Abstract
Background:
Improvement and maintenance of bulbar function are important goals of disease-modifying treatments (DMTs) for spinal muscular atrophy (SMA), but standardized and validated measures for assessing bulbar function do not exist, nor does a widely accepted definition of bulbar function in SMA. As such, the impact of DMTs on bulbar function has not yet been comprehensively evaluated.
Objective:
We conducted a systematic literature review (SLR) to identify evidence about the impact of DMTs for SMA on bulbar function.
Methods:
We used the Preferred Reporting Items for Systematic Reviews and Meta-Analysis guidelines to conduct this review. Embase® and MEDLINE® databases were searched through August 10, 2023. Eligible studies included patients with SMA who were treated with any DMT and reported bulbar function outcomes. Non-English studies were excluded.
Results:
We identified 51 studies (across 83 publications) that evaluated SMA DMTs and bulbar function for more than 1600 patients. The ability to feed orally, the ability to tolerate liquids, and the need for nutrition support were commonly reported. Most infants treated with any DMT before SMA symptom onset preserved bulbar function. Infants, children, and adults treated after SMA symptom onset experienced variable results in terms of bulbar function outcomes.
Conclusions:
The definition and assessment of bulbar function are not standardized. Therefore, the tools, scales, methods, and timing used for bulbar function assessments varied among studies. Larger prospective studies using standardized and age-based assessments with longer follow-up periods are needed to assess the clinical stability of bulbar function for patients with SMA who receive DMTs.
Keywords
Introduction
Spinal muscular atrophy (SMA) is a neurodegenerative disease that results in motor neuron loss and, consequently, progressive muscle weakness and atrophy because of the deletion of the survival motor neuron 1 (SMN1) gene. SMN1 directs the production of the survival motor neuron (SMN) protein, which is essential for maintenance and survival of motor neurons in the brain and spinal cord.1–3 The survival motor neuron 2 (SMN2) gene partially restores the production of SMN, and the presentation and severity of SMA correlate with the number of copies of SMN2, with fewer copies associated with earlier onset and more severe presentation of SMA.1,2,4,5 SMA phenotypes are increasingly described according to age at symptom onset, SMN2 copy number, and maximum motor milestones achieved,6–12 though the historical descriptions of SMA types 1 through 4 are still widely used. Type 1 is the most common and is a very severe phenotype of SMA with symptoms usually appearing during the first 6 months of life. 4
Although muscle weakness affecting gross motor function (involving large muscle groups and movements of the entire body) is critically important in the diagnosis and progression of SMA, bulbar function – the ability to swallow, maintain airway protection, and communicate verbally 13 – is increasingly a focus of SMA care. Swallowing and vocal control are complex processes requiring refined integrity of cranial nerves V, VII, IX, X, and XII to guide the appropriate extent and timing of contraction of over 30 paired muscles within the head and neck. Adding to this complexity is that although swallowing and vocalization rely on the same shared head and neck anatomy, the two processes cannot occur together. Instead, the aerodigestive tract must be re-oriented into two unique postures to facilitate each function successfully.14–18 Given the high complexity in this task, even the slightest perturbation to neuromuscular integrity can cause a cascade of functional impairments such as choking with meals, respiratory infections, need for alternative nutrition, inability to be understood by unfamiliar listeners, impaired health-related quality of life, and even death.2,12,14,19–27 For patients with SMA types 1 or 2, respiratory complications, often related to impaired swallowing, are the most common cause of mortality.2,28,29 Symptoms of bulbar dysfunction may not appear or progress at the same rate or to the same extent as gross motor dysfunction in SMA. 16
The availability of disease-modifying treatments (DMTs), including an antisense oligonucleotide (nusinersen) and a small-molecule drug (risdiplam) that affect SMN2 splicing, as well as a gene therapy (onasemnogene abeparvovec) that delivers a functional human SMN gene to restore expression of full-length SMN protein, has changed the landscape of SMA treatment. 30 Clinical trials have demonstrated improved survival and motor function, as well as achievement of motor milestones such as sitting without support, standing alone, and walking alone, after administration of DMTs in infants with SMA.9,31–41 Improvement and maintenance of the components of bulbar function are important goals of DMTs for SMA, but standardized and validated measures for assessing bulbar function do not exist, 14 nor does a widely accepted definition of bulbar function in SMA. As such, the impact of DMTs on bulbar function has not yet been comprehensively evaluated. Current assessments of bulbar function are often limited to one component instead of comprehensive bulbar integrity, and a lack of expertise in bulbar function and physiology in the setting of SMA limits assessments. 42 Expert consensuses have been developed and recommendations put forth regarding the comprehensive assessment of bulbar function in SMA, but none are yet widely accepted nor applied.13,42,43 The objective of this systematic literature review (SLR) was to identify published evidence to understand the impact of DMTs for SMA on bulbar function.
Methods
This SLR was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. This SLR on the impact of DMTs on bulbar function was conducted as part of a three-part review that also examined published evidence of the physiology and pathophysiology of SMA.
Data sources
For this review, full-text studies were identified by screening abstracts located by searching Embase® and MEDLINE®. Searches were conducted from database inception to August 10, 2023. In addition, the reference lists of identified literature were hand-searched to identify relevant publications not detected by the database searches. The search strategies are detailed in the Supplementary Material (Supplemental Table 1).
Study eligibility
Study eligibility was determined according to study design, population/indication, comparator intervention, and language. Clinical trials, observational studies, and case studies or reports that describe the effects of nusinersen, risdiplam, or onasemnogene abeparvovec on bulbar function were included. Complete inclusion/exclusion criteria are included in the Supplementary Material.
Study selection and data synthesis
All records retrieved from the literature search were screened based on each citation's abstract. Each citation was screened by two independent reviewers, and any discrepancies between reviewers were reconciled by a third independent reviewer. Citations that did not match the eligibility criteria were excluded at this first-level screening. If this first screening was unclear, citations were included. Duplicate citations (related to overlap in the coverage of the databases) were excluded at the abstract screening stage. Full-text copies of all references that could potentially meet the eligibility criteria were obtained, and the eligibility criteria were applied to the full-text citations. Each full text was screened by two independent reviewers, and any discrepancies between reviewers was reconciled by a third independent reviewer. Data from the included studies were extracted by one reviewer and the quality and completeness of the data were thoroughly checked by the second reviewer.
Ethical compliance
This article is a systematic literature review based on published articles, and as such, does not require institutional review board approval.
Results
The initial searches as part of this SLR yielded 7431 citations. After review and exclusion, a total of 51 studies across 83 publications were identified that evaluated approved SMA therapies (nusinersen, onasemnogene abeparvovec, and risdiplam) and reported bulbar function data (Figure 1, Table 1).9,13,14,32,34,35,38–41,43–114
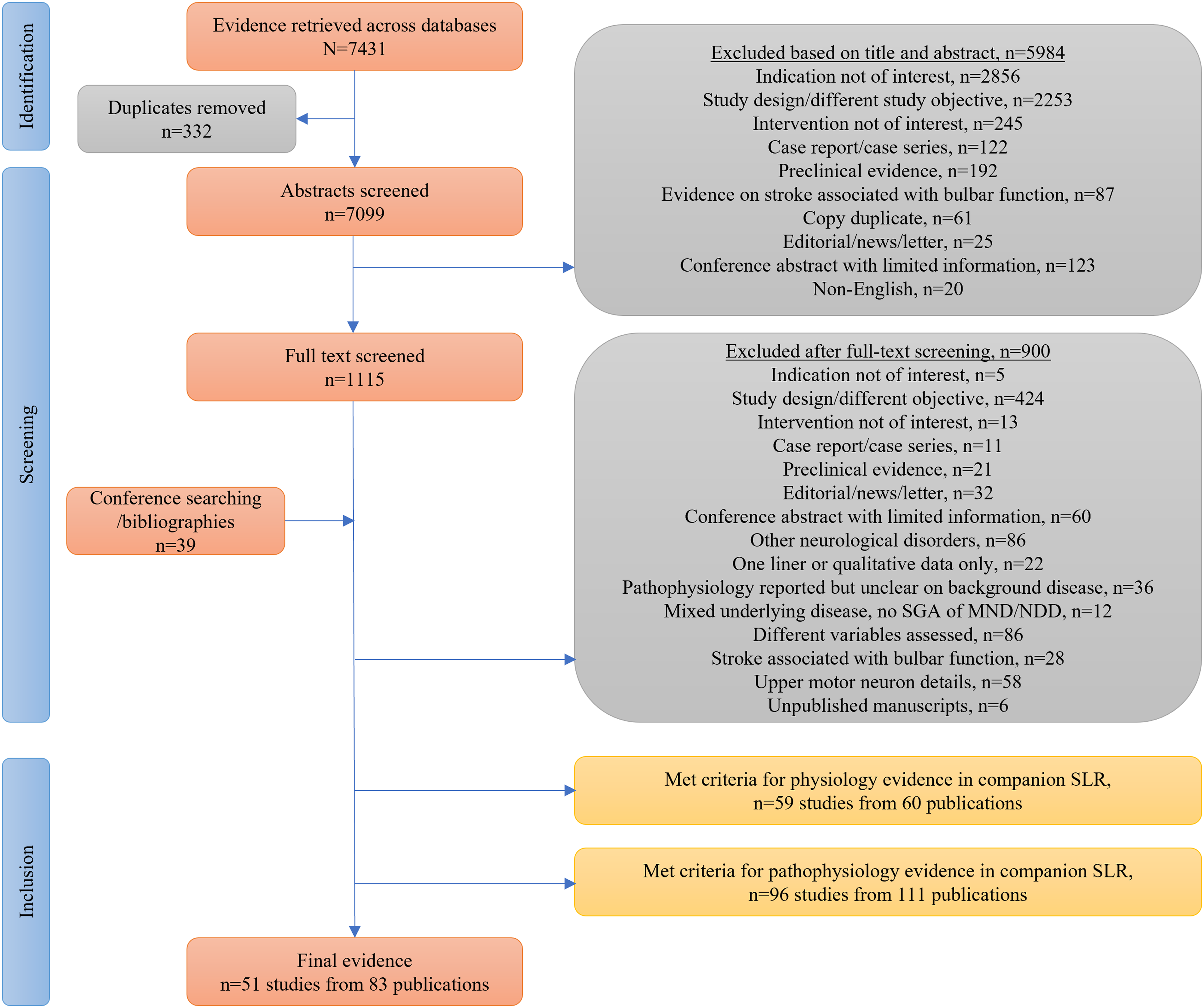
PRISMA diagram.
Summary of SMA studies reporting bulbar function.
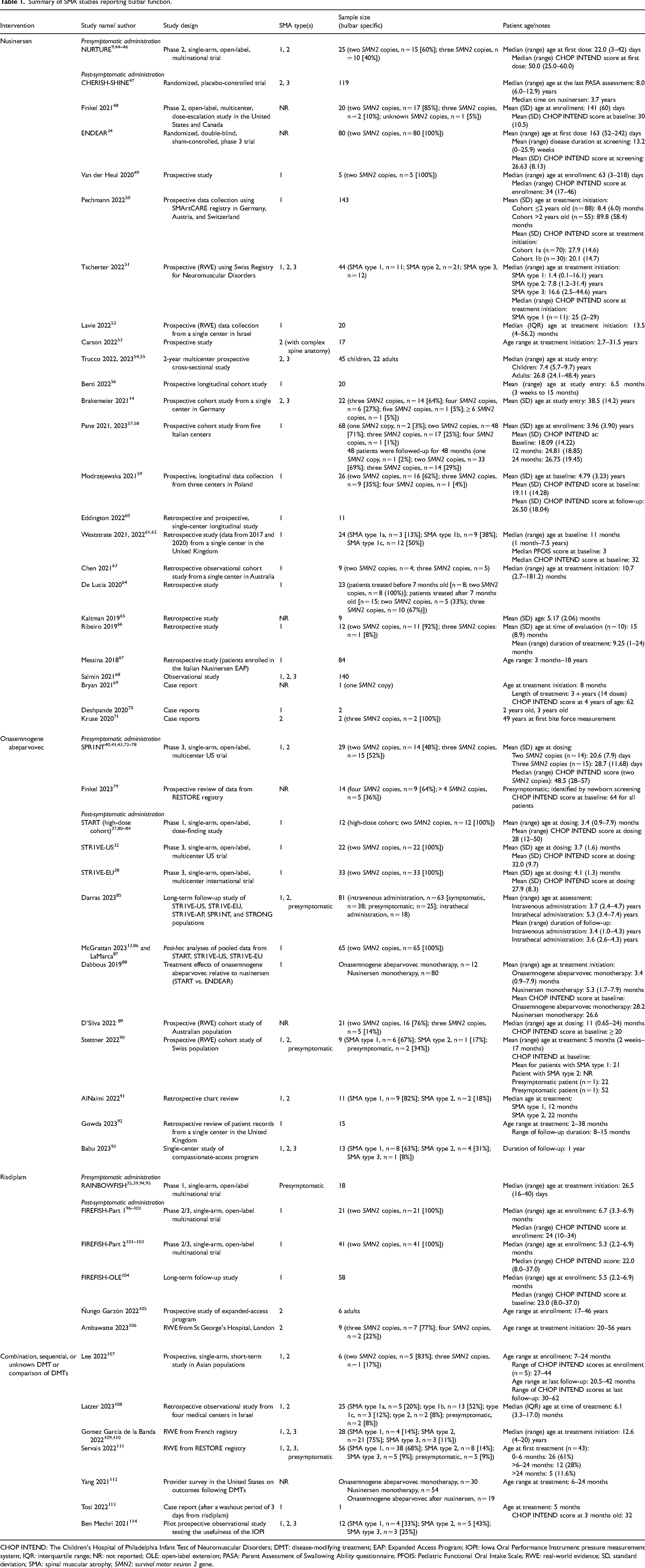
CHOP INTEND: The Children's Hospital of Philadelphia Infant Test of Neuromuscular Disorders; DMT: disease-modifying treatment; EAP: Expanded Access Program; IOPI: Iowa Oral Performance Instrument pressure measurement system; IQR: interquartile range; NR: not reported; OLE: open-label extension; PASA: Parent Assessment of Swallowing Ability questionnaire; PFOIS: Pediatric Functional Oral Intake Scale; RWE: real-world evidence; SD, standard deviation; SMA: spinal muscular atrophy; SMN2: survival motor neuron 2 gene.
More than two-thirds of the studies included patients with SMA type 19,13,32,37,38,40,41,43–46,49–52,56–64,66–68,70,72–78,80–88,90–93,96–104,107–111,113,114 and most patients in the studies that reported SMN2 copy number had two copies of SMN2.9,13,32,34,37,38,40,43–46,48,49,57–59,63,64,66,72,74–78,80–84,86,87,89,96–103,107 The majority of studies evaluated DMTs that were initiated in patients who were already symptomatic, though one clinical trial for each available DMT evaluated effects of presymptomatic administration.9,35,39–41,43,44–46,72–78,94,95 Two reports of real-world evidence (RWE)90,111 and one retrospective observational study 108 included both presymptomatic and symptomatic administration: one 90 is presented in the “Symptomatic administration of DMTs” section because of the small number of children who were presymptomatic, and the others108,111 are presented in the “Combination, sequential, or unknown DMTs” section because of the use of multiple DMTs.
The definition and assessment of bulbar function are not standardized, and the tools, scales, and methods used for assessments of bulbar function, as well as the timing of assessments and length of follow-up, varied among studies identified in this SLR (Table 2). Common components of bulbar function assessed in the studies included the ability to feed orally, the ability to tolerate liquids, and the requirement for nutrition support. Communication was rarely assessed. Study details are listed as available; due to inconsistencies in assessments and reporting, the same information was not reported for each study and descriptions of baseline information, tools used, and outcomes achieved are not uniform throughout this report. The need for non-oral nutrition support was a common assessment across studies, and Table 3 lists studies that report details on baseline and post-baseline nutrition needs. A summary of the scales and tools used is provided in Supplemental Table 2.9,14,44–47,50,54–56,61–63,68,88,89,96–105,107,113–131
Overview of bulbar function assessed across the studies.
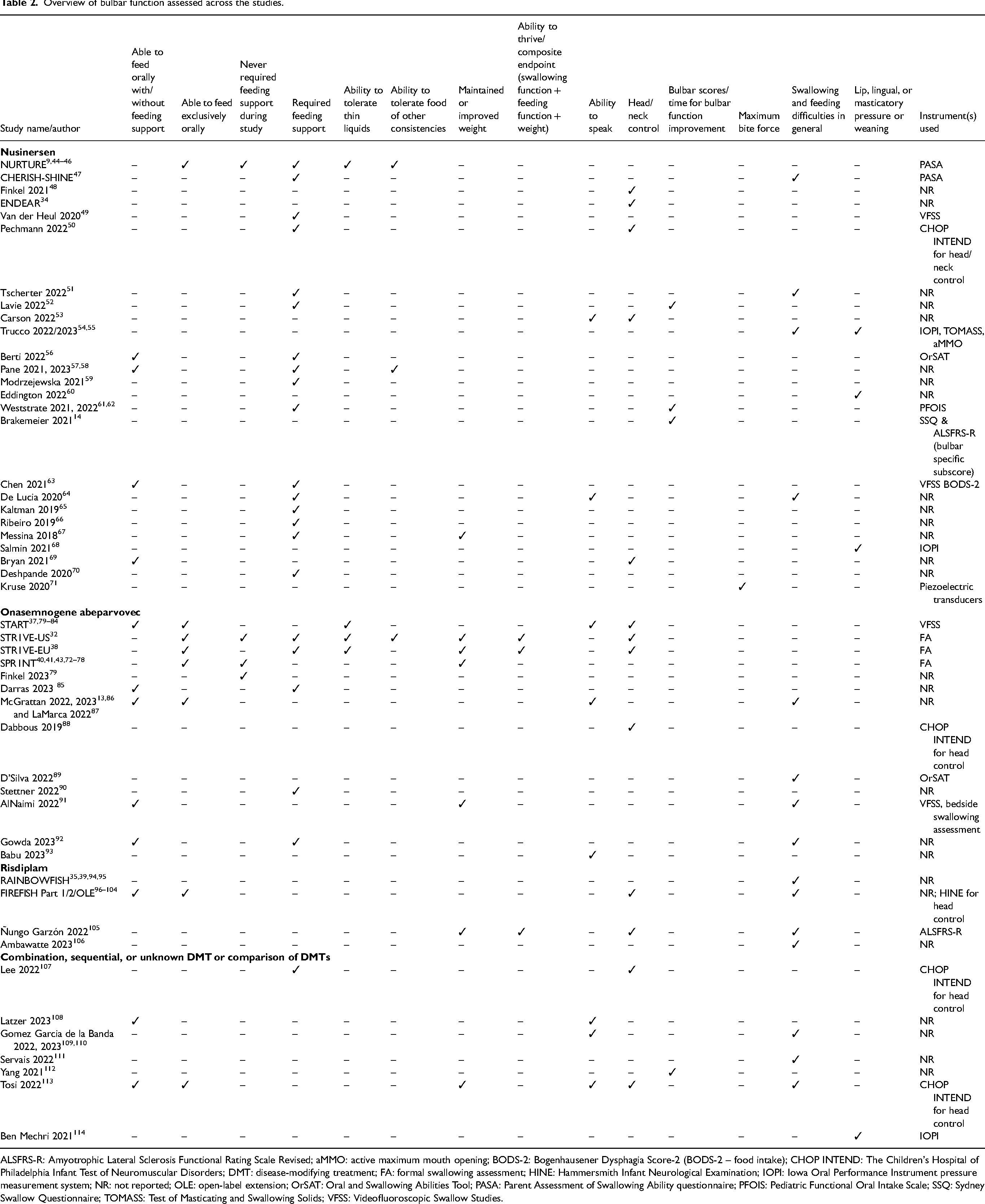
ALSFRS-R: Amyotrophic Lateral Sclerosis Functional Rating Scale Revised; aMMO: active maximum mouth opening; BODS-2: Bogenhausener Dysphagia Score-2 (BODS-2 – food intake); CHOP INTEND: The Children's Hospital of Philadelphia Infant Test of Neuromuscular Disorders; DMT: disease-modifying treatment; FA: formal swallowing assessment; HINE: Hammersmith Infant Neurological Examination; IOPI: Iowa Oral Performance Instrument pressure measurement system; NR: not reported; OLE: open-label extension; OrSAT: Oral and Swallowing Abilities Tool; PASA: Parent Assessment of Swallowing Ability questionnaire; PFOIS: Pediatric Functional Oral Intake Scale; SSQ: Sydney Swallow Questionnaire; TOMASS: Test of Masticating and Swallowing Solids; VFSS: Videofluoroscopic Swallow Studies.
Studies detailing requirements for non-oral nutrition support at baseline and post-baseline.
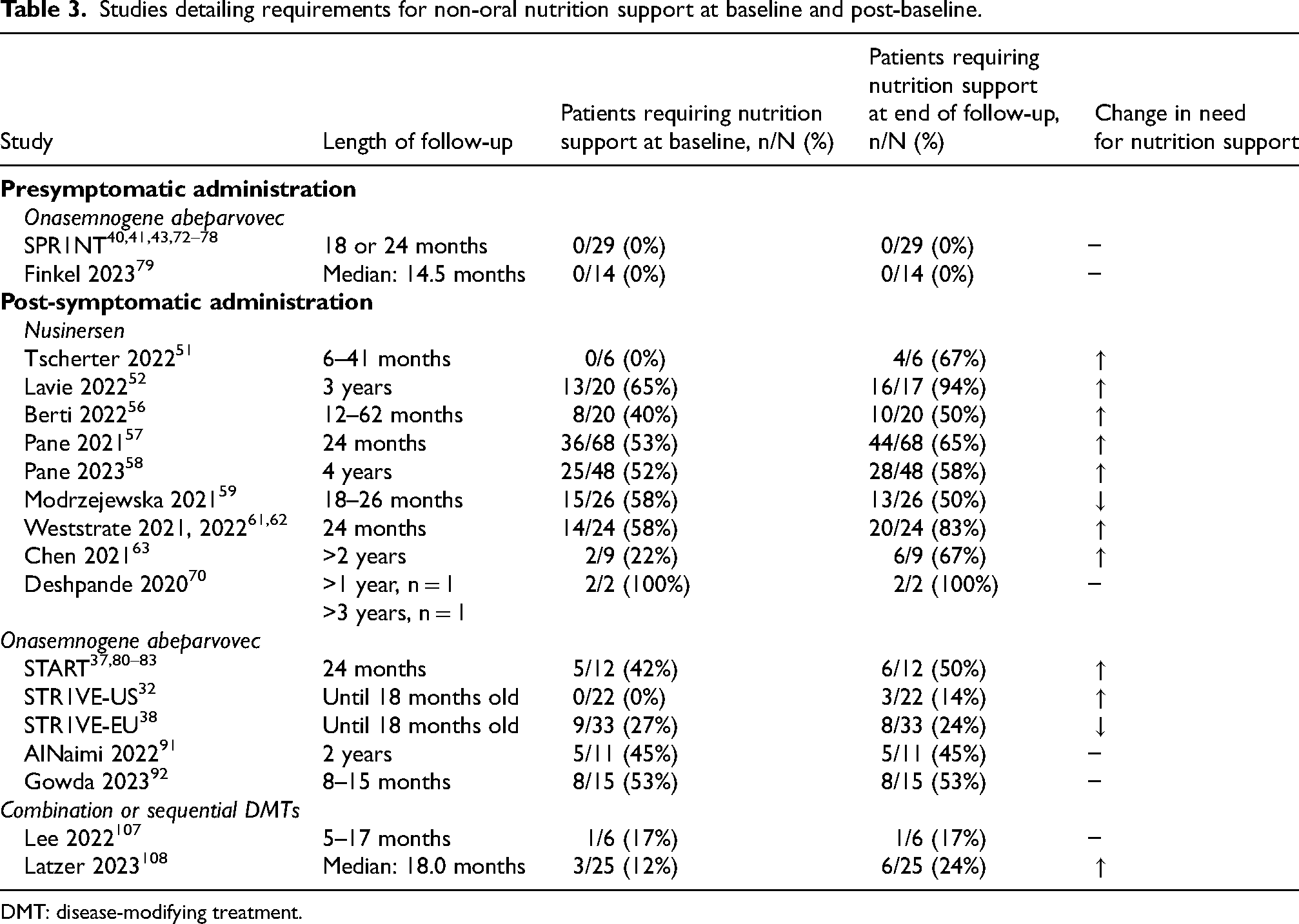
DMT: disease-modifying treatment.
Presymptomatic administration of disease-modifying treatments
Nusinersen evidence
NURTURE is an ongoing, phase 2, single-arm, open-label study assessing intrathecal nusinersen in clinically presymptomatic infants with a genetic diagnosis of SMA. Four publications identified in this SLR described outcomes associated with bulbar function observed in NURTURE.9,44–46
Twenty-five infants (two SMN2 copies, n = 15; three SMN2 copies, n = 10) were enrolled in NURTURE.9,44–46 Nusinersen was administered before the age of 6 weeks (median [range] age at first dose, 22.0 [3.0–42.0] days; median [range] Children's Hospital of Philadelphia Infant Test of Neuromuscular Disorders [CHOP INTEND] score at first dose, 50.0 [25.0–60.0]). 9 Swallowing functions were evaluated using the Parent Assessment of Swallowing Ability (PASA) questionnaire and a neurologic assessment tool, the Hammersmith Infant Neurologic Examination (HINE). The PASA is a 33-item questionnaire that uses a Likert-type scale to rate items related to general feeding, drinking liquids, eating solid foods, and parental assessment of swallowing concerns. The HINE is a standardized neurological assessment that evaluates nerve function, movements, reflexes and reactions, posture, and tone to identify movement disorders; swallowing is evaluated on a scale of 0 to 3 points, with greater scores indicating better function. At the time of an interim analysis (median [range] age, 3.8 [2.8–4.8] years), all children (n = 25) were alive. On average, parents who completed the PASA consistently rated their children as never-to-rarely experiencing difficulty in general feeding, drinking liquids, and eating solid foods. In addition, most parents were not concerned about their child choking (88% [21/24]) or aspirating (96% [23/24]) while eating.44,45 According to parent reports, in the 7 days prior to the interim analysis, two patients (8%), both with two SMN2 copies, required tube feeding exclusively; two others (8%), also with two SMN2 copies, required nutrition support with supplemental tube feeding.44,45 On the HINE, most children (88% [12/15 with two SMN2 copies, 10/10 with three SMN2 copies]) achieved the maximum score of 3, indicating good sucking and swallowing abilities. 46 The remaining three children (all with two SMN2 copies) achieved a score of 1, indicating poor sucking and/or swallowing, and all three required the placement of a gastrostomy tube. 46
Onasemnogene abeparvovec evidence
SPR1NT was a phase 3, open-label, multicenter global trial including 29 presymptomatic infants with a genetic diagnosis of SMA and two (n = 14) or three (n = 15) copies of SMN2. Ten publications identified in this SLR described bulbar function outcomes in SPR1NT.40,41,43,72–78
Infants in SPR1NT received onasemnogene abeparvovec at an age younger than 6 weeks old (mean [SD] age at dosing: two SMN2 gene copies [n = 14], 20.6 [7.9] days; three SMN2 gene copies [n = 15], 28.7 [11.68] days; median [range] CHOP INTEND score at dosing: two SMN2 copies [n = 14], 48.5 [28–57]; CHOP INTEND scores were not available for children with three SMN2 copies). Children with two copies of SMN2 were followed until 18 months of age, and those with three copies were followed until 24 months of age. Bulbar function was assessed with three endpoints — swallowing, nutrition intake, and airway protection — as individual endpoints and as a composite endpoint in a post-hoc analysis of SPR1NT. Specifically, the ability to swallow safely was defined as the absence of clinician-identified (clinical or fluoroscopic) markers of physiologic swallowing impairment. Nutrition intake was assessed through achievement of full oral nutrition (defined as not requiring a feeding tube for nutrition support and receiving 76–100% of nutrition via oral intake). Airway protection was defined as the absence of adverse events relating to pulmonary stability (i.e., aspiration or aspiration pneumonia). At all interim and final analyses, 100% (29/29) of children swallowed without clinical or fluoroscopic indicators of physiologic impairment, received all nutrition by mouth (Table 3),72–75 and maintained pulmonary stability without aspiration or aspiration pneumonia.43,78 A long-term follow-up study that included children from SPR1NT confirmed that, of 25 children treated presymptomatically with intravenous onasemnogene abeparvovec, none required non-oral nutrition support after a mean (range) follow-up of 3.4 (1.0–4.3) years. 85
A study of real-world evidence (RWE) from the United States (collected through the RESTORE registry) evaluated the use of nutrition support in 14 newborns with four or more SMN2 copies after presymptomatic treatment with onasemnogene abeparvovec. 79 The median (range) age at administration of onasemnogene abeparvovec was 3.5 (1–11) months, and the CHOP INTEND score for all patients was 64. The median (range) duration of follow-up at the time of this analysis was 14.5 (2.2–28.6) months. No non-oral nutrition support was reported for any patient during the entire observation period (Table 3).
Risdiplam evidence
RAINBOWFISH is an ongoing, phase 2, open-label, single-arm, multicenter study of risdiplam in presymptomatic infants with genetically diagnosed SMA, regardless of SMN2 copy number. Four reports identified in this SLR described outcomes associated with bulbar function observed in RAINBOWFISH.35,39,94,95
Eighteen infants were enrolled in RAINBOWFISH. Treatment was initiated at an age younger than 6 weeks (median [range] age at first dose, 26.5 [16–40] days). At the first interim analysis, seven of the infants had received treatment for at least 12 months. Of these, all (100% [7/7]) were alive and maintained swallowing and feeding abilities. 94
Symptomatic administration of disease-modifying treatments
Nusinersen evidence
Twenty-seven publications were identified that collected bulbar function data along with nusinersen administration for symptomatic patients with SMA.14,34,47–71
CHERISH was a randomized, sham procedure–controlled study of patients likely to develop SMA types 2 or 3 based on symptom onset after the age of 6 months. SHINE was the open-label extension (OLE) of CHERISH. Overall, 119 children were included in an assessment of swallowing using the PASA. At the time of the analysis, the children had a median (range) age of 8.0 (6.0–12.9) years, and they had been receiving nusinersen for a median of 3.7 years. 47 Swallowing was evaluated using the PASA at multiple assessments during the nearly 4 years before the interim analysis. Over time, parents consistently rated their children as never to rarely experiencing difficulties with general feeding, drinking liquids, or eating solid foods, and no notable differences were noted according to age at first nusinersen dose. Only three of 119 participants (3%) required tube feeding at one or more PASA assessments, with two still requiring tube feeding, though not exclusively, at the last assessment. 47
A prospective cohort study of five infants (median [range] age at inclusion of study, 63 [3–18] days; median [range] CHOP INTEND score at enrollment, 34 [17–46]) evaluated sucking and swallowing after the initiation of nusinersen in combination with best supportive care (as outlined in current standard-of-care recommendations 12 ). 49 Two of the infants were presymptomatic at the time of initiation, and three already showed symptoms of fatigue during feeding and unsafe swallowing at the time of treatment initiation, including weak suck and cough, as well as wet breathing while eating or drinking. Swallowing was assessed using clinical swallow evaluations and videofluoroscopy. The clinical swallow evaluation included the completion of a feeding-related checklist evaluating the length and frequency of nursing sessions; the presence of sweating when eating or drinking; coughing, clearing the throat, and wet breathing during oral feeding; frequency of spitting up food or milk during feeding; the presence of abdominal breathing patterns; respiratory rate; weakness of voice and cough; and frequency of respiratory infections. There were no reports of outcomes evaluated during the videofluoroscopic exam or details pertaining to the clinical indication for the exam. Sucking and swallowing functions improved initially in three of five infants with reported bulbar dysfunction. However, between the ages of 8 to 12 months, all five infants experienced respiratory infections and coughing or wet breathing while feeding. 49
The SMArtCARE registry is a disease-specific registry that collects longitudinal data on patients with SMA. A prospective study of patients in the SMArtCARE registry who achieved the ability to sit after initiation of nusinersen included 143 patients with early-onset SMA who were followed for up to 38 months. 50 Patients were divided according to their age at treatment initiation: ≤2 years (n = 88) and >2 years (n = 55). At the last observation, the probability of the need for tube feeding increased from 24% at baseline to 53% at 38 months in the younger cohort (n = 67) and from 44% to 51% in the older cohort (n = 33). The requirement for tube feeding was substantially associated with SMN2 gene copy number. Patients with three SMN2 copies had a lower likelihood of requiring tube feeding than patients with two SMN2 copies during treatment with nusinersen. 50
Another real-world study of registry data described 44 patients (median [range] age at treatment initiation, 9.4 [0.1–44.6] years). 51 At the time of analysis, children had received nusinersen for a range of 6 to 41 months. At baseline, children with SMA type 1 who were less than 18 months old were evaluated for swallowing and feeding difficulties; none required non-oral nutrition support at that time. After 8 to 30 months of treatment, four of six (67%) required nutrition support with a feeding tube (Table 3). These same six children underwent a language evaluation with either the Griffith Mental Development Scale (n = 4) or the Bayley Scales of Infant and Toddler Development (n = 2). Three children (50%) had age-appropriate language development at baseline. At last assessment, two patients (33%) did not speak at all. Of the four patients (67%) who were able to articulate some sounds, only one (17%) had age-appropriate articulation.
A prospective database study in Israel assessed the impact of nusinersen on facial changes and feeding methods for patients with SMA type 1 (n = 20; median [range] age at time of treatment initiation, 13.5 [4–56.2] months) over a 3-year period. 52 Four patients (20%) had facial deformities at baseline. After 3 years of treatment, 15 of 17 surviving patients (88%) displayed facial changes, including open mouth (n = 12), nasal flattening (n = 8), extreme facial weakness (n = 7), facial asymmetry (n = 4), mandibular retraction (n = 3), and midface hypoplasia (n = 2), all of which can impact respiratory and feeding abilities. Most of these changes occurred in patients using noninvasive ventilation, with the exception of extreme facial weakness with open mouth, which also occurred in patients using invasive ventilation. Fifteen patients (88%) were fed by gastrostomy, one (6%) by nasogastric tube due to family's preference over gastrostomy, and one (6%) was fed exclusively orally. Of the 16 patients with non-oral nutrition support, four (25%) maintained at least some oral feeding initially, but all demonstrated clinical or radiologic evidence of aspiration and were advised to stop oral feeding by the end of 3 years of follow-up (Table 3). 52
A prospective study by Berti and colleagues assessed swallowing abilities for patients treated with nusinersen using the Oral and Swallowing Abilities Tool (OrSAT). 56 This study included 20 infants with SMA type 1 with varying swallowing abilities at the start of treatment. At baseline (mean [range] age at initiation, 6.5 months [3 weeks to 15 months old]), 12 infants (60%) were fed exclusively orally, and the remaining eight (40%) fed exclusively by feeding tube, including four who required tracheostomy. After nusinersen treatment for 12 to 62 months, 10 of 12 orally fed children (83%) demonstrated an age-appropriate increase in OrSAT scores with continued feeding exclusively by mouth. The four infants who required tube feeding and tracheostomy remained stable without any improvement after treatment. The four infants who required tube feeding but not tracheostomy showed an increase in OrSAT scores after treatment, indicating partial functional improvement, and three (75%) of these infants gained the ability to feed at least partially by mouth (Table 3). 56
A prospective cohort study assessed bulbar function in 22 adults with SMA types 2 or 3 who were receiving nusinersen. 14 Bulbar function was assessed with the Amyotrophic Lateral Sclerosis Functional Rating Scale Revised (ALSFRS-R) and the Sydney Swallow Questionnaire. Overall, no significant changes were observed in bulbar function during the 14 months of follow-up. Individual results varied, with similar numbers of patients experiencing improvement and worsening of bulbar function. For patients without bulbar dysfunction at baseline, no new dysfunctions were observed. 14
Reports from an expanded access program in Italy described bulbar function in 68 patients (mean [SD] age at time of assessment, 3.96 [3.90] years) with SMA type 1.57,58 At baseline, 32 patients (47%) were fed orally, and 36 (53%) were tube fed. The mean (SD) CHOP INTEND score at baseline was 18.09 (14.22). At 24 months of follow-up, 24 patients (35%) remained orally fed, and 44 (65%) were tube fed (Table 3); the mean (SD) CHOP INTEND score was 26.75 (19.45). Seven of eight patients who had a feeding tube inserted after baseline were still able to swallow and eat solids or semi-solids orally. The differences between baseline and 24-month assessments were not statistically significant. 57 In addition, 4-year follow-up results of 48 patients (mean [SD] age, 3.3 [3.6] years) from the same study reported that 20 of 23 children (87%) who fed orally at baseline maintained the ability to feed orally, and three of 23 (13%) lost the ability to feed orally and subsequently required nutrition support with tube feeding. Two of 20 children (10%) who were already receiving tube feeding at baseline regained the ability to be partially fed orally (Table 3). 58
A prospective study described 26 patients with SMA type 1 (mean [range] age at time of assessment, 4.79 [2–15] years) who were treated with nusinersen for a median of 21 months. 59 The percentage of children fed by nasogastric tube or percutaneous endoscopic gastrostomy decreased from 15 (58%) at baseline to 13 (50%) at the time of follow-up (Table 3).
A single-center retrospective study of 24 infants with SMA type 1 assessed bulbar function using the Pediatric Functional Oral Intake Scale and videofluoroscopic swallowing assessments along with the need for non-oral nutrition support at baseline and 6, 12, and 24 months after nusinersen initiation.61,62 At baseline, the median (range) age of patients was 11 months (1 month to 7.5 years), and the median CHOP INTEND score was 32. Feeding and swallowing complications continued and/or deteriorated for most patients, and the percentage of patients who required nutrition support with tube feeding increased from 58% (14/24) at baseline to 83% (20/24) 2 years after nusinersen initiation (Table 3). Four patients (18%) were exclusively orally fed for the duration of the study.61,62
An Australian single-center, retrospective, observational study included nine patients with SMA type 1 (two SMN2 gene copies, n = 4; three SMN2 gene copies, n = 5; median [range] age 10.7 [2.7–181.2] months) who were treated with nusinersen and followed for a minimum of 2 years (a total of 270.5 patient months). 63 All patients with two SMN2 copies (n = 4) were exclusively orally fed at baseline but lost this ability and required gastrostomy after a median nusinersen treatment time of 30 months. Gastrostomy was indicated because of failure to thrive (n = 2) or aspiration and poor feeding (n = 1) or was anticipatory (n = 1). Three patients with three SMN2 copies who were exclusively orally fed at baseline remained orally fed after a median treatment time of 28 months; the remaining two patients with three SMN2 copies required gastrostomy at baseline and follow-up (Table 3). 63
A case report described a 4-year-old girl with one gene copy of SMN2 who was clinically diagnosed with SMA at 6.5 months of age. At the time of the report, she had been treated for more than 3 years with nusinersen (14 doses). The child was able to feed herself during the entire observation period. 69
Another case report described two patients with SMA type 1 treated with nusinersen. 70 One was a 2-year-old who had received nusinersen since the age of 7 months; the other was a 3-year-old who had received nusinersen since the age of 3 months. Despite treatment, they did not demonstrate improvements in their ability to swallow and were unable to feed orally throughout the study (Table 3). 70
Another case report described two adult (age 49 years) monozygotic female twins with infantile-onset SMA type 2 (three gene copies of SMN2). At the time of nusinersen administration, both patients had facial deformities and limitations to mouth opening that impacted feeding abilities. Masticatory function was assessed via maximum bite force measurement at baseline and 1 year after nusinersen initiation. 71 During the 2 months in which the four loading doses were administered, maximum bite force increased gradually from baseline and reached a statistically significantly greater level compared with baseline for both patients. Bite forced increased slightly but not significantly in one patient after the first maintenance dose of nusinersen, which was administered 4 months after the last loading dose. Four months later, after the second maintenance dose, no additional increases were observed. 71
Two other author groups54,55,68 described the importance of using objective, validated tools for assessing long-term bulbar function related to SMA. Trucco and colleagues54,55 evaluated the use of the Iowa Oral Performance Instrument (IOPI), the Test of Masticating and Swallowing Solids (TOMASS), and active maximal mouth opening (aMMO). Perceived orobulbar burden was assessed with the SMA-Health Index (SMA-HI). The IOPI is a system that investigates the strength of the lips, tongue, and muscles used for chewing by measuring the maximum pressure an individual can apply with the lips, tongue, and palate or between molars; the TOMASS is a widely used, standardized, quantitative assessment of solid bolus ingestion, and aMMO measures mandibular range of motion. In all, 45 children (median [interquartile range {IQR}] age, 7.42 [5.69–9.69] years) and 22 adults (median [IQR] age, 26.80 [24.10–48.42] years) with SMA types 2 or 3 were included. All were receiving nusinersen. At baseline, among the children, 15 (47%) had reduced tongue strength, 19 (43%) had reduced mouth opening, and 19 (50%) required longer than normal time to eat a cracker. In contrast, only two (12%) perceived problems with chewing or swallowing. For adults, seven (33%) had reduced tongue strength, eight (38%) had reduced mouth opening, and six (29%) required a longer time to eat. Only five (24%) reported perceived difficulty with chewing or swallowing. After 16 months, tongue strength, mouth opening, and time to eat remained stable in both children and adults. 55 Overall, the authors reported that orobulbar function was independent of motor functional status and that a multimodal approach to orobulbar function is needed to assess swallowing and mastication despite patient perception.54,55 Salmin and colleagues 68 described the IOPI and the SMA-HI as potential tools to track changes over time in orofacial and orobulbar functions related to SMA. The observational, multicenter study of 140 patients was ongoing at the time of publication, but preliminary findings demonstrate that IOPI and SMA-HI scores correlated to nutritional and swallowing abilities of patients and may be useful for tracking changes in orobulbar functions for patients with SMA. 68
Other studies included in this SLR34,48,53,60,64–67 collected information on bulbar function-related outcomes but lacked specifics on timing and/or methods of assessments or offered only qualitative reports of outcomes. The available data are presented in Tables 1 and 2.
Onasemnogene abeparvovec evidence
Eighteen publications were identified that collected bulbar function data along with administration of onasemnogene abeparvovec after the onset of SMA symptoms.13,32,37,38,80–93
START was a phase 1 dose-finding clinical trial of onasemnogene abeparvovec for patients with SMA. Positive findings were confirmed by the phase 3 STR1VE-US and STR1VE-EU trials. All three trials were conducted for symptomatic patients with SMA type 1. Eight reports describe findings related to bulbar function in START, STR1VE-US, and STR1VE-EU.32,37,38,80–84
START included 15 patients with SMA type 1 (mean [range] age at dosing, 3.4 [0.9–7.9] months; mean [range] CHOP INTEND score at dosing, 28 [12–50]).37,.80–84 Patients received a single dose of intravenous onasemnogene abeparvovec: three patients received a low dose (6.7 × 1013 vg/kg of body weight), and 12 received a greater, therapeutically equivalent dose (2.0 × 1014 vg/kg).37,80–84 Swallowing was assessed by a fluoroscopic examination at screening, every 6 months starting at Month 6, and at the end of the study (24 months after infusion). At baseline, four of 12 patients (33%) in the therapeutic-dose cohort could safely swallow thin liquids, as assessed by videofluoroscopic studies, and seven (58%) were able to feed orally without non-oral nutrition support. At 24 months of follow-up, 10 (83%) were able to swallow thin liquids, and 11 (92%) could safely feed orally. Of the seven patients who were able to feed orally without any nutrition support at baseline, six (86%) continued to feed exclusively orally (Table 3). In addition, 11 (92%) attained the ability to speak.37,80–83 After a mean of 83.5 months post-dosing, all patients were alive (mean [range] age, 7.1 [6.6–7.9] years). Four of 10 patients (40%) who received the therapeutic dose and had data available did not require any non-oral nutrition support. 84
STR1VE-US was a phase 3, single-arm, single-dose, open-label, multicenter trial. Patients (n = 22) younger than 6 months of age received a one-time intravenous infusion of onasemnogene abeparvovec (mean [SD] age at dosing, 3.7 [1.6] months; mean [SD] CHOP INTEND score at dosing, 32.0 [9.7]) and were assessed once per week, beginning at Day 7 post-infusion for 4 weeks and then once per month until the end of the study (age 18 months or early termination). 32 At baseline, all (22/22 [100%]) patients were able to swallow thin liquids and feed exclusively orally. Nine patients (41%) met the composite endpoint criteria (ability to thrive) comprising ability to maintain swallowing function (defined as having a normal, functional, or safe swallow of thin or very thin liquids during a formal swallowing test), feeding function (defined as independence from non-oral nutrition support), and weight as compared with untreated historical controls (defined as having a weight greater than the third percentile according to World Health Organization growth standards). Fifteen patients received no nutrition support at any time during the study, and four received intermittent or temporary (6–91 days) nutrition support during the study but did not require any at study end. Three patients (14%) used non-oral nutrition support at age 18 months or at time of withdrawal from the study (two underwent gastrostomy tube placements, and one was on nutrition support [nasojejunal tube] at the time of premature discontinuation [death]) (Table 3). Twelve patients (55%) were able to swallow thin or very thin liquids, and seven (32%) could swallow consistencies other than thin or very thin liquids. In addition, 14 (64%) maintained weight appropriate for age. 32
Similar to STR1VE-US, STR1VE-EU was a phase 3, single-arm, open-label, multicenter, multinational trial of one-time intravenous infusion onasemnogene abeparvovec for infants younger than 6 months old with SMA type 1 (mean [SD] age at dosing, 4.1 [1.3] months; mean [SD] CHOP INTEND score at dosing, 27.9 [8.3]). 38 STR1VE-EU used broader eligibility criteria than STR1VE-US and included patients with more severe disease at baseline (i.e., using nutrition support or requiring up to 12 h of non-invasive ventilatory support, or both). Of the 33 patients enrolled in the study, one died and 32 continued the study. At baseline, nine patients (27%) required non-oral nutrition support, with the remaining 23 included in the ability to thrive population (intact swallowing population). At 18 months of age, seven (30%) of these patients met the criteria for ability to thrive: 15 patients (65%) maintained weight consistent with their age, 20 (87%) were fed exclusively orally, and nine (39%) were able to tolerate thin liquids (Table 3). 38
Recent post-hoc analyses assessed bulbar function from pooled data from START, STR1VE-US, and STR1VE-EU.13,86,87 These analyses were unique because they applied a composite endpoint representing bulbar function that comprised swallowing, oral nutrition, pulmonary stability, and communication. In total, 65 patients were included in the pooled analyses. Communication was only assessed for patients from STR1VE-US and from native English-speaking families in START who had a CHOP INTEND score of 60 or greater: 20 patients from STR1VE-US had communication assessed at baseline, and 20 patients (four from START and 16 from STR1VE-US) had communication assessed post-baseline). 13 Overall, the majority of patients met each of the individual endpoints by the end of the study. At the last evaluated time point, 92% (60/65) of patients had evidence of a normal swallow according to clinical or fluoroscopic markers of physiologic swallowing impairment, and 75% (49/65) achieved full oral nutrition; 92% (60/65) maintained pulmonary stability evidenced by no events of pneumonia or aspiration pneumonia; and 95% (19/20) were able to establish verbal communication (defined as being able to vocalize at least two different vowel sounds). 13 Taken together, 75% (15/20) of patients who had evaluable data achieved the composite endpoint of having a normal swallow, achieving full oral nutrition, demonstrating no evidence of pulmonary instability, and representing expressive communication abilities. 13
Across Australian RWE, onasemnogene abeparvovec demonstrated improvement in swallowing abilities for three of seven patients (43%) with bulbar dysfunction at baseline (median [range] age at dosing, 11 [0.65–24] months; CHOP INTEND score for all patients, ≥20). 89 No change was observed in the other four children (57%) during the first year after treatment.
A small study of patients with presymptomatic SMA (n = 2) or symptomatic SMA type 1 (n = 6), or type 2 (n = 1), evaluated motor and bulbar function following onasemnogene abeparvovec treatment. 90 Mean (range) age at treatment was 5 months (2 weeks–17 months) and mean (range) follow-up was 270 (68–482) days. For patients with SMA type 1, mean CHOP INTEND score increased from 21 at baseline to 47, and four patients (67%) were dependent on non-oral nutrition support at last follow-up.
A retrospective chart review from Qatar showed favorable results with onasemnogene abeparvovec for 11 patients (SMA type 1, n = 9; SMA type 2, n = 2) who received treatment between 12 and 22 months of age: 6 of 11 patients (55%) demonstrated safe swallowing before treatment and continued to demonstrate normal swallow function post-treatment. 91 The remaining five patients (all with SMA type 1) required nutrition support with enteral feeding before treatment and remained on enteral feeds post-treatment during the 2-year follow-up period (Table 3).
In a cohort study from the United Kingdom, 15 patients (age range at time of treatment, 2–38 months) treated with onasemnogene abeparvovec were followed for 8 to 15 months. 92 Six children fed exclusively orally before treatment, and five (83%) of them were able to continue exclusive oral feeding after treatment. Six children received nutrition support with exclusive tube feeding before treatment, and one (17%) of them received a mixture of oral and tube feedings after treatment. Of two children who received both oral and tube feedings before treatment, one received exclusive oral feeding after treatment, and the other required exclusive tube feeding (Table 3). 92
Other studies included in this SLR88,93 collected information on bulbar function-related outcomes after onasemnogene abeparvovec administration but lacked specifics on timing and methods of assessments or offered only qualitative reports of outcomes. The available data are presented in Tables 1 and 2.
Risdiplam evidence
Eleven studies were identified in this SLR that reported outcomes associated with bulbar function after initiation of risdiplam after the onset of SMA symptoms.96–106
FIREFISH was a phase 2/3, single-arm, open-label, two-part, multicenter, multinational trial evaluating risdiplam for infants aged 1 to 7 months with SMA type 1. Part 1 was a dose-finding study and included 21 infants (low-dose cohort, n = 4; high-dose cohort, n = 17; median [range] age at enrollment, 6.3 [3.3–6.9] months; median [range] CHOP INTEND score at enrollment, 24 [10–34]) .94–100 In Part 1, no infant lost the ability to swallow during the 12-month period after risdiplam initiation. At the time of evaluation, 19 infants were alive, of which 18 (94.7%) (three from the low-dose cohort and 15 in the high-dose cohort) were able to feed orally, either exclusively (n = 15) or in combination with a feeding tube (n = 3).97,99 At the 24-month assessment, 14 patients were alive. All 14 (100%) were able to swallow, and 13 were able to feed orally (exclusively, n = 12; in combination with tube feed, n = 1). 99
FIREFISH Part 2 was a confirmatory study with 41 infants (median [range] age at enrollment, 5.3 [2.2–6.9] months; median [range] CHOP INTEND score at enrollment, 22.0 [8.0–37.0]) who received risdiplam for 12 months at the dose selected in Part 1.101–103 At 12 months, 38 infants (93%) were alive, of which 95% (n = 36) maintained the ability to swallow, with 89% (n = 34) able to feed orally (exclusively, n = 28; in combination with tube feeding, n = 6). According to the swallowing assessment of the Clinical Global Impression of Change conducted at 12 months, 71% (n = 29) of the infants maintained or improved their ability compared with baseline: three infants (7%) had “very much improved” swallowing, 12 (29%) had “much improved” swallowing, four (10%) had “minimally improved” swallowing, and ten (24%) had “unchanged” swallowing. 103 Fifty-two patients who completed Parts 1 or 2 and maintained the ability to swallow were enrolled in the OLE. At 36 months, 48 of these patients were able to swallow, 34 were able to feed orally, and 30 were able to feed exclusively orally. 104
In a prospective study from Spain, six patients with SMA type 2 (17–46 years old) were followed for 12 months after treatment with risdiplam. Three patients (50%) reported improved swallowing according to the ALSFRS-R, and two (33%) demonstrated clinically meaningful improvements (>5%) in body mass index. Five patients had clinically meaningful improvements in the bulbar domain of the Egen Klassifikation 2 scale. None of the patients demonstrated clinically meaningful deterioration in any measure. 105
One additional study evaluated the use of risdiplam for nine adults with SMA type 2. The published abstract lacks details related to bulbar function endpoints but demonstrates that risdiplam was well-tolerated. The available data are provided in Table 1. 106
Combination, sequential, or unknown DMTs
Seven studies were identified in this SLR that reported outcomes associated with bulbar function after sequential or combination DMTs or compared two different DMTs,107–113 and one study was identified that evaluated unknown DMTs for SMA. 114
A study of six patients (SMA type 1 [two SMN2 copies], n = 5; SMA type 2 [three SMN2 copies], n = 1; age range 7–24 months) who received nusinersen followed by onasemnogene abeparvovec and were followed for 5 to 17 months evaluated the ability to feed orally. 107 At baseline, only one patient (20%) with SMA type 1 received nutrition support with tube feeding, and he was still dependent on tube feeding at the end of the observation period. All other children maintained the ability to feed exclusively by mouth (Table 3).
A retrospective observational study of 25 patients with presymptomatic SMA (n = 2) or symptomatic SMA type 1 (n = 21) or type 2 (n = 2) evaluated real-world outcomes after a median (IQR) of 18.0 (12.4–18.3) months following onasemnogene abeparvovec treatment. 108 Eight patients had received nusinersen, and one had received risdiplam before onasemnogene abeparvovec treatment. Three patients (12%) required nutrition support with exclusive tube feeding before onasemnogene abeparvovec treatment. At the last follow-up, one of these three patients (4%) gained the ability to feed orally. Another four patients (16%) lost the ability to feed orally (Table 3). In addition, all 10 patients (40%) who were able to speak at baseline maintained this ability. Of the 15 (60%) who were unable to speak at baseline, 10 (40%) were able to speak at follow-up and five (20%) remained unable to speak.
RWE obtained from a French national registry assessed risdiplam for 28 patients with SMA (median [range] age, 12.6 [4–20] years; SMA type 1, n = 4; SMA type 2, n = 21; SMA type 3, n = 3), the majority of whom (20/28 [71%]) had been previously treated with nusinersen.109,110 Over 1 year of follow-up, seven of 15 patients (47%) reported improved mandibular mobility, and improved speech and swallowing ability was seen in one 3-year-old child with SMA type 1. 110
A single abstract explains the preliminary outcomes observed from data collected by the RESTORE registry for patients with SMA who received DMTs either sequentially or in combination. 111 CHOP INTEND scores reportedly increased among most patients with available data (n = 8/10 [80%]) but no correlations were made with a specific DMT or a specific bulbar function outcome.
Improvement in bulbar function after DMT administration was assessed via a survey of health care providers in the United States who administered onasemnogene abeparvovec or nusinersen to patients with SMA aged 6 to 24 months. 112 Based on provider assessment, mean time to improvement in bulbar function was shorter for patients who received onasemnogene abeparvovec compared with those receiving nusinersen monotherapy. Mean (SD) time to achieving improvements was 2.4 (2.0) months for patients who received onasemnogene abeparvovec monotherapy, 4.1 (3.5) months for those who received onasemnogene abeparvovec after nusinersen, and 5.6 (3.1) months for those receiving nusinersen monotherapy. The tool used to assess improvement was not reported. 112
In a case report, an infant with symptomatic SMA type 1 was switched to onasemnogene abeparvovec at 5 months after a washout period of 3 days from risdiplam. 113 Nine months after onasemnogene abeparvovec treatment, the patient's neurocognitive and speech profile was normal. The child fed by mouth, had no swallowing problems, chewing fatigue, or drooling, and had appropriate body weight for age. 113
Lastly, a small prospective observational study included children with SMA type 1 (n = 4), SMA type 2 (n = 5), or SMA type 3 (n = 3) who were receiving either nusinersen or risdiplam and an age-matched cohort of normally developing children. 114 The study evaluated the usefulness of the IOPI in SMA. All patients with SMA exhibited lower scores than normally developing children, and a negative correlation between maximum pressure applied by the mouth and SMA type was observed. This study demonstrated the usefulness of the IOPI system for measuring outcomes related to bulbar function, but correlations with motor function and other SMA-related sequelae remain unclear.
Discussion
This SLR identified 51 studies across 83 publications that evaluated the impact of DMTs for SMA on bulbar function: 31 studies of nusinersen, 29 of onasemnogene abeparvovec, 15 of risdiplam, seven of combination or sequential DMTs, and one of unknown DMT. Common bulbar functions assessed across studies of nusinersen, onasemnogene abeparvovec, and risdiplam included the ability to feed orally, the ability to swallow liquids, and the need for non-oral nutrition support. Communication was rarely assessed.
In the publications included in this SLR, presymptomatic treatment for SMA led to the preservation of full oral nutrition for most patients. Specifically, full oral nutrition was maintained in 84% of patients treated presymptomatically with nusinersen44,45 and 100% of those treated with onasemnogene abeparvovec.72–75 Results from studies of risdiplam did not explicitly report what percentage of patients were receiving full oral nutrition, but 100% of patients maintained swallowing abilities, 96 which indicates promising results. It is unclear if the improved outcomes associated with onasemnogene abeparvovec compared with nusinersen are a result of the DMT or subclinical differences in baseline bulbar integrity, given that the nusinersen results were based on a lesser percentage of patients with three copies of SMN2 than those treated with onasemnogene abeparvovec (40% vs. 60%).9,40,41
In contrast to these uniformly promising outcomes for patients treated presymptomatically, outcomes among patients treated after the onset of SMA symptoms are more variable. Although study results from this SLR consistently demonstrate that patients treated after symptom onset tended to have poorer bulbar outcomes than those treated presymptomatically, differences in sample characteristics and reporting structure pertaining to timing of post-symptomatic treatment, bulbar involvement, and outcomes across studies impede the ability to clearly elucidate the magnitude of these differences or how they are altered by type of DMT. Specifically, outcomes after nusinersen indicated high rates of post-treatment bulbar impairment, with only 6% to 50% of patients achieving full oral nutrition at follow-up.51,52,56–59,61–63,70 This contrasts with results from patients treated with onasemnogene abeparvovec, where the majority of studies reported that 50% to 100% of patients were receiving full oral nutrition after treatment,32,37,38,79–83,91 with only a single study reporting less than half (47%) of patients receiving full oral nutrition. 92 Though fewer investigations were available examining bulbar outcomes after risdiplam, available results indicated that 57% to 89% of patients were receiving full oral intake at follow-up.99,101,103–105
When interpreting these results, it is important to note that patients treated with nusinersen generally had already experienced very severe disease progression at the time of DMT administration. For studies that reported average age at time of DMT administration, the majority of investigations examining outcomes after symptomatic nusinersen treatment included children with an average age older than 1 year.14,47,50–55,57–59,68,70,71 This differs from the majority of investigations examining outcomes after onasemnogene abeparvovec, in which the average age at time of treatment administration in most studies was 4 months or younger,32,37,38,79–84,88 and risdiplam, in which the average age at administration was often 5 to 6 months old.98–105 These differences in age at administration of DMT likely have significant implications for bulbar outcomes, as previous research on patients with untreated SMA type 1 indicates that bulbar degradation typically occurs between 5 and 12 months of age, 132 with early phases of degradation likely not causing clinical symptoms identifiable without instrumental assessment. Future investigations comparing outcomes across DMTs using validated pre- and post-physiologic and functional swallowing outcomes are critical in elucidating differences in treatment effects.
Several international multidisciplinary teams of experts in bulbar function and SMA have been assembled and engaged in series of meetings to develop recommendations for the assessment of bulbar physiology and function in SMA. To date, no guidelines have been widely accepted, and no instruments have been validated. According to the expert panels, key domains for inclusion in assessment tools for bulbar function include oral intake, facial structure and oral motor strength, swallowing, communication, airway protection, and fatigability.13,42,43 Overall, the publications included in this SLR demonstrate a lack of consistency in timing of assessments, length of follow-up, age of patients, instruments used, clinicians and experts involved in examinations, and outcomes reported. These inconsistencies highlight the need for reliable, validated, objective measures of bulbar function over time for patients with SMA as well as a multidisciplinary approach to SMA care.
Additional research is needed to facilitate the integration of these bulbar assessments into clinical care. Specifically, elucidating the relationship between a patient's motor and bulbar function is a key area for future investigation. Previous reports of correlations between motor and bulbar function in natural history patients have frequently resulted in providers foregoing bulbar assessment among patients with strong motor function. Recent reports suggest such a practice may result in many patients with bulbar deficits being missed, as motor and bulbar integrity may experience different extents of deficits and present at different rates. 16 Research is also needed to understand what changes in bulbar function are associated with disease progression and which are strictly normal age-related changes in physiology. For example, the studies that describe details of nutrition support before and after DMT administration (Table 3) indicate a clear pattern of increased use of non-oral nutrition support over time. In this review, only two of the 18 studies listed in Table 3 reported a decrease in the need for nutrition support over time, six reported no change in the need for support, and 10 reported an increase in the need for support. This need for nutrition support may not reflect a degradation in disease state but only increased nutritional needs related to growth that oral feedings alone cannot meet. In addition, outcomes that are reported in qualitative terms do not provide a complete picture of physiologic functions, such as the presence of penetration when swallowing. Likewise, the ability to feed orally is often based on clinician recommendations and parental reports, which are highly variable and can be influenced by personal experiences. Reporting validated outcomes would remove this clinical subjectivity.
The findings of this review should be interpreted cautiously due to several limitations. While we collected and present available bulbar function data for each of the currently approved DMTs, robust comparisons are not possible due to differences in assessments of bulbar function in terms of instrument used, timing of assessment, definitions of bulbar function, length of follow-up time, and training of assessor. To date, no head-to-head comparisons of the effects of DMTs on bulbar function have been conducted. Patients included in the studies also varied in terms of age at assessment, age at symptom onset (if not presymptomatic), presentation of SMA, and number of SMN2 copies. As such, comparisons among studies must be interpreted cautiously, as direct comparisons are not possible without a better understanding of the patient populations being evaluated.
Larger prospective studies using standardized and age-based assessments with longer follow-up periods are needed to assess the clinical stability of bulbar function for patients with SMA who receive DMTs. Clinicians who treat patients with SMA must consider all components of bulbar function as part of the overall treatment plan.10,12 While components of bulbar function in SMA are routinely evaluated in clinical practice, no standard, comprehensive definition of bulbar function exists, and bulbar function endpoints are not consistently measured with objective scales specific for SMA. A comprehensive definition of bulbar function has been put forth by a consensus of multidisciplinary experts,13,43 but it has not yet been widely applied. The definition of and vocabulary related to bulbar function, as well as scales and tools to measure bulbar function for patients with SMA, should be standardized and applied consistently to guide clinical decision-making and improve outcomes. Broadly, standardization and consistency of motor function assessment should be applied to other neuromuscular disorders that are treated with gene therapy. Onset and duration of DMTs, disease-specific considerations, age-related physiology, and individual variations in presentation impact disease progression and must be considered by the multidisciplinary care team.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602241303373 - Supplemental material for Systematic literature review of the impact of spinal muscular atrophy therapies on bulbar function
Supplemental material, sj-docx-1-jnd-10.1177_22143602241303373 for Systematic literature review of the impact of spinal muscular atrophy therapies on bulbar function by Katlyn McGrattan, Katie Walsh, Lesa Mehl , Simarjeet Kaur and Keith W Dilly in Journal of Neuromuscular Diseases
Supplemental Material
sj-docx-2-jnd-10.1177_22143602241303373 - Supplemental material for Systematic literature review of the impact of spinal muscular atrophy therapies on bulbar function
Supplemental material, sj-docx-2-jnd-10.1177_22143602241303373 for Systematic literature review of the impact of spinal muscular atrophy therapies on bulbar function by Katlyn McGrattan, Katie Walsh, Lesa Mehl , Simarjeet Kaur and Keith W Dilly in Journal of Neuromuscular Diseases
Footnotes
Abbreviations
Acknowledgments
Medical writing and editorial support were provided by Jennifer Gibson, PhD, of Kay Square Scientific, Newtown Square, PA, USA. This support was funded by Novartis Gene Therapies, Inc. All article processing fees/open access fees were provided by Novartis Gene Therapies, Inc.
Ethics statement
This article is a systematic literature review based on published articles, and as such, does not require institutional review board approval.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This analysis was funded by Novartis Gene Therapies, Inc. The study sponsor contributed to the design and conduct of the study, and collection, management, analysis, and interpretation of the data.
Declaration of conflicting interests
Data availability
The data summarized from this review are from published articles and are publicly available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
