Abstract
Background:
In the Netherlands a centralised approach to respiratory care for patients with Amyotrophic Lateral Sclerosis is used based on national guidelines. Patients with Amyotrophic Lateral Sclerosis are referred to one of 4 centres for Home Mechanical Ventilation.
Objective:
Our aim was to evaluate the respiratory care according to the Dutch guideline by evaluation of reasons for starting non-invasive ventilation, timing of initiating and survival in patients with Amyotrophic Lateral Sclerosis using non-invasive ventilation.
Method:
A retrospective chart-review was performed of 323 patients, who had been referred to centres for Home Mechanical Ventilation in 2016–2018. Data collected included symptoms of hypoventilation, forced vital capacity, blood gasses, criteria for (not) initiating non-invasive ventilation, and survival. Kaplan-Meyer curves and Multivariate Cox proportional hazard regression were used in the analysis.
Results:
The main criteria used for initiating non-invasive ventilation were hypercapnia (77%) and the presence of orthopnea and/or dyspnoea (25%). Median survival after starting non-invasive ventilation was 11 months, and was shorter for patients with bulbar disease onset and older age. The proportion of the total disease duration that was spent on non-invasive ventilation was not significantly affected by age, sex or site of disease. Seventy nine percent of the patients who didn’t start non-invasive ventilation had reached a joint decision with their caregivers and/or physicians.
Conclusion:
Key outcomes of the Dutch centralised respiratory care approach have shown that most patients were initiated on non-invasive ventilation due to presence of hypercapnia and/or dyspnoea/orthopnea, which is according to the Dutch guidelines. Half of patients spent at least 33% of their disease duration on non-invasive ventilation. To help find the optimal criteria and timing for non-invasive ventilation it would be useful for other countries to share their key outcomes as well.
Keywords
Introduction
Respiratory failure is the main cause of death in patients with Amyotrophic Lateral Sclerosis (ALS).1,2 Non-invasive ventilation (NIV) is recommended to relieve symptoms of hypoventilation, and has been shown to improve quality of life and prolong survival in patients with ALS.3–5 As there is no evidence based consensus on the optimal indication for, or timing of NIV, recommendations in current guidelines are mainly based on expert opinion, 6 resulting in considerable differences between countries.7–9 Furthermore, common practices of prescribing non-invasive ventilation use differ among international ALS specialists. 6 More evidence for the optimal timing of NIV is needed in order to achieve more robust and uniform guideline recommendations.
However, conducting randomised controlled trials to expand the evidence base for the timing of NIV is difficult, as patients may be reluctant to be randomised into an ‘early’ or ‘late’ NIV group10,11 or in order to achieve the estimated sample size. 12
So far, several studies, most of them retrospective, have investigated the effect of NIV on survival, but lacked information on NIV initiation practices; which could therefore not be evaluated.13–15 Furthermore, one study provided a detailed description of the timing of NIV and whether initiation practices adhered to NIV guidelines. 16 They evaluated criteria used to initiate NIV as symptoms, PaCO2, forced vital capacity (FVC), maximal inspiratory pressures and time spent with SpO2 < 90% at night. They reported that in most cases, NIV was initiated according to the national NIV guidelines. In this study the authors’ concluded that NIV was initiated quite late in the disease course, because the majority of the patients started NIV at the stage of symptomatic daytime hypoventilation.
In recent years, several studies have been carried out to evaluate the differences between “early” and “late” initiation of NIV. A randomised controlled trial has recently been published on this topic. 12 Although not all of the results in this study reached statistical significance, they state that all of the data analysed is in favour of early NIV. In the attempt to improve NIV practices and respiratory care in general, (national) population-based studies could be conducted, as they provide a reliable representation of current NIV practices in a specific population or country. 17 By doing so, we can evaluate the adherence to NIV guidelines, identify shortcomings in respiratory and/or NIV care, and optimally inform patients with ALS about the expected benefits of NIV in their country specific care setting. In addition, national population-based NIV data could be compared between countries and be used to optimise NIV practices.
Respiratory care for patients with ALS in The Netherlands
There are 38 certified multidisciplinary care teams throughout the Netherlands, specialised in the management of ALS. As a result all patients with ALS receive specialised care close to home. After diagnosis by a neurologist, the patients are referred to one of the multidisciplinary care teams, which consist of at least a physiotherapist, occupational therapist, speech therapist, psychologist, dietician and social worker. Each team works under the guidance of a rehabilitation physician and according to Dutch guidelines for the management of ALS. 18 One of the responsibilities of a care team is regular monitoring of respiratory function, through physical examination, spirometry and blood gas analysis. As described in the Dutch guidelines for ALS, the patients will be referred to a centre for Home Mechanical Ventilation (HMV) when they want to get additional information about respiratory care and (non) invasive ventilation, experience signs of respiratory dysfunction or show a decline on a pulmonary function test. The centres for HMV are spread over four regions and cover all patients on HMV in the Netherlands. The organisation of centres for HMV in the Netherlands is described in the article of Van den Biggelaar et al. 19
After referral, the centre for HMV will take over respiratory monitoring from the referring multidisciplinary care team, examine patients for signs of nocturnal hypoventilation and provide comprehensive information about NIV. Once signs and/or symptoms of respiratory insufficiency are detected, a shared-decision on NIV is made.
The strength of this unified approach is that all patients in The Netherlands have access to the best quality respiratory care, irrespective of where they receive multidisciplinary care.
NIV guidelines
The Dutch guidelines for long-term mechanical ventilation20,21 recommend that patients with ALS should be referred electively, if onset of respiratory dysfunction is expected in the near future, or when patients want information about the benefits and drawbacks of chronic NIV. The guidelines also describe that patients should be referred if at least one of the following criteria is met: partial pressure of blood carbon dioxide (PCO2) > 45 mmHg (or >6 kPa), bicarbonate concentration >30 mmol/L, FVC < 50% of predicted, peak cough flow <300 L/min, recurrent airway infections, signs or symptoms of nocturnal hypoventilation (orthopnea, nightmares, disturbed sleep, night sweats, morning headache, nocturnal dyspnoea, hypersomnolence, concentration difficulties). Measurement of pCO2 can be assessed both while being awake or during sleep by an arterial/capillary blood sample or by a transcutaneous measurement.
The criteria used for the initiation of NIV are the presence of symptoms due to hypercapnia (pCO2 > 45 mmHg or >6.0 kPa) and/or symptoms of orthopnea. In case of hypercapnia without symptoms, the physician may consider waiting to start NIV. This decision is made together with the patient and close monitoring is mandatory.
Currently, there is very limited data on this approach to NIV care for patients with ALS in The Netherlands. For this reason, we aim to determine the criteria used for, and the timing of referral to a centre for HMV and (not) initiating NIV, and the survival after starting NIV in a population-based cohort of patients with ALS in The Netherlands.
Methods
Study design, setting and population
In this retrospective, national population-based study, we extracted data from electronic health records (EHRs) of two centres for HMV based in the University Medical Centres of Utrecht and Groningen in the Netherlands. Two-thirds of all Dutch patients with ALS who are referred to a centre for HMV, are treated at the two above-mentioned centres of HMV. In all centres for HMV the same guidelines are used.20,21 We included all consecutive records of patients diagnosed with ALS, including subtype progressive muscular atrophy (PMA), who were referred to a HMV centre between January 2016 and December 2018. No exclusion criteria were applied. The final EHR chart review was performed on 28 June 2021.
Evaluation of NIV practices
To evaluate NIV practices, we explored the criteria used for referral to a centre for HMV, initiation of NIV and not using NIV. In addition, we determined the time from disease onset to NIV and survival after initiation of NIV, and assessed which patient and clinical characteristics were associated with these two outcomes. We extracted the following data: age, gender, diagnosis, site of disease onset, NIV use, gastrostomy, Riluzole use, diagnostic delay, as well as signs and symptoms at first referral to a centre for HMV and at initiation of NIV, including nocturnal symptoms (frequent awakenings, night sweats, nightmares, difficulty getting to sleep), daytime symptoms (waking up tired, morning headaches, concentration problems, daytime sleepiness, fatigue), dyspnoea, orthopnea, PCO2, bicarbonate, oxygen saturation, and the FVC in upright and supine position (including FVC at diagnosis).
Analyses
Quantitative data are presented either as median and interquartile range (IQR), or mean and standard deviation (SD); categorical data as the number with percentage. Differences in patient and clinical characteristics between subgroups, based on diagnosis and NIV use, were evaluated using an independent t-test for continuous variables or the χ2 test for categorical variables. The time to NIV and survival after NIV were determined using Kaplan-Meier curves. In order to assess the effect of patient and clinical characteristics on time to NIV and survival after NIV, multivariate Cox proportional hazard regression analyses were applied to estimate hazard ratios (HRs). Adjusting covariates were gender, age, diagnosis, site of onset, Riluzole use and gastrostomy. Sub-groups were created for gender (male/female), site of onset (bulbar/spinal), age at diagnosis (<60, 60–70, >70 years), age at initiation (<60, 60–70, >70 years), FVC at diagnosis and at referral (<50, 50–80, >80%), diagnostic delay (<6, 6–12, 12–18, >18 months), time from disease onset to NIV (<12, 12–24, 24–36, >36 months), dyspnoea and/or orthopnea at initiation (yes/no), hypercapnia at initiation (yes/ no). The effect of FVC at diagnosis on time to NIV or survival after NIV will only be assessed in patients with spinal onset, as the performance of FVC in patients with bulbar dysfunction may be unreliable.22,23
P-values lower than 0.05 were considered to be significant. All data were analysed with SPSS version 24.
Results
We included data from 323 consecutive patients referred to a centre for HMV between 2016 and 2018, 284 (88%) of whom were diagnosed with ALS and 39 (12%) with PMA. In this period five patients with ALS received invasive ventilation. Invasive ventilation does not fall within the scope of the present study. No patients were excluded from the analyses. In our cohort, 197 (61%) patients started using NIV. All non-users of NIV and 88% of NIV users had died at time of the final EHR search. Patients with ALS who used NIV were younger and more of them had undergone gastrostomy compared to those who did not use NIV (Table 1). The quantitative outcomes measures that were recorded at referral and initiation of NIV are presented in Table 2. The study flow diagram is shown in Figure 1.
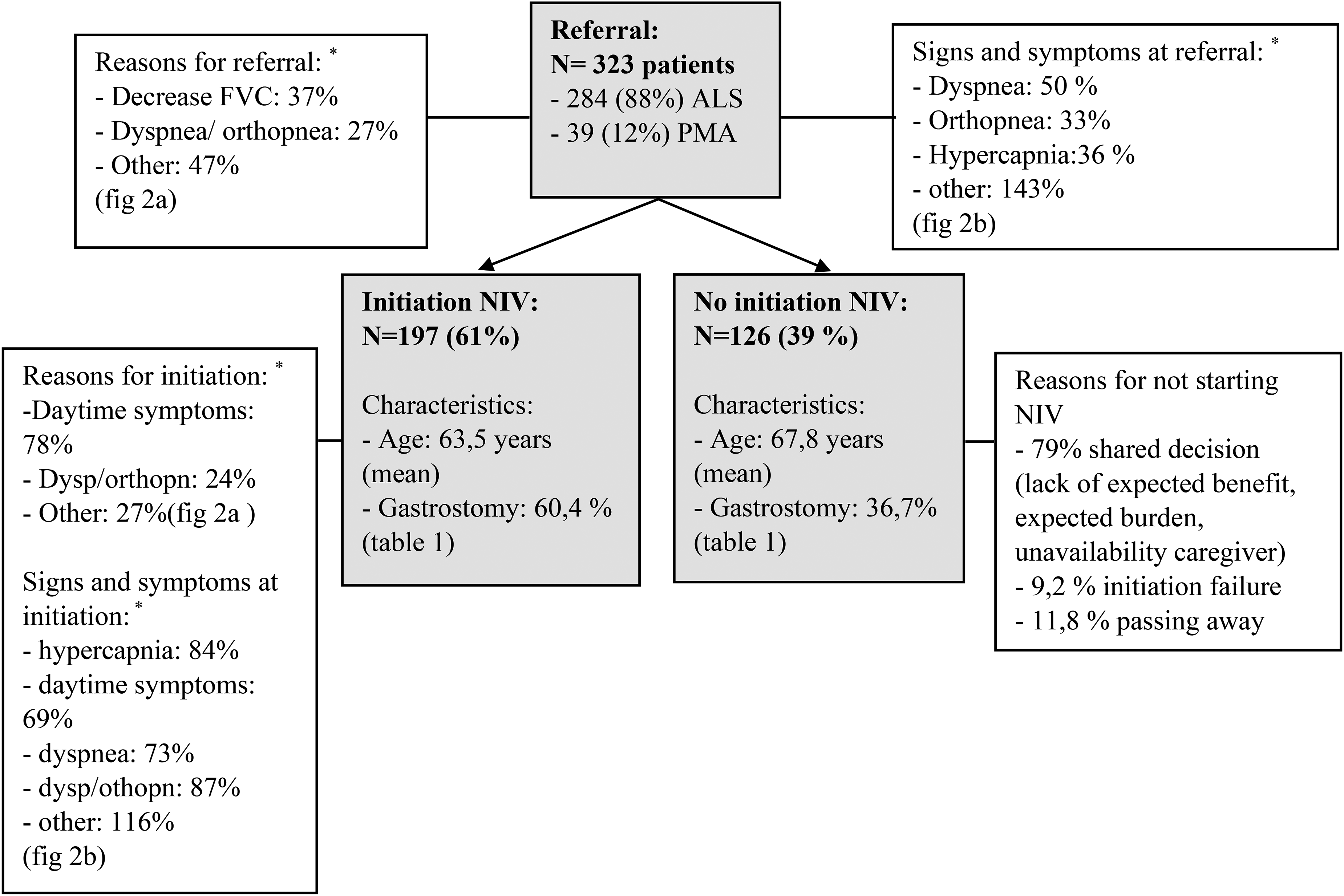
Flowdiagram for the retrospective study.
Characteristics of all patients with ALS.
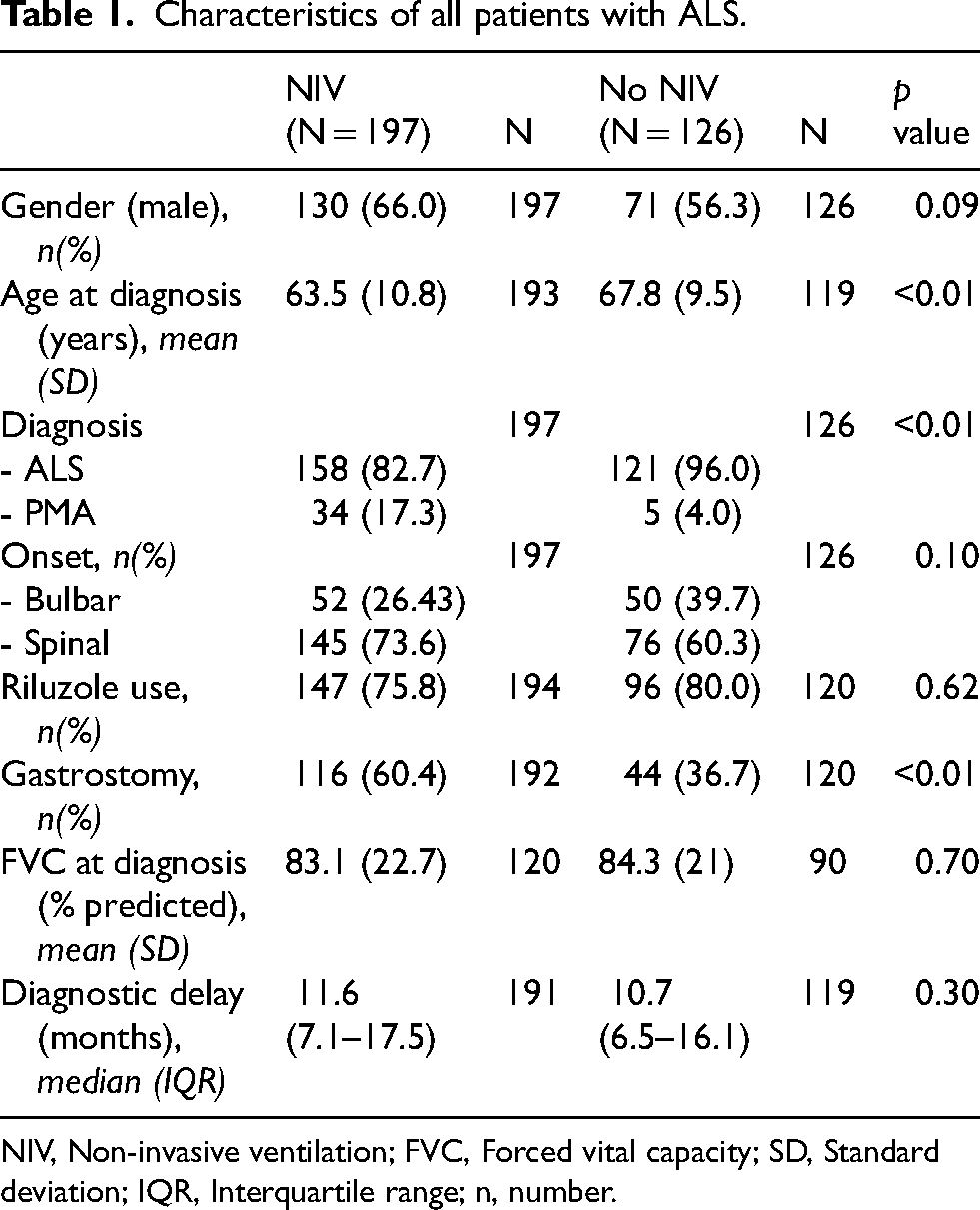
NIV, Non-invasive ventilation; FVC, Forced vital capacity; SD, Standard deviation; IQR, Interquartile range; n, number.
Respiratory measures at time of referral and initiation.
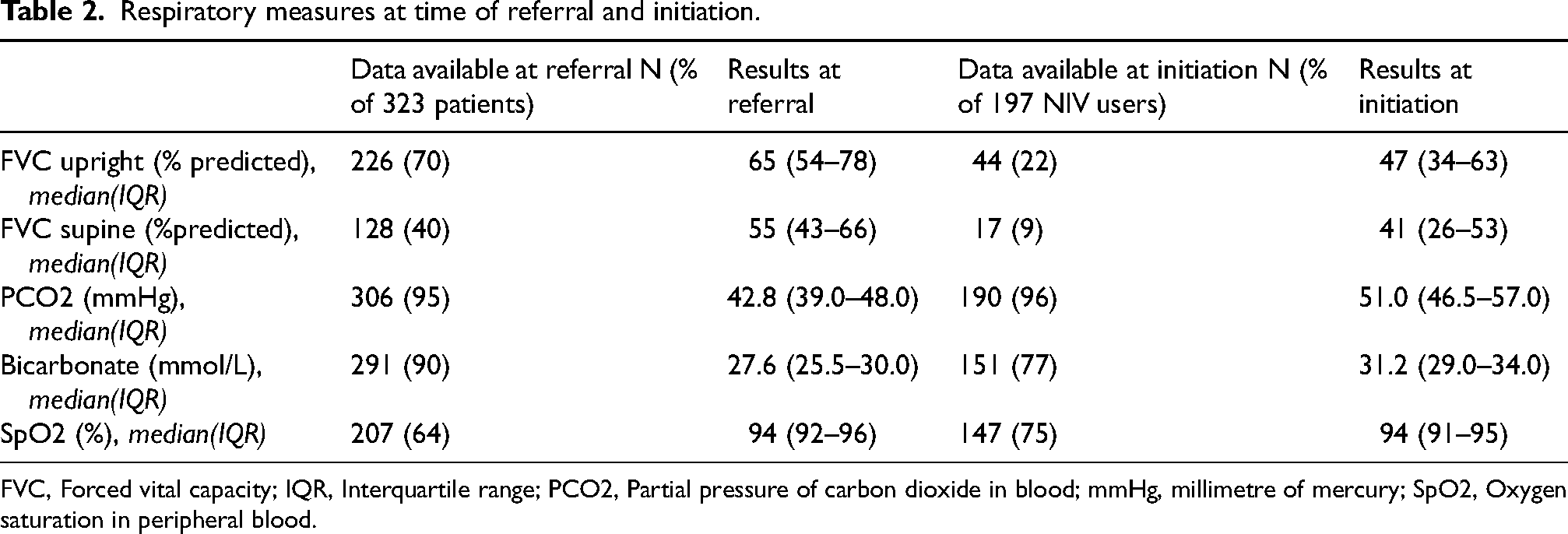
FVC, Forced vital capacity; IQR, Interquartile range; PCO2, Partial pressure of carbon dioxide in blood; mmHg, millimetre of mercury; SpO2, Oxygen saturation in peripheral blood.
NIV referral and initiation practices
The main reasons for referral to a centre for HMV, apart from an informative consultation were a decrease in FVC (37%) and the presence of dyspnoea/orthopnea (27%), (Figure 2(a)). In 24% of referrals, patients were deliberately referred early, i.e. without indication for NIV, to a centre for HMV in order to be informed about the benefits and drawbacks of NIV. The main reasons for starting NIV were daytime symptoms (78%) (Figure 2(a)). Furthermore, the criteria that were leading for initiating NIV were PCO2 > 45 mmHg (84%) or the presence of orthopnea and/or dyspnoea (87%) (Figure 2(b)). Dyspnoea and orthopnea were present in 50% and 33% of patients at referral, respectively, and in 73% and 57% at initiation of NIV, respectively (Figure 2(b)). At referral, 36% of patients had daytime or nocturnal hypercapnia and at initiation, 84%. In the group of patients with normal PCO2 at initiation of NIV, 28 of 29 (97%) reported dyspnoea and/or orthopnea.
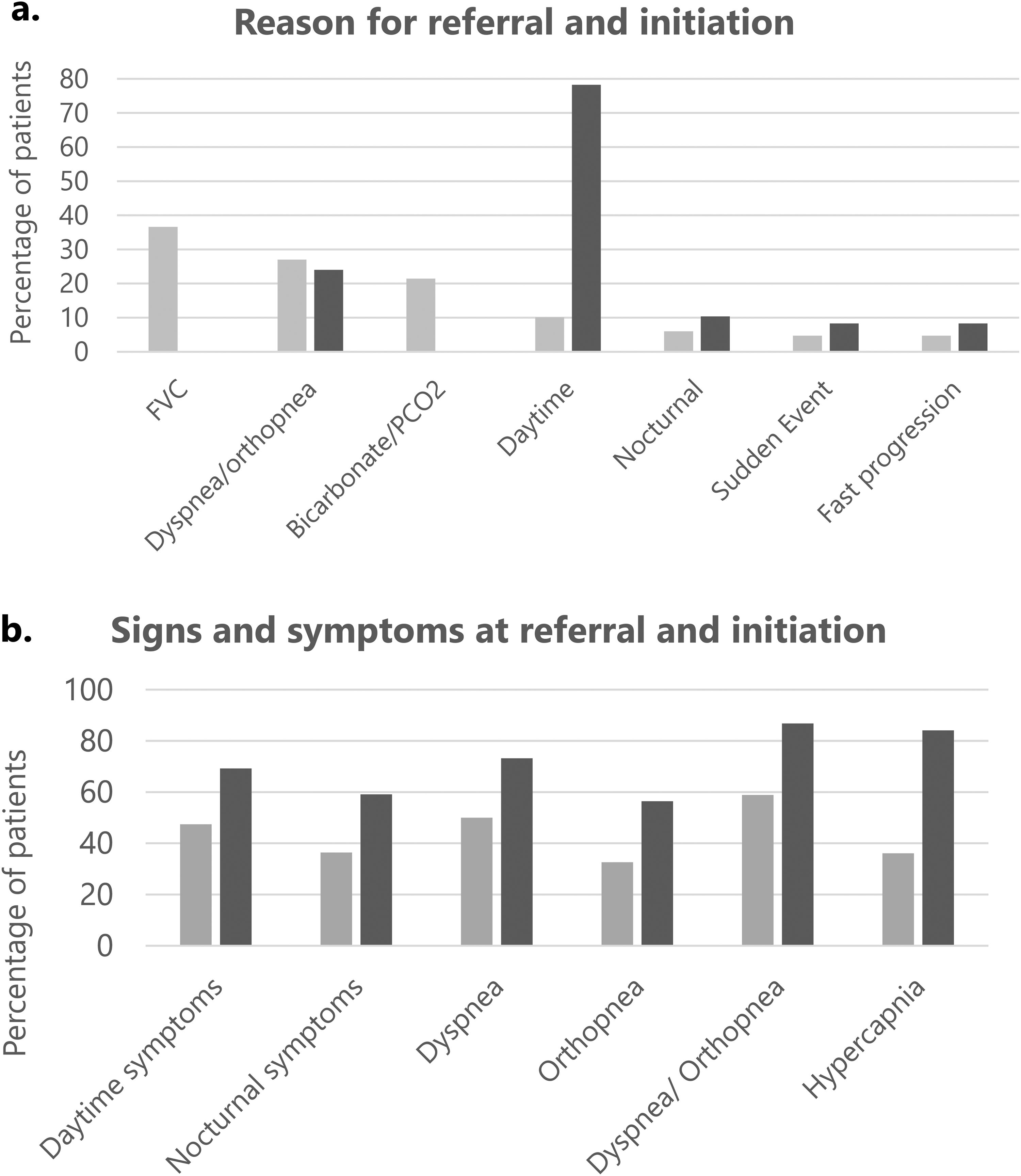
The presence of signs and symptoms and the criteria used for referral and initiation of NIV. (a) Reason for referral and initiation, (b) signs and symptoms at referral and initiation. NIV, non-invasive ventilation; FVC, forced vital capacity; PCO2, partial pressure of blood carbon dioxide; Sudden event, hypercapnia-induced coma, dyspnoea due to a fall, choking or pneumonia; Fast progression, fast overall disease progression not specifically related to respiratory decline; Nocturnal symptoms, frequent awakenings, night sweats, nightmares, difficulty getting to sleep; Daytime symptoms, waking up tired, morning headaches, concentration problems, daytime sleepiness, fatigue. As there may be multiple criteria for referral or initiation (max 3), the total of the percentages exceeds 100%.
Generally, patients in our cohort were reluctant to start NIV when they had no or only mild signs or symptoms of respiratory insufficiency. Of 126 patients who did not use NIV, 79% had reached this decision through a shared decision-making process with their caregivers and/or a physician. Decisions were mostly based on lack of expected benefit to quality of life, expected burden of NIV on caregivers/family, lack of expected benefit to survival due to bulbar impairment or rapid disease progression, and unavailability of a caregiver at home, requiring a move to a nursing home. Other reasons for non-use of NIV were an initiation failure (9.2%) and passing away prior to the decision to start NIV (11.8%).
Median time from disease onset to NIV was 22.5 months. In patients with a bulbar disease onset, time to NIV was significantly shorter compared to patients with a spinal onset (19 vs 23.7 months, HR = 1.77, p = 0.002), when corrected for gender, age, Rilozule and gastrostomy (Figure 3(a) and Table 3). Besides bulbar onset, a diagnostic delay <12 months (HR = 1.72, p < 0.001) and age at diagnosis >70 (HR = 1.81, p = 0.004) were independently associated with shorter time to NIV. Due to the high percentage of missing FVC data (39%) at diagnosis, the FVC was excluded from the analysis.
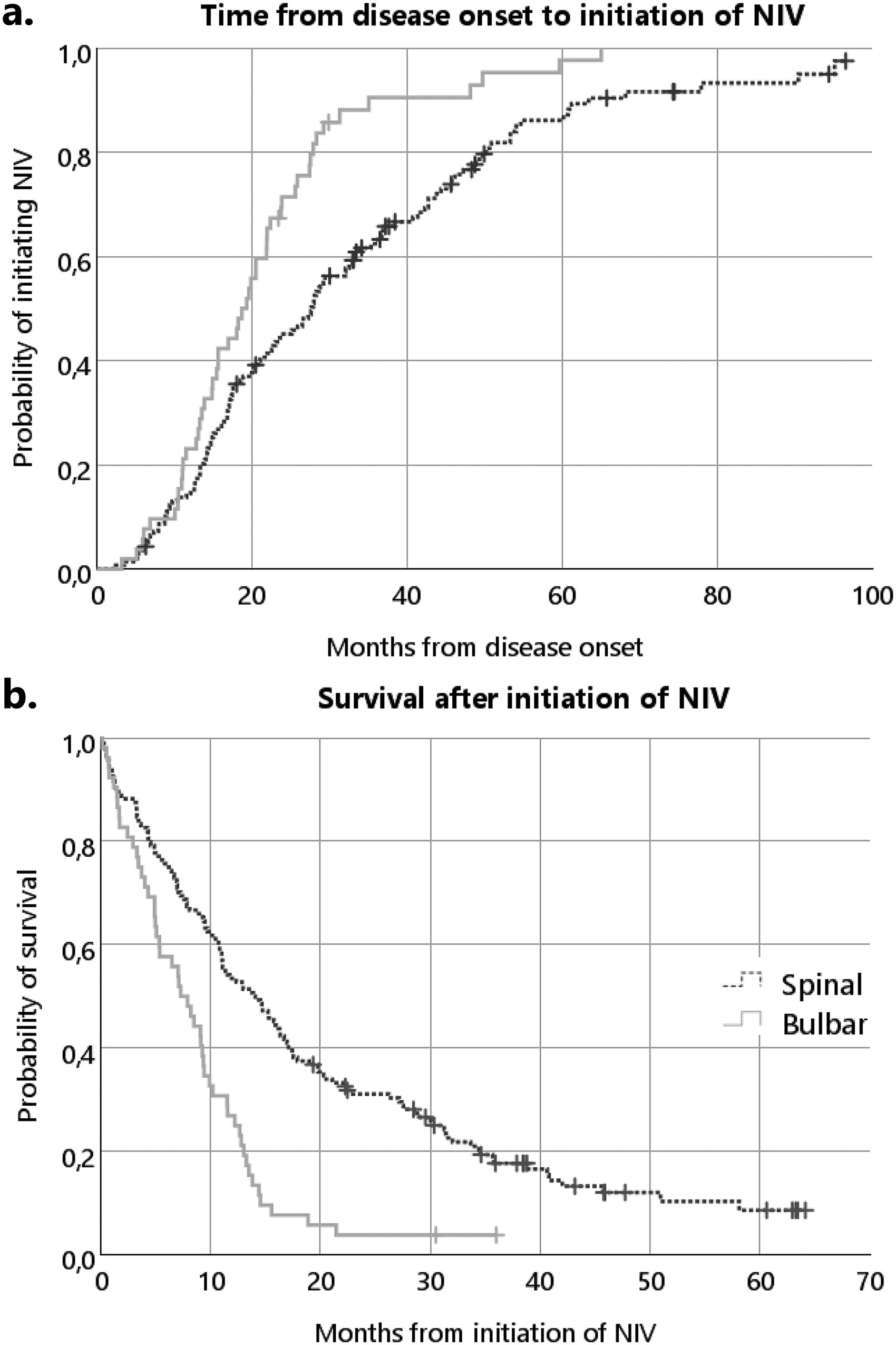
Kaplan Meier curves for time to NIV and survival after NIV. NIV, non-invasive ventilation. (a) Time from disease onset to NIV initiation, with separate lines for site of onset, (b) survival after NIV, with separate lines for site of onset.
Time variables for patients initiated on NIV.

ALS, amyotrophic lateral sclerosis; PMA, progressive muscular atrophy; NIV, non-invasive ventilation; IQR, interquartile range.
Median time from referral to initiation of NIV was 2.0 (IQR = 0.7–6.9) months. Of all 197 NIV users, 78 (40%) had received an indication for NIV during the first consultation at the centre for HMV. Factors associated with a shorter time between referral and initiation, were age > 70 (HR = 1.61, p = 0.032) and the presence of hypercapnia (HR = 2.61, p < 0.001). Due to the high percentage of missing upright and supine FVC data (30 and 60%, respectively) at referral, these variables were excluded from the analysis.
Survival after initiation of NIV
In this cohort of Dutch ALS patients referred to a centre for HMV, patients using NIV had a median survival from disease onset of 37.6 months (IQR = 23.4–56.2). The median survival after starting NIV was 11.1 months (IQR = 4.9–21.9). Patients with a bulbar disease onset had a shorter survival after NIV compared to those with spinal onset (7.6 vs 14.0 months, HR = 2.0, p < 0.001) (Figure 3(b) and Table 3). A median of 33% (IQR = 17.8–50.4%) of patients’ total disease duration was spent using NIV, which was not significantly affected by age, sex or site of disease onset. Besides a spinal disease onset, age at NIV initiation <70 (HR = 0.70, p = 0.018) and a time from disease onset to NIV >24 months (HR = 0.55, p = 0.002) were also independently associated with longer survival after NIV in patients with ALS (Figure 3(c) and Table 3).
Discussion
This population-based study in The Netherlands showed that in a large cohort of consecutive patients who were referred to a centralised centre for HMV, most of them started NIV due to presence of hypercapnia, and/or symptoms of orthopnea/dyspnoea, which was according to Dutch guidelines for initiating NIV. The median survival after the initiation of NIV was 11 months, which was one-third of patients’ total disease duration. Despite that survival after NIV was shorter in patients with a bulbar disease onset and older age, the proportion of the total disease duration spent using NIV was similar across all subgroups of patients. Lastly, most patients who did not use NIV, had reached a joint decision with their caregiver(s) and/or physician not to start NIV.
Of all NIV users, 85% were hypercapnic at time of NIV initiation; this is higher than the 58% and 76% reported in previous studies.16,24 Besides hypercapnia, symptoms of dyspnoea and orthopnea were present in most patients at initiation; this has also been reported in other studies.16,24 These studies did not report the survival after NIV, which could therefore not be compared to the present study. Similarly, there are several studies that have reported the survival after NIV or survival gain due to NIV in patients with ALS, but did not report the presence of signs or symptoms at initiation or details about the criteria used for initiating NIV.1,14,15,24–26 This shows, that there is a need for studies that include both details about the timing of NIV as well as survival, so that data between countries can be compared to help determine the optimal timing of NIV In addition more studies are needed to asses quality of live after starting NIV as this an important outcome for patients. The presence of respiratory symptoms (e.g. dyspnoea or orthopnea) may be a positive predictor for NIV adherence, since patients are likely to be more motivated to use NIV to relieve respiratory symptoms.10,27 Accordingly, in consultation reports, we found that most patients in our cohort were reluctant to allow initiation of NIV at the time of no or mild signs or symptoms of respiratory insufficiency. This observation is in line with the early termination of a randomised controlled trial on the effect of early initiation of NIV, due to insufficient enrolment.10,11 In contrast, two studies showed that early initiation of NIV is feasible and does not reduce immediate acceptance or short-term adherence and also the RCT of Sarasate et al. stated that all of the data analysed is in favour of early NIV.12,28,29 However, long-term data on this topic is still lacking. Older studies showed that early initiation of NIV can affect caregiver burden, since the complexity and intensity of care will increase at an earlier stage.30,31 Moreover, a recent article has highlighted that long-term use of NIV can be accompanied by psychosocial issues and have impact on social and professional life, which in turn may lead to overburden of treatment, and can cause adverse outcomes. 32 Future studies could determine the optimal timing of NIV from the perspective of patients with ALS and evaluate the burden of NIV on patients and their caregivers.
Most patients in the present study who did not use NIV, reached the decision not to start NIV jointly with their caregiver(s) and/or physician. This emphasises the importance of informing patients adequately about the advantages and drawbacks of NIV, as this allows them to make a well-informed decision on whether or not to start with NIV. In The Netherlands most knowledge on and experience with NIV is concentrated in the centralised centres for HMV. For this reason one out of four patients in our cohort had been referred early (i.e. prior to the indication for NIV) to one of the centres for HMV for an informative consultation. This subgroup of patients had significantly more time to the initiation of NIV compared to patients who were referred based on signs or symptoms, and had so more time to come to a well-informed decision on NIV.
Furthermore, we found that NIV was used less often by women, especially when we compared the male/female ratio of patients with ALS using NIV (1.8/1) with the male/female ratio of patients with ALS in the Netherlands (1.2–1.3/1). 33 Correspondingly, studies from Italy, United States, Scandinavia and Japan have reported lower NIV acceptance and adherence among women, that NIV was underused by women, and that fewer women than men chose to transition from NIV to invasive ventilation.13,34–36 As argued in the above-mentioned studies, women may worry more about the impact of mechanical ventilation on their family and experience more anxiety than men; they may receive a lower level of caregiving from their male spouse, or be more likely to live without a spouse.13,34,35 Gender differences in NIV use require further investigation.
Strengths & limitations
A strength of the present study is the large, real-world sample of consecutive patients with ALS in a clinical setting in the Netherlands in which initiation of NIV in patients with ALS is relatively standardised. We chose to collect the data from the two largest centres for HMV. Two-thirds of all Dutch ALS patients who are referred to a centre for HMV are treated at these centres. As all Dutch centres for HMV in the Netherlands work according to the Dutch guidelines we are confident that our sample is representative of the target population.
A limitation is the retrospective design, which led to missing data, such as FVC, progression rate based on ALSFRS-R, severity of bulbar dysfunction at initiation of NIV, hours of NIV use per day/night, use of airway clearance techniques, and quality of life. These are factors that can have a significant impact on the success of NIV. We could not determine whether these factors affected compliance with NIV or survival with NIV in our cohort. The large percentage of missing FVC data could not be explained and is one of the known drawbacks of every retrospective design. It is likely that we missed a subset of patients in our cohort, since not all patients with ALS are referred or want to be referred to a centre for HMV. In addition, provider bias regarding the risks and benefits of NIV may influence the decision making of health care professionals and, therefore, may influence clinical outcomes. For this reason we were not able to perform a survival analysis between patients on NIV and patients not on NIV. The missing data and risk for provider bias may limit the comparability of our findings to those of previous studies.
Reflection on current practices in The Netherlands
When evaluating the Dutch approach, some questions may rise. The timing of initiation of NIV in the Netherlands appears to be relatively late, which may have a negative impact on survival. The current approach mainly focuses on relieve of symptoms and not on results of FVC. In our experience, patients don’t want to start earlier in the course of their disease when they are not suffering from symptoms of respiratory insufficiency. There might be a general tendency for patients in the Netherlands to postpone the use of aids, possibly because it is a confrontation with the worsening of the disease as well as it is more difficult to maintain the NIV if it takes a long time while the disease deteriorates. Apparently, patients seem to feel that something should be done about their symptoms as soon as they experience them. As long as there are no symptoms, people will wait longer, possibly leading to a shorter survival time. Although there is no recent literature to support this, in our experience patients are reluctant to increase the burden of the caregiver when NIV is started, which also might play a role.
On the other hand, our results show that some patients pass away prior to the decision to start NIV. The recent RCT of Sarasata et al. 12 showed that all of their data analysed is in favour of early NIV and they demonstrated there was good tolerance and compliance with early NIV without quality of sleep impairment. Perhaps patients should be referred earlier to the centre of HMV in the Netherlands and it might be considered to start NIV earlier. The difficulty is that it is not obvious which is the better outcome: maintaining quality of life or prolonged survival. Prolonged survival is particularly desirable if accompanied by maintaining quality of life.
Another point of discussion is the lack of FVC. Although FVC is not a criterium of initiation of NIV in the Dutch guideline, stricter criteria could be considered to prevent patients from being referred too late. Failure to perform FVC can lead to missing patients developing respiratory insufficiency. However, it should be evaluated whether FVC is the right criterium as it is difficult to perform in bulbar patients and because of the limited value of only upright FVC. Supine FVC might be more valuable in this respect.
Given the limitations, several questions remain unanswered. To better understand the outcomes of Dutch ALS patients after starting NIV, a prospective cohort study was initiated in 2022. 37
Clinical implications
The obtained insights from this study on the reasons for (not) using NIV, timing of NIV, and survival after NIV in patients with ALS, may help healthcare professionals to better inform patients about the timing and expected effect of NIV from the moment they are referred to a centre for HMV in the Netherlands. In turn, this will support patients in making a well-informed decision whether or not to use NIV. Furthermore, the finding that two of the main reasons for patients not to start with NIV were related to the expected burden of NIV on the patient and informal caregivers, indicates that future research could investigate the effect of NIV on care burden and quality of life of patients with ALS and their family, including the impact of ‘early’ initiation of NIV. 32
Conclusion
This study showed that nearly all patients with ALS were either hypercapnic and/or symptomatic with dyspnoea or orthopnea at time that NIV was initiated at one of the centralised centres for HMV, which was according to the Dutch guidelines. Despite differences in survival with NIV between subgroups of patients, on average patients spent one third of their total disease duration using NIV, irrespective of age, gender or site of symptom onset. Moreover, we have found that patients may be reluctant to start NIV when they experience no or only mild respiratory symptoms, or when treatment burden of NIV is expected to be high. Therefore future studies could investigate patients’ perspectives on the optimal timing of NIV and treatment burden of NIV. Given the differences in international NIV guidelines and initiation practices, comparison of population-based NIV data between countries will provide valuable insights in the timing and effect of NIV in different settings and populations. Therefore, we would like to invite other countries to share their key outcomes in respiratory care of patients with ALS. Due to the limitations of this prospective study and to gain more insight into the population of patients with ALS in The Netherlands, a prospective cohort study was initiated in 2022. 37
Footnotes
Acknowledgements
This study was funded by the Netherlands ALS Foundation.
ORCID iDs
Ethical approval
Ethical approval was waived by the Ethics Committee of the UMC Utrecht, since the study was retrospective and all data was collected during routine care.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
Leonard H. van den Berg PhD, is an Editorial Board Member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
Data availability statement
The data supporting the findings of this study are not publicly available due to privacy or ethical restrictions.
