Abstract
Background:
Spinal Muscular Atrophy (SMA) is a progressive neuromuscular disorder that in its most severe form, causes profound swallowing deficits. There remains a paucity of research systematically elucidating the biomechanical and functional correlates. This void limits the ability to evaluate the effects of disease-modifying treatments on swallowing.
Objective:
Elucidate characteristics of swallowing biomechanics and function among untreated patients with SMA.
Methods:
Infants with SMA who had not received disease modifying therapy when they underwent a videofluoroscopic swallow study (VFSS) were retrospectively identified from 13 international children's hospitals. Infants were eligible if they exhibited symptoms by six months old, or in cases where they detected prior to symptom onset, if they had two copies of SMN2. Eligible infants underwent medical record review to gather oral intake status and need for suctioning for secretion management. Digital files of VFSS’ were extracted and analyzed using BabyVFSSImP™. Non-parametric t-tests were used to compare swallowing biomechanics based on viscosity and the presence of dysphagia symptoms.
Results:
62 infants were included in this investigation with average age at initial VFSS 4.4 ± 3.11 months. A high proportion of infants exhibited profound deficits in swallowing biomechanics that impeded extraction of the bolus from the nipple, clearance of the bolus from the pharynx, and prevention of the bolus from entering the airway. Deficits were significantly more prevalent in infants referred for VFSS due to dysphagia symptoms than asymptomatic infants referred as part of high-risk referral (t ≤ 2.5, p ≤ 0.03). A high proportion of infants relied on suctioning for secretion management (34%; N = 21) and alternative nutrition (39%; N = 24), with those relying on these supports exhibiting significantly worse swallowing biomechanics than those who did not (t ≤ 2.8, p ≤ 0.01).
Conclusions:
Profound deficits in swallowing biomechanics and function are common in infants with SMA. Though clinical symptoms may aid in identifying infants with the most profound swallowing deficits, their absence in cases of more mild deficits inhibits the ability to use these clinical markers as a valid metric of swallowing integrity.
Introduction
Spinal Muscular Atrophy Type 1 (SMA Type 1) is a progressive neuromuscular disorder that left untreated, results in loss of neuromuscular control of swallowing within the first 6 months of life.1–7 These swallowing deficits have historically been one of the leading causes of infant morbidity and mortality, as they led to aspiration of secretions and nutrients and caused respiratory failure.7–12 Despite the widespread appreciation of these swallowing deficits within the clinical community, there remains a paucity of research systematically elucidating the physiologic and functional correlates of this degradation.
What is known about swallowing degradation is based on a limited number of investigations composed of small sample sizes that rarely report on swallowing systematically using validated metrics.4,13 Swallowing requires coordinated contraction of 30 paired muscles regulated by five cranial nerves to enable bolus extraction from the nipple, closure of the larynx to prevent the bolus from entering the lungs, and rapid clearance of the bolus from the pharynx to enable progression of nutrients down the digestive tract. 14 Findings from available investigations suggest there are physiologic deficits in all of these critical abilities among infants with SMA type 1.9,11,14 The specifications of the underlying physiologic dysfunction that lead to these deficits is poorly understood.
A greater number of investigations have reported on the functional implications of these physiologic deficits, though with similar limitations in sample size. These investigations indicate a rapid degradation in swallowing, which results in complete reliance on non-oral alternative nutrition (ex. g-tube) and external suctioning for secretion management by 12 months of age.7–9,11,12 No previous investigations have elucidated the association between these functional and physiologic deficits.
This void in our understanding of swallowing among untreated infants with SMA 1 has important implications in the current era of SMA management where disease modifying therapies are routinely administered and control groups are no longer available to test treatment effects on swallowing.4,15 Such a reference point is also important for families, clinicians, and other stakeholders as they navigate aerodigestive risk and decision-making, particularly when considering additional disease modifying therapies. The aim of this investigation is to examine and describe the swallowing biomechanics and function among historically untreated patients with SMA to enable future comparison of outcomes following the provision of disease modifying therapies.
Materials and methods
This is a retrospective multicenter investigation including 13 international children's hospitals in the United States, New Zealand and Brazil. Select participants included in this investigation may have also been reported in previously published cross-sectional investigations by Hurst Davis et al. (2014) and Yuan et al. (2017).10,12
Participant identification
Infants ≤ 12 months old with genetically confirmed SMA who had not received disease modifying therapy at the time they underwent a clinically indicated videofluoroscopic swallow study (VFSS) were retrospectively identified. This included children receiving the aforementioned procedures between 2000–2024. Infants diagnosed with SMA prior to symptom onset (ex. newborn screen) were included if they had two copies of SMN2 on genetic testing, while those diagnosed with SMA after symptom onset were included if they were less than or equal to six months old at the time of symptom onset. These criteria were selected as the majority of infants went on to receive disease modifying therapy at some point after their VFSS exam, some prior to symptom onset, which prohibits historic classification approaches that utilize the highest motor milestone achieved. Further, patients evaluated prior to the development of disease modifying therapy often did not undergo refined genetic testing to determine SMN2 copy number. As our goal was to characterize the most severe phenotype, and historically 80% of untreated infants with two copies of SMN2 end up having SMA Type 1 phenotype, infants treated prior to symptom onset were included if they had two copies of SMN2 (6). Infants were excluded from the investigation if they did not have a digital copy of their videofluoroscopic swallow study available for prospective analysis, or if their videofluoroscopic swallow study was completed at <15 pulses per second.
Data collection
Eligible infants underwent medical record review by clinicians with expertise in neuromuscular disorders to gather information pertaining to demographics, SMA diagnostics and medical management. Patient and procedure specifications for each VFSS exam were collected by Speech Pathologists with bulbar expertise. This included age, reason for VFSS referral, and correlates of swallowing function including whether the child required external supports such as suctioning for secretion management (Y/N) and their oral intake status as classified by the Children's Eating and Drinking Activity Scale (CEDAS). 16 CEDAS is a 6-point scale categorizing oral intake based on the presence and extent of tube use and the need for compensatory interventions like thickening. 16 Oral intake status and secretion management scores were then categorized as to whether or not the scores were indicative of profound impairment, which for this investigation was characterized by CEDAS scores of 1–3 (reliance on alternative nutrition), and reliance on external supports for secretion management. Children's Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP INTEND) scores taken closest to the VFSS were recorded. CHOP INTEND is a scale rating 16 areas of motor integrity on a 4-point scale for a best possible score of 64/64. 17 Notably, CHOP INTEND scores were not available for all infants as the study included infants who underwent swallow studies prior to the time that CHOP INTEND had been developed or validated in the examiner's language.
Digital files of VFSS exams from eligible infants were extracted from each site, de-identified, and shared with the core lab (University of Minnesota) for prospective analysis of swallowing biomechanics. VFSS exams were conducted using barium contrast, with infants in varying positions ranging from side lying to upright, and in varying seating modalities ranging from the fluoroscopy bed to a tumble form chair. Although all exams were conducted by fluoroscopically visualizing a sample of swallows, the timing of fluoroscopic evaluation and the number of swallows varied across patients and sites. This study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the University of Minnesota Institutional Review Board. The need for obtaining informed consent was waived because of the retrospective study design.
Data analysis
De-identified exams were scored independently by two Speech-Language Pathologists blind to the infant's background information using BabyVFSSImP© on a high-definition liquid retina XDR display with 3024 x1964 native resolution at 254 pixels per inch. Rating clinicians had between 12–17 years of experience in VFSS analysis and had achieved 80% reliability criterion in the analysis of VFSS exams using BabyVFSSImP© prior to initiating analysis.
BabyVFSSImP© is a VFSS analysis metric that ranks performance within 21 physiologic components of oropharyngeal swallowing biomechanics.18,19 Each physiologic component is ranked on an ordinal scale ranging from zero, representing the highest level of swallow function, and increasing in number to represent the lowest level of swallow function. For the purposes of this investigation, physiologic component scores were assigned using the overall impression methodology, which assigns a score for each physiologic component based on the worst performance across all swallows of a given consistency. Composite overall impression scores, representing the worst performance across all presented consistencies and exams, as well as domain scores, summarizing performance into five physiologic domains, were then calculated. Supplemental Table One provides a listing of all BabyVFSSImP© components, score variants, domains, and scores demarcating profound impairment for analyses. Inter-rater reliability was checked on 20% of exams, with discrepancies between raters resolved by consensus.
Statistical analysis
Inter-rater reliability of BabyVFSSImP© ratings was calculated using weighted Cohen's kappa coefficients. Characteristics of swallowing biomechanics and function were summarized using descriptive statistics including median (inter-quartile range) for domain scores and age, and proportions for individual components. Non-parametric t-tests were completed to test differences in profound deficits in swallowing biomechanics of summed domain scores between viscosities and reasons for VFSS referral (studentized Wilcoxon for unpaired and Fisher sign tests for paired data). Fisher's exact test was used to test differences in profound impairments within specific component scores. Effect sizes of summed domain scores were calculated using cliff's delta (δ) and specific component scores with odds ratios (OR). All analyses were performed in R, with use of the nptest package 20 for all non-parametric t-test analyses, and an alpha level of 0.05 for all tests. Visualization of changes in swallowing biomechanics by infant age were depicted with scatter plots containing the polynomial regression LOESS smoothing curve in ggplot2. Given the rare nature of the disorder and required retrospective methodology, sample size was determined based on feasibility, using the largest number of exams accessible, rather than to achieve a specific power. A full export of the analysis can be found in the Supplemental Data File.
Results
Sample demographics and characteristics
Sixty-two infants were included in this investigation. The majority of infants were diagnosed following symptom onset (92%, N = 57) and had two copies of SMN 2 (66%, N = 41). Comorbidities were rare, though included a history of prematurity (13%, N = 8), tracheomalacia (2%, N = 1), and tracheoesophageal fistula (2%, N = 1). All infants born prematurely were born ≥30 weeks gestation, with the average age at delivery being 35 weeks gestation (range 30–36 weeks). CHOP INTEND scores were available for 47% (N = 29) of infants, with a median score of 30 (21.5) out of 64. At the time of VFSS 21% (N = 12) used non-invasive ventilation (NIV). Table 1 provides a full report of sample demographics and characteristics.
Sample demographics and characteristics (N = 62).

Values reflect median (interquartile range) and N (proportion).
Proportions reflect the number of participants out of the sample with data available for the outcome of interest. They may not add to 100% due to those reported as unknown and rounding errors.
Outcomes reflect worst performance on any VFSS exam.
*Symptomatic in reference to clinical diagnosis method differs from those referenced throughout the manuscript as ‘symptomatic’ for swallowing impairments specifically.
Infants underwent between one and three swallow studies without disease modifying treatment, though most (81%, N = 50) had only one. Median age at the time of the initial swallow study was 3.37 (4.86) months. Almost all initial exams were completed due to clinical signs or symptoms of swallowing impairment (84%, N = 52) as characterized by caregiver concerns about their child's feeding performance (21%, N = 13) or physician-identified signs of impairment including respiratory morbidities or nutritional compromise that may be attributable to swallowing impairment (58%, N = 36). Although the majority of exams were performed due to dysphagia signs or symptoms (symptomatic), a subset of exams (16%, N = 10) was performed as part of a routine high risk VFSS referral upon diagnosis of SMA (asymptomatic). Half of these asymptomatic referrals were participants identified on newborn screen. Median age at time of initial exam was significantly lower among infants referred as part of high-risk referral (1.15 (2.10)) when compared to infants referred due to symptoms (4.35 (4.83)) (t = −2.97, p = 0.01, δ = 0.56).
Swallow biomechanics
Raters achieved scores that corresponded to a Landis-Koch category of moderate or greater (kappa > 0.4) agreement in their reliability of analyzing all BabyVFSSImP© components. All but 3 infants (95%) initiated a swallow response to the presentation of barium during the exam, however the integrity of the corresponding biomechanical processes was often so profoundly impaired that bolus did not adequately clear from the pharynx into the esophagus and likely would not be perceived as a swallow clinically. These profound impairments were observed across both thin and nectar liquids, where there were no significant differences in domain scores (t ≤ 4.11, p ≥ 0.08) (Table 2). Differences in swallowing were, however, observed across infants based on the reason for VFSS referral (Table 3). Infants referred with dysphagia signs or symptoms exhibited significantly worse swallowing performance (higher scores) than those who had no dysphagia symptoms (t ≤ 2.49, p ≤ 0.03) although infants in both groups exhibited profound deficits in swallowing biomechanics. Specifications of these deficits are outlined below. Supplemental Table 1 provides a full listing of BabyVFSSImP© component scores.
Swallowing biomechanics BabyVFSSImP© domain scores by consistency.
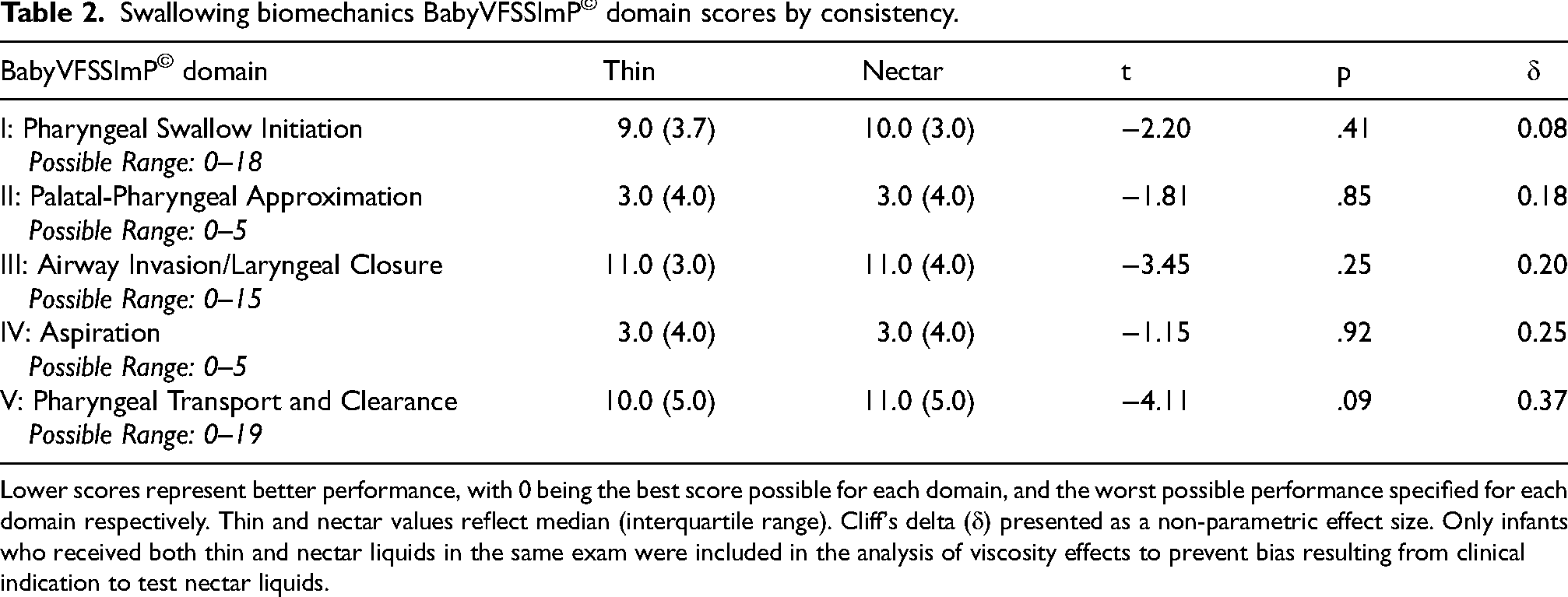
Lower scores represent better performance, with 0 being the best score possible for each domain, and the worst possible performance specified for each domain respectively. Thin and nectar values reflect median (interquartile range). Cliff's delta (δ) presented as a non-parametric effect size. Only infants who received both thin and nectar liquids in the same exam were included in the analysis of viscosity effects to prevent bias resulting from clinical indication to test nectar liquids.
Swallowing biomechanics BabyVFSSImP© domain scores by reason for referral.
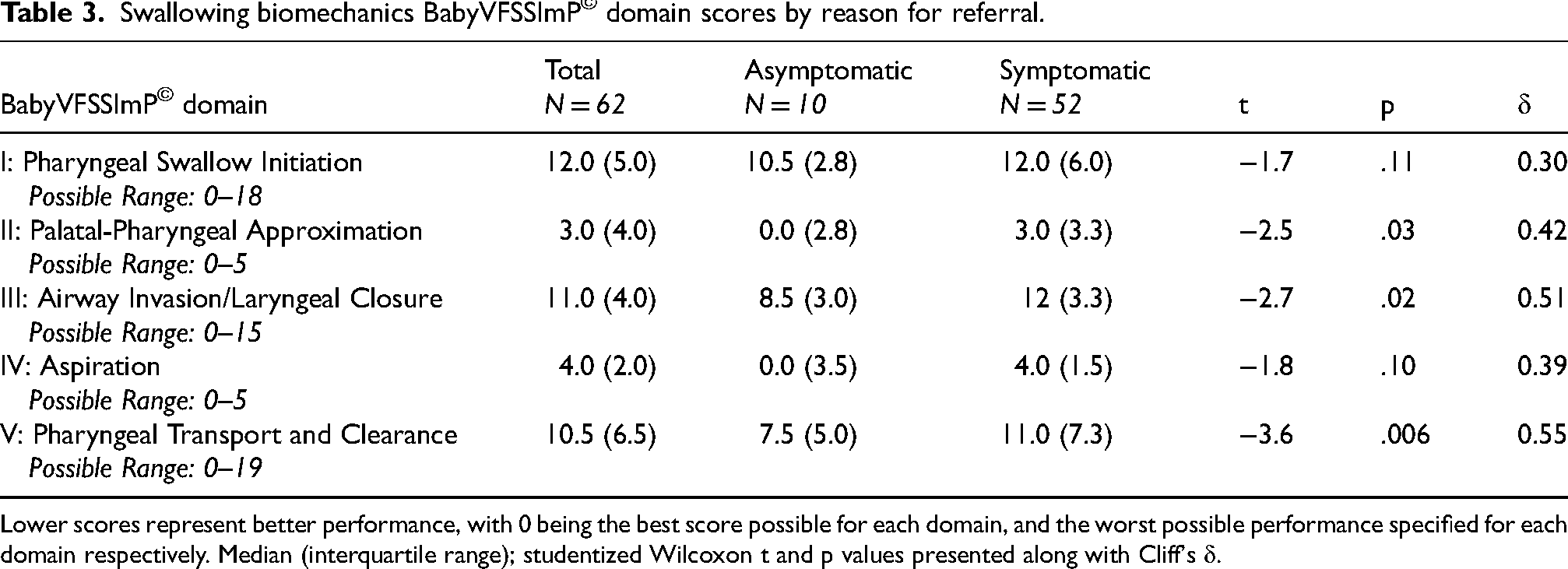
Lower scores represent better performance, with 0 being the best score possible for each domain, and the worst possible performance specified for each domain respectively. Median (interquartile range); studentized Wilcoxon t and p values presented along with Cliff's δ.
Bolus Extraction: Infants exhibited profound impairments in sucking biomechanics as characterized by 25% (N = 15) unable to extract a bolus from the nipple due to the absent initiation of nutritive sucking, or physiologic deficits impeding bolus removal despite their attempts.
Bolus Clearance: High rates of profound impairment were observed in components pertaining to the clearance of the bolus from the pharynx. This included deficits in the integrity of soft palate elevation (52%, N = 32), tongue base retraction (50%, N = 31), pharyngeal stripping wave (23%, N = 14), and pharyngoesophageal segment/upper esophageal sphincter opening (47%, N = 29). The combined effects of these impairments in bolus clearance resulted in a majority to no clearance of the bolus from the pharynx during the swallow in 39% (N = 24) of infants. Infants who were referred with dysphagia symptoms were significantly more likely to have profound impairments in soft palate elevation (Symptomatic 58%, Asymptomatic 20%, p = 0.04; OR = 0.19) and pharyngoesophageal segment opening (Symptomatic 54%, Asymptomatic 10%, p = 0.01, OR = 0.10).
Airway Protection: The aforementioned deficits in bolus clearance, paired with high rates of incomplete early (98%, N = 61) and late (94%, N = 58) laryngeal closure resulted in high rates of bolus airway entry. This was characterized by penetration in 97% (N = 60) of infants. Aspiration was also extremely common, occurring in 76% (N = 47) of infants.
Swallowing function
Reliance on suctioning for secretion management (34%, N = 21) and alternative, non-oral nutrition (ex. g-tube) (31%, N = 19) was highly prevalent within the sample. Those relying on either of these supports had significantly worse integrity in the swallowing components that facilitate bolus clearance from the pharynx (Suctioning Reliance (t = −2.79, p = 0.01, δ = 0.42), Alternative Nutrition (t = 3.44, p = 0.003, δ = 0.48)). Table 4 provides a full listing of parameters of swallow function.
Swallowing function.

Timing of degradation and SMN2 copy number influences
Profound impairments in swallowing biomechanics were rarely seen within the first month of life for all biomechanical measures except for penetration and aspiration, which were frequent (penetration 86%, aspiration 43%). The prevalence of profound impairments increased thereafter, with degradation from a non-profound impairment score to profound impairment score over a median of 1.8 (3.21) months. Examination of how swallowing biomechanics differed in the first three months of life by SMN 2 copy number revealed remarkably worse swallowing in the infant with one copy SMN 2. This was characterized by sucking without the ability to extract the bolus, absent opening of the pharyngoesophageal segment/upper esophageal sphincter, absent clearance of the bolus from the pharynx, and more than trace aspiration at two and a half months old, whereas none of these profound impairments were observed in the infant with three copies of SMN2 when evaluated at the same age.
Although profound impairments were common among older infants, this severity of impairment did not always manifest. Among the six infants who were evaluated between ten to twelve months of age, two exhibited more mild impairments in pharyngoesophageal segment opening, tongue base retraction, and pharyngeal stripping wave. One of these infants had three copies SMN2, while the other had two. Table 5 provides a full record of the cases where profound deficits were observed by age of VFSS, with Figure 1 illustrating the change in the proportion of infants with profound impairments in pharyngeal residue by age.
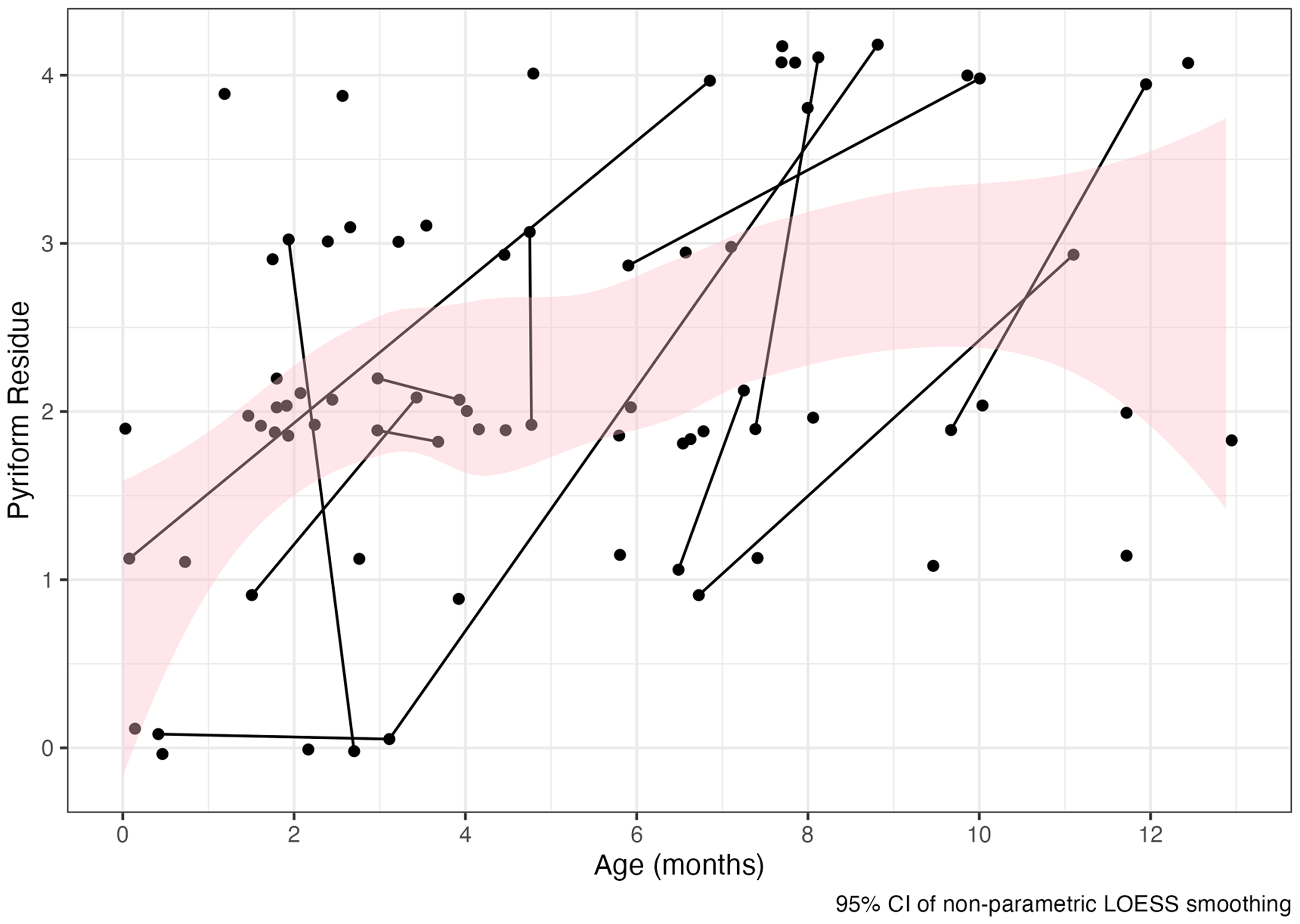
Change in pyriform residue based on age. Lower scores representing higher integrity biomechanics. Scores of 3 (majority of bolus remaining) or 4 (no clearance of the bolus) indicate profound impairment.
Infants exhibiting profound impairments in swallowing biomechanics and function by age (months).
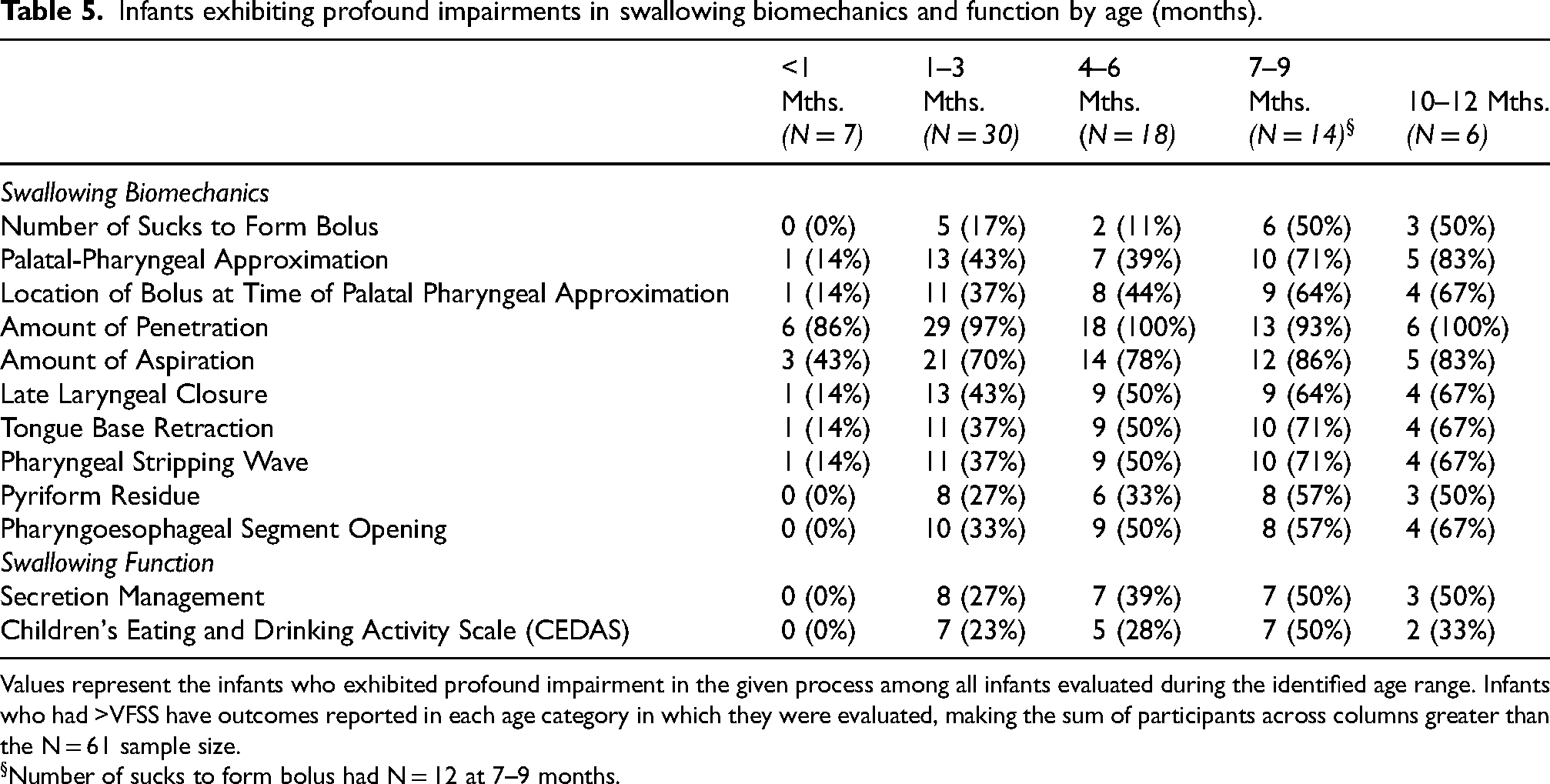
Values represent the infants who exhibited profound impairment in the given process among all infants evaluated during the identified age range. Infants who had >VFSS have outcomes reported in each age category in which they were evaluated, making the sum of participants across columns greater than the N = 61 sample size.
Number of sucks to form bolus had N = 12 at 7–9 months.
Discussion
This investigation is the first to systematically characterize swallowing biomechanics and function in a large sample of untreated infants with SMA. This fills a critical void in the current era where routine administration of disease-modifying treatments prohibits the prospective collection of these natural history data points and, therefore, impedes the ability to test disease modifying therapy effects. It also provides a reference point for families and providers who are grappling with symptomatic presentations, delayed access to treatments, or questions about maintaining treatments. Results from this investigation highlight three key findings. First, without disease modifying treatments infants with SMA frequently suffer from profound impairments in swallowing biomechanics and function. Second, although infants with profound swallowing deficits often show some clinical signs or symptoms of these impairments, in some cases these indicators are absent. Third, degradation resulting in profound impairments occurs at variable rates within the first year of life.
We demonstrated untreated infants with SMA regularly suffer from profound deficits in oropharyngeal swallowing biomechanics that impede the extraction of milk from the nipple, its clearance from the pharynx, and their ability to protect the airway. These general themes of impairment are consistent with reports by Durkin et al. (2008), Choi et al. (2020), and Zang et al. (2023) who examined swallowing biomechanics in symptomatic patients with SMA 1.9,11,21 Although comparison of the exact proportions of infants presenting with these deficits across previous investigations is limited by the small sample sizes (N ≤ 10) and frequent use of non-standardized and unvalidated metrics, past investigators did report high proportions of patients exhibiting impairments in bolus formation,11,21 pharyngeal residue,11,21 and aspiration.9,11,21 Not only do our results support these general themes in a large sample of patients using standard and reliable metrics, but they identify the underlying physiologic deficits and corresponding severity levels responsible. These included profound deficits in laryngeal closure, epiglottic inversion, tongue base retraction, pharyngeal stripping wave, and pharyngoesophageal segment/upper esophageal sphincter opening that resulted in minimal clearance of the bolus from the pharynx and aspiration. These physiologic deficits were found to be strongly associated with metrics of bulbar function, where nearly a third of the infants relied on external supports such as suctioning for secretion management as well as alternative nutrition to meet nutritional needs. Both of these outcomes were significantly associated with higher (worse) domain scores in Domain V: Bolus Transport and Clearance.
Another notable finding pertains to the observation that many infants who had no signs or symptoms of swallowing impairment exhibited impairments on VFSS examination. In some cases these deficits were profound; contributing to aspiration in 40% of asymptomatic infants and minimal clearance of the bolus from the pharynx in one. More common score variants, however, were those that many clinicians consider ‘mild’ deficits. For example, 70% of asymptomatic infants exhibited a score of two (narrow) for tongue base retraction, a component with score variants spanning 0 for the highest physiologic integrity and 4 for the worst. Similarly, high rates of ‘mild’ score variants were observed for epiglottic inversion, pharyngoesophageal segment/upper esophageal sphincter opening, pharyngeal residue, and penetration, indicating early changes occur, leading to more profound dysfunction as time goes on. Unfortunately the lack of normative VFSS data on healthy non dysphagic infants makes delineation of mild impairment from normal variant significantly more difficult, the uncertainty regarding their interpretation certainly warrants close clinical monitoring, as such observations may be the first indicators that the start of bulbar degradation has occurred. Cumulatively, these results showing ‘silent’ swallowing degeneration underscores the importance of cautious interpretation of non-instrumental clinical swallow assessment results among infants with SMA. We recommend that greater emphasis is placed on instrumental assessment findings. This includes the use of routine instrumental swallowing assessments, that in the clinical arena can be used to help guide aerodigestive management, and in the research arena can be used to demonstrate disease-modifying therapy effects. Determination of the exact timing and frequency that instrumental assessments should be conducted is beyond the scope of this investigation, and requires careful consideration of these results in the context of other variables such as payer requirements, access to specialized equipment, disease modifying therapy effects, and clinical status. A recommendation that is within the scope of this investigation, and carries through across all the aforementioned variables, is that if having a clear understanding of swallowing integrity is critical to a patient's clinical course or research investigation, an instrumental assessment should be employed.
Previous investigations have reported specifications pertaining to the timing of ‘silent’ degradation in swallowing biomechanics that takes infants from full oral intake without symptoms of impairment to complete dependence on alternative nutrition and hourly suctioning for secretion management over a period of one to four months.7–9,11,12 Our findings underscore the variability in timing of degradation. Profound impairments were rarely seen during the first month of life, though they were commonly observed in the months thereafter. The ability to delineate the exact timing of this degradation is limited by the fact that the majority of infants in the current investigation only underwent an isolated VFSS at which time they had already lost the majority of swallowing biomechanics and function. However, examination of the changes in swallowing among infants who underwent multiple VFSS exams provides greater insight into the onset and rate of degeneration of brainstem motor neurons. This indicates infants encountered degradation from a non-profound to a profound impairment score, on average, over a two-month time period, and in some cases as quickly as two days. For many this change initiated early, as evident by the high proportion of infants who exhibited profound impairments within the first three months of life, but for others it occurred much later, with some infants not exhibiting profound impairments at ten to twelve months. Interestingly, those infants who did not exhibit profound impairments between ten to twelve months had SMN 2 copy numbers ranging from two to three. These results hold clinical significance in the development of protocols guiding the timing of standardized instrumental swallow assessments to evaluate the integrity of swallowing biomechanics among high risk SMA patients to allow earlier treatment, and improved health outcomes in cases where impairment is seen.
Limitations
The development of pharmaceuticals aimed at halting neuromuscular degeneration and enabling infant survival has transformed SMA management. Paired with the adoption of routine newborn screening for SMA in many countries throughout the world, many infants with SMA 1 now get disease-modifying treatment prior to symptom onset. Despite the clear benefits of this paradigm shift, this changing landscape has posed a challenge for testing the effect of disease-modifying treatments, as prospective data collection using an untreated control group is not ethically indicated. As such, methods of testing disease-modifying therapy effects are limited to retrospective methodologies such as those used in this investigation, which come with inherent limitations.
The greatest of these limitations is that the data utilized as part of this investigation was based on VFSS exams that were done as part of routine, clinical care and did not follow any standardized assessment protocol governing which infants underwent exams, how the exams were administered, and the timing that they were completed. This includes the use of select VFSS exams that were completed at 15 pulses per second, whereas 30 pulses per second is optimal to maximize validity of exam results. Such limitations must be considered in the interpretation of these outcomes. To assist in interpretation, we have provided detailed results in associated tables and Supplemental Materials pertaining to specific outcomes for patients who underwent exams due to dysphagia symptoms separate from those who underwent exams as part of routine high risk referral. Caution should be taken in future investigations to compare proportions from the sample most closely reflecting their VFSS methodology.
A further limitation in the interpretation of results from the current investigation is the lack of clear normative cut-points to delineate normal biomechanics from impaired. To circumvent this knowledge gap, we have summarized results pertaining to the proportion of patients exhibiting clear-cut profound impairments in the text, and we have provided the proportion of infants scoring within each score variant in Supplemental Material. This will enable future investigators to compare how these ‘mild’ score deviations change based on the timing, and type of disease modifying treatment provided and as more normative data emerges.
Conclusions
Swallowing deficits pose profound health risks for infants with SMA Type 1. Unfortunately, the specifications of these deficits have not been systematically evaluated previously, making it difficult to demonstrate the effect and value of disease-modifying therapies on swallowing. Results of this investigation document the nature and timing of swallowing degradation in a large, international database of patients with SMA. We found a high proportion of infants with SMA experience profound deficits in swallowing biomechanics and function within the first year of life without the receipt of disease-modifying treatments. Though clinical symptoms may aid in identifying many infants with the most profound swallowing deficits, their absence in cases of mild deficits limits use of these clinical markers as a valid metric of swallowing integrity. Such information is essential to inform families of aerodigestive risks allowing for simultaneous, proactive clinical support while they pursue SMN directed treatments. Future studies utilizing supplemental metrics to evaluate swallowing biomechanics are warranted to further characterize deficits in untreated patients and examine the effects of disease-modifying therapy.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602241308762 - Supplemental material for Characterization of swallowing biomechanics and function in untreated infants with spinal muscular atrophy: A natural history dataset
Supplemental material, sj-docx-1-jnd-10.1177_22143602241308762 for Characterization of swallowing biomechanics and function in untreated infants with spinal muscular atrophy: A natural history dataset by Katlyn Elizabeth McGrattan, Robert J Graham, Alicia Hofelich Mohr, Anna Miles, Jacqui Allen, Juliet Ochura, Kayla Hernandez, Katie Walsh, Vamshi Rao, Melanie Stevens, Lindsay Alfano, Mackenzi Coker, Carmen Leon-Astudillo, Leann Schow Smith, John Brandsema, Hiba Farah, Julia Welc, Deborah Salle Levy, Miranda Clements, Whitney J Tang, Tina Duong, Carolina Tesi Rocha, Graham Schenck, Heather McGhee, Keeley Nichols, Ashley Brown, Allison Brown, Diana Castro and Basil T Darras in Journal of Neuromuscular Diseases
Supplemental Material
sj-html-2-jnd-10.1177_22143602241308762 - Supplemental material for Characterization of swallowing biomechanics and function in untreated infants with spinal muscular atrophy: A natural history dataset
Supplemental material, sj-html-2-jnd-10.1177_22143602241308762 for Characterization of swallowing biomechanics and function in untreated infants with spinal muscular atrophy: A natural history dataset by Katlyn Elizabeth McGrattan, Robert J Graham, Alicia Hofelich Mohr, Anna Miles, Jacqui Allen, Juliet Ochura, Kayla Hernandez, Katie Walsh, Vamshi Rao, Melanie Stevens, Lindsay Alfano, Mackenzi Coker, Carmen Leon-Astudillo, Leann Schow Smith, John Brandsema, Hiba Farah, Julia Welc, Deborah Salle Levy, Miranda Clements, Whitney J Tang, Tina Duong, Carolina Tesi Rocha, Graham Schenck, Heather McGhee, Keeley Nichols, Ashley Brown, Allison Brown, Diana Castro and Basil T Darras in Journal of Neuromuscular Diseases
Supplemental Material
sj-docx-3-jnd-10.1177_22143602241308762 - Supplemental material for Characterization of swallowing biomechanics and function in untreated infants with spinal muscular atrophy: A natural history dataset
Supplemental material, sj-docx-3-jnd-10.1177_22143602241308762 for Characterization of swallowing biomechanics and function in untreated infants with spinal muscular atrophy: A natural history dataset by Katlyn Elizabeth McGrattan, Robert J Graham, Alicia Hofelich Mohr, Anna Miles, Jacqui Allen, Juliet Ochura, Kayla Hernandez, Katie Walsh, Vamshi Rao, Melanie Stevens, Lindsay Alfano, Mackenzi Coker, Carmen Leon-Astudillo, Leann Schow Smith, John Brandsema, Hiba Farah, Julia Welc, Deborah Salle Levy, Miranda Clements, Whitney J Tang, Tina Duong, Carolina Tesi Rocha, Graham Schenck, Heather McGhee, Keeley Nichols, Ashley Brown, Allison Brown, Diana Castro and Basil T Darras in Journal of Neuromuscular Diseases
Footnotes
Acknowledgements
The authors would like to express their deepest appreciation to Biogen for their continuous dedication to improving bulbar function in patients with SMA and their support of this work. This work reflects many international clinicians who dedicated their time to making this work possible. We are eternally grateful for your collaboration, and your pure passion towards improving health outcomes in your patients with particular gratitude towards Louise Bax, Hana Church, and Sally Dunaway Young.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was supported by grant funding provided by Biogen (US-SPN-12066).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All listed authors received salary support from their listed institutions and were funded by the aforementioned Biogen grant to execute this work. Additional conflicts of interest include grant support from Biogen (BTD), Roche (KM, BTD, CLA), Biohaven (CLA), Novartis (BTD), Scholar Rock (BTD), Fibrogen (BTD), NIH (BTD), SMA Foundation (BTD), Ionis Pharmaceuticals, Inc. (BTD), and Sarepta Pharmaceuticals (BTD). The following authors have received consulting support for Biogen, Roche (KM), and Novartis, and advisory board membership for Novartis (BTD), Biogen (BTD), Sarepta (BTD), Scholar Rock (BTD), Roche (KM, BTD) and Astellas Pharmaceutical (RG).
Data availability statement
A html file of the analyses is available in the supplemental material. The full dataset is not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
