Abstract
INTRODUCTION
Drug discovery programs are demanding that all assays and sample analysis be done at ever higher throughput rates simultaneously with lower costs and better results. The rapid sample throughput of MALDI-TOF Mass spectrometry has made this a preferred tool for sophisticated analysis of complex biological samples. However, in order to have optimized results and spectra that are easily interpreted, intelligent sample simplification and purification pre-Mass Spec is necessary. Obvious tools for this are the various chromatography media, including reverse phase and ion-exchange resins. For high throughput screening, rapid and simple methods are needed for sample preparation to keep up with the MALDI-TOF. Previous work has shown that MultiScreen® Resist 96-well filter plates, loaded with adsorptive resins, have excellent well-to-well reproducibility in sample recovery (<10% CV) and >90% salt removal. These plates can be conveniently loaded with the chromatography media optimum for the samples to be processed and at sample volumes and recoveries consistent with MALDI-TOF requirements.
To demonstrate the selectivity and versatility of 96-well mini-column sample preparation, both reverse phase and ion-exchange resins were used. Data presented shows initial determinations of column capacity and recovery using bovine serum albumin (BSA) and cytochrome c. Methods and results for complex mixtures are demonstrated by fractionation of trypsinized peptides from cytochrome c and mouse serum. Trypsinized biological samples were fractionated using a step-wise elution procedure for identification by HPLC and/or MALDI-TOF.
Materials and Methods: 96-well mini-chromatography columns were prepared following the manufacturer's instructions by loading dry media into MultiScreen Resist 0.4mm filter plates (MAR4N0410, Millipore Corporation, Bedford MA) using either a 25μl or a 45μl MultiScreen Column Loader (Millipore Corporation, Bedford MA). Media used included reverse phase resins of a) 300 Å 6–10 um C18 (Waters-Microbondapak, Waters Corporation, Milford, MA); b) 300 Å 15pm or 40 μm C4 (Amicon-Matrex, Millipore Corporation, Bedford, MA); or c) R2 styrene-divinylbenzene(DVB), C4 like (PE-PerSeptive Biosystems, Framingham, MA). Also included was a strong cation exchange (SCX) resin, the AG-50w-x 2 sulphonated-styrene-divinylbenzene, 100–200 mesh (Bio-Rad Laboratories, Hercules, CA).
After dry loading, media in the mini-columns was packed for separations by adding 200 μl of isopropanol (IPA) to each well and centrifuging at 3000 × g for 5 minutes. Residual IPA was removed when wells were equilibrated in 200 μl of their respective binding solutions, either 0.1 % TFA (reverse phase resins) or 20 mM HCl (SCX resin) and centrifuged. Samples were loaded in 200 ml of appropriate binding solution and centrifuged. Loosely bound sample, salts, and detergents were removed by performing a 200 μl wash step with the respective binding solutions prior to elution. Resin-bound peptides were eluted in 50% acetonitrile (ACN) or sequentially by applying a step gradient. Peptide recovery from all resins was carried out using either 1 × 100 μl, 2 × 50 μl, 2 × 25 μl per well of ACN in 0.1% TFA. Step-gradient elution was performed by increasing desorption strength (10, 30 and 50% ACN for C18 or 5, 15 and 50% for DVB) of eluant, followed by an additional 300 μl rinse step of the same eluant prior to the next gradient step. In the case of SCX resin, step gradient elution by pH was performed in 50mM ammonium acetate: ACN (1:1) at indicated pH. Packing, binding and elution steps require centrifugation for optimum performance. Wash steps can be done more rapidly using vacuum filtration. In all cases one must take care not to disturb the packed bed with the pipetting equipment. MALDI-TOF mass spectroscopy data was acquired on a Voyager STR (PE-PerSeptive Biosystems, Framingham, MA).
RESULTS AND DISCUSSION:
Table 1 shows that, as expected, the smaller particle size resin gives improved capacities. An advantage of these mini-columns is that flow restrictions associated with small particle size media columns are not present, and thus the enhanced capacity can be used without detrimental results. The capacities are not linear going from the 25μl to 45μl columns, because the 25μl column has essentially no bed depth. However, the capacity is sufficient for most MALDI-TOF sample prep, and the smaller volumes are both less expensive and have less hold-up volume.
Capacity of MultiScreen Mini-column for various media.
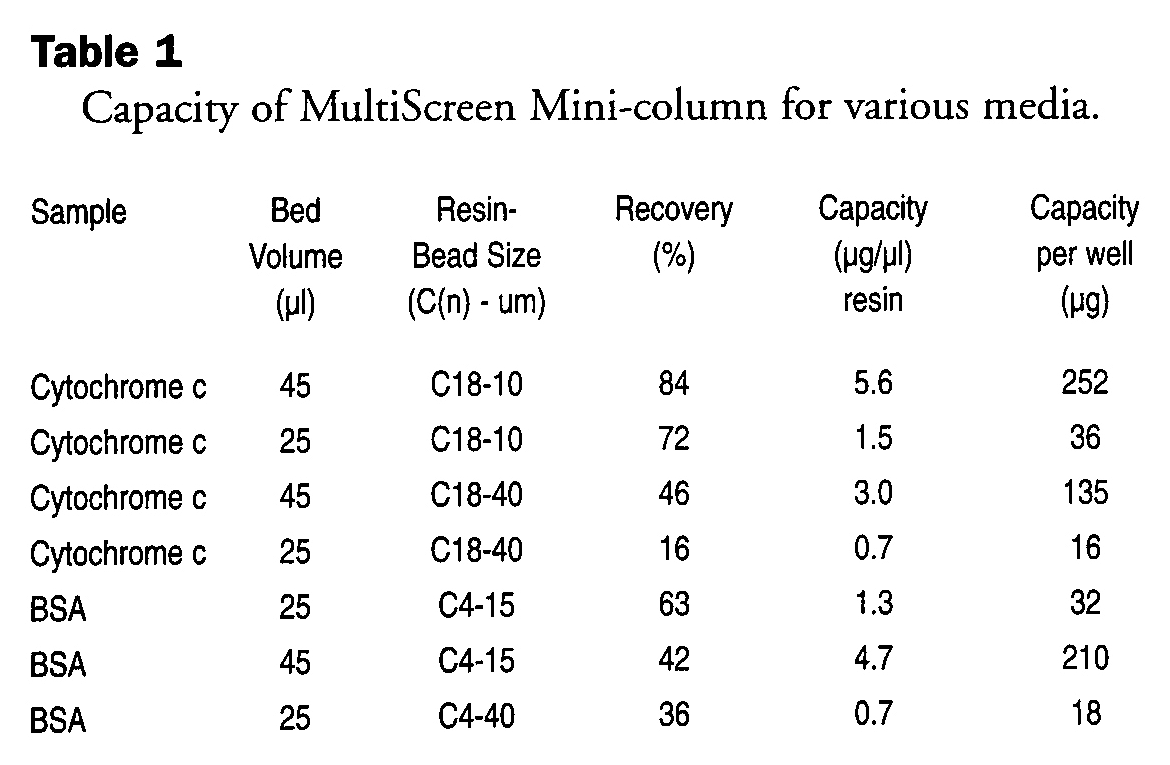
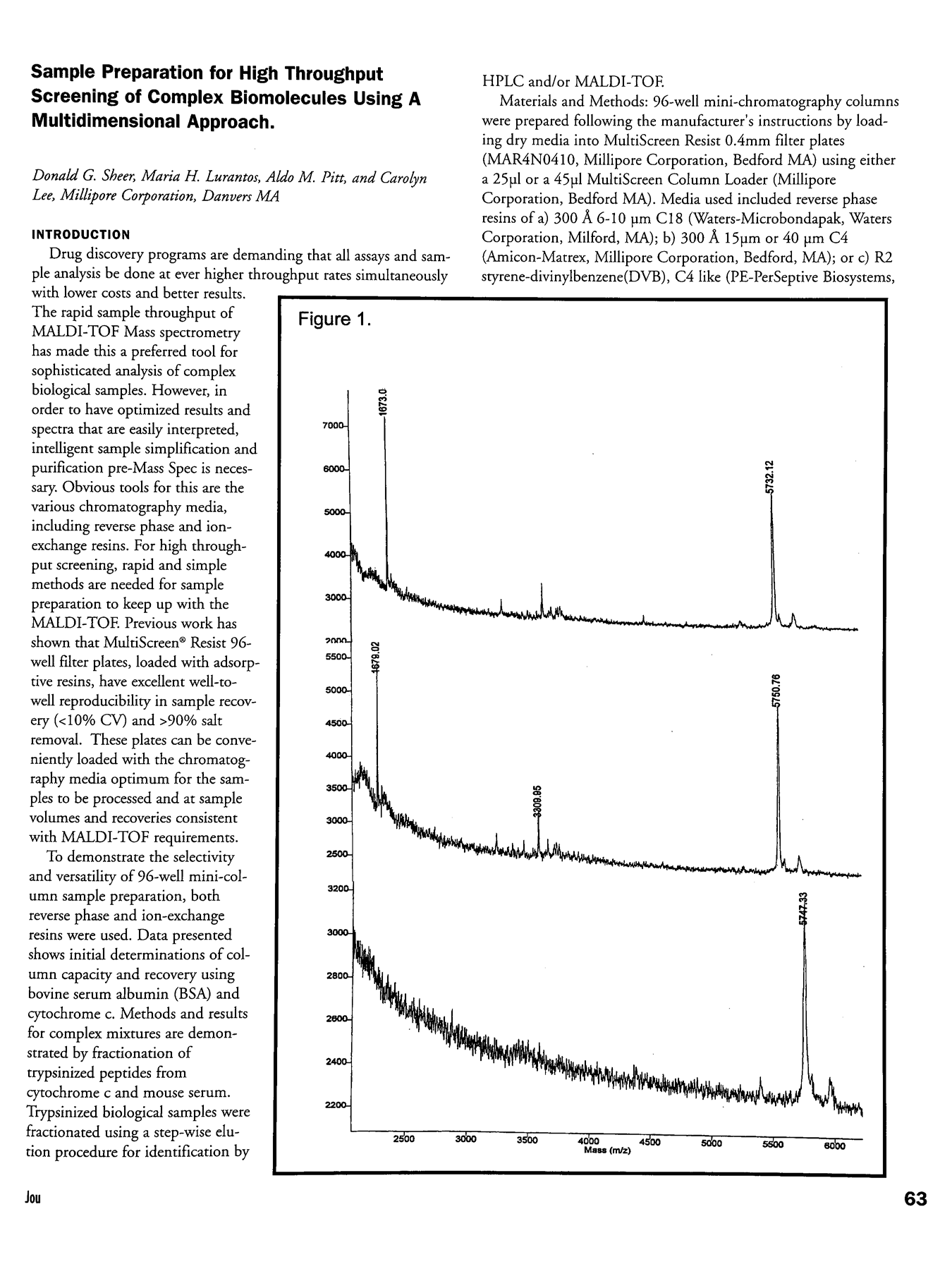
Figure 1 demonstrates that loadings of as low as 0.062μg onto a 25μl C18 column result in excellent MALDI-TOF spectra. HPLC analysis of recovery (data not shown) was 44%, 53%, 65% for the 3 loadings. Thus, desalt and concentration normalization were achieved without serial dilutions, eliminating the need to optimize the MALDI-TOF loading for each sample. Samples were eluted after washing in 2 × 25 μl.
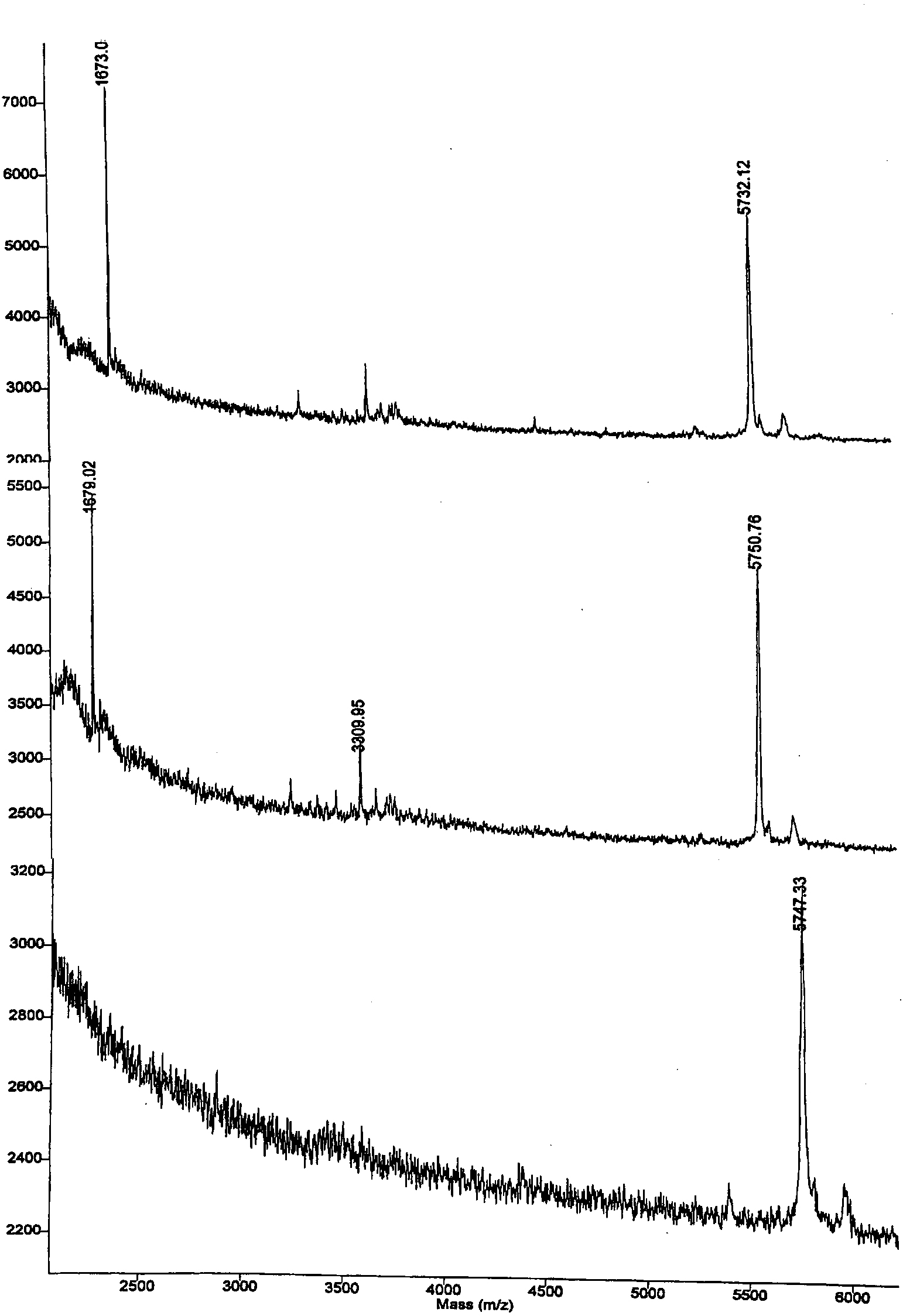
Sensitivity and Recovery for Insulin, as demonstrated by MALDI-TOF after bind, wash and elute on C18 25μl Mini-column. TOP:0.25μg; MIDDLE:0.125μg; BOTTOM:0.062μg insulin loading.
Reproducibility and full recovery from the columns of applied samples was demonstrated in data not shown for trypsinized mouse serum over C18 columns and trypsinized cytochrome c over SCX columns in addition to the insulin, BSA and cytochrome c samples.
The ability to simplify spectra containing multiple peptides prior to MALDI-TOF greatly eases the interpretation of the spectra, in addition to increasing the likelihood of the entire sample “flying”. Stepwise chromatography in the MultiScreen mini-column plates provides an easy method for accomplishing this in a fast and cost effective way. This is shown in Figure 2 with analytical HPLC data for a cytochrome c tryptic digest processed using C18 minicolumns and step-wise elution. Further optimization of the step gradients could have more completely separated the hydrophilic from the hydrophobic peptides. In Figure 3 this is shown by a series of step gradients using different media applied to a mouse serum tryptic digest. It is shown that, by using a combination of cation exchange (SCX), C18 and Styrene Divinyl Benzene, the peptides can be widely separated into simpler spectra before MALDI-TOF, and peptides unresolved in the control spectra are resolved after sample preparation.
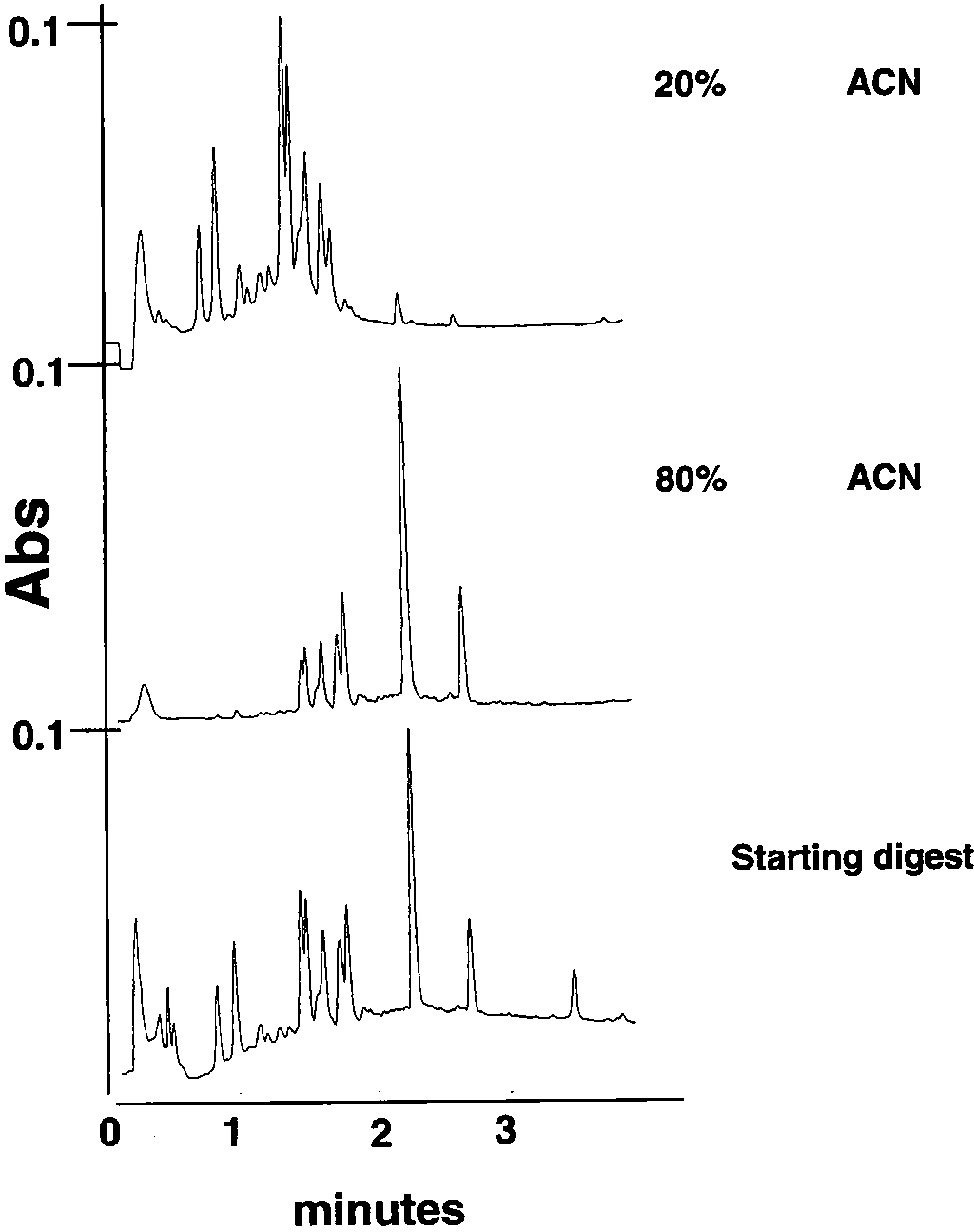
Analytical HPLC after step gradient elution of Cytochrome C tryptic digest using C18 25μl mini-columns.
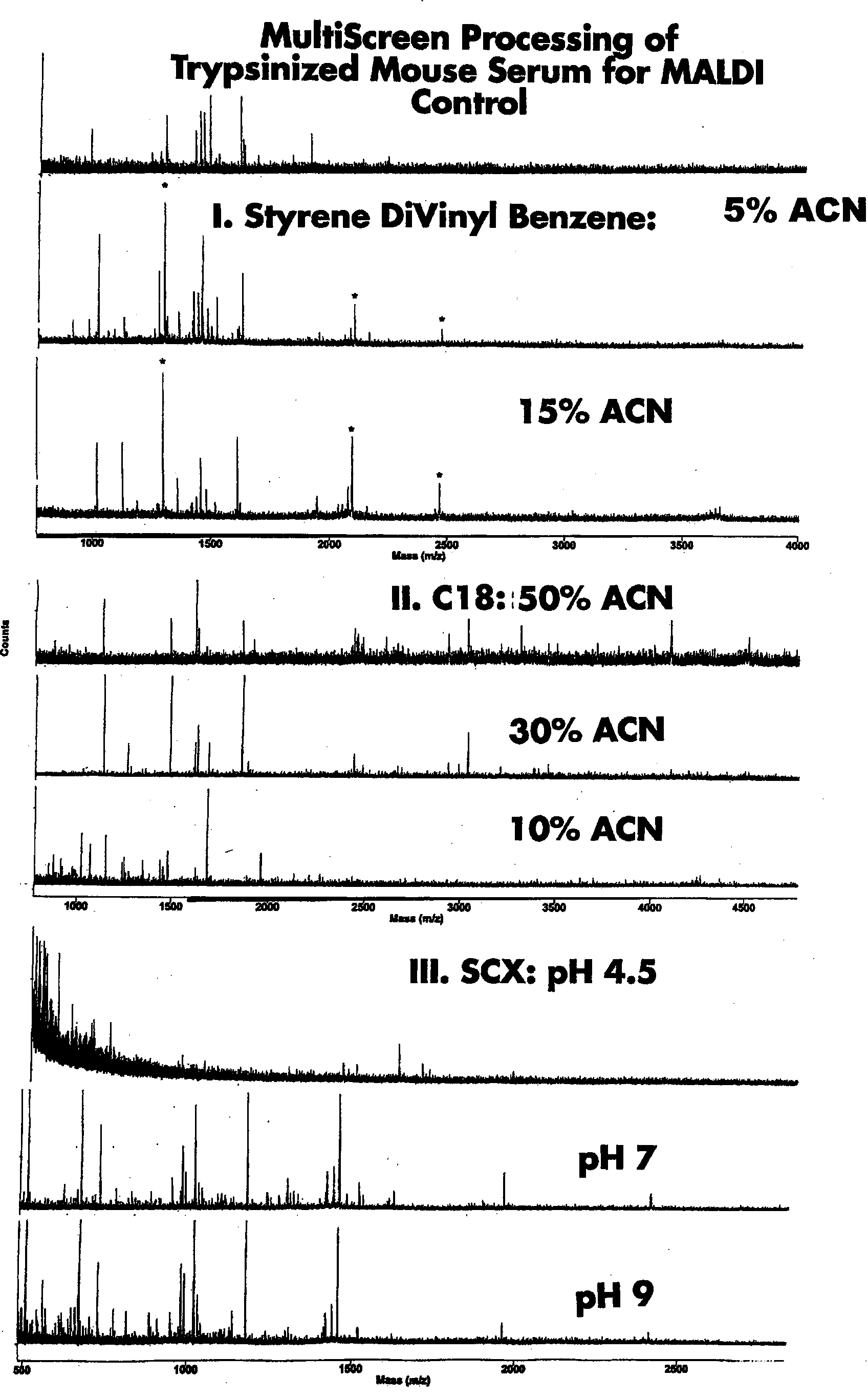
Comparison of step gradient methods for simplifying MALDI-TOF spectra of Trypsinized mouse serum.
CONCLUSIONS
Direct sample analysis by MALDI-TOF mass spectrometry and analytical HPLC of eluates from 96-well mini-column plates provided reproducible results and simplified spectra while maintaining sample fidelity. MALDI-TOF sensitivity and resolution were increased enabling previously unresolved peptides to be detected. Desalting and concentration normalization were accomplished simultaneously in simple bind, wash and elute protocols. Step wise elutions resulted in useful fractionations with either ion exchange or reverse phase resins, eliminating the need for more expensive and slower preparative columns, which also tend to have higher void volume losses. The ability to choose the best resin for the separation required and then rapidly prepare hundreds of identical columns adds cost effectiveness and convenience. Rapid sample prep using 96-well mini-columns prepared with the MultiScreen separations system results in clean, easy to interpret spectra.
