Abstract
Background
Three medications have been approved for spinal muscular atrophy (SMA) treatment. No head-to-head clinical trials have directly compared the efficacy of nusinersen and risdiplam. We compare the efficacy of them in Type 2 and 3 SMA patients, with 6 months of follow-up.
Methods
A multicenter cohort study was conducted. Demographic, genetic and clinical findings containing Hammersmith Functional Motor Scale Expanded (HFMSE) and revised upper limb module (RULM) scales were gathered. An increase of at least 3 points in HFMSE and RULM scores was considered a positive response.
Results
73 (58.4%) children received risdiplam, and 52 (41.6%) received nusinersen non-randomly, based on clinical decision. The difference in HFMSE and RULM scores compared to the baseline was significant in both groups (P-value <0.001). However, there was no significant difference between mean difference changes in HFMSE and RULM scores between the two groups. 80.4% of patients in the risdiplam group and 72% in the nusinersen group achieved the 3-point cutoff after 6 months, and there is no significant difference between the two groups (P-Value:0.33).
Conclusions
This study showed that both medications significantly changed the HFSME and RULM after 3 and 6 months of follow-up. However, there was no significant difference between the two drugs according to the HFSME.
Introduction
5q-linked spinal muscular atrophy (SMA) is an inherited neuromuscular disorder characterized by the degeneration of alpha motor neuron cells in the anterior horn of the spinal cord.1–3 This degeneration leads to progressive hypotonia and muscular weakness. SMA is the second most common autosomal recessive disorder, with an incidence rate of one in 10,000 live births on average. 4 In most cases, a homozygous deletion within the survival motor neuron 1 (SMN1) gene is confirmed and can be used as a sensitive diagnostic test. 5 The clinical phenotype of SMA is categorized into three types based on the age of onset and the highest motor function achievement. 6 However, these categories are seen more as a spectrum than distinct subtypes. Additionally, there is a Type IV for adult-onset cases and a Type 0 for patients with prenatal onset, resulting in death within weeks. 6 The severity of SMA is in part determined by the number of SMN2 copies which give rise to a small percentage of functional SMN protein compensating for the lack of SMN1. 7
To date, three treatment options have been approved by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) 8 : Nusinersen, onasemnogene abeparvovec, and risdiplam. Nusinersen and risdiplam were approved and available in 2017 and in 2020 respectively. Other medications are yet in the pipeline and are currently being investigated. Although nusinersen and risdiplam have some differences, they are approved for type 2 and 3 SMA treatment. 8 onasemnogene abeparvovec is currently limited to infants and not considered for older and heavier children. 9 Risdiplam (EVRYSDI®) is administered orally in liquid form and is an SMN2 splicing modifier, whereas Nusinersen (SPINRAZA®) is an SMN2-targeting antisense oligonucleotide administered through intrathecal injection. 10 Due to their distinct mechanisms of action, they may have varying effects. Risdiplam exhibits a broader systemic effect on other organs, while nusinersen primarily targets the central nervous system.11,12 The availability of two medications for children with type 2 and 3 SMA raises the question of choosing the most appropriate treatment for these patients. Most studies comparing these two medications have been conducted indirectly through systematic review and meta-analysis,8,10 as no head-to-head clinical trials have directly compared the efficacy of nusinersen and risdiplam. None of these indirect studies could conclusively determine the superiority of one medication over the other in type 2 and 3 SMA, primarily due to substantial differences in study populations and unadjusted confounding factors. However, in type 1 SMA, risdipalm has shown better efficacy with fewer adverse side events.8,10 Therefore, we conducted a prospective cohort study using real-world data to compare the efficacy of risdiplam and nusinersen in Type 2 and 3 SMA patients aged 2 to 10 years, based on the HFMSE and RULM scale after 6 Months of follow-up.
Materials and methods
Study oversight
This study was conducted as a multicenter cohort study based on Iranian Registry of SMA (IRSMA), 13 with a three and six-month follow-up. All patients with SMA (Spinal Muscular Atrophy) with an indication for at least one of two drugs entered this study. The study was approved by the Ministry of Health ethics committee in Iran and conducted following the principles of the Helsinki Declaration (IR.NREC.1401.005). Additionally, all parents of participating patients signed an informed consent form indicating their participation in the study per the approved protocol and all instructions and follow-ups. Data collection was performed by 21 academic centers across Iran, considering the prevalence of SMA patients. All authors ensure the completeness and accuracy of the data and adherence to the study protocol.
Patients
All registered patients with a confirmed genetic diagnosis (deletion of both SMN1 copies) and having one to four copies of SMN2, aged between two to ten years, were enrolled in this study. The exclusion criteria included prior treatment with other SMA drugs in the last four months, undergoing stem cell therapy, continuous ventilation requirement (more than 16 h per day in the past 21 days without infection history), the presence of a severe and life-threatening condition, severe acute physical problems within the last month (e.g., severe anemia or cachexia), other concomitant genetic disorders, recent fever causing difficulties in patient evaluation, the use of oral salbutamol or another beta-2 adrenergic agonist in the last six months, recent treatment with chloroquine, hydroxychloroquine, retigabine, vigabatrin, thioridazine, riluzole, carnitine, creatine, sodium phenylbutyrate, valproate, or hydroxyurea within the last three months, a history of bacterial meningitis, and non-compliance of parents for regular visits and implementation of rehabilitation protocols as per the given instructions.
Study procedures
All patients were divided into two categories based on the following criteria: The first category included patients who were unable to take one of the two drugs, nusinersen and risdiplam, for various reasons, such as severe scoliosis without a suitable area for intrathecal injection or those with anesthesia contra indications who received risdiplam. Patients with swallowing difficulties without a percutaneous endoscopic gastrostomy (PEG) were placed on nusinersen. These patients were placed in their respective groups, and all their information was collected. The second category included patients with no specific criteria for allocating both drugs. After providing complete explanations to parents about the prescription, follow-ups, and potential side effects, the choice of treatment was left to the patient.
Prescription of medications protocol is as follows: In the risdiplam group: Above two years old and weighing less than 20 kilograms, 0.25 milligrams per Kg daily, and above two years old and weighing over 20 kilograms, 5 milligrams daily. In the nusinersen group, Intrathecal injection with a single vial (12 mg dose) is done on days 1, 15, 29, 64, and then every four months.
After obtaining informed consent, patient demographic information, genetic data, swallowing and nutrition status, respiratory condition, scoliosis, and patient function based on the HFMSE and RULM test were completed. In addition to recording patient performance based on the HFMSE and RULM questionnaires, all patients should continue their routine rehabilitation program.
End points
Four endpoints were considered based on previous studies. The first key endpoint was the average change in HFMSE scores after 3 and 6 months of treatment and a comparison between both groups. HFMSE is a reliable and valid measurement scale to assess overall motor functional capabilities in SMA patients (ambulant and non-ambulant) from 30 months to adulthood. 14 It comprises 33 items, ranging from 0 to 66, with higher scores indicating better motor function. The second key endpoint was an increase of at least 3 points compared to the baseline HFMSE score.10,14 The third endpoint was the revised upper limb module for SMA (RULM), which rates only the upper limb but correlates very well with the ability to perform everyday activities. It is a sensitive test for distal muscle strength and contains 19 elements with a maximum score of 37 points. The last endpoint was an increase of at least 3 points compared to the baseline RULM score15,16
Additionally, the mortality and morbidity in both groups were recorded. If a patient did not attend the scheduled appointment, they were followed up by telephone up to three times within a maximum of one week. If the patient did not participate, they were considered missing. If a patient visited another center, their data were added to the patient's previous data with the coordination of the primary center. The national health insurance system also rechecked the patients’ national identification numbers at the end of the study. If a patient had passed away, the date and cause of death were added to the data after follow-up, telephone contact, and review of the electronic health records.
Statistical analysis
Patients who could complete the three and six-month follow-ups during recruitment were included in the final analysis. Demographic and clinical characteristics were summarized using frequencies (percentage) for categorical variables and mean (standard deviation (S.D.)) or median (1st - 3rd quartile) for continuous variables. To compare the effect of risdiplam to nusinersen on HFMSE and RULM score at 3- and 6-months follow-up, we used regression analysis adjusted for baseline values and probable confounding factors such as HFMSE and RULM score before treatment, age at the SMA diagnosis, age at the start of treatment, sex, SMN2 copy number, scoliosis and using of assistive devices. We also used a T-test to compare the intragroup effectiveness of each drug at 3- and 6-month follow-up times. To compare the clinical effectiveness of two drugs, we used a cutoff value of a 3 point increase and more (6-month score compared to baseline score) to divide patients into responder and non-responders and used logistic regression analysis adjusted for confounding variables to compare the clinical effectiveness of two drugs. We also checked the effect of risdiplam versus nusinersen at 6 months periods after treatment with a generalized linear mixed model adjusted for baseline score and other probable confounding variables like HFMSE and RULM score before treatment, age at the SMA diagnosis, age at the start of treatment, sex, SMN2 copy number, scoliosis and using of assistive devices. In the final model the confounder variables which had P-value less than 0.2 remained in the model. Stata software version 17 was used.
Results
Of 165 children with type 2 and 3 SMA, the mean age at enrollment was 80.03 ± 31.39 months, and 45.5% were female (Table 1). 115(69.7%) were type 2, and 50 (30.3%) were type 3.SMN2 copy number 3 was the most prevalent (50.3%), and eighty-two (50.3%) patients had scoliosis (Table 1). Seven (4.3%) children needed non-invasive ventilation (NIV); none of the other children didn’t require any respiratory support. In both groups there was one patient with non-oral feeding condition. Table 1 shows the demographic and clinical characteristics of the patients at the baseline in two groups, and there is a significant difference between the two groups for the age at the start of the treatment, gender, scoliosis, assistive device usage, RULM and HFMSE variables (Table 1). The children in the nusinersen group were on average 12 months younger at the start of treatment, far fewer had scoliosis, and about 10 points better in HFSME and RULM Of these patients. 95 (57.2%) children received risdiplam, and 70 (42.2%) received nusinersen. Of 165 patients, 40 were excluded due to prior treatment with other SMA drugs in the last four months (16 patients), undergoing stem cell therapy (3 patients), invasive or continuous non-invasive ventilation requirement (5 patients), other concomitant genetic disorders (1 patient) and non-compliance of parents for regular visits (15 patients). Total 125 patients were considered for final analysis. 73 and 52 patients in the risdiplam and nusinersen groups respectively (Figure 1).
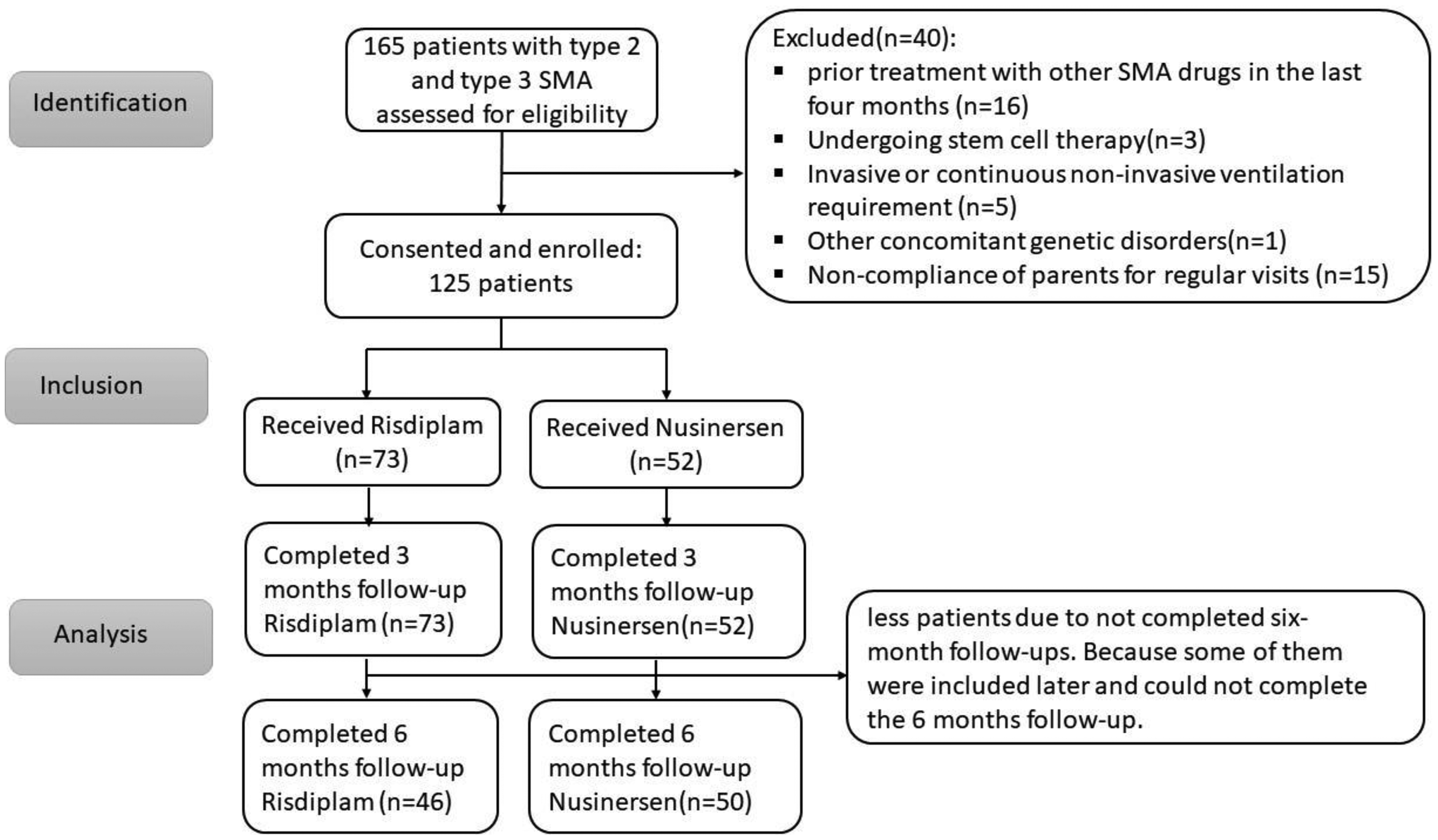
STROBE chart.
Demographic and clinical characteristics of the patients at baseline.
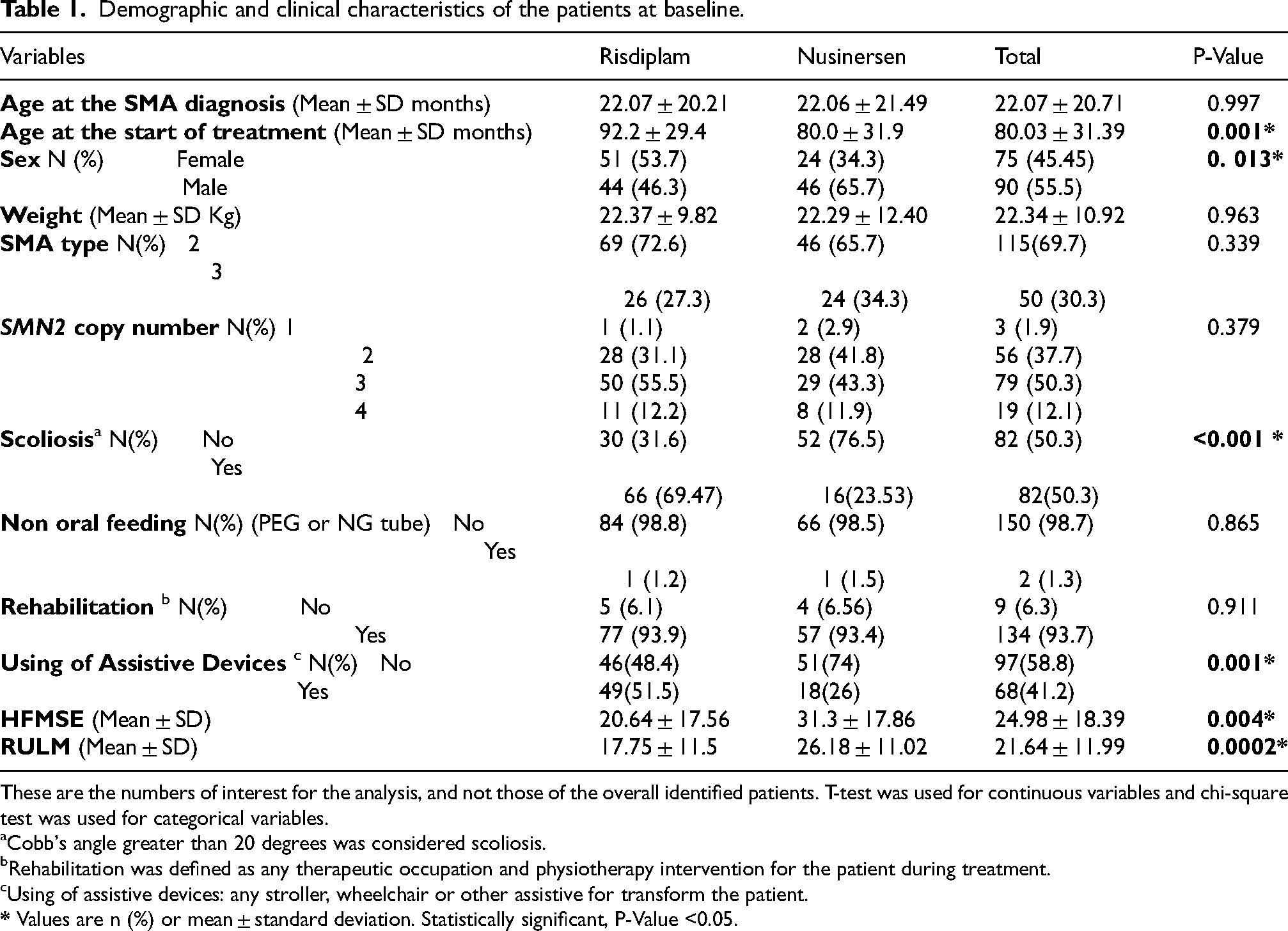
These are the numbers of interest for the analysis, and not those of the overall identified patients. T-test was used for continuous variables and chi-square test was used for categorical variables.
Cobb's angle greater than 20 degrees was considered scoliosis.
Rehabilitation was defined as any therapeutic occupation and physiotherapy intervention for the patient during treatment.
Using of assistive devices: any stroller, wheelchair or other assistive for transform the patient.
Outcomes
HFMSE: The difference in HFMSE score after 3 and 6 months of treatment compared to the baseline was significant in both groups (P-value <0.001). (Table 2 and Figure 2).
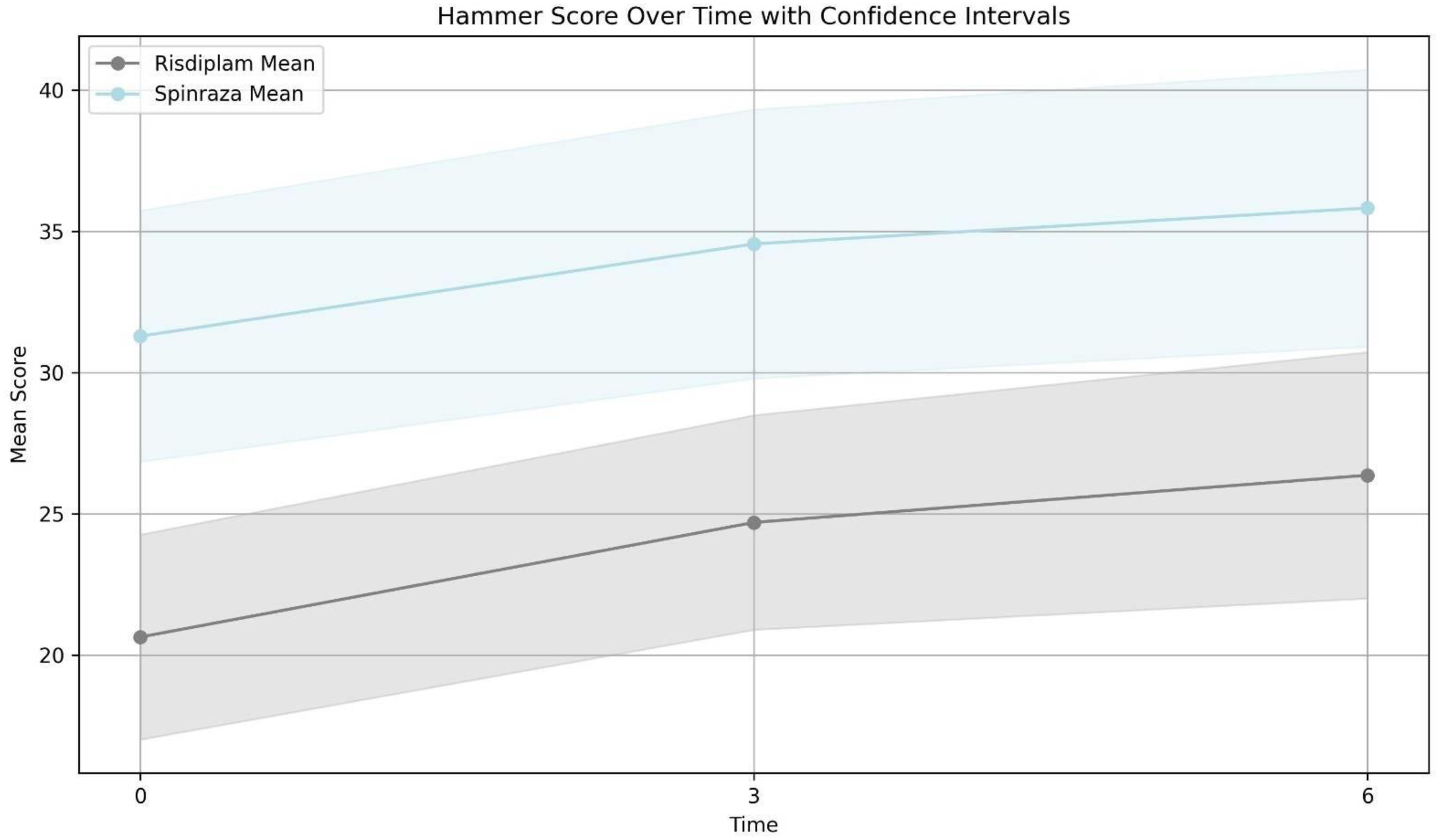
Comparison of changes in the mean of HMFSE after three months and six months between two groups. Risdiplam group mean of HMSFE score; before treatment: 20.66 (90 patients), 3 months after treatment: 24.69 (73 patients) and 6 months after treatment:26.36 (46 patients). Nusinersen group mean of HMSFE score; before treatment: 31.29 (62 patients), 3 months after treatment: 34.88 (54 patients) and 6 months after treatment:35.82 (51 patients).
Comparison of changes in the mean of HMFSE after three and six months between two groups.
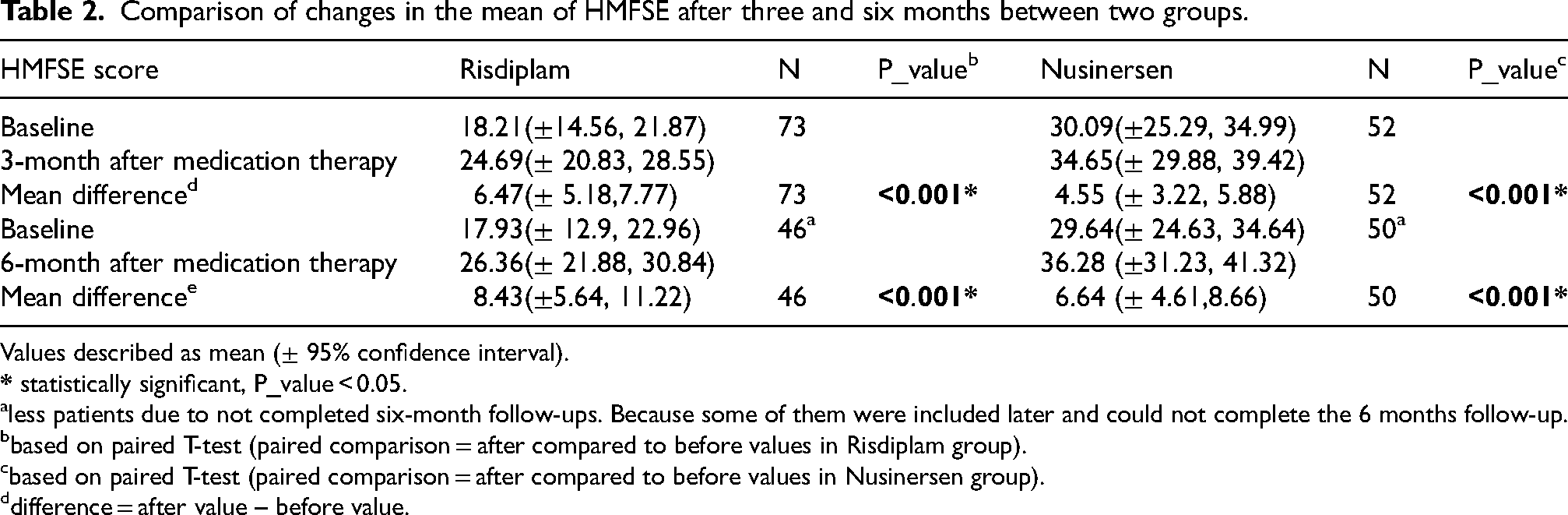
Values described as mean (± 95% confidence interval).
less patients due to not completed six-month follow-ups. Because some of them were included later and could not complete the 6 months follow-up.
based on paired T-test (paired comparison = after compared to before values in Risdiplam group).
based on paired T-test (paired comparison = after compared to before values in Nusinersen group).
difference = after value – before value.
RULM: The difference in RULM score after 3 and 6 months of treatment compared to the baseline showed significant changes in both groups (P-value <0.001). (Table 3 and Figure 3)
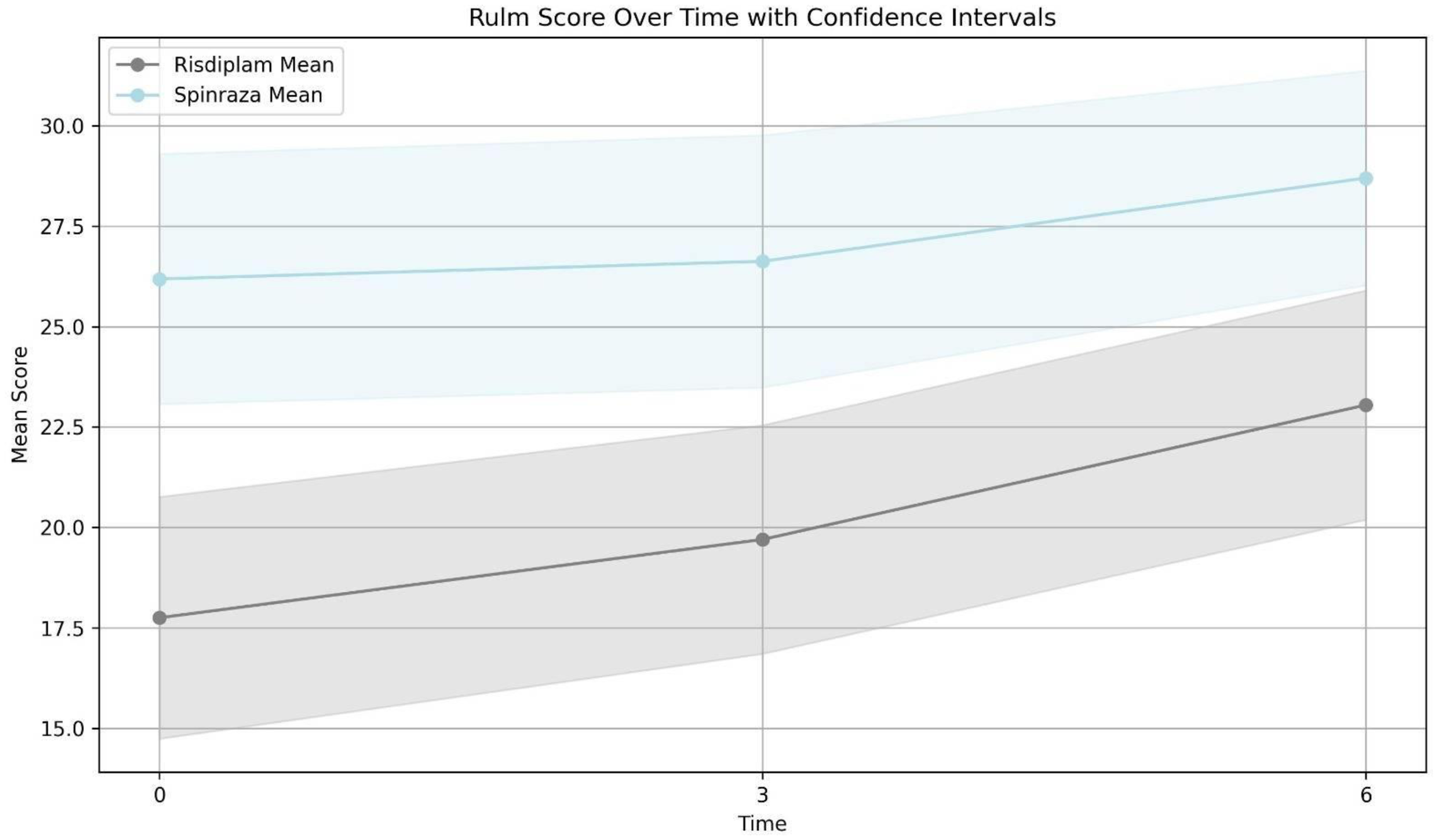
Comparison of changes in the mean of RULM after three months and six months between two groups. Risdiplam group mean of RULM score; before treatment: 17.75 (56 patients), 3 months after treatment: 19.7 (50 patients) and 6 months after treatment:23.04 (42 patients). Nusinersen group mean of RULM score; before treatment: 26.18 (48 patients), 3 months after treatment: 26.62 (43 patients) and 6 months after treatment:28.7 (47 patients).
Comparison of changes in the mean of RULM after three and six months between two groups.
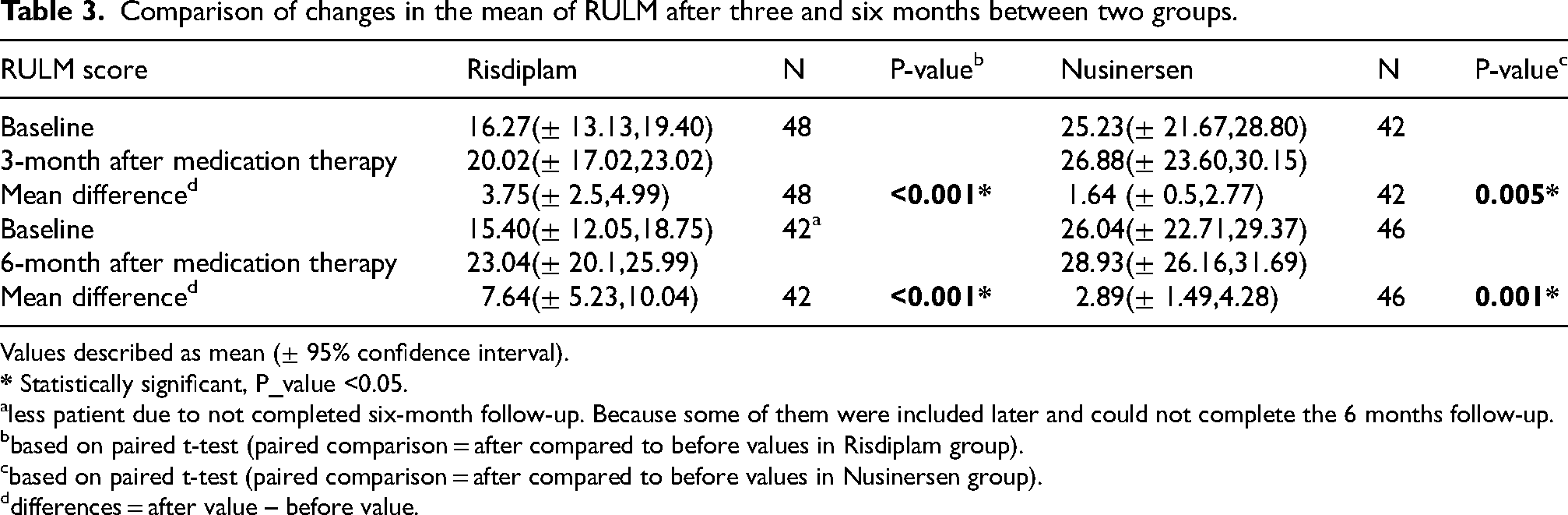
Values described as mean (± 95% confidence interval).
less patient due to not completed six-month follow-up. Because some of them were included later and could not complete the 6 months follow-up.
based on paired t-test (paired comparison = after compared to before values in Risdiplam group).
based on paired t-test (paired comparison = after compared to before values in Nusinersen group).
differences = after value – before value.
According to the results of the linear regression model one, in Table 4, after adjusting for the baseline value of the HMFSE and RULM scores, no significant difference was observed between the two types of drugs used after three and six months in this study. This result was repeated in the second model after adjusting for other confounding factors (age at the SMA diagnosis, age at the start of treatment, sex, SMN2 copy number, scoliosis and using of assistive devices.).
The results of univariate and multiple linear regression models adjusted for confounding factors to compare the effect of two treatments on HMFSE and RULM at three and six months.
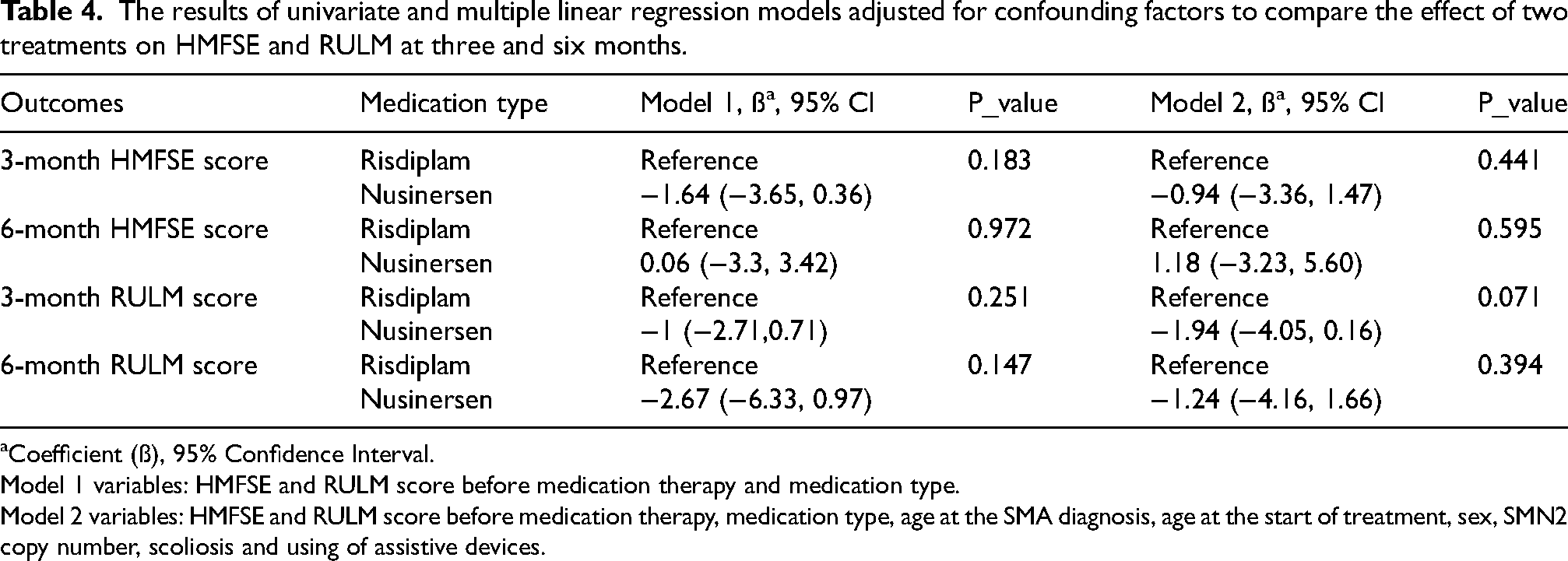
Coefficient (ß), 95% Confidence Interval.
Model 1 variables: HMFSE and RULM score before medication therapy and medication type.
Model 2 variables: HMFSE and RULM score before medication therapy, medication type, age at the SMA diagnosis, age at the start of treatment, sex, SMN2 copy number, scoliosis and using of assistive devices.
With control over the age of treatment, age of diagnosis, the number of SMN2 gene copies, and disease type, there was no significant difference in HMFSE and RULM scores between the two groups at 3 and 6 months. (P-value >0.05)
Minimal clinically important differences (MCID) were considered 3 points for HFSME, and according to the analysis of this factor, 73 patients (76%) achieved this cutoff, 37 patients(80.4%) in the risdiplam group and 36 patients (72%) in nusinersen group. There is no statically significant difference between the two groups (P-Value:0.33). The odds of HMFSE clinical improvement (3 points) after 6-month follow-up in the risdiplam group compared to spinraza group was 1.59 (CI: 0.61, 4.15 and P-value: 0.335). And, this ratio was not changed after adjusting for variables such as baseline HMFSE, copy number, treatment and diagnosis age.
Minimal clinically important differences (MCID) were considered 3 points for RULM, too; 50 (56%) patients achieved this cutoff total, 32 (76%) patients in the risdiplam group and 18 (39%) patients in the nusinersen group. The MCID ≥ 3 is more frequent in the risdiplam group compared to the nusinersen group (P-Value:0.001). In the risdiplam group, the chance of 3 points changes of RULM (MCID) was 4.9 more than the nusinersen group. (Odds ratio:4.9, 95% CI: 1.97,12.54 and P-value: 0.001); however, it was changed after adjusting with baseline RULM, copy number, treatment and diagnosis age and this chance disappeared (Odds ratio:1.6, 95% CI: 0.46, 5.5 and P-value: 0.45).
Mixed effect analysis revealed that patients in the nusinersen group had 2.3 lower RULM scores than the risdiplam group after a 6-month follow-up (95% CI: −4.53, −0.13 and P-value: 0.038). This analysis for HMFSE score revealed 0.49 higher score in the nusinersen group which was not statistically significant (95% CI: −2.66, 3.65 and P-value: 0.759).
Mortality and morbidity report
Mortality
Totally in both groups, only one patient died who was in the nusinersen group. He was an SMA Type 2 patient (4-year-old) had a history of respiratory problems with decreased FVC in oscillometry test 17 and mild scoliosis. He had normal oral feeding but was hospitalized multiple times for respiratory infections. HMFSE score and RULM score before treatment were 11 and 26, respectively. He received intrathecal nusinersen with four bolus doses. However, he developed a severe respiratory infection, leading to respiratory arrest after eight days, causing death. Based on the mortality committee report, the cause of his death was related to a respiratory infection due to the underlying disease.
Side effects of medication
Two patients had minor complications. A 9-year-old patient experienced abdominal cramps after risdiplam, which resolved spontaneously. A 7-year-old patient had back pain and nausea after a nusinersen injection, managed with hydration and anti-nausea medication in subsequent injections.
Discussion
To the best of our knowledge, there are currently no head-to-head trials comparing the efficacy of treatments for SMA types 2 and 3. Most available studies are indirect and involve systematic reviews to compare these two drugs.8,10 It is unlikely that a side-by-side blinded, randomized, controlled will be conducted and real world data has shown to provide valuable results about the effectiveness of these treatments across a variety of ages and endpoints.18–20 Our study is the first of its kind, involving both nusinersen and risdiplam treatment groups prospectively at the same time and in the same healthcare setting. The assignment to the 2 treatment groups was not random but based on pre-specified, clinical criteria of scoliosis and PEG feeding. This led to the risdiplam group being on average 12 months older at treatment start and scoring about 10 points lower in both RULM and HMFSE at baseline. However, in analysis, we adjusted for confounding factors to compare the effect of two treatments (Table 4). We observed that patients in both groups experienced improved HMFSE after treatment, with most patients (76%) reaching the cutoff point compared to their baseline values. Improvement of HMFSE and RULM score in the risdiplam group was more than nusinersen group. However, it is not statistically significant difference between nusinersen and risdiplam in SMA types 2 and 3 after three and 6-month too.
Risdiplam and nusinersen are both medications used to treat spinal muscular atrophy (SMA) in both adults and children. 21 They function by targeting SMN2 (survival of motor neuron 2) and increasing SMN protein levels throughout the body, albeit through slightly different mechanisms.21–23 One significant distinction between these drugs is that risdiplam is administered orally, while nusinersen is administered via intrathecal injection. 21 The systemic effects of risdiplam hint at potential differences in efficacy in this context.11,12 In a systematic review and indirect treatment comparison study, it was suggested that risdiplam is a superior alternative to nusinersen in treating type 1 SMA. 24 However, their study did not reveal any significant differences between the two drugs based on HFMSE. 24 Robust conclusions regarding the comparative efficacy of risdiplam and nusinersen in type 2 and 3 SMA were hindered by some enrollment criteria that could not be adjusted, limiting comparability on HFMSE endpoints. 24 In the study about Matching-adjusted indirect comparison (MAIC) in SMA patient, it was observed an uncertain indirect comparison of these two drugs due to differences in baseline characteristics that could not be adjusted for through Matching-adjusted indirect comparison (MAIC). 10 Nevertheless, our study showed that after adjusting for enrollment data, no significant difference was observed between the type of drug used and the HMFSE after three and six months of follow-up.
HFMSE has recently been shown to be a key outcome measure in SMA studies due to the high content validity and clinical significance of the items included in the scale. 14 Determining the minimal clinically important difference (MCID) values could help to evaluate treatment efficacy. In Judit's study, Minimal clinically important differences for the HFSME were considered three points. 8 By considering the MICD cutoff, two-thirds of our patients (75%) achieved the 3-point cutoff in the HFMSE. Judit mentioned in his systematic review study that treatment of SMA type 2 to 4 stabilized the health status in the trials of nusinersen. 8 The result for risdiplam was not concluded due to a lack of appropriate studies for evaluation. 8 However, in our study, the percentage of patients who reached the cutoff point 3 is similar, and we did not find any statistically significant difference between the two groups.
In our study, the RULM mean difference in the risdiplam group was similar in both groups. According to other studies, the MCID cutoff point was considered 3 for RULM.15,16 In our study, the chance of 3-point RULM (MCID) changes was 4.5 more than the nusinersen group; however, this chance disappeared after adjusting with the baseline of RULM. This could be related to two hypotheses: the lower score changed to a higher score more easily than the higher score or the systemic effect of risdiplam. However, more studies are needed for more evaluation. In a systematic review and indirect treatment comparison study, Ribero and his colleagues showed similar responses with comparable improvements in RULM scores over the same period. 24 In this study, analyses provided insufficient evidence to draw concrete conclusions on the relative efficacy of RULM change from baseline due to a very wide confidence interval. 24
After comparing the two group's for complications, there is one mortality in the nusinersen group who had a history of respiratory problems and admission. The etiology of his death was related to the respiratory infection and underlying disease. Ribero's study showed less serious adverse events in type 1 SMA in the risdiplam group; however, this result was not concluded in type 2 and 3 due to insufficient evidence to draw concrete conclusions on the relative safety. 24
Most of the indirect systematic reviews comparing the medications for type 2 and 3 SMA, mentioned several limitations.,8,10,24 including heterogeneous populations and definitions of certain patient characteristics, different outcome measures and length of follow-up. We conducted this study by patients with similar characteristics and outcome measures in the three and 6-month follow-ups. However, this study also had some limitations. This clinical study was without randomization and blinding. Therefore, it can cause selection bias and potential known and unknown confounders in causal interpretations. However, efforts were made to calculate and mitigate the impact of confounding factors. Another limitation is inter-rater bias consideration, which acknowledges potential bias due to the variety of patient reception centers and differences in the individuals measuring patients’ scores. This is a short prospective cohort study, and some of patient added to this study late and not completed the follow-up for children two to ten years of old. Therefore, prolonged study in other ages is recommended.
In conclusion, this study showed that both medications significantly changed the HFSME and RULM after 3 and 6 months of follow-up. However, there is no significant difference in type 2 and 3 SMA between risdiplam and nusinersen groups according to the HFSME after 3 and 6 months. However, a study with longer follow-ups including other subtypes (e.g., invasive ventilator dependency) with matched baseline characteristics is needed for better evaluation.
Footnotes
Acknowledgments
We want to thank the Iranian Ministry of Health and their committed team, consisting of Younes Panahi, Seyed Heidar Mohamadi, and Saeed Karimi, for their generous provision of medications for all patients, as well as to the SBMU and the TUMS registry secretariat. We also wish to thank Professor Hanns Lochmüller from Ottawa, Canada, for his valuable insights and discussions on our study before its submission. Additionally, we appreciate the TREAT-NMD neuromuscular network for supplying a valuable tool for collecting and analyzing patient data, which has been vital to our research.
Data availability statement
The data that support the findings of this study can be provided upon request from the corresponding authors. However, the data are not publicly accessible due to privacy concerns or ethical restrictions.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
