Abstract
Background
Endurance exercise improves aerobic capacity (VO2peak) and motor symptoms in people with early Parkinson's disease (PD). Some people with PD exhibit signs of chronotropic incompetence (CI), which may impact exercise-induced benefits.
Objective
We investigated whether CI in people with early PD influences the change in motor signs, VO2peak, and peak heart rate (HR) following 6 months of endurance exercise.
Methods
We performed secondary analyses of the Study in Parkinson's Disease of Exercise (SPARX), which randomized people with early PD into a high-intensity endurance exercise [80–85% of peak HR], moderate-intensity endurance exercise [60–65% of peak HR], or usual care group. MDS-UDPRS Part 3 score, VO2peak, and heart rate (HR) response to maximal cardiopulmonary exercise testing (CPET) were analyzed at baseline and following 6 months of exercise. Participants were divided into three groups: 1) normal chronotropic response at baseline, 2) CI at baseline, and 3) taking medications with a known negative chronotropic effect regardless of CI status.
Results
Data from 119 individuals (64.0 ± 9.0 years, 57.1% male, 0.3 years since diagnosis [median]) were analyzed. There were no differences among the groups in change in MDS-UPDRS motor score (p = 0.953), VO2peak (p = 0.965), or peak HR (p = 0.388). People randomized into the high-intensity group improved VO2peak compared to usual care (p < 0.001adj) regardless of CI status.
Conclusions
Baseline CI did not alter responses to endurance exercise in those with early PD, suggesting that the beneficial effects of endurance exercise on disease progression and VO2peak in people with early PD apply equally to people with CI.
Plain language summary
Exercise training, such as walking on a treadmill, helps improve fitness and symptoms of Parkinson's disease (PD) in those who have recently been diagnosed with PD. We aimed to determine if these beneficial effects of exercise were also experienced by those with PD who may have a hard time increasing their heart rate during an exercise test. This study looked at the effects of different intensities of 6 months of treadmill exercise and the ability to get one's heart rate up to where one might expect based on age during exercise testing on motor symptoms of PD and exercise capacity. Regardless of one's ability to increase their heart rate to an expected value based on age, individuals with early PD experienced benefits from 6 months of treadmill exercise training. This suggests that the beneficial effects of exercise training on disease progression and exercise capacity in early PD apply equally to those with normal or abnormal heart rate responses to exercise testing.
Introduction
Parkinson's disease (PD) is a neurodegenerative disease characterized by motor and nonmotor symptoms. 1 Motor symptoms include bradykinesia, tremor, rigidity, gait impairment, and postural instability, while nonmotor symptoms include depression, apathy, cognitive impairments, autonomic dysfunction, and more. 1 Cardiovascular autonomic dysfunction in PD can manifest as abnormal blood pressure and heart rate responses to standing (i.e., orthostatic hypotension) and during exercise (i.e., chronotropic incompetence; CI).2,3 Autonomic dysfunction in PD is associated with more rapid disease progression. 4 Recent data demonstrate that CI is present in a subset of people with early PD, and that this CI is not attributable to poor effort. 5
Abnormal hemodynamic responses to maximal acute endurance exercise have been reported in PD using both treadmill and cycle ergometer.6–10 The abnormal heart rate response in PD during cardiovascular function testing is thought to be related to cardiovascular autonomic impairment. 11 An insufficient heart rate response during exercise testing, because of CI, may be a major limiting factor in the exercise capacity of people with PD and may negatively impact exercise-induced benefits.
There are no disease-modifying medications for PD, but there is substantial evidence that endurance exercise has favorable effects on motor symptoms and may slow the progression of the disease.12–14 While the response of skeletal/somatic motor function and mobility to endurance exercise training in people with PD has been studied,12–14 little attention has been paid to the effects of CI on exercise efficacy. What remains unknown is whether endurance exercise has the same effect on motor signs, exercise capacity, or heart rate response to maximal exercise in people with early PD who demonstrate CI compared to those with a normal chronotropic response. There are several possible reasons why CI may influence exercise-induced benefits in PD negatively: 1) accurately prescribing exercise in individuals with CI is challenging, and target zones based on a percentage of measured peak HR may be too low to improve cardiorespiratory fitness, 2) exercise-induced benefits in PD may rely on the appropriate stimulation of the cardiovascular system to match increased metabolic demands from working muscles, 3) people with PD with cardiovascular autonomic dysfunction may be at higher risks of complications during exercise secondary to exercise-induced hypotension or exaggerated post-exercise hypotension, which could lead to falls, syncope, and injury. 15
To determine whether CI influences the rate of progression of motor symptoms and change in peak aerobic capacity (VO2peak) following 6 months of treadmill exercise in people with early PD, we performed a secondary subgroup analysis of the Study in Parkinson's Disease of Exercise (SPARX, NCT01506479) clinical trial. SPARX was a multicenter, randomized, controlled, single-blinded, Phase II study designed to test the effects of high-intensity treadmill endurance exercise on the motor symptoms of PD in participants within 5 years of diagnosis not yet taking dopaminergic medication. 16 Our goal was to investigate whether baseline CI in people with early PD influenced changes in motor signs as measured by the Movement Disorders Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) Part 3 score, VO2peak, and peak HR following 6 months of endurance exercise training.
Methods
Standard protocol approvals, registrations, and consent
The trial was approved by the Institutional Review Boards of participating sites (University of Colorado, University of Illinois at Chicago, Northwestern University, Rush University Medical Center, University of Pittsburgh). Study participants provided written informed consent before participation in any study procedures. Detailed study procedures have been previously reported. 16
Study participants
The SPARX trial recruited participants with idiopathic PD within five years of diagnosis. Participants were 40–80 years old, had a Hoehn and Yahr stage of 1–2, were not taking dopaminergic medication upon enrollment, and were not expected to begin dopaminergic medication within 6 months of enrollment. Exclusion criteria included uncontrolled cardiovascular disease, an abnormal stress test, failure to meet the United Kingdom Brain Bank criteria for idiopathic Parkinson's disease, and participation in moderate-to-vigorous physical activity more than 3 days per week. 17
Health history and clinical assessments
Participant clinical and demographic data were obtained via self-report questionnaires and participants were assessed by trained clinicians at baseline. Study neurologists administered the Movement Disorders Society Unified Parkinson's Disease Rating Score (MDS-UPDRS).
Medication usage
The use of medications with a negative chronotropic effect (i.e., beta-blockers and others) was documented during the baseline health history and medical records review and was again confirmed on the day of the cardiopulmonary exercise test (CPET). 5 Although all participants enrolled in SPARX were naïve to dopaminergic medications, 21 participants started dopaminergic medications before Month 6 clinical assessments and CPET. For Month 6 analyses, participants who started taking dopaminergic medications had their last medication-naïve clinical assessments imputed. All CPETs were performed at Month 6 regardless of medication status. When possible, the Month 6 CPET was conducted off medication, with a 12-hr washout of dopaminergic medications.
Cardiopulmonary exercise testing
Participants completed a CPET on a treadmill at baseline and after 6 months of the study intervention. 18 The protocol was individualized to each participant by determining treadmill speed during the warm-up stage of the test. The protocol consisted of a constant walking speed with grade increased by 2% every two minutes until volitional fatigue was reached. Expired gas analyses were performed to measure oxygen consumption and carbon dioxide production (ParvoMedics TrueOne 2400, Salt Lake City, UT for Chicago and Denver; AEI Technologies, MOXUS, Bastrop, TX for Pittsburgh). HR, blood pressure (BP), and rating of perceived exertion (RPE) were recorded at the end of each two-minute stage.
Heart rate monitoring
HR was monitored with an electrocardiogram (ECG) before, during, and after the CPET. HR values were obtained prior to the test in the supine, seated, and standing positions. HR was recorded during the last 30 s of each stage during the CPET. Peak HR was defined as the highest HR achieved during the CPET. CI was defined as a failure to achieve 85% of age-predicted maximal heart rate (APMHR) (calculated as 220-age). 19
Screening, baseline testing, and randomization
Participants who met screening criteria underwent baseline assessments. Participants were randomly assigned to high-intensity treadmill exercise (80%-85% peak HR), moderate-intensity treadmill exercise (60%-65% peak HR), or usual care. The usual care group was instructed to maintain baseline exercise habits throughout the 6 months. Full screening, baseline testing, and randomization procedures have been previously reported. 12
Heart rate response groups
We stratified participants based on their chronotropic response to baseline CPET and whether they were taking chronotropic medications. 5 The first group consisted of participants who were not taking negative chronotropic medications and did not display evidence of CI (PDnon-chrono). The second group consisted of those not taking negative chronotropic medications but demonstrating CI (PDchrono), defined as having a measured peak HR less than 85% of age-predicted maximal HR. The third group was defined as those taking medications with a known negative chronotropic effect (PDchronomed), regardless of chronotropic response to baseline CPET. These groups have been previously detailed by Griffith and colleagues. 5
Exercise intervention
Treadmill exercise was prescribed for 4 days per week for 6 months. Each exercise session included 5 to 10 min of warm-up, 30 min of treadmill exercise in the prescribed HR zone, and 5 to 10 min of cool down. Exercise frequency and intensity were increased during weeks 1 to 8 to reach these target exercise prescriptions. Thereafter, target HR was maintained by adjusting treadmill speed and/or grade, as necessary.
Participants used a HR monitor (Polar, Lake Success, NY) to record the intensity of all exercise sessions. All sessions in weeks 1 to 2 were supervised at the study site. Thereafter, participants completed at least one on-site exercise session per month, during which HR data for the past month were downloaded. Protocol adherence was ensured by monthly conference calls with study coordinators.
Statistical analyses
Baseline continuous variables were compared using a one-way analysis of variance (ANOVA) with a Bonferroni correction (adjusted p-values indicated with the subscript adj), and categorical variables were analyzed with a Chi-squared test. Month 6 change scores (i.e., Month 6 – baseline) were compared using a two-way ANOVA, with baseline chronotropic group and intervention arm as the independent variables. Post hoc analyses between sets of two intervention arms were performed using independent samples t-tests.
Results
Sample size determination
A total of 128 participants were enrolled into SPARX, with 9 participants being lost to follow-up prior to completion of Month 6 clinical assessments (Figure 1). Therefore, data from 119 participants were included in the final analyses. MDS-UPDRS data were available for all 119 participants, 21 of whom had started dopaminergic medications (62% were in the PDnon-chrono group, 14% in the PDchrono group, and 24% in the PDchronomed group). Of these 21 participants, 7 individuals had baseline MDS-UPDRS data imputed for Month 6, 7 individuals had Month 3 MDS-UPDRS data imputed for Month 6, and 7 individuals had special assessment MDS-UPDRS data (i.e., off-schedule clinical visits immediately prior to starting medication between months 3 and 6) imputed for month 6. Data from n = 112 and n = 114 were available for change in VO2peak and peak HR, respectively.
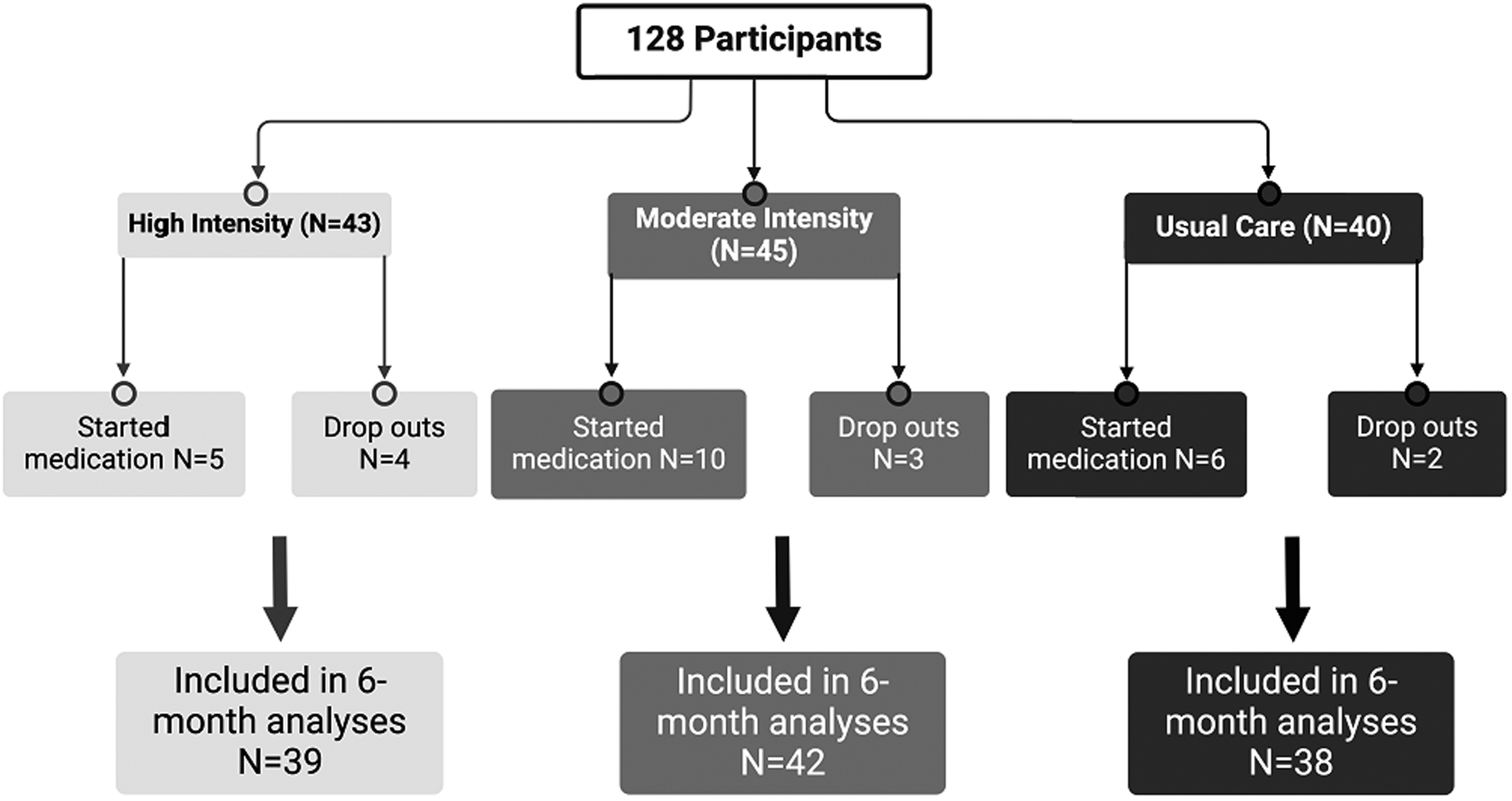
Number of participants remaining in each intervention arm following 6 months of endurance exercise.
The final analysis included 84 participants without baseline CI, 10 participants with baseline CI, and 25 participants who were on negative chronotropic medications (Figure 1).
Baseline demographic and clinical characteristics
Baseline demographic and clinical characteristics of study participants are shown in Table 1. There were no demographic differences between the study groups at baseline, other than a significantly greater proportion of male participants in the PDchrono group compared to other groups (Chi-square p = 0.025). There was a difference in resting (F[2,116] = 9.689, p < 0.001) and peak HR (F[2,116] = 27.487, p < 0.001) among the 3 groups. Post hoc testing showed that the mean resting HR in the PDnon-chrono group was higher than the PDchrono group (p = 0.002adj) and the PDchronomed group (p = 0.006adj). Post hoc testing also showed that the mean peak HR in the PDchrono group was lower than both the PDnon-chrono (p < 0.001adj) and PDchronomed (p = 0.029adj) groups.
Baseline demographic and clinical characteristics of SPARX individuals who completed the 6 month intervention.
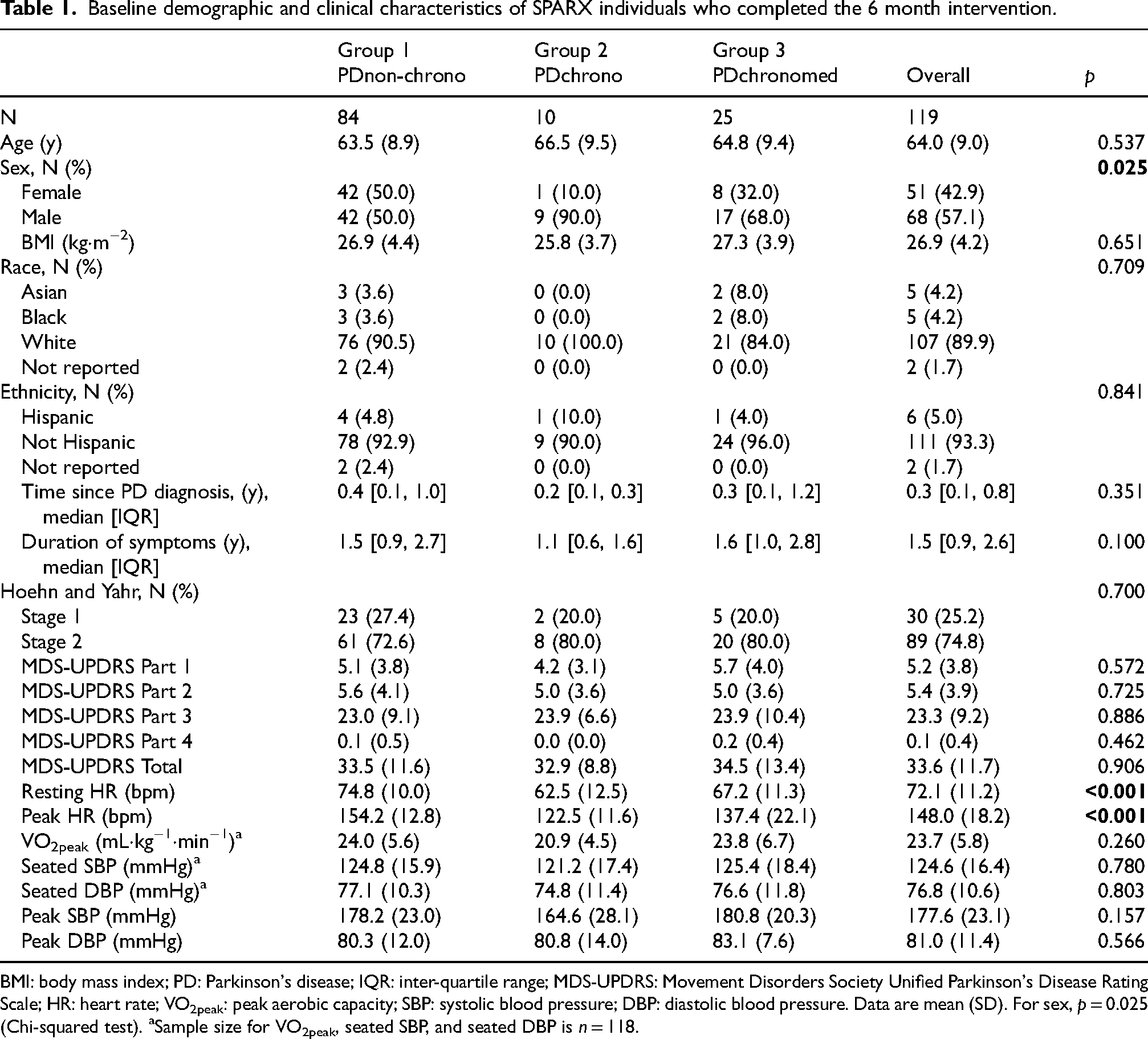
BMI: body mass index; PD: Parkinson's disease; IQR: inter-quartile range; MDS-UPDRS: Movement Disorders Society Unified Parkinson's Disease Rating Scale; HR: heart rate; VO2peak: peak aerobic capacity; SBP: systolic blood pressure; DBP: diastolic blood pressure. Data are mean (SD). For sex, p = 0.025 (Chi-squared test). aSample size for VO2peak, seated SBP, and seated DBP is n = 118.
Intervention characteristics
Compliance with the exercise prescription and adverse events in SPARX were previously reported. 12 The intervention arms showed no difference in compliance as defined by number of days per week performed by the high intensity and moderate intensity groups, and each group's mean exercise intensity fell within its prescribed range. There were 2 serious adverse events, both documented in the moderate intensity group and confirmed to be unrelated to the exercise intervention.
Change in motor signs
Figure 2 depicts the change in MDS-UPDRS Part 3 (Month 6 – baseline) based on the randomized exercise group and chronotropic status group. There was no significant interaction between baseline chronotropic status and intervention arm (F[4110] = 0.535, p = 0.711n). There were no significant differences in change in MDS-UPDRS Part 3 based on chronotropic status (F[2116] = 0.048, p = 0.953). Participants in the high-intensity exercise group significantly reduced their MDS-UPDRS Part 3 score compared to those in the usual care group (p = 0.034). There was no difference between the high-intensity and moderate-intensity exercise groups (p = 0.402), or between the moderate-intensity and usual care groups (p = 0.147). 12 Pre, post, and change data for MDS-UPDRS Part 3 are included in Supplemental Table 1.

Effect of endurance exercise on change in MDS-UPDRS part 3 score (Month 6 – baseline) for each chronotropic group. A positive change score represents an increase in MDS-UPDRS Part 3 score following 6 months of endurance exercise. Sample size for change in MDS-UPDRS part 3 score is n = 119. Data are mean ± SE.
Change in exercise capacity
Figure 3 depicts the change in VO2peak (Month 6 – baseline) based on the randomized exercise group and chronotropic status group. There was no significant interaction between baseline chronotropic status and intervention arm (F[4103] = 0.343, p = 0.849). There were no significant differences in change in VO2peak based on chronotropic status for the entire sample (F[2109] = 0.035, p = 0.965). There was a significant difference in the change in VO2peak among the 3 randomized groups (F[2109] = 3.676, p = 0.029). Post hoc testing showed that the mean change in VO2peak in the high-intensity exercise group (1.92 ± 2.87 mL/kg/min) was greater than individuals in the usual care group (−1.30 ± 2.53 mL/kg/min, p < 0.001adj). There were no statistically significant differences in change in VO2peak for those participating in the moderate-intensity group when compared to either high-intensity exercise (p = 0.085adj) or usual care (p = 0.230adj). Pre, post, and change data for VO2peak are included in Supplemental Table 1.
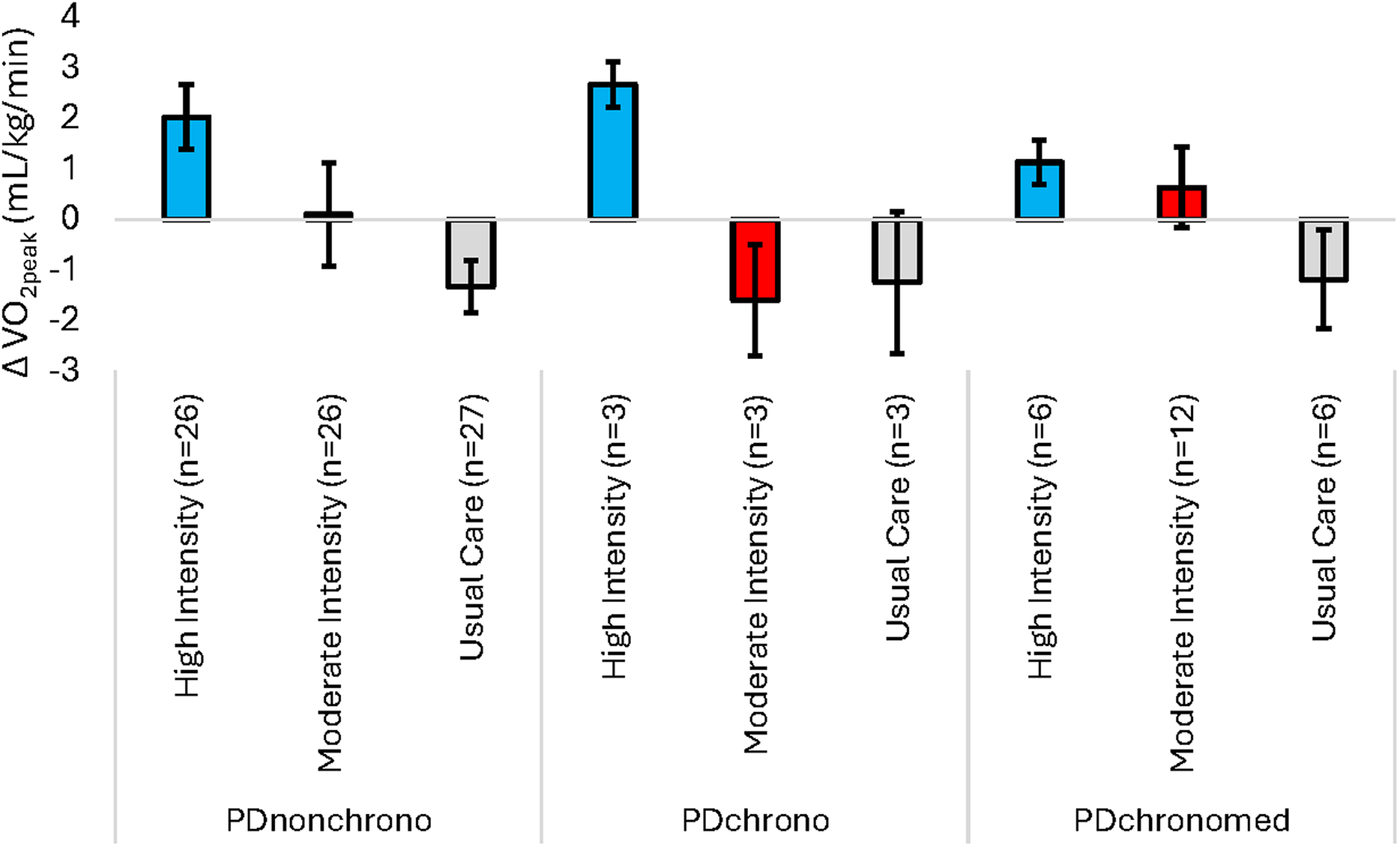
Effect of endurance exercise on change in VO2peak (Month 6 – baseline) for each chronotropic group. A positive change score represents an increase in VO2peak following 6 months of endurance exercise. Sample size for change in VO2peak is n = 112. Data are mean ± SE.
Change in peak heart rate
Figure 4 depicts the change in peak HR (Month 6 – baseline) based on the randomized exercise group and chronotropic status group. There was no significant interaction between baseline chronotropic status and intervention arm (F[4105] = 0.919, p = 0.456). There were no significant differences in change in peak HR based on baseline chronotropic status (F[2111] = 0.956, p = 0.388) or randomized group (F[2111] = 2.073, p = 0.131). Pre, post, and change data for peak heart rate are included in Supplemental Table 1.
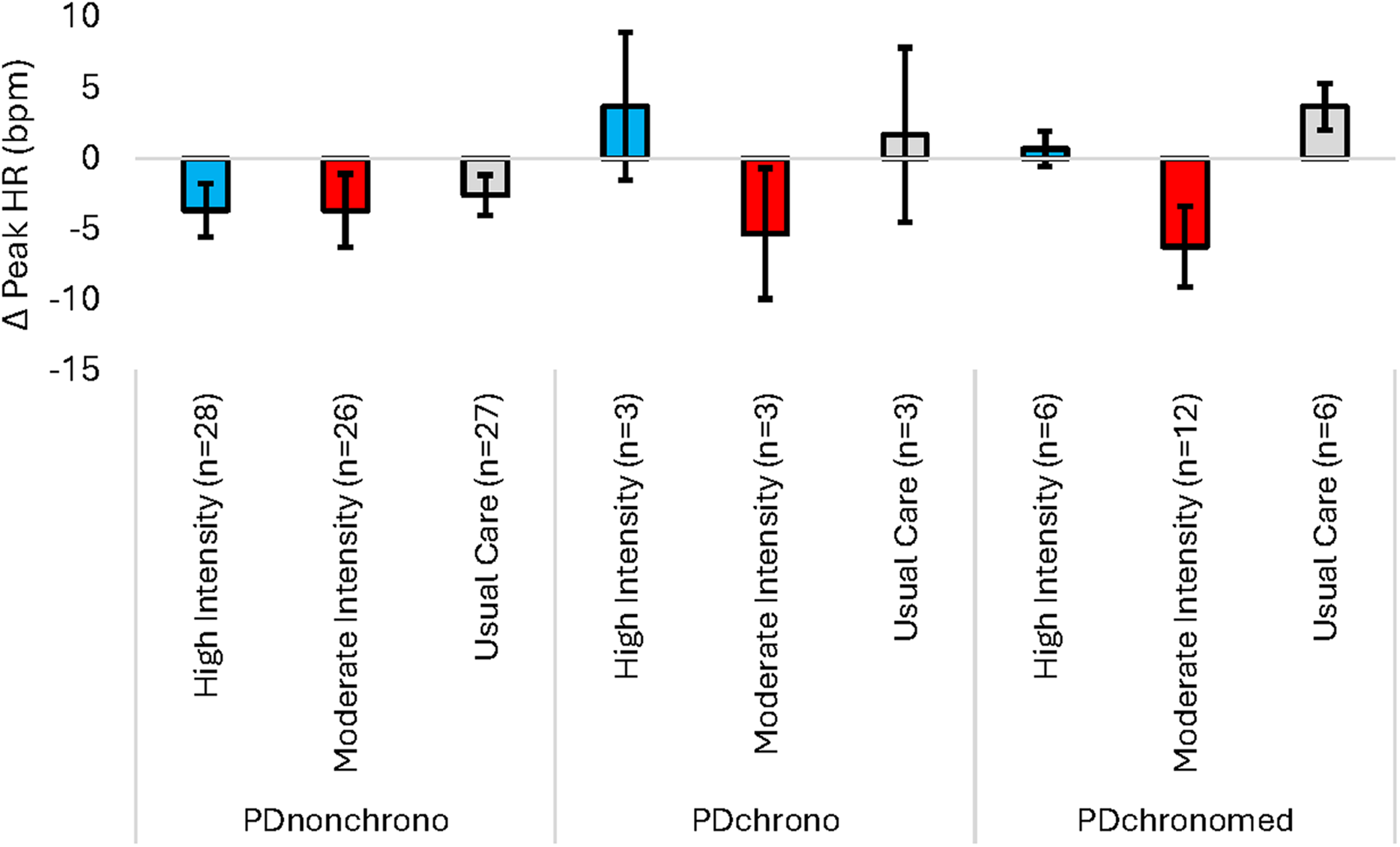
Effect of endurance exercise on change in Peak HR (Month 6 – baseline) for each chronotropic group. A positive change score represents an increase in Peak HR following 6 months of endurance exercise. Sample size for change in peak HR is n = 114. Data are mean ± SE.
Discussion
The results of this study suggest that chronotropic incompetence does not influence the changes in MDS-UPDRS Part 3 score, VO2peak, and peak HR following 6 months of treadmill endurance exercise in people with early PD. These results suggest that the previously reported beneficial effects of endurance exercise on disease progression and exercise capacity in people with early PD 12 apply equally to people with CI.
We previously demonstrated that high-intensity endurance exercise slowed the progression of motor signs when compared to moderate-intensity exercise or usual care. 12 This finding has been replicated by two subsequent studies.20,21 The present study suggests a benefit of high-intensity endurance exercise training on motor function in people with early PD regardless of the presence or absence of CI. It may even be that the motor function of those with CI benefits to a greater extent than those without CI, particularly following participation in high-intensity endurance exercise (Figure 2); however, these data did not reach statistical significance, and additional research is required to further explore this possibility.
Multiple studies have found that participation in endurance exercise training improved VO2peak in adults both with12,22–24 and without PD.25–30 Improvements in VO2peak may lead to reductions in fatigability and improved health outcomes in individuals with PD. 31 Our data confirm this effect and demonstrate that high-intensity endurance exercise was more effective than moderate-intensity endurance exercise for improving exercise capacity regardless of the presence of CI. It is possible that endurance exercise training for 6 months improves central hemodynamic parameters (i.e., stroke volume) in people with PD. This is a well-known adaptation to endurance exercise training and might be more important compared to a change in peak HR. Therefore, prescribing endurance exercise based on a percentage of measured peak HR during CPET may be sufficient in PD and is inclusive of those with PD and CI in whom prescribing endurance exercise based on age-predicted maximal HR is not feasible.
Our previous research showed that prevalence of CI in people with recently diagnosed PD may be between 10–20%, although estimates of CI prevalence are difficult to ascertain due to the potential for chronotropic medications to impact maximal HR during CPET. 5 Our current findings suggest that peak HR during CPET following 6 months of endurance exercise training in people with early PD remains unchanged from baseline. Some studies in clinical populations with reduced peak HR have shown an increase in peak HR following exercise training. Keteyian and colleagues showed an increase of 12 bpm in people with heart failure and baseline CI following a 24-week exercise training program. 32 Jin and colleagues reported an increase of 11 bpm following 12 weeks of exercise training in a study sample of 30 individuals with type 2 diabetes who reached a mean peak HR at baseline of 80.4% of APMHR. 33 The differential response of peak HR to exercise training between our findings in people with PD and these studies of people without PD may be influenced by inherent pathophysiological or clinical differences between these populations and PD, lower baseline fitness levels of patients in these studies compared to our cohort, or differences in autonomic plasticity among these populations. Assessment of peak HR via CPET has demonstrated high test-retest reliability in clinical populations,34,35 although data suggest high within-subjects variability. 36 Additional research is required to explore potential factors that may influence whether peak HR can be increased in people with PD and CI in response to endurance exercise training.
Importantly, while most people in our study completed their Month 6 CPET off dopaminergic medication, some were on dopaminergic medication during the assessment. Available data in the literature suggest a possible cardioinhibitory effect of levodopa at rest.37,38 However, the effect of dopaminergic medications on the heart rate response to acute and chronic endurance exercise in PD requires further investigations.
Nine of the initial 128 individuals (7.0%) were lost to follow-up during the 6 month study. However, there were no differences in attrition from the study or initiation of dopaminergic therapy based on exercise intervention arm or baseline chronotropic status group. This suggested that baseline CI does not necessarily predispose people with early PD to a particularly poor experience with endurance exercise training or increase the likelihood of development of symptoms that may lead to pharmaceutical management of PD. These findings, combined with the similar impact of endurance exercise training on exercise capacity, motor function, and peak HR between the three chronotropic groups, support the conclusion that all individuals with PD benefit from endurance exercise. Therefore, exercise prescriptions should be individualized based on peak HR as measured by CPET, even in the presence of CI.
Strengths of this study included enrollment of people with medication naïve PD to minimize potentially confounding effects of medication, the application of an exercise intervention that has been demonstrated to improve VO2peak and slow the progression of PD as measured by the MDS-UPDRS Part 3 score, 12 and objective assessment of effort during CPET. 5 The primary limitation of this study was the relatively small sample sizes for certain study subgroups, a result of analyses that require grouping participants by both intervention arm and baseline chronotropic status. Additional research with a more robust study sample is required to further understand the potential impacts of CI on the response to exercise in PD and the subsequent progression of clinically relevant outcomes. Further, this study did not collect data on cardiac output or stroke volume, limiting our ability to determine the effect of exercise training on these parameters in PD.
Future research can build upon this work by including additional measures and analyses of autonomic function, such as HR recovery following exercise to study potentially differential responses to endurance exercise training in PD patients with CI. These measures have been incorporated in studies investigating autonomic function following maximal exercise testing39,40 and endurance exercise training sessions. 41 Complementary cardiovascular assessments such as standardized orthostatic blood pressure measurements may also contribute to a more thorough understanding of autonomic function and how it may change following participation in endurance exercise.
Conclusion
In this secondary analysis, we demonstrated that people with early PD, regardless of chronotropic status, may benefit from high-intensity endurance exercise. The resulting reduction in the MDS-UPDRS Part 3 score and improvement in VO2peak provide clinical and physiological support for incorporating endurance exercise training as part of a comprehensive approach to PD management in those with and without CI.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241308813 - Supplemental material for Effects of 6 months of endurance exercise on motor function, exercise capacity, and autonomic function based on presence of autonomic dysfunction in individuals with early Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241308813 for Effects of 6 months of endurance exercise on motor function, exercise capacity, and autonomic function based on presence of autonomic dysfunction in individuals with early Parkinson's disease by Garett J Griffith, Niyati Mehta, Guillaume Lamotte and Kathleen E McKee, Erin Suttman, Jacob M Haus, Elizabeth Joslin, Katherine Balfany, Wendy M Kohrt, Cory L Christiansen, Edward L Melanson, Lana M Chahine, Demetra D Christou, Charity G Patterson, Daniel M Corcos in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
ORCID iDs
Funding
The Study in Parkinson's Disease of Exercise (SPARX) trial was funded by grant NINDSR01 NS074343. Support for this paper was provided by U01 NS113851 from the National Institute of Neurologic Disease and Stroke. The research was further supported by the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the National Institutes of Health under award number T32HD101395.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data sharing is not applicable to this article as this is a secondary analysis, and no datasets were generated during this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
