example, annual pulmonary function testing forms the current standard of care in assessment of lung volumes (FVC and FEV1) and respiratory muscle strength (MIP, MEP). While such serial testing aims to monitor disease progression and enable early identification and treatment of pulmonary complications, 59 it does not provide insight about how ventilatory parameters respond to exercise. Our findings demonstrate CPET is sensitive in detecting ventilatory inefficiency during exercise, and reveal a degree of respiratory muscle weakness and fatigue that is not captured by respiratory strength measures done once at rest. Likewise, ECG and echocardiograms are part of standard DMD clinical assessment, however they are not always adequate to detect early, asymptomatic disease progression (preclinical cardiomyopathy). 60 Overall, our results highlight the potential of CPET to optimize clinical care for DMD patients and support the need for further longitudinal investigation across a spectrum of disease severity to demonstrate its utility and ability to provide additional value compared to traditional measures when predicting mortality.
Study limitations and considerations
We were careful to screen for abnormal cardiac function and history of rhabdomyolysis, and asked patients to report signs or symptoms of exercise-induced muscle damage following testing. There were no cardiac or other adverse events during CPET and there were no patient reports of damage following CPET. However, we did not measure the impact of peak-effort exercise on markers of muscle damage (such as serum creatine kinase, CK) in our cohort. There have been longstanding concerns that exercise can exacerbate damage in dystrophic muscle, warranting analysis of such markers in future studies using CPET. While an increase in CK may occur after an acute bout of maximal effort cycling exercise in dystrophinopathies,61,62 chronic cycle training for 12–24 weeks had no impact on CK levels. 57 In line with this, the significance of a potential CK increase given persistently high levels in dystrophic muscle is not currently known. Overall, there have been very few adverse events reported with CPET in previous studies performed in adolescents with dystrophinopathies 13 and other neuromuscular disease patients, 16 supporting its use for intermittent monitoring. While CPET can also be conducted on a treadmill, we propose the use of cycling exercise (which is non-weight bearing) as a safer modality of testing for dystrophic patients.
Prior to this study, there was no information regarding feasibility of VO2 peak assessment in non-ambulatory patients with dystrophinopathies. Here we demonstrate for the first time that non-ambulatory boys with DMD can reach RER values consistent with maximal-effort exercise, providing the possibility to quantify VO2 peak. Their lower values compared to ambulatory boys can be partly accounted for by the arm-cycling modality, which is known in healthy individuals to elicit a lower VO2 peak (∼70% of that achieved during leg cycling 4 ) given the smaller recruited muscle mass. However, even when assuming a 30% higher VO2 peak with leg-cycling, 4 they are below the threshold for functional disability. Our study only included 5 non-ambulatory boys, and whether these findings translate to a larger cohort needs to be assessed. Our overall patient cohort spanned an age range of 6 to 16 years, limiting generalization of findings to boys younger or older. We chose a minimum age of 6 years based on our experience and knowledge that very young children may not cooperate or understand testing instruction, stopping prematurely before reaching maximal criteria. 30 The safety and feasibility of CPET in older DMD boys with greater disease severity should also be considered for future research, although previous studies support the use of CPET in older adolescents and adults with dystrophinopathies.13–15 We did not detect age-related differences in VO2 peak, likely due to the low number of patients in each specified age group. Finally, our study did not include measures of lean muscle mass. To account for fibrofatty changes within dystrophic muscle, it would be valuable to consider body composition with VO2 normalization in future studies.
Conclusions
These data demonstrate that the majority of DMD patients (>80%) are capable of attaining maximal effort criteria, supporting the feasibility of CPET as a non-invasive, quantitative, repeatable and physiologically integrative approach. We report markedly impaired VO2 peak attributable to limitations not only in skeletal (locomotor and respiratory) muscle but also in the cardiorespiratory responses to exercise, and propose CPET as a functional marker of disease impact in DMD. Collectively, these findings support continued investigation of CPET as an objective and potentially more sensitive approach than current motor outcomes for assessing exercise capacity and cardiorespiratory reserve, monitoring disease progression and determining whether any given intervention has integrated physiological benefits.
Footnotes
Acknowledgements
The authors thank the boys and their parents for their participation and valuable contribution.
Funding
The research reported in this publication was supported by funding awarded to T. Taivassalo from the Muscular Dystrophy Association (award number MDA875732) and the Department of Defense, Congressionally Directed Medical Research Programs (CDMRP), Award Mechanism, # MD180023.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author.
Description and significance of CPET variables assessed in this study. Normal responses are indicated. HR = heart rate SV = stroke volume A-vO2 difference = difference in oxygen content between arterial and venous blood Increases ∼ linearly with exercise intensity to reach plateau at maximal effort Wide range of peak values in healthy individuals Disability threshold for VO2 peak is <18 mL/kg/min
42
Between 45–65% VO2 peak in untrained Lower values reflect increased reliance on glycolysis and suggest muscle metabolic limitation to peak exercise
5
Linear increase Normal slope = 10 mL/min/watt
5
Linear increase with exercise intensity until VT Generally not limiting to peak exercise 30–40 liters of air per liter of oxygen uptake is normal at peak exercise
62
Calculated as linear regression function, excluding the non-linear data after onset of acidosis-related increase in ventilation <30 is considered normal (slightly higher with advanced age)63 Values >34 associated with worse prognosis in cardiopulmonary diseases MVV = maximum voluntary ventilation MVV is estimated as FEV1 × 35 at rest Describes how closely peak VE approaches MVV Reduced VR suggests pulmonary limitation Ventilation is considered limiting when VR <15–20%
61
HR reserve = age-predicated maximal HR − resting HR >0.80 at peak RER Lower values reflect inability of HR to increase commensurate with increased activity
30

CPET variable
Definition
Description
Normal response / values to incremental peak exercise
VO2 peak (L/min or mL/kg/min)
Volume of oxygen uptake during exercise; VO2 peak refers to the highest VO2 an individual can consume
Commonly referred to as cardiovascular fitness, VO2 (mL/min) = (HR × SV) × a-vO2 difference as per Fick equation
5
VT
Ventilatory threshold
Point at which minute ventilation increases disproportionally relative to VO2, often described by a change in slope of VCO2 plotted against VO2
RER peak
Respiratory exchange ratio
VCO2/VO2
O2 pulse (mL/beat)
Oxygen pulse = VO2/HR
Illustrates central cardiac adaptation to exercise and is a surrogate for SV and a-vO2 difference during exercise
ΔVO2/ΔWR
Oxygen cost of work; Ratio of VO2 to resistance in watts
Reflects the efficiency of the metabolic conversion of chemical potential energy to mechanical work and the mechanical efficiency of the musculoskeletal system
28
VE (L/min)
Ventilation
Product of tidal volume and respiratory rate
VE/VO2
Ventilatory equivalent of oxygen uptake
Ventilatory cost of oxygen uptake and marker of ventilatory efficiency
VE/VCO2 slope
Relationship between ventilation (y axis) to VCO2 (x-axis)
Liters of air breathed to eliminate 1 liter of CO2, marker of ventilatory efficiency
VR
Ventilatory reserve = peak VE/MVV
Peak exercise ventilation divided by predicted MVV
Vt elevation
Tidal volume relative to forced vital capacity
Tidal volume recruitment at peak exercise
CI
Chronotropic index
Change in HR from rest to peak exercise relative to HR reserve
