Abstract
Background
Spinal muscular atrophy is a rare, genetic neuromuscular disorder. Disease-modifying therapies, when administered early, have shown improved outcomes, leading to the implementation of numerous newborn screening programs for spinal muscular atrophy.
Objective
The aim of this study was to evaluate the progress in implementing newborn screening for spinal muscular atrophy and therapy accessibility worldwide, after the first paper published in 2021.
Methods
We conducted a survey, contacted experts from 143 countries worldwide, gathered responses from 86 experts from 80 countries.
Results
By 2023, 31 countries reported established programs, 33 in the beginning of the year 2024; identifying approximately 1176 cases of spinal muscular atrophy. Additionally, the availability of disease-modifying therapies has expanded. At least one therapy is now accessible in 62 countries. Challenges, such as lack of governmental support, resource constraints, and varying healthcare priorities continue to impede implementation in some countries.
Conclusions
The data shows a significant increase in the implementation of newborn screening programs since 2021. The experts are still expressing a strong need for equitable access to standard of care for all the patients globally. Despite all setbacks, collaborative efforts have played a crucial role in newborn screening for spinal muscular atrophy implementation and currently 7% of world newborns are being screened, projections indicate an estimated 18% screening rate by 2028.
Keywords
Introduction and context
Spinal muscular atrophy (SMA) associated with 5q is an autosomal recessive disease caused by lack of functional survival motor neuron (SMN) protein. Recent meta-analysis of newborn screening has estimated the incidence of SMA at 1 in 14,848 births worldwide. 1 Three disease modifying therapies, nusinersen, onasemnogene abeparvovec, and risdiplam, dramatically change the natural history of the disease.2,3 All data so far demonstrate that efficacy of these treatments is highly dependent on the timing of administration. Optimally, treatment should be initiated before the patient exhibits symptoms of the disease; treatment initiation before symptom onset results not only in better patient outcomes but also in significant cost savings.4–8 Therefore, several newborn screening (NBS) programs for SMA have been established in recent years. 9
In 2021, we conducted a survey of 87 experts from 82 countries to evaluate the status of NBS for SMA worldwide and the likely evolution in the coming years. 9 Only one country (Taiwan) had a SMA NBS program that screened all newborns. Eight countries had SMA NBS programs that screened some newborns (USA, Germany, Belgium, Australia, Italy, Russia, Canada, and Japan). At that time, less than 2% of newborns worldwide were screened for SMA. Thirty-seven respondents from countries without SMA NBS programs reported plans to establish SMA NBS in their countries, and since then reports describing pilot programs or national programs have been published.10–14 The aim of this study was to evaluate progress in implementation and execution of SMA NBS since 2021. We also aimed to identify the obstacles and challenges faced during implementation and maintenance of SMA NBS and to determine whether the COVID-19 pandemic disrupt plans for NBS implementation as it globally disrupted patient care. 15
Methods
We used the same method used in our first survey 9 but adapted the questionnaire to better address progress in SMA NBS implementation and maintenance (Appendix 1). In addition to the experts who responded to the first survey, we sent the questionnaire to others identified through the website of the World Federation of Neurology. 16 The number of newborns in the world was calculated using data from the United Nations. 17
We conducted comparisons between two groups using an unpaired t-test, setting the threshold for statistical significance at p < 0.05. The statistical analysis and graphical representations were performed using IBM's SPSS Statistics software (version 27) and GraphPad Prism (version 10).
Results
Survey respondents
The questionnaire was sent to experts in 143 countries (74% of countries identified by United Nations). These were the countries in which we were able to identify an expert. Responses were obtained from 86 experts from 80 different countries (55% of contacted countries) on six continents. Experts from almost all European countries and from 80% of countries with high income responded to the survey. 18 The response rate was similar to that in our first project in 2021. 9
Of the 86 respondents, 53 identified themselves as pediatric neurologists, 10 as adult neurologists, 13 as geneticists, 10 as researchers, 5 as newborn screening specialists, 8 as pediatricians, 2 as pediatrics neurology fellows, 1 as a patient advocacy group member, 1 as a pediatric pulmonologist, and 1 as a policy maker and project manager (multiple options possible). Three independent respondents provided data for three separate regions of China (Hong Kong Special Administrative Region, Taiwan and mainland, and the Peoples’ Republic of China) and they are presented as separate countries. For six countries (Colombia, Romania, Serbia, Norway, Iceland and Tanzania) two independent respondents provided the data for the whole country, the data were combined for the analysis. Three respondents did not complete the survey, their partial answers were included in the results section below.
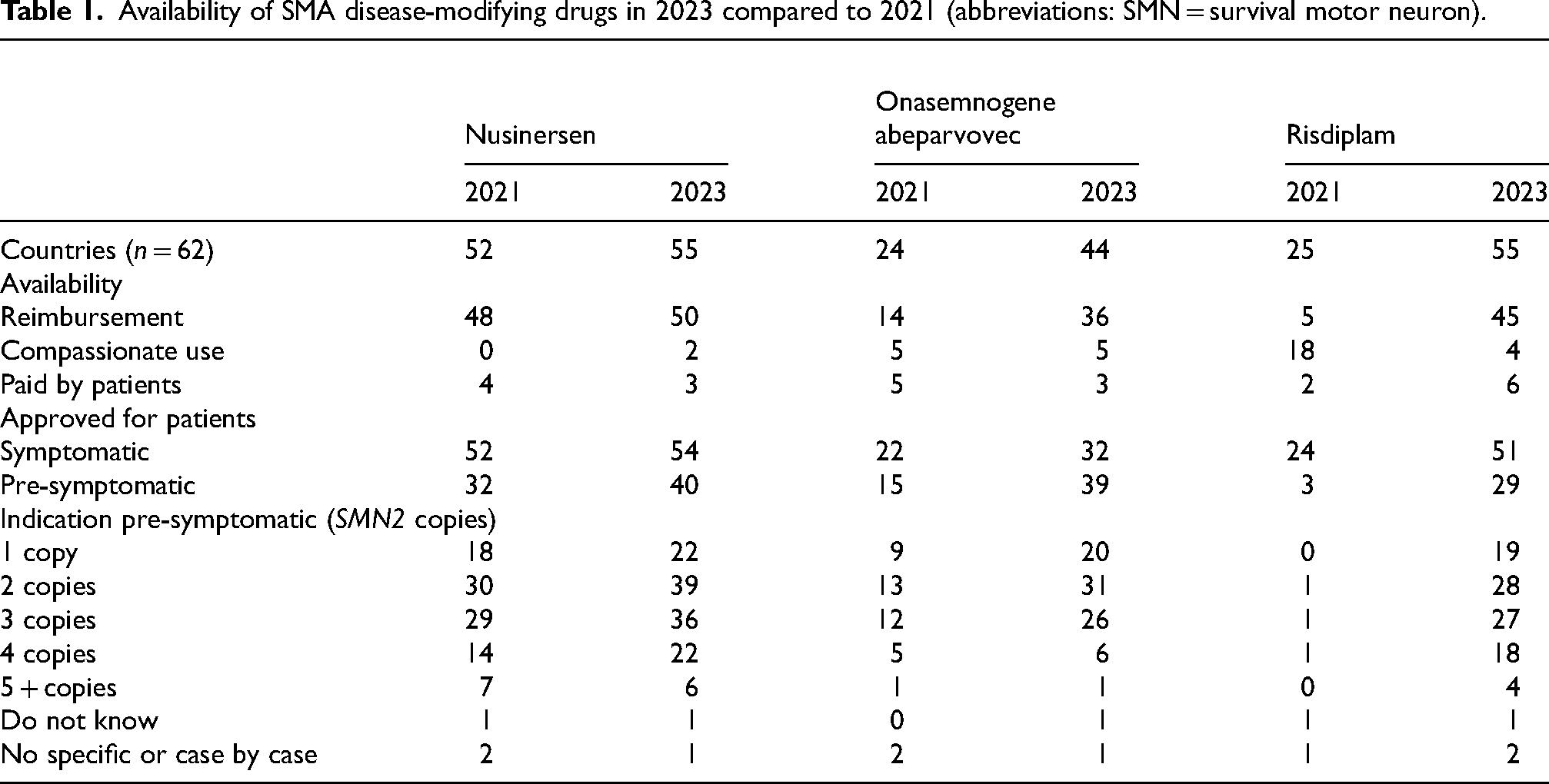
Availability of SMA disease-modifying therapy
The number of countries in which nusinersen, onasemnogene abeparvovec or/and risdiplam were available in 2021 9 and 2023 are compared in Table 1 and presented in Figure 1. As of 2023, our survey indicated that one or more drugs have been approved for treatment of SMA in 62 countries: 39 countries provide three treatment options, 14 countries offer two, 9 countries have one treatment option, and no treatment options are available in 18 countries. Differences may exist in insurance coverage across states or parts of the country, the following data reflecting the prevailing situation regarding treatment availability at the time of the survey. In some cases, treatments were either donated or covered by pharmaceutical companies, while some respondents indicated their availability within their respective countries.
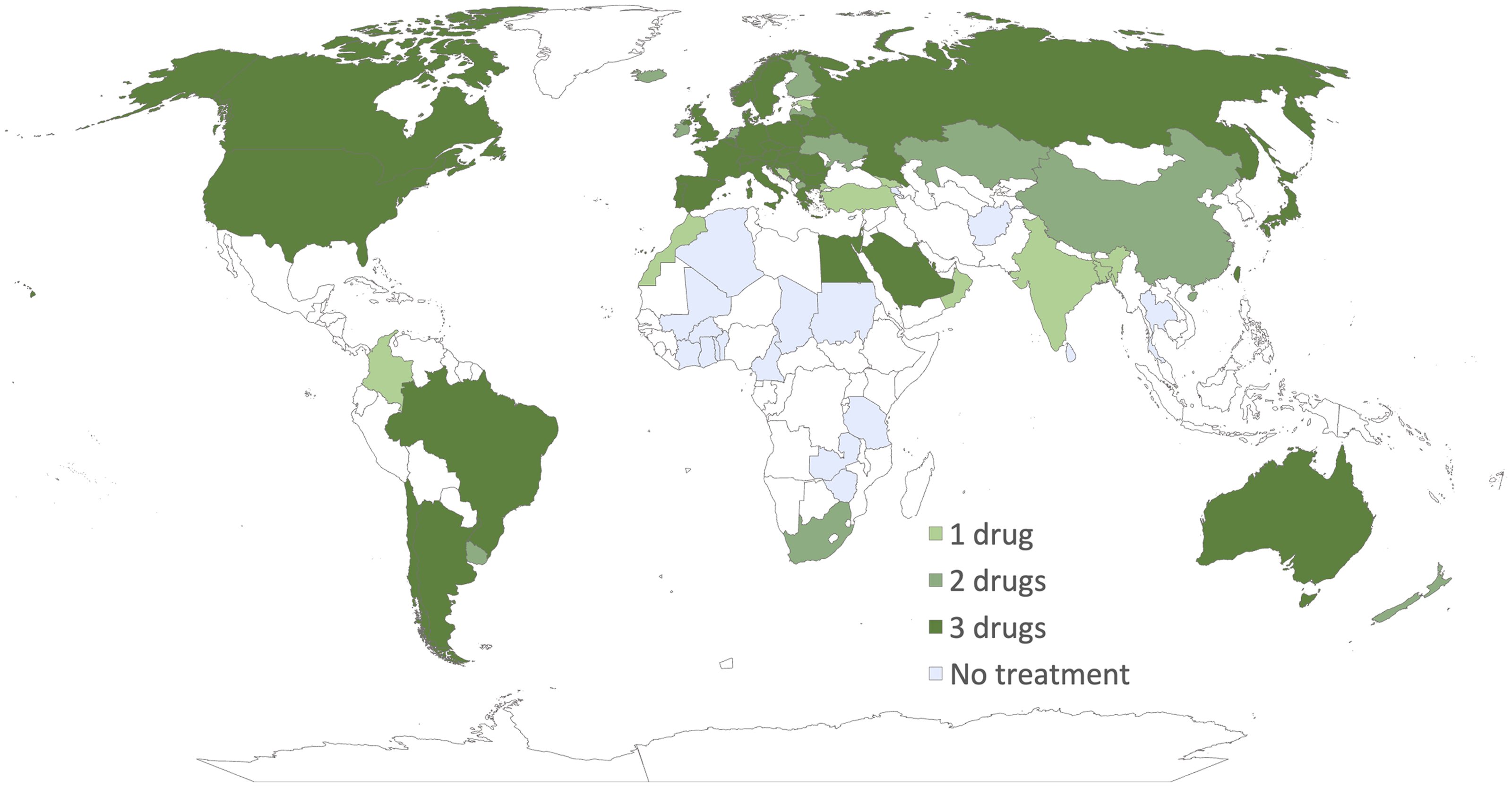
Availability of treatments around the world. White-marked countries indicate no available data.
Availability of SMA disease-modifying drugs in 2023 compared to 2021 (abbreviations: SMN = survival motor neuron).
Countries with SMA NBS
In 2023, experts from 31 countries reported that SMA NBS had been implemented in their country (Table 2 and Figure 2), 22 countries more than in 2021. In 2021 only Taiwan screened all newborns, whereas Belgium, the USA, Germany, Australia, Italy, Russia, Canada, and Japan screened only some newborns. The number of countries with implemented SMA NBS programs has increased each year since 2014 (Figure 3(a)) as have the number of newborns diagnosed with SMA through SMA NBS (Figure 3(b)). As per the respondents’ reports, by 2023, a total of 1,176 newborns with SMA had been identified through SMA NBS.
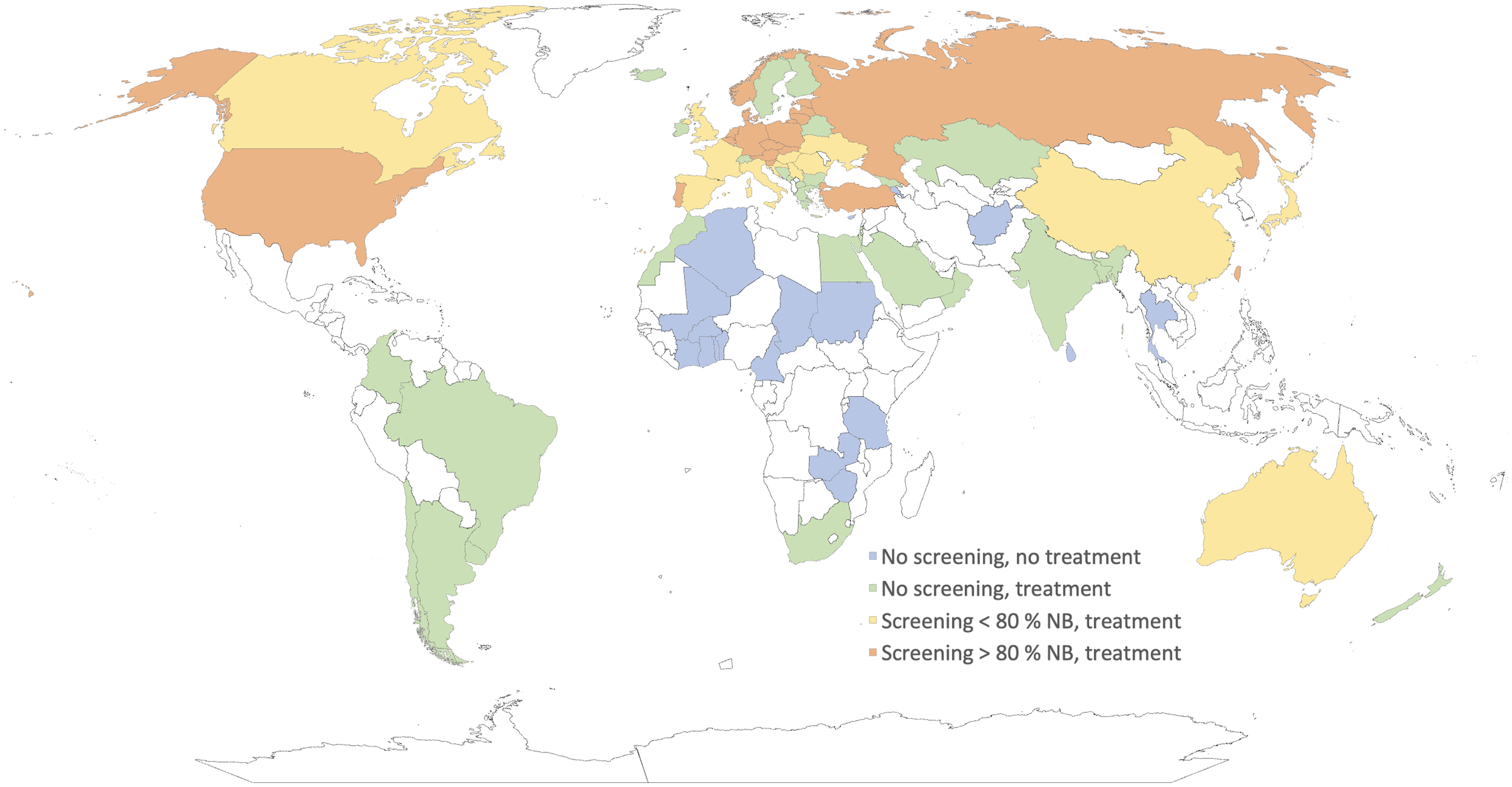
Availability of SMA treatments and SMA NBS around the world (categorized by countries screening below and above 80% of newborns). White-marked countries indicate no available data. NB = newborns.
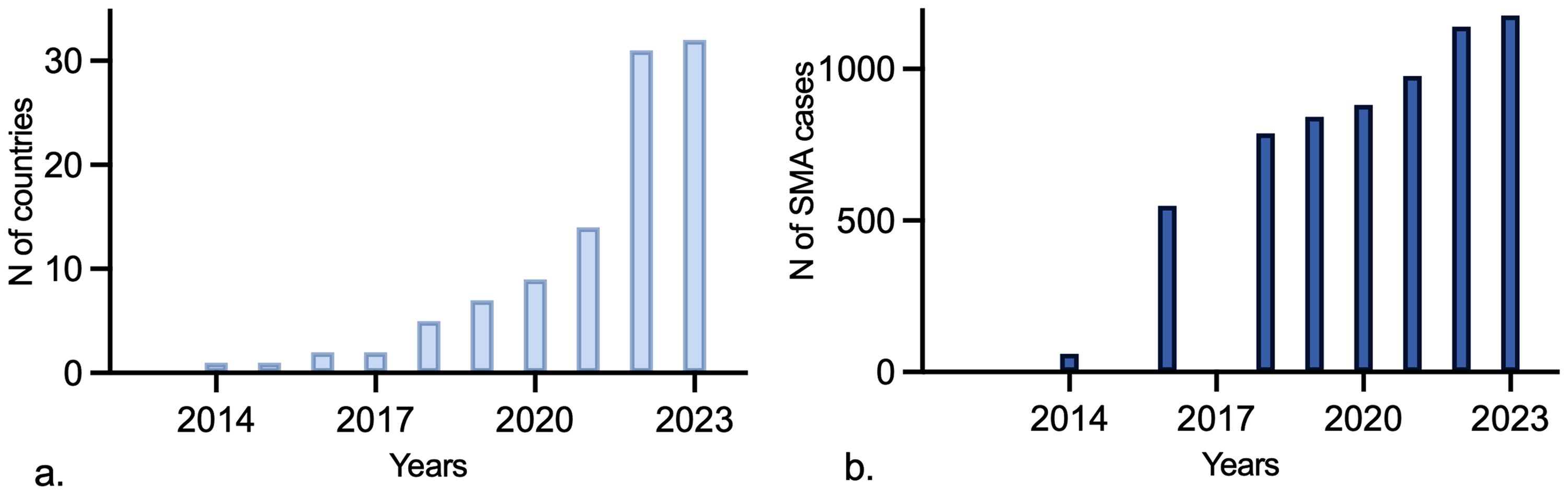
A: Number of countries with SMA NBS programs by year. b: Number of SMA cases identified by SMA NBS worldwide by year.
Summary of SMA NBS programs in countries by alphabetical order. The number of cases, false positives and negatives represent data up to the year of 2023. W: whole country; P: part of country; NB: newborns, Pilot: pilot program; Official: official program; Cases: number of SMA cases identified; pos: positive; neg: negative; Acad: academical funds/grants; P: funded by parents; G: funded by government; HI: funded by health insurance; Ph: funded by pharmaceutical company; Reg: Regional health funds; Char: Charity Foundation; US NBS: usual NBS laboratory; Gen lab: genetic laboratory; qPCR: quantitative polymerase chain reaction; ddPCR: droplet digital polymerase chain reaction; MLPA: multiplexed ligation-dependent probe amplification; var methods: various methods; NK: not known; HKSAR: Hong Kong Special Administrative Region.
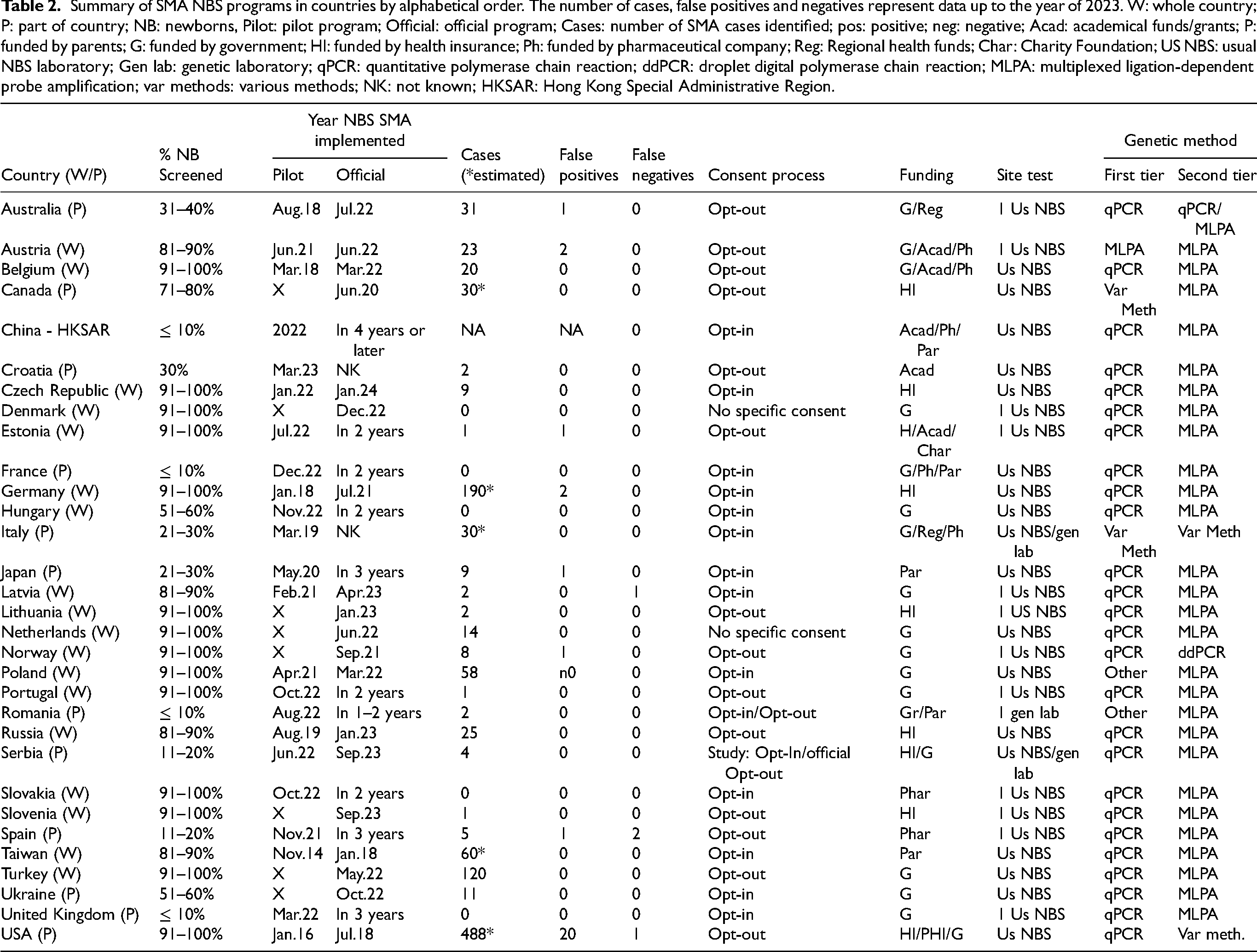
Table 2 presents a comprehensive list of countries with implemented SMA NBS. Further elucidation for select countries is provided herewith: In Belgium, the initial funding for SMA NBS stemmed from Academia and Pharmaceutical entities during the pilot phase. However, subsequent to its official implementation, governmental funding now sustains the program. Canada exhibits significant disparities in SMA NBS implementation among its provinces. This discrepancy arises due to healthcare decisions, including drug reimbursement and newborn screening, being autonomously administered at the provincial level rather than at a federal level. The Czech Republic initiated its pilot SMA NBS program in March 2023, successfully screening over 12,000 newborns since its inception.
By the process of publishing this paper, Luxembourg and Switzerland started with SMA NBS in the first half of 2024, and Canada and Serbia now screen their entire populations.
The experts surveyed noted that the following factors positively influenced the maintenance and expansion of the SMA NBS programs: (i) patient associations/organizations (n = 7), (ii) cooperation on different levels (n = 7), (iii) shared experiences by other states or countries (n = 6), (iv) pilot studies and already established screening for metabolic diseases (n = 4), (v) support by pharmaceutical or private companies (n = 4), (vi) access to disease-modifying therapies (n = 3), (vii) economic analyses (n = 2), (viii) media coverage (n = 1), and (ix) government support (n = 1). The negative influences and challenges mentioned were (i) lack of cooperation with the authorities (n = 6), (ii) lack of human resources (n = 3), (ii) bureaucracy (n = 4), (iii) fear of genetic screening and academic conflicts (n = 2), (iv) financial limitations (n = 2), (v) lack of awareness about SMA (n = 1), and (vi) inequality between provinces (n = 1). Thirteen of the respondents thought that the COVID-19 pandemic did not influence the maintenance or implementation of the SMA NBS programs, whereas 8 respondents thought that the pandemic had a negative influence.
The methods used in the initial SMA screen in NBS programs in the countries surveyed are quantitative polymerase chain reaction (qPCR), which is used by 27 countries, high-resolution melt analysis PCR, which is used by two countries and various states or regions in Italy and Canada, and multiplexed ligation-dependent probe amplification (MLPA) and droplet digital PCR (ddPCR), both used by one country. MLPA is used as a second-tier confirmatory screen by most countries (n = 26).
Based on the global number of births estimated by the United Nations for 2021 (approximately 130 million), around 7% of newborns worldwide, roughly 10 million, are currently being screened for SMA. Countries that have initiated pilot screening programs aim to double the number of newborns screened within 5 years.
Countries without NBS for SMA
Experts in 17 of the 45 countries without current SMA NBS programs expect that such a program could potentially be implemented. No plans were reported by experts from 24 countries, and 4 were uncertain about the future implementation of these programs in their respective countries. In countries where SMA NBS has not yet been implemented, respondents to the question of how important they thought it was to introduce SMA NBS in their country was 85 on average on a scale of 0 (not important) to 100 (extremely important). The perception of the importance was lower in experts from countries where no NBS is planned in comparison with those from countries where NBS is planned (rating of 77 vs. 95, p < 0.01). Table 3 summarizes the reasons experts gave for not having a plan to implement SMA NBS and the barriers to current implementation in countries that have plans to implement SMA NBS. The reasons most often cited were “lack of financial resources” and “not a healthcare priority in our country”. In countries with plan for implementation, organizational issues are the biggest foreseen obstacle. Other reasons given included lack of human resources and governmental support. In Thailand, carrier screening has been proposed as an alternative plan to NBS, and in Israel carrier screening has already been implemented instead of NBS.
Obstacles to implementation of SMA NBS in countries without a plan and in countries who have a plan to implement the screening.

Sixteen experts, who are all part of the SMA NBS World Study Group, have planned to introduce SMA NBS in their countries since the first meeting of the Study Group in 2021, but have not been successful. The obstacles reported by these experts were (i) no governmental support (n = 5), (ii) no treatments yet available for SMA (n = 2), (iii) logistic and organizational problems (n = 2), (iv) lack of financial resources (n = 2), (v) lack of human resources (n = 2), (vi) carrier screening already established (n = 1), (vii) underestimation of work needed for SMA NBS implementation (n = 1), and (viii) COVID-19 pandemic (n = 1). The experts surveyed stated that a number of measures can help countries with no SMA NBS establish this screen (Table 4). The measure most often selected was the collection of economic data and a cost-benefit analysis.
The measures that can be helpful for countries for establishing SMA NBS.

Experts from seven countries expect that SMA NBS will be implemented within 1 year (Iceland, Sweden, the rest of China, Kuwait, Luxembourg, Qatar, and Switzerland). Switzerland and Luxembourg already started in 2024. Experts from eight countries expect that this will occur within 2 years (Brazil, Macedonia, Oman, Finland, Georgia, the Republic of Ireland, New Zealand, and Bulgaria). The expert from Israel expects that SMA NBS will be implemented there within 3 years, and the experts from Colombia and Egypt estimate 4 years or longer. These predictions are shown graphically in Figure 4. Eleven experts estimate that a pilot program will first be initiated, and 10 experts think that no pilot will precede the official screening program. According to these projections, 18% of the newborns worldwide will be screened for SMA in 2028. Figure 5 illustrates the difference between the projection that we established in 2021 and the actual evolution of SMA NBS programs.
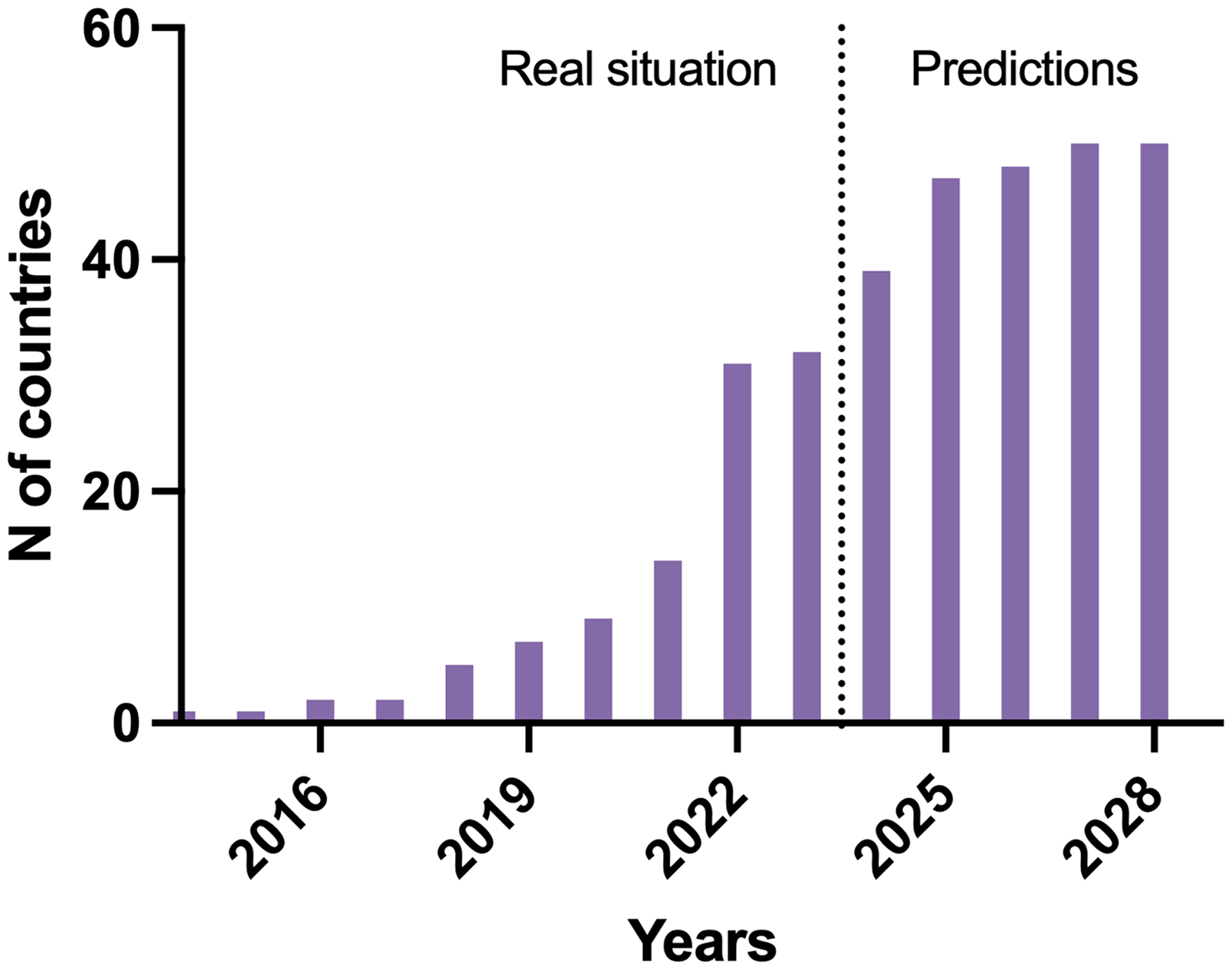
The number of countries with SMA NBS and additional predictions for future years.
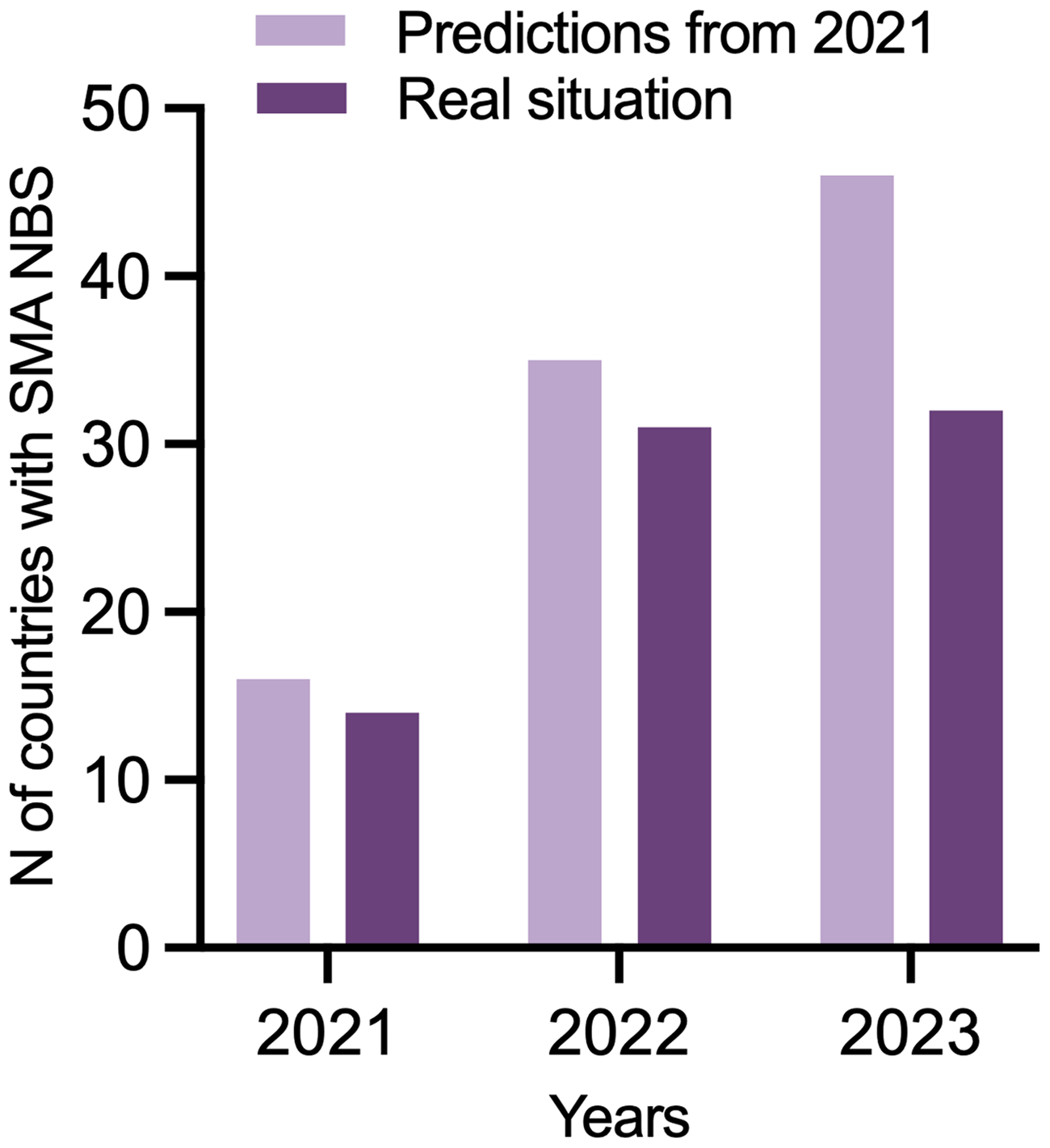
The predictions for SMA NBS implementation from 2021 versus the number of countries with SMA NBS in 2023.
When asked what test will be used in first-tier SMA NBS, 11 respondents predicted that qPCR will be used, 2 predicted that MLPA in 2 will be used, 1 that ddPCR will be used, and 4 were not sure. Ethical committee approval or approval by the Health and Social Ministry specific to the SMA NBS is required in 6 countries, and no ethical committee approval is needed to establish SMA NBS in 7 countries. Two respondents did not specify what will be needed.
Carrier screening and other genetic NBS programs
SMA carrier screening is available for all who request it (covered by health insurance) in three countries where SMA NBS is also implemented. In five countries, carrier screening is available for a limited number of parents (covered by health insurance). Parents can pay for carrier screening in five countries. One respondent did not know whether there was carrier screening available in their country, and in one country there is no carrier screening but genetic counseling is offered for families who have a risk of having a child with SMA.
In 60 countries, metabolic/endocrine NBS is performed, and in 9 countries there is no NBS for any diseases. In Belgium, there is an opt-in pilot NBS program for 165 genetic diseases, 19 and in Italy, a pilot genetic screening has been implemented.
Discussion
Our survey of 86 experts from 80 different countries indicates that disease-modifying treatments for SMA and SMA NBS have become more widely available compared to 2021. The number of countries that have implemented SMA NBS has more than tripled in the last 2 years (31 countries in 2023, 33 in 2024 vs. 9 in 2021). The results of the first survey in 2021 predicted that in two years (2023) there would be 24 countries where at least half of the newborns would be screened. 9 Currently, 21 countries have implemented SMA NBS with at least half of the newborns in countries with SMA NBS programs screened on average. Therefore, although our predictions overestimated the speed of the SMA NBS implementation, the actual numbers do not lag far behind our predictions. This is encouraging, especially since the COVID-19 pandemic disrupted the implementation of SMA NBS in several countries. Based on our data, 7% of the world's population of newborns is currently being screened for SMA compared to 2% in 2021. Projecting 5 years in the future, 18% of newborns will be screened for SMA in 2028.
Many experts stated that joint efforts with patient advocacy groups helped to establish SMA NBS in their respective countries, evidence that patient organizations are important collaborators when it comes to improving the lives of patients with rare diseases. 20 In Europe, the SMA NBS Alliance founded by SMA Europe called for all European countries to offer a test for SMA to all newborns by 2025. 21 The SMA Screening Alliance in the UK has also been instrumental in the development of an “evaluation in service program” or SMA NBS.
The experts who responded to our survey reported several obstacles to the implementation and maintenance of SMA NBS; obstacles were similar to those reported in 2021. The experts from countries that do not have a plan to establish SMA NBS mostly mentioned the lack of financial resources, which was not unexpected in some low- or middle-income countries. The challenge of rare disease management in these countries is broadly recognized.22,23 Lack of information and adequate training is also an obstacle. Although many experts mentioned the absence of international consensus or health economic data as an obstacle to the NBS implementation, a consensus actually exists, and several health economic studies from different countries have now been published. For example, 5-year follow-up data from the NURTURE study have been published.24–27 Governmental support is frequently reported as an unmet need and the importance of political willingness has been underlined in several US States and Australia.13,28 That implementation is possible even under difficult circumstances was demonstrated in Ukraine, where SMA NBS was implemented in 2022 while the country was at war. 29
SMA NBS constitutes one of the most reliable NBS programs. Only 29 false-positive cases have been reported of approximately 10 million total tests per year, most of them in the USA where MLPA is not conducted on the dried blood spot to confirm PCR results. Of these, 50% were described previously. 9 As approximately 1176 positive cases have been identified, this gives a positive predictive value of 97%, which is remarkable for such a rare condition. True false negative cases have been reported in the USA. 30 The methods used in the SMA NBS do not detect patients with heterozygous mutations, thus patients with deletions of one allele and point mutations are not considered as false negatives. Only some programs report these patients (Belgium, Spain, and Taiwan), which is facilitated by healthcare organisation in these countries. It must be noted that these patients may be identified several years after the end of a NBS data collection- so that their true prevalence could be difficult to estimate. As mentioned, 5% of patients have a single allelic deletion combined with a point mutation, which cannot be detected with the current methodology used in SMA NBS. It is crucial to continue promoting SMA awareness and the limitations of SMA NBS to ensure that cases with clinical manifestations of SMA are not overlooked and are properly reported.
Many screening projects incorporate other metabolic/endocrine diseases other than SMA in their NBS screening panels, most commonly severe combined immunodeficiency and cystic fibrosis. Increasingly, molecular genetic analyses are also being conducted in NBS programs.31–34 In Liege, Belgium, the “Baby Detect” program screens for more than 165 genetic diseases in the newborns from a few drops of blood taken after birth. 35 Taiwan added Duchenne muscular dystrophy to the national screening program in 2021 and have already screened more than 50,000 newborns. 36
Our study has several limitations. First, the progress on SMA NBS implementation in the last year has been rapid, and this fast-moving target will probably be imperfectly described at the time of publication. We have tried to obtain as much up-to-date information as possible from respondents and also to get predictions for the coming years. A second limitation comes from the diverse backgrounds of the respondents. We were unable to identify experts from 50 countries, but it is highly unlikely that SMA NBS has been implemented in these countries. Finally- the survey does not include questions on the countries specific NBS organisation, turn over and costs.
Conclusion
The progress of implementation of SMA NBS was slightly slower than predicted in 2021. Nevertheless, we estimate that 7% of the world newborns are currently being screened for SMA and a realistic expectation is that about 14% will be screened by 2028. Efforts for equitable access to standard of care, NBS, and disease-modifying therapy should be pursued by all stakeholders.
SMA NBS world study group
The SMA NBS World Study Group is composed of academics from the following countries and regions, listed alphabetically by family name:
Mina Abbasi (Sweden), Thierry Adoukonou (Benin), Madara Auzenbaha (Latvia), Patricia Balazova (Slovakia), Nina Barisic (Croatia), Fahad Bashiri (Saudi Arabia), laila Bastaki (Kuwait), Tawfeg BenOmran (Qatar), Guenther Bernert (Austria), Patricia Borde (Luxembourg), Peter Born (Denmark), Nina Butoianu (Romania), Claudia Castiglioni (Chile), Feriha Catibusic (Bosnia Herzegovina), Sophelia Chan (China), YinHsiu Chien (Taiwan), Kyproula Christodoulou (Cyprus), Milda Dambrauskiene (Lithuania), Tamara Dangouloff (Belgium), Donniphat Dejsuphong (Thailand), Marieke Dekker (Tanzania), Gouba Elodie (Burkina), Michelle Farrar (Australia), Monika Gos (Poland), Herve Guinhouya (Togo), Jana Haberlova (Czech Republic), Charles Hammond (Ghana), Beatrice Ilomo (Tanzania), Pirjo Isohanni (Finland), Nelica Ivanovic Radovic (Montenegro), David Jacquier (Switzerland), Bosanka Jocic Jakubi (Oman, Serbia), Gwendoline Kandawasvika (Zimbabwe), Kaoutar Khabbache (Morocco), Jan Kirschner (Germany), Sherry Kodsy (Egypt), Natella Kostandyan (Armenia), Angeeric Kouameassouan (Ivory Coast), Ruzica Kravljanac (Serbia), Vincent Laugel (France), Ivan Litvinenko (Bulgaria), Sigurdur Marelsson (Iceland), Hugh McMillan (Canada), Sandra Mesa (Colombia), Inaam Mohamed (Sudan), Sayed Hamid Mousavi (Afghanistan), Elena Neagu (Romania), Yoram Nevo (Israel), Seraphin Nguefack (Cameroon), Gina O’Grady (New Zealand), Nataliia Olkhovych (Ukraine), Declan O’Rourke (Republic of Ireland), Damjan Osredkar (Slovenia), Katrin Ounap (Estonia), Dimitri Poddighe (Kazakhstan), Juan Prieto (Colombia), Vanessa Romanelli (Brazil), Narayan Saha (Bangladesh), Kayoko Saito (Japan), Foksouna Sakadi (Chad), Modibo Sangare (Mali), Manuela Santos (Portugal), Mary Schroth (United States), Laurent Servais (United Kingdom), Leanid Shalkevich (Belarus), Aspazija Sofijanova (Macedonia), Asbjoorg Stray-Pedersen (Norway), Renu Suthar (India), Lena Szabo (Hungary), Nana Tatishvili (Georgia), Meriem Tazir (Algeria), Brynja Thorarinsdottir (Iceland), Danilo Tiziano (Italy), Eduardo Tizzano (Spain), Haluk Topaloglu (Turkey), Ludo Van der Pol (Netherlands), Gabriel Vazquez (Argentina), Dmitry Vlodavets (Russian Federation), Sean Wallace (Norway), Jithangi Wanigasinghe (Sri Lanka), Jo Wilmshurst (South Africa), Hui Xiong (China), Diana Yorio (Uruguay), Dimitrios Zafeiriou (Greece), Stanley Zimba (Zambia).
Footnotes
Abbreviations
Acknowledgements
The authors thank Jacky Wyatt for language editing.
Data availability statement
The data supporting the findings of this paper are available on reasonable request from the corresponding authors. The data are not publicly available due to privacy reasons.
Funding
EV has served as a consultant for Roche and Novartis. The work by EV was partly funded by the European Union's Horizon 2020 research and innovation program under the EJP RD COFUND-EJP N° 825575.
TD has given lectures sponsored by Biogen and Roche.
DO has given lectures and has served as a consultant for Biogen, Novartis Gene Therapies (formerly AveXis), PTC, Medis and Roche.
LS has given lectures and has served as a consultant for Roche, Biogen, Novartis, BioHaven, Zentech, Scholar Rock, and Illumina.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Correction (April 2025):
The article has been updated to correct the name of a member of the ‘the SMA NBS World Study Group’ working group from “Sayedhamid Mousavi” to “Sayed Hamid Mousavi”.
