Abstract
Keywords
Introduction
Spinal muscular atrophy (SMA) is a genetic disorder marked pathologically by dysfunction and degeneration of motor neurons in the spinal cord and brainstem, and clinically by weakness of the limbs, trunk, and respiratory muscles – but also by impairment of oral motor functions. 1 SMA manifests clinically across a wide range of severity and age of onset but common to all is a single genetic cause: bi-allelic pathologic variants of SMN1. For prognostic and care need purposes the range of impairment has historically been sorted into 5 clinical types (Types 0–4), delineated by age of symptom onset and highest level of motor function achieved. 2 Since 2016, disease-modifying therapies (DMT) have successfully targeted the underlying cause of diminished SMN protein, resulting in improved survival and improvement, stabilization or slowed decline of motor function with the greatest impact associated with DMT prior to clinical symptom onset or early in the disease course.1,3–9
The profound impact of therapeutic advancements on survival rates coupled with new phenotypes of SMA have led to an increased recognition of the importance of SMA-specific patient-reported outcomes (PROs) that track emerging trajectories of the disease progression, enable identification of clinically meaningful burdens, and capture information on the patient's perspective about the impact of SMA and its treatment.10–16 Optimizing the practice of bulbar assessment and management across the spectrum of SMA is well-recognized as a critical need for improving clinical care, advancing research, and facilitating approval of new treatments.12,16–20 Two recurring themes that highlight what is needed to advance the field are the standardized, validated clinical tools that identify and quantify impairments in bulbar status, and validated PRO's specifically designed to assess the burdens associated with bulbar problems experienced by patients, treated and untreated with DMT.10,11,16,19–22 While there are many validated metrics of motor outcomes in SMA,23–26 comparable scales of impaired bulbar function have not been developed.17,27,28 Existing assessment tools are typically limited to the evaluation of isolated components of bulbar dysfunction (e.g., impaired swallowing, chewing, phonation, speech, and salivary control).10,29,30 There is an urgent need for validated, agreed-upon, and easily accessible assessment tools that harmonize the isolated components of bulbar function and improve understanding of their interactive contributions to a corresponding assortment of clinically important complications, including feeding difficulty, excessively long mealtimes, growth and nutritional compromise, aspiration pneumonia, and communication challenges.17,31,32 Each of these consequences is widely variable over the range of SMA phenotype severity and age.1,17,31,33–35 Substantial improvements in SMA appendicular function following initiation of DMT have been demonstrated, particularly if initiated before degeneration accrues, but a lack of reliable, quantitative and meaningful outcome measures has undermined the ability to document the effect of DMT upon bulbar function. There is, however, early evidence that infants who inherit severe SMA genotypes and are now surviving due to early DMT later manifest emerging features of bulbar dysfunction as they grow and develop.4,10,22,33,34,36–40 To address important unmet clinical and research needs, standardized validated tools are necessary to identify and then to quantify bulbar status across the range of SMA, treated and untreated with DMT.21,23
The objectives of this study are to develop and validate an SMA-Bulbar Scale that captures and quantifies patient-reported experiences with bulbar dysfunction. We hypothesized that the best first step to understanding and developing meaningful metrics of bulbar dysfunction would be made by probing the experiences of adults with SMA with a comprehensive self-report survey. We further hypothesized that analysis of the accumulated survey data would enable identification of underlying common elements that may simplify further bulbar dysfunction investigations, thus enabling more powerful approaches to clinically and scientifically relevant bulbar dysfunction research. We expected that higher dysfunction on an overall bulbar scale would be associated with greater overall disease severity as measured by SMA “type”, more severe SMA genotype, and older age in those who either have not received DMT or only received it well after significant progression of the disease. We also wished to evaluate how this scale of bulbar function was associated with a validated scale of feeding level status. 41
Methods
Bulbar scale development and additional areas assessed on the study survey
This project involved the partnership of academic investigators with the U.S.-based patient advocacy organization Cure SMA, which has developed one of the largest patient-reported data repositories in the world. 42 The initial generation of items to be assessed in an online survey was based upon a comprehensive literature review of bulbar function in SMA, and the collaboration of patients, caregivers, health care providers, and pharmaceutical companies. A team of two healthcare providers with expertise in SMA and four members of Cure SMA research and clinical care staff used a consensus-driven process for the inclusion and wording of specific survey items. They participated in multiple rounds of discussions using an iterative approach to refine the clarity of survey items and ensure thematic inclusion of clinically relevant items characterizing bulbar function in SMA. Next, the survey was revised based upon input from five adults affected with SMA and three caregivers for persons with SMA, who provided feedback on the importance of each proposed survey item, the length of the survey, the ease of understanding the wording of questions, and whether any items were missing that should have been included. This version of survey was further revised after receiving input from an adult patient who volunteered during a clinic appointment. As a result, late in the process an additional question was added to capture difficulty “saying words clearly”. For each item in the survey tool, participants were asked to score the frequency of the experience using a rank-ordered scale ranging from 1 to 7, with 1 representing “Never” and 7 representing “Always”.
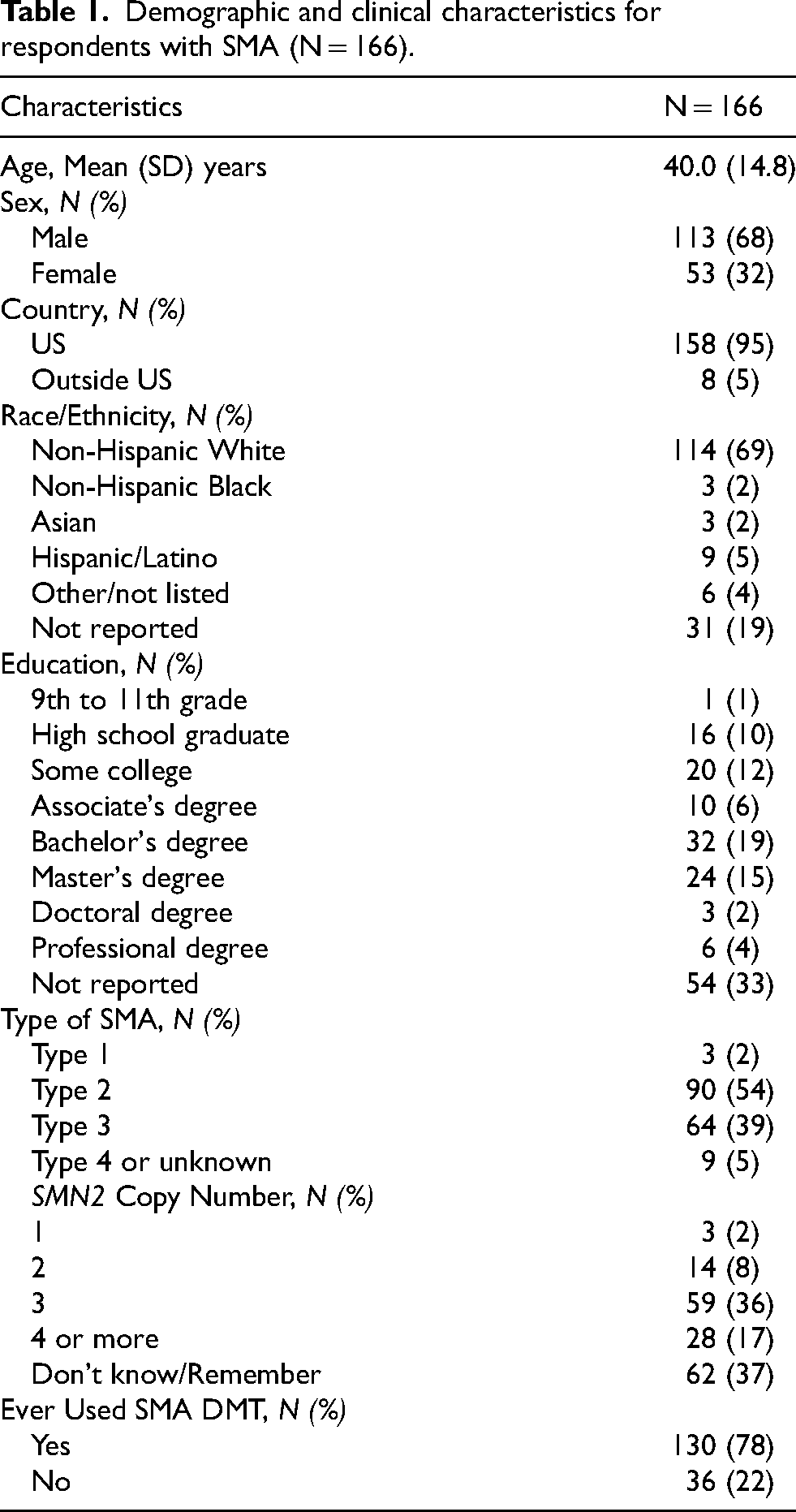
The study survey included items on demographics (e.g., sex, race, education, nationality) and SMA characteristics (SMA “type”, SMA genotype severity as assessed by SMN2 copy number, and history of SMA treatment with DMT). In addition, we assessed feeding levels using modified wording for descriptions in the validated Functional Oral Intake Scale for Children (FOIS-C), derived from the validated FOIS for adults.41,43 The FOIS-C is comprised of 5 levels ranging from 1 to 5, with 1 signifying the most restrictive and 5 the least restrictive level of feeding. Our wording modifications were aimed to: (1) improve readability for self-reporting given that the FOIS-C was designed for clinicians, and (2) capture typical feeding adaptions commonly used by individuals with SMA. The creators of the initial scale hypothesized that survey-reported bulbar related experiences would cluster into six domains: swallowing, breathing and feeding, chewing and eating, mealtimes, mealtime concerns or worries, and articulation and phonation.
Participants
Adults with SMA and caregivers (18 years and older) of persons affected with SMA who were listed in the Cure SMA membership database were invited to participate via an email from Cure SMA. The email provided a description of the study purpose, and a link to the electronic survey that was open for five weeks (October 22 to November 3, 2021). Initial emails with links to the survey were sent out on October 22, and follow-up emails were sent on November 4, 15, and 19, 2021. The Cure SMA membership database is comprised of individuals who previously contacted Cure SMA for information or resources. Details of the Cure SMA database are described in Belter et al. 42
The research conducted for this project was considered exempt by the WCG Institutional Review Board. Survey participants were offered entry into a drawing to receive a $100 e-gift card for their participation. Participants received information about the study on the first page of the survey and were notified that their participation was voluntary. Completion of the survey indicated consent.
Statistical analysis
Surveys were evaluated for completeness; analysis was restricted to only those surveys returned with a complete data set of sex, DMT history, designation of feeding levels according to the FOIS-C, 41 and all 31 items of the Bulbar Scale (Figure 1). Participant demographic and clinical characteristics were characterized using frequencies and percentages, or means and standard deviations, as appropriate to the data. Bulbar Scale items were characterized descriptively using frequencies and percentages of scale response option endorsements, as well as each item's mean score and standard deviation. Skewness and kurtosis were also evaluated for each scale item.
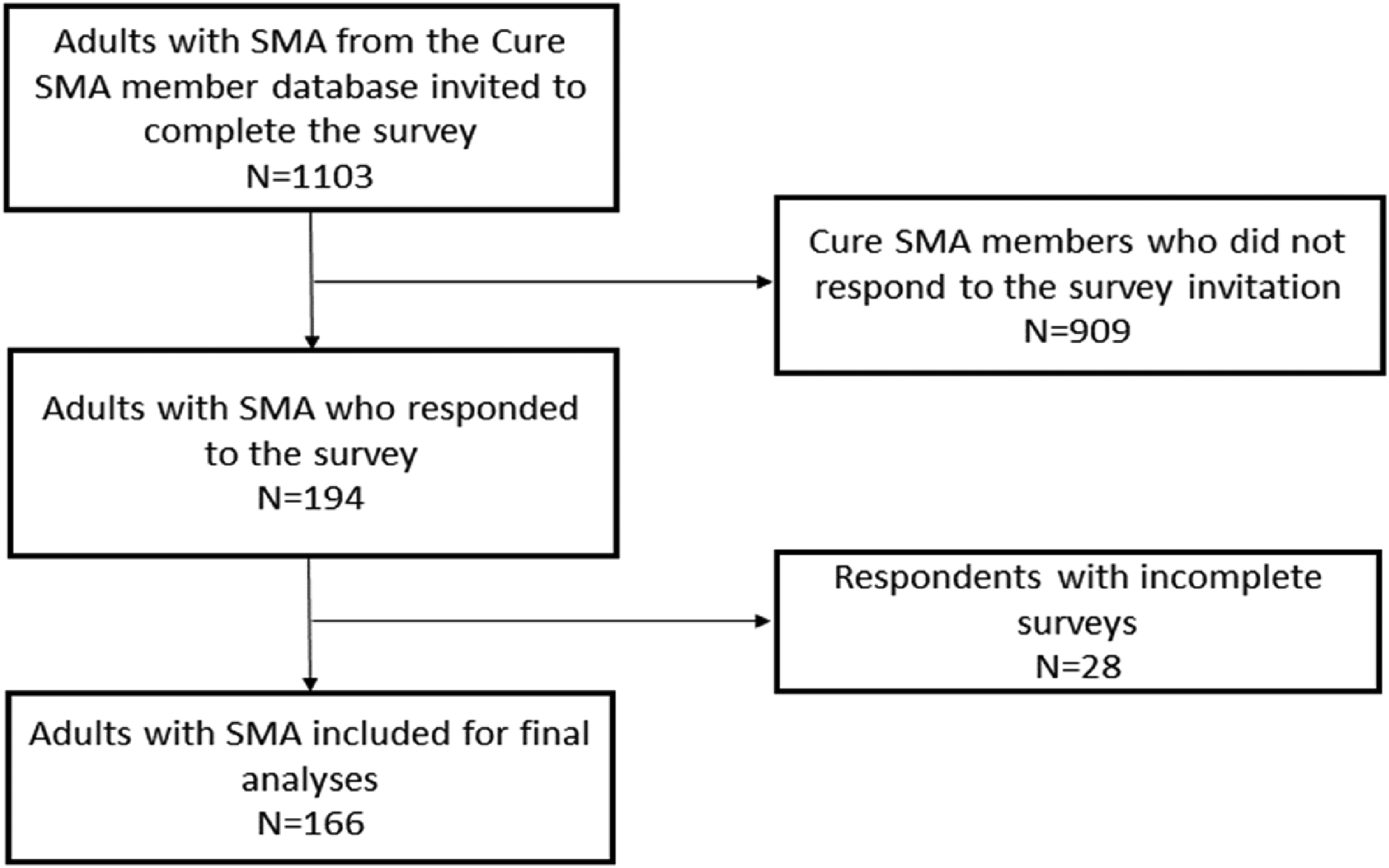
Flow chart of participant recruitment and inclusion in final analyses.
The Bulbar Scale was originally designed as a scale grouped by expert opinion into six clusters of function. A six-factor model was thus evaluated initially, with confirmatory factor analysis (CFA) using weighted least square mean and variance (WLSMV) adjusted estimator. Model fit was evaluated using the recommended cutoffs for the chi squared test (non-significant), comparative fit index (CFI > .95), Tucker-Lewis Index (TLI > .95), root mean square error of approximation (RMSEA ≤ .06), and standardized root mean squared residual (SRMR < .08). 44 By this process, however, model fit was poor (see Preliminary factor structure evaluation section).
An exploratory factor analysis (EFA) was thus conducted to identify better means for clustering Bulbar Scale responses. To determine if it was appropriate to proceed with the EFA, Barlett's test for sphericity and the Kaiser-Meyer-Olkin factor adequacy test were evaluated. Eigenvalues, scree plot, and parallel analysis were examined to inform the number of factors to extract. If multiple factor solutions were suggested by these preliminary evaluations, the research team examined each solution to determine which one best represented bulbar function from the perspective of expert opinion. Factors were extracted using principal axis factoring with oblique (promax) rotation due to expected correlations between factors. 45 Items were retained on a factor if the loading was >.40. If cross-loadings occurred with <.20 difference, the investigators collaboratively evaluated the item to consider the conceptual and clinical significance of the item in relation to the measurement of bulbar function, whether to retain it on the scale, and, if retained, on which factor it best fit.
The factor solution suggested by the EFA was then evaluated in a CFA using WLSMV adjusted estimator, which is appropriate for ordinal response options with skewed item distributions.44,46 Model fit was evaluated using the recommended cutoffs for the chi squared test (non-significant), comparative fit index (CFI > .95), Tucker-Lewis Index (TLI > .95), root mean square error of approximation (RMSEA ≤ .06), and standardized root mean squared residual (SRMR < .08). 44 Robust fit indices were reported when available. Model fit was further evaluated using the dynamic fit indices method given potential issues arising from applying traditional fit indices cutoffs to Likert-type responses.47,48 Internal consistency was examined with Cronbach's alpha reported for each factor and the overall Bulbar Scale. Item-total correlations were evaluated for the items on each factor.
Construct validity of the Bulbar Scale was preliminarily evaluated by examining differences in sum scores by key patient characteristics – specifically, age, sex, SMA type, SMN2 copy number, history of an SMA DMT, and typical feeding level during the survey month – using Wilcoxon rank sum test or Kruskal-Wallis rank sum test as appropriate to the independent variable. Statistical significance was assumed when p < .05.
All analyses were performed using R Statistical Software (version 4.2.0; R Core Team 2022). For the EFA and CFA analyses the psych and lavaan packages, respectively, were used.49,50 To generate the dynamic fit indices the dynamic package was used. 51
Results
Of the 192 adults with SMA who responded to the survey, the 166 (86%) respondents (
Demographic and clinical characteristics for respondents with SMA (N = 166).
Feeding levels when surveyed and ever achieved: reported by 166 adults with SMA. a

Modified descriptions of feeding levels reported in Functional Oral Intake Scale for Children FOIS-C. 24
Note: Our wording for feeding level descriptions was modified for ease of reading and clarity for non-clinicians. For example, our description for Level 5 reads, “Oral feedings. Everything by mouth. No restrictions. (Age or developmentally appropriate diet)”, which was modified from the Level 5 description “Total oral diet with no restrictions” reported by Yi & Shin (2020).
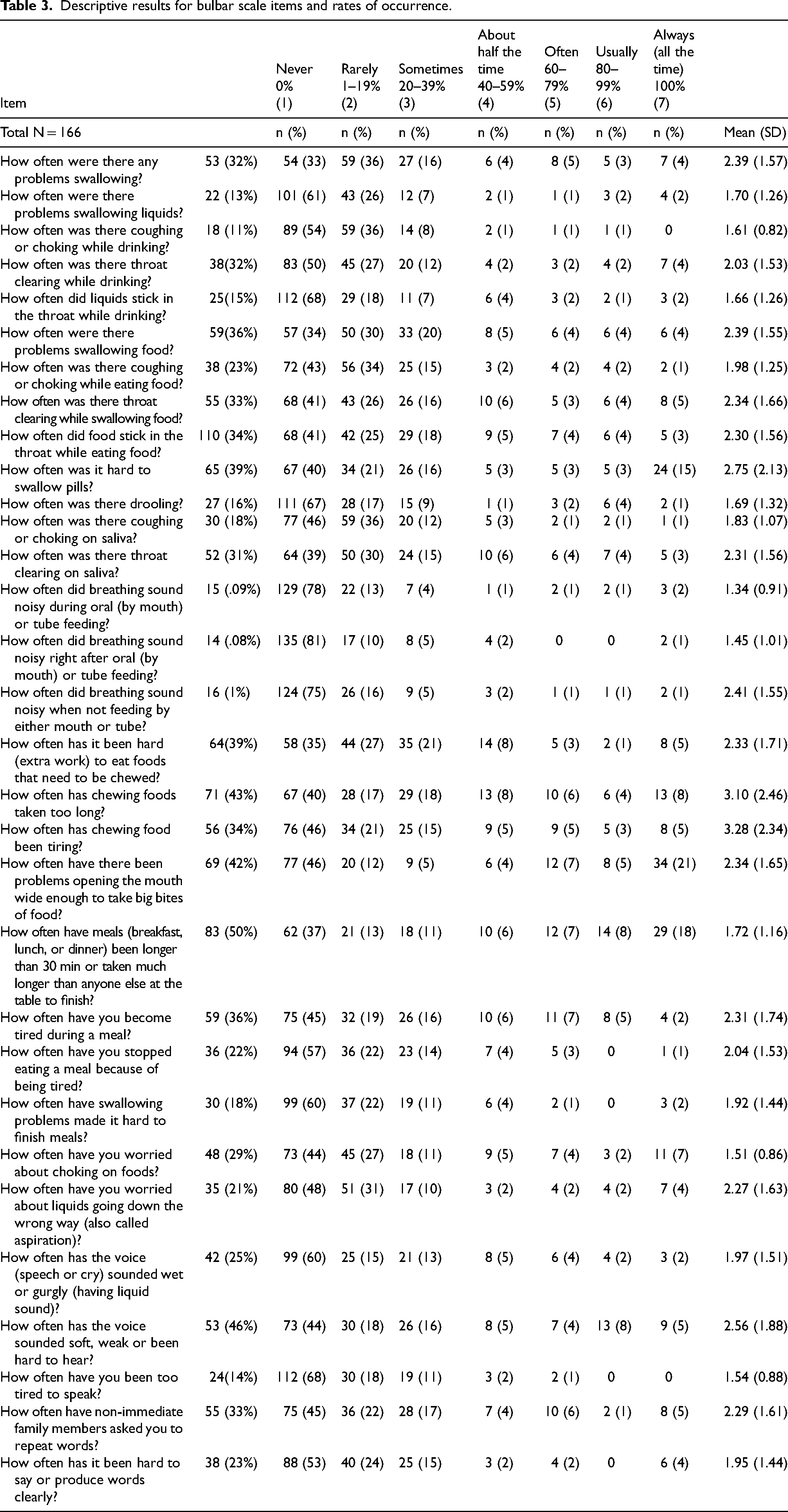
Table 3 shows the frequencies of item endorsement on the Bulbar Scale and each item's mean and standard deviation. Participants endorsed each of the scale options for most items, although, there were six items for which no one endorsed “Often”, “Usually”, or “Always (all the time).” There was some evidence of positive skew with the majority of participants endorsing “Never,” “Rarely,” “Sometimes” or “About half the time.” Skewness ranged from .50–3.78 and kurtosis ranged from 1.66–20.32. Specific manifestations of bulbar problems were characterized by varying rates of occurrence. The most common problems reported as occurring greater than 80% of the time (i.e., usually or always) included prolonged meals for 26%, difficulty opening their mouths wide enough to take big bites for 25%, and swallowing pills for 18% of the respondents.
Descriptive results for bulbar scale items and rates of occurrence.
Preliminary factor structure evaluation
The original six factor solution was evaluated using CFA. Although the RMSEA (.04) and SRMR (.07) were acceptable, the majority of fit indices were not acceptable (X2 = 529.25, p < .001; CFI = .86; TLI = .85). Modification indices were examined but suggested modifications were not clinically or theoretically justified. Thus, the decision was made to use EFA to examine other alternative factor structures.
Exploratory factor analysis
Examination of Bartlett's test for sphericity (χ2 = 5270.85, p < .001) and the Kaiser-Meyer-Olkin test (0.93) indicated that it was appropriate to proceed with the EFA. Evaluation of eigenvalues >1 and the parallel analysis suggested four factors and the scree plot suggested two or three factors.
The three-factor model accounted for 62% of the variance and was determined to be the most appropriate solution from a theoretical and clinical standpoint. (Table 4) Evaluation of the items on each factor revealed distinct concepts including Swallowing Function (Factor 1), Mealtimes and Communication (Factor 2), and Breath, Articulation-Phonation, and Secretion Management (Factor 3). There were three cross-loading items; the research team evaluated each item and determined that from a theoretical framework and clinical application one item (How often was it hard to swallow pills?) fit best on the Swallowing Function factor and two items (How often have non-immediate family members asked you to repeat words? and How often has it been hard to say or produce words clearly?) fit best on the Breath Sounds, Articulation-Phonation, and Secretion Management factor.
Exploratory factor analysis factor loadings: three-factor model.
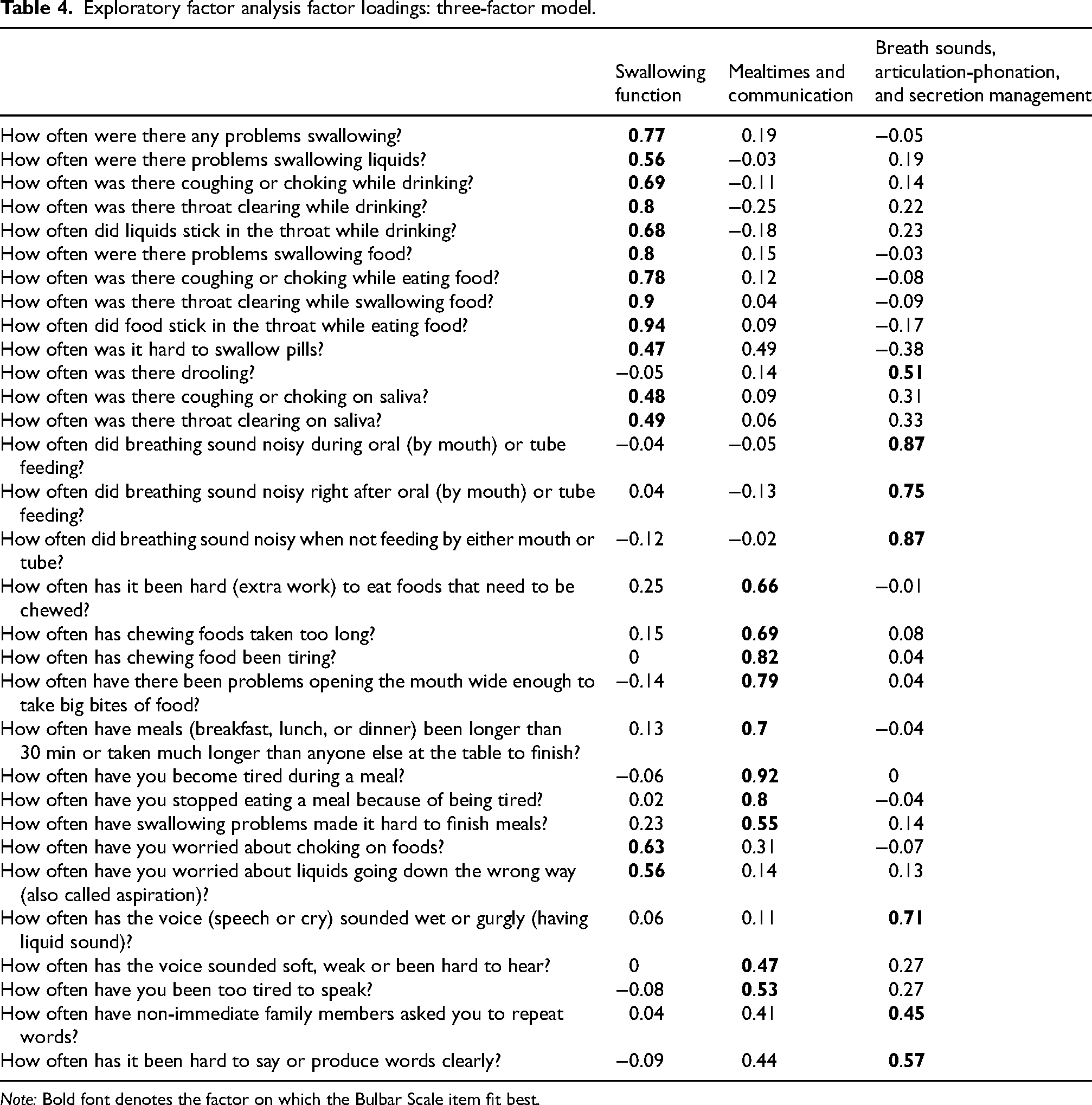
Note: Bold font denotes the factor on which the Bulbar Scale item fit best.
The two-factor model accounted for 57% of the variance and had no cross-loadings; two items did not have any loading >.40 and therefore would have been excluded. The research team determined that the two-factor solution did not provide enough measurement precision to be clinically informative. The four-factor model accounted for 67% of the variance but the factors were not clearly interpretable. See Supplemental File for detailed results of the two and four factor EFA models.
Confirmatory factor analysis of the three-factor solution
The three-factor solution was evaluated in CFA (Table 5). Evaluation of the model fit indices indicated that the chi squared test was statistically significant (X2 = 541.65, p < .001) while the other fit indices were also acceptable (CFI = .99, TLI = .99, RMSEA = .04, SRMS = .07). We also evaluated model fit using dynamic fit indices and found that the CFI was acceptable based on the dynamic fit index cutoffs (CFI > .96), but the SRMR and RMSEA were not acceptable (SRMR < .062, RMSEA < .022). The three factors were positively and significantly correlated with one another (ps < .01). Within each of the three factors, the items demonstrated strong internal reliability (α = .89–.95). Item total correlations were also high (.55–.88).
Confirmatory factor analysis for 31 common bulbar problems in adults with SMA: three-factor model.

Construct validity
There were significant differences on the Overall Scale (summative) score, as well as for Swallowing Function, Mealtimes and Communication, and Breath Sounds, Articulation-Phonation, and Secretion Management subscales, with higher scores of bulbar dysfunction endorsed by participants with some degree of feeding restrictions (Feeding Levels 2–4) compared to no restrictions (Feeding Level 5) on the FOIS-C. (Table 6) There were no significant differences on scores based on sex, age, or use of an SMA DMT.
Associations between bulbar scale scores and participant characteristics (n = 166).
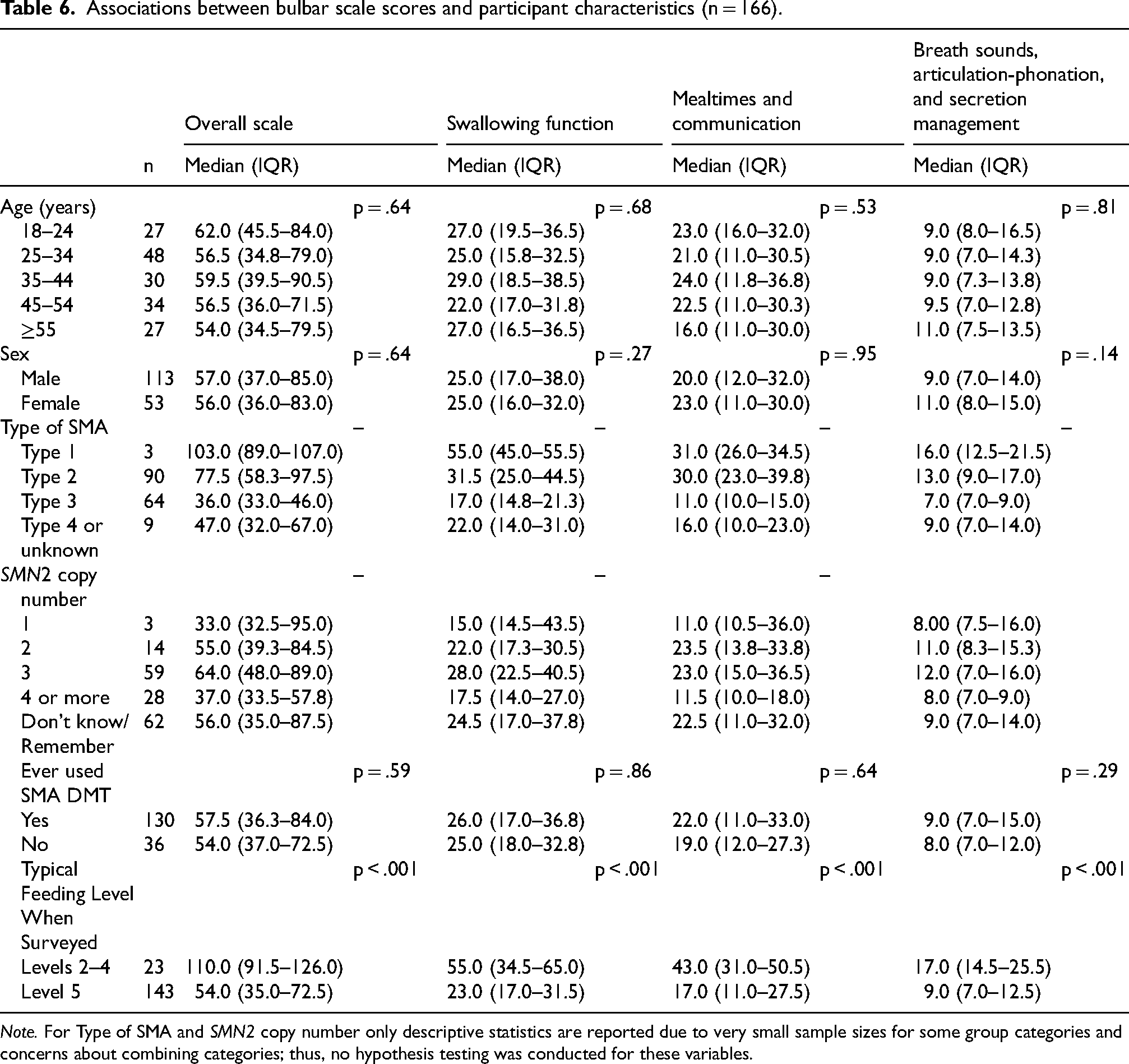
Note. For Type of SMA and SMN2 copy number only descriptive statistics are reported due to very small sample sizes for some group categories and concerns about combining categories; thus, no hypothesis testing was conducted for these variables.
Due to small sample sizes within group categories and inappropriateness of combining categories, we did not statistically evaluate differences on the scale based upon SMA type. Descriptive characterization, however, identifies the not surprising trend that those participants with the most severe “Type 1” phenotype, when compared to those with milder or unknown assigned SMA type, have the highest reports of bulbar dysfunction. Analysis of any effect of SMA genotype (characterized by copy number of the partially functional SMN2 gene) was undermined by a high percentage of “unknown” responses, and by clustering of the reported SMA genotypes to just one category (3 copies of SMN2). (Table 6)
Discussion
We report development and preliminary validation of a patient-reported outcome scale that quantifies the array of manifestations of bulbar dysfunction experienced by adults with SMA. Bulbar functions and dysfunctions – involving verbal communication, deglutition, airway protection, and individual inconvenience and discomfort – are multidimensional, difficult to measure and variably independent or interactive with one another.12,17,27,28 The importance of bulbar functions to human experience, when combined with the complexity of evaluation and expression of their associated dysfunctions, has spawned development of an assortment of individual outcome assessments differing in character and means. These encompass a range of subjective and objective methodologies, focused and global approaches, and instrumented and clinician evaluations.17,27,28 Notably, the resulting diverse outcomes lack a common reference other than neuroanatomic localization to the lower brainstem – a feature that underpins the “bulbar” label but that otherwise does not organize or consolidate the many associated dysfunctions. A cacophony of meaningful but poorly cross-validated bulbar outcomes undermines the power of research protocol design. This scale is intended to approach this discord by starting with the described experiences of adults with common bulbar dysfunctions due to SMA, followed by factor analysis to identify those domains of various described experiences that cluster in order to simplify their understanding. We expect the scale will have immediate value as a clinical guide to monitor and direct individual care, but also as a means to further research – either as an outcome itself, or to aid clinical trial design as a marker of potential risk, a variable for eligibility, or stratification analysis. This effort to simplify the measurement of bulbar dysfunction in a disease such as SMA, where the functional deficits relate to a restricted and fairly patterned impairment just of motor power, may provide a beginning point, or a basis for comparison, for studies of other neurologic disorders in which bulbar dysfunction is multifactorial. 17
Isolation of three coherent domains of bulbar dysfunction
Exploratory and confirmatory factor analyses reduced our initial hypothesis of six possible independent factors that contribute to the experience of bulbar dysfunction to just three distinct meaningful dimensions of bulbar dysfunction: (1) Swallowing Function, (2) Mealtimes and Communication, and (3) Breath Sounds, Articulation-Phonation, and Secretion Management. The three-factor CFA model was supported by traditional fit indices cutoffs, 44 except for the chi-squared test that is especially undermined by sensitivity to small sample size and non-normality. 46 Examination of the dynamic fit index cutoffs – a new method advancing traditional fit index methodology that is appropriate for Likert-type scales47,48,51 – has acceptable fit on the CFI but not the RMSEA and SRMR, which were stricter than traditional cutoffs. 44 There is little consensus on how to interpret goodness-of-fit indices. 46 A holistic examination of the range of fit indices of this study identified a reasonably good fit for the three-factor model.
Application of the scale to bulbar problems experienced by now-prevalent population of adults with SMA
In line with previous research, our data identified bulbar problems throughout the spectrum of age and weakness found in adults living with SMA, with greater burdens experienced by participants having more substantial feeding restrictions as defined by the FOIS-C.10,34,52,53 Against our initial prediction, our sample did not support a relationship between bulbar dysfunction scores and SMA early phenotype, except that a ceiling effect of high bulbar dysfunction scores was evident in those with early onset “Type 1” severe SMA. Analysis of a hypothesized relationship between SMA genotype and bulbar function was undermined by incomplete data and relative homogeneity of the available data. Upon further consideration however, we suspect that a relationship between SMA genotypes and phenotype, at least in the more mild chronic forms of SMA, may not be evident or too small to discern in the present adult population given wide range of age, disease duration, and time to treatment with DMT. “Ever used DMT treatment” similarly failed to discriminate bulbar dysfunction scores, likely for the same reason that many of the adult respondents were untreated for decades before limited availability first emerged in 2017. Lack of relationship between these variables in the present adult population does not undermine value of the scale; instead, it points to the value of this scale as one means to measure the combined impact of the many contributors to SMA dysfunction. In this cross-sectional study, 10% reported worsening FOIS-C defined feeding levels, consistent with the slow progression of dysfunction previously reported in adults.10,34,54 We expect that early administration of DMT, before substantial weakness emerges, may over time lead to a salutary change in the prevalence of bulbar dysfunction in adults with SMA.
Meaningful comparisons about the prevalence of common bulbar problems in our study with those in the existing literature are hampered by differences in research design, populations studied, and methodologic approaches to assessing the presence of bulbar problems. Nonetheless, the individual items (problems) on the Bulbar Scale reflect the common themes of bulbar dysfunction previously reported in adults with SMA including: swallowing dysfunction, prolonged mealtimes with challenges biting and chewing, and communication difficulties related to articulation-phonation problems.10,17,18,22,28,32,35,55 In addition, the concurrence of problems captured within each of the scale's subscales is consistent with both plausibility and published reports.28,34 For example, adults experiencing swallowing problems have also reported solid food dysphagia and the sensation of food sticking, both of which potentially reflect findings of pharyngeal residue observed on instrumental swallow assessments.10,56,57 Similarly, the co-occurrence of jaw and mastication problems with prolonged mealtimes suggests that problems related to the oral management of solid foods may contribute to inefficient or lengthy meal times.18,28,34,57
Most important, but potentially most difficult, will be to compare by retrospective and prospective means the results of this scale and the other outcomes measures to downstream medical complications of bulbar dysfunction. Also valuable will be to understand the relationship between reported bulbar dysfunction and other measures of SMA motor performance. Perhaps most ambitious, the bulbar scale may be used to track the population of individuals with SMA treated with DMT to identify both positive trends, but also those areas where the benefit from early DMT remains limited.54,58 Early evidence suggests that those with the most severe SMA genotypes, even when treated early, nonetheless manifest some bulbar dysfunction. 4
Our next steps in scale development will involve multicenter collaborations to facilitate administration of this scale to a larger number of subjects who are representative of the broader community of persons with SMA. A larger, representative sample with the addition of more specific information on disease trajectory, SMA genotype, and objective details about treatment history (instead of relying on recalled information), will support rigorous evaluation of how bulbar scale scores are related to these important patient characteristics. These data will enable us to improve the clarity and efficiency of the current scale by removing redundant items, confirming that the remaining items align with the intended constructs, and as appropriate, organizing items hierarchically. Additional investigations are needed to compare our scale to other bulbar outcome measures including quantifiable clinical metrics, instrumental swallowing evaluations (e.g., Videofluoroscopic Swallow Studies and Fiberoptic Endoscopic Evaluations of Swallowing), and subjective measures of bulbar dysfunction in SMA.17,18,28,55,57,59,60
We believe this scale has the potential to identify and quantify the impact of specific bulbar problems experienced by adults with SMA and thereby, provide insights into the patient's perspective that can guide clinical care and the development of meaningful endpoints in clinical trials. Clinicians and researchers can assess whether treated and untreated patients perceive their problems as improving, remaining stable, or worsening. In addition, by correlating traditional, objective clinical indices of bulbar function (e.g., range of jaw opening, tongue strength, or maximum phonation time) with items on the scale, cut-off points for detecting early-stage impairments may be potentially identified, allowing for the prompt initiation of interventions and minimizing the impact of disease progression.18,28,55,61
Limitations
This study has several limitations. The survey was conducted among actively engaged members in the Cure SMA community and therefore results may not be representative of the entire SMA community. 42 Another limitation is that our sample included only a small number of respondents with the most severe phenotype, which may have influenced our findings by leading to a higher-than-expected proportion of oral feeders and worse (higher) bulbar scores among more mildly affected participants. Reevaluation with a more representative sample is needed to better understand the differences in feeding status and bulbar scores based on SMN copy number. Though, as noted above, we have reason to believe that disease severity, including bulbar dysfunction, should lessen with the increasing use of DMT. With these improvements, our current sample of respondents may become more representative of the overall population treated with DMT. Confirming this scale with adult populations accrued prospectively and independent of functional status will help to validate the scale more broadly. In this survey, we were unable to determine the influence of family, personal aides, healthcare providers, or therapies focused on bulbar function on the reported experiences. For example, we do not have the benefit of data about whether subjects participated in speech and swallowing therapies, or if these interventions impacted the targeted problems. However, our scale provides quantifiable metrics that could be valuable in assessing the effectiveness of these and other therapies in future clinical trials. An assessment of test-retest reliability of the scale will improve the value of this scale over the present cross-sectional assessment through follow-up study over time. Finally, given that the focus of this scale is specifically on bulbar function, we did not assess factors that are known to influence bulbar processes (e.g., fatigue).17,18,55
Conclusions
We describe the first partially-validated patient reported outcome of the bulbar function experience of adults with SMA. A high prevalence of bulbar impairments and their profound influence on daily functioning highlight the necessity for this scale. Factor analysis identified three coherent domains of bulbar dysfunction, potentially simplifying future research into bulbar function assessment by providing a common anchor useful to the development of more powerful bulbar function outcomes. It also holds promise for identifying and tracking bulbar problems in individual clinical practice, enabling improved treatment before the development of bulbar-dysfunction complications. Taken together, this scale can contribute to informing practice guidelines and advancing the regulatory science needed for the evaluation and development of therapeutic interventions.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602251325741 - Supplemental material for Patient-reported assessment of bulbar function in spinal muscular atrophy (SMA): Validation of a self-report scale
Supplemental material, sj-docx-1-jnd-10.1177_22143602251325741 for Patient-reported assessment of bulbar function in spinal muscular atrophy (SMA): Validation of a self-report scale by Maureen A Lefton-Greif, Lisa Belter, Jill Jarecki, Mary Schroth, Yong Zeng, Thomas O Crawford and Cyd Eaton in Journal of Neuromuscular Diseases
Footnotes
Acknowledgments
We express our appreciation to patients and their families for their time and energies contributed to this research and Cure SMA for its invaluable support. Cure SMA provided funding for the IRB oversight and incentives, and owns the membership database that was used for patient recruitment.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ML-G, YZ, and EA have no conflicts of interest to report. LB and MS are employees of Cure SMA. JJ was an employee of Cure SMA when this survey was created. TOC has been an advisor/consultant to Cure SMA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
