Abstract
Five years ago, drug discovery involved carefully planned synthesis of single organic molecules. The overall synthetic approach had not changed much since the 19th century; individual chemists made individual compounds in round-bottom flasks. These compounds were then screened for biological activity in an attempt to develop a lead compound that could be taken into animal trials. A typical medicinal chemist might manage to synthesize 30 novel compounds in a year for biological screening. Since the annual cost of a chemist, including salary, lab space, instrumentation, management overhead, etc. is approximately $250,000, this meant that each compound synthesized cost approximately $8,000!
There had always been a strong demand for larger numbers of novel organic compounds for biological assay, both for random screening and structure-activity optimization. Once the pharmaceutical companies automated high-throughput screening (HTS), the demand for more compounds further intensified. Many pharmaceutical companies began internal efforts to improve the productivity of chemical synthesis, and a number of commercial vendors began providing automated equipment for organic synthesis, Initially, many companies planned to synthesize and to screen “pools” containing several compounds at once. While these efforts at pooled synthesis still continue, analysis of the results of pooled screens have not been as successful as was first hoped. False positive and negative assay results are quite common with pools, and identifying the particular component within the pool that was responsible for a positive result is often difficult (the “deconvolution” problem). The past several years has seen a definite swing in most companies' strategies toward automated synthesis of single compounds, running many reactions in parallel in separate reaction vessels. Solid-phase organic reactions are preferred when possible, since these usually result in relatively fewer impurities than solution-phase reactions. The improvement in the efficiency of parallel synthesis when compared to a traditional output of 30 compounds per chemist per year is startling: Pfizer, for example, recently announced the parallel synthesis of a library of 500,000 single compounds! [1]
Drug discovery may be viewed as a sequential process, including such steps as reaction design, procurement of starting materials, synthesis, purification, characterization, solvent evaporation, sample storage, and biological screening. At any one time, one or more of these steps may be a bottleneck in the overall process. The recent improvements in the efficiency of synthesis has moved the bottleneck downstream, to purification. This paper describes a new instrument, the Parallex™ HPLC, that will eliminate the purification bottleneck (and will of course move the bottleneck elsewhere, probably to solvent evaporation).
Why Purify?
Some companies routinely take compounds directly from synthesis to high-throughput biological screening with only minor purification (perhaps filtration and solid-phase extraction). In this strategy, the solid-phase reactions that can be used are limited to those that are usually driven nearly to completion; the hope is that almost all members of a library can be synthesized at a purity level suitable for assay (typically > 80%) using multi-step reactions. Only those compounds that show a positive assay result in these preliminary biological screens are further purified and then characterized, typically by mass spectrometry and NMR.
One problem with this strategy is that, within any individual parallel-synthesis library, the purity standards can be quite uneven. Even if the library achieves the desired average purity level of 80%, individual compounds may be much less pure. Why, in spite of this problem, do some companies attempt to avoid a purification step? The answer is simple: traditional purification is difficult and expensive.
The Parallex HPLC
The deveopment of Biotage's new Parallex HPLC (Figure 1) makes high-throughput preparative HPLC straightforward and relatively low-cost. Purification can now be a routine step in the preparation of organic libraries.
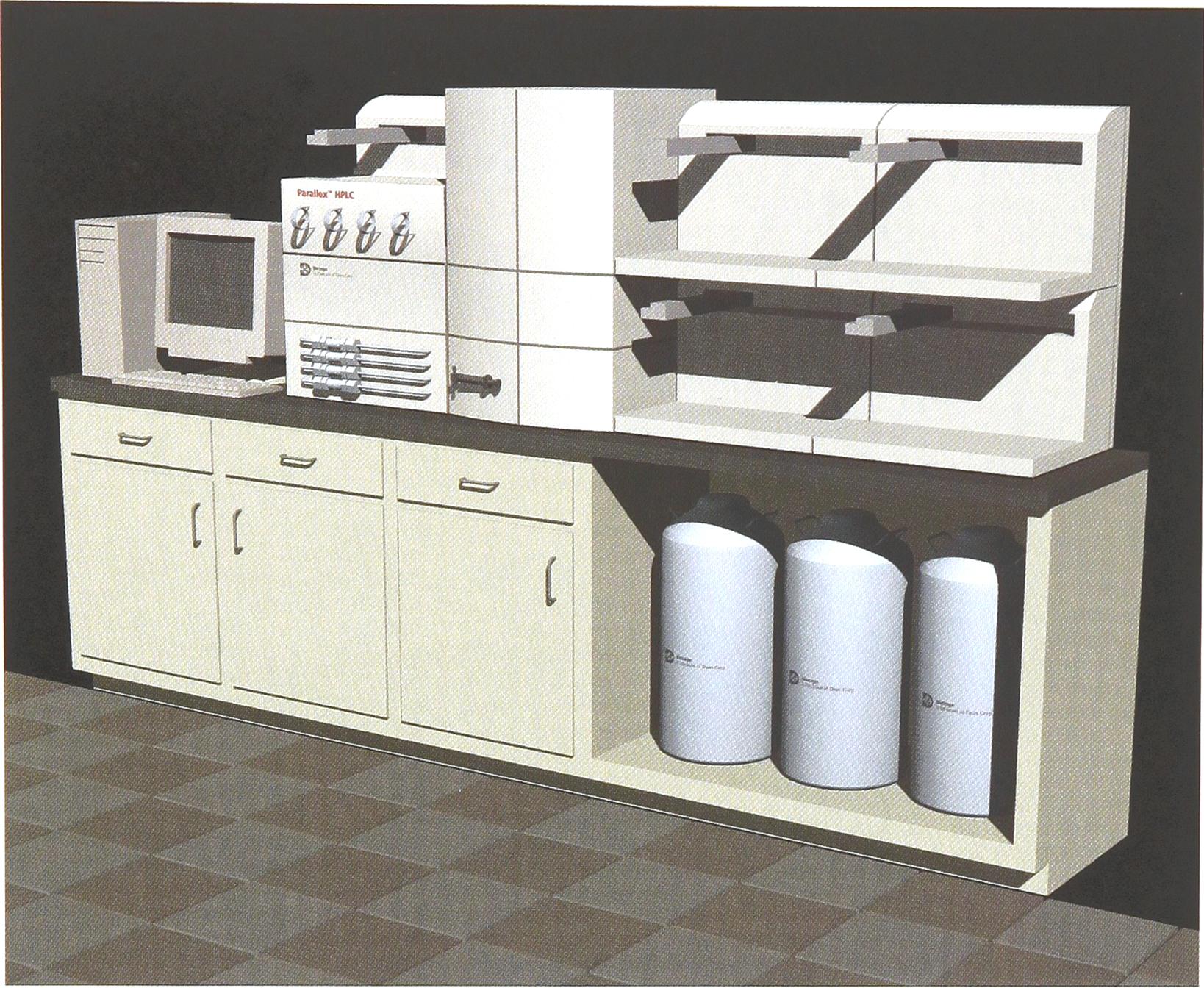
The Parallex HPLC is a fully automated purification system that runs four (4) columns simultaneously, uses a microtiter plate format and features Microsoft Access software for data management.
The advantages of routine purification include:
While many purification techniques are used for low-throughput applications, preparative HPLC seems to be the technique best-suited for high-throughput automation. The Parallex HPLC has been optimized for crude sample weights of 5–200 mg, the range that seems to be of interest for most parallel purification efforts. In developing the Parallex HPLC, we have emphasized four design goals that have not been important in the design of traditional HPLC instrumentation:
Figure 2 shows a flow diagram of the Parallex HPLC. Four columns operate in parallel, each with its own pump head to ensure uniform flow rates through all columns regardless of variations in backpressure among the columns. All columns draw from a single low-pressure mixer running a programmable binary gradient between solvents A and B. All four injection loops are rotated into the flow stream simultaneously, and they consequently run the identical gradient.
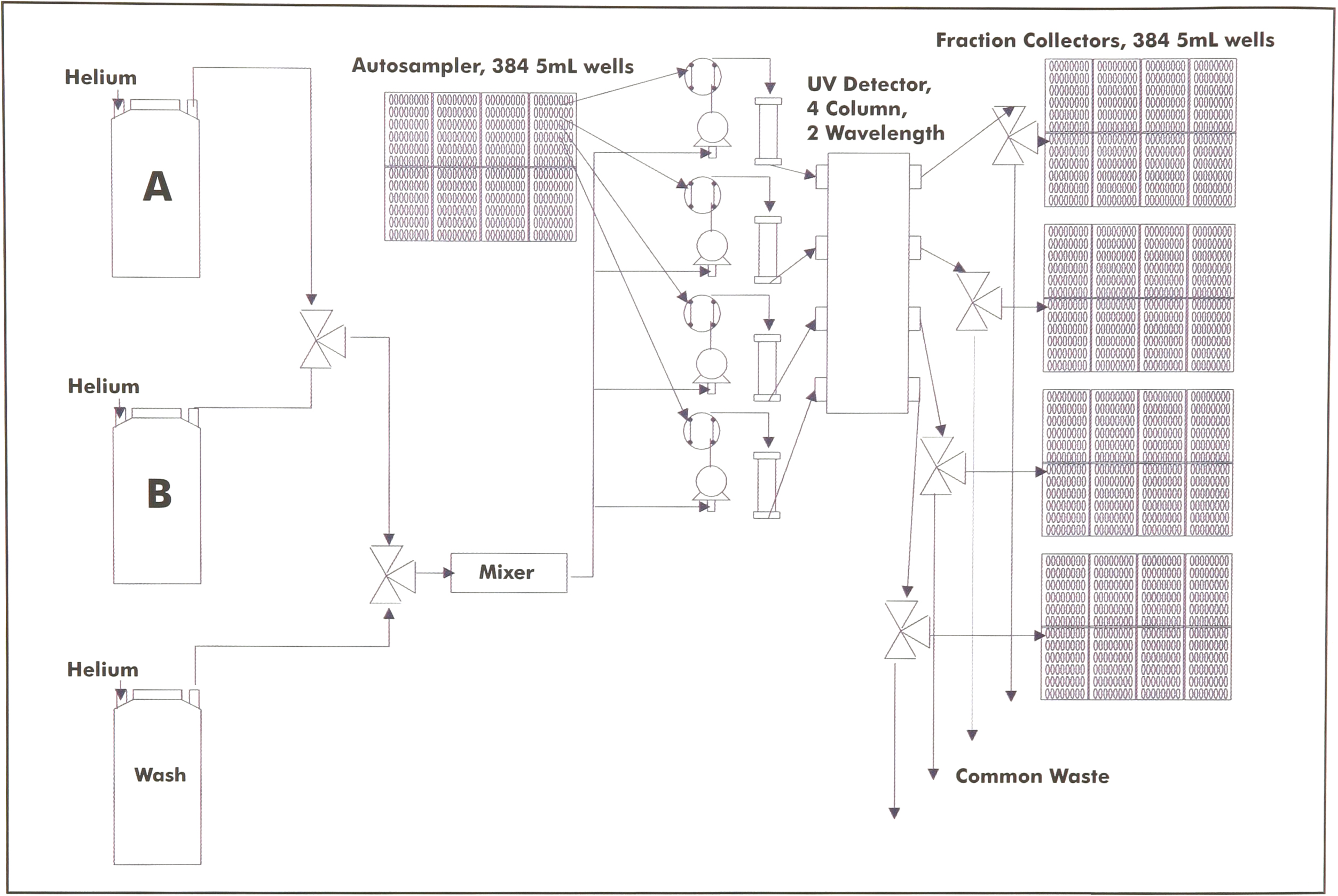
Schematic flow diagram for the Parallex HPLC.
Samples are introduced in deep-well microtiter plates, and collection is in deep-well plates as well. Depending on injection volume and flow rates, either 96-well 2 ml plates or 48-well 5 ml plates may be used. Up to eight plates may be loaded on the autosampler and on each of the four fraction collectors, giving a total of either 384 5 ml samples or 768 2 ml samples that may be loaded. The Parallex HPLC uses UV detection for slope and threshold decisions on fraction collection. The UV detector multiplexes a single deuterium lamp across all four channels. Each column has its own flow cell (Figure 3) combined in a single housing, with simultaneous detection of two UV wavelengths and a ratio. Each column has its own fraction collector with an independent x-y arm filling the appropriate wells in the collector's microtiter plates. The only solvent collected is that lying under one or both UV peaks, thereby minimizing the amount of solvent that must be collected.

The four UV flow cells integrated into a single housing.
One of the most significant problems in handling this volume of samples is information management. Both sample and collection plates are tracked by barcode. All sample and fraction information is tracked in Microsoft Access™. The Parallex HPLC software runs under Windows NT and provides a data channel to both liquid handling systems and to mass spectrometers or other characterization devices. Figure 4 shows the Parallex Explorer screen that integrates information on physical devices (instruments, autosampler and collector trays, microtiter plates), sample and fraction wells, textual information from the database, the status of the instrument, and chromatograms.
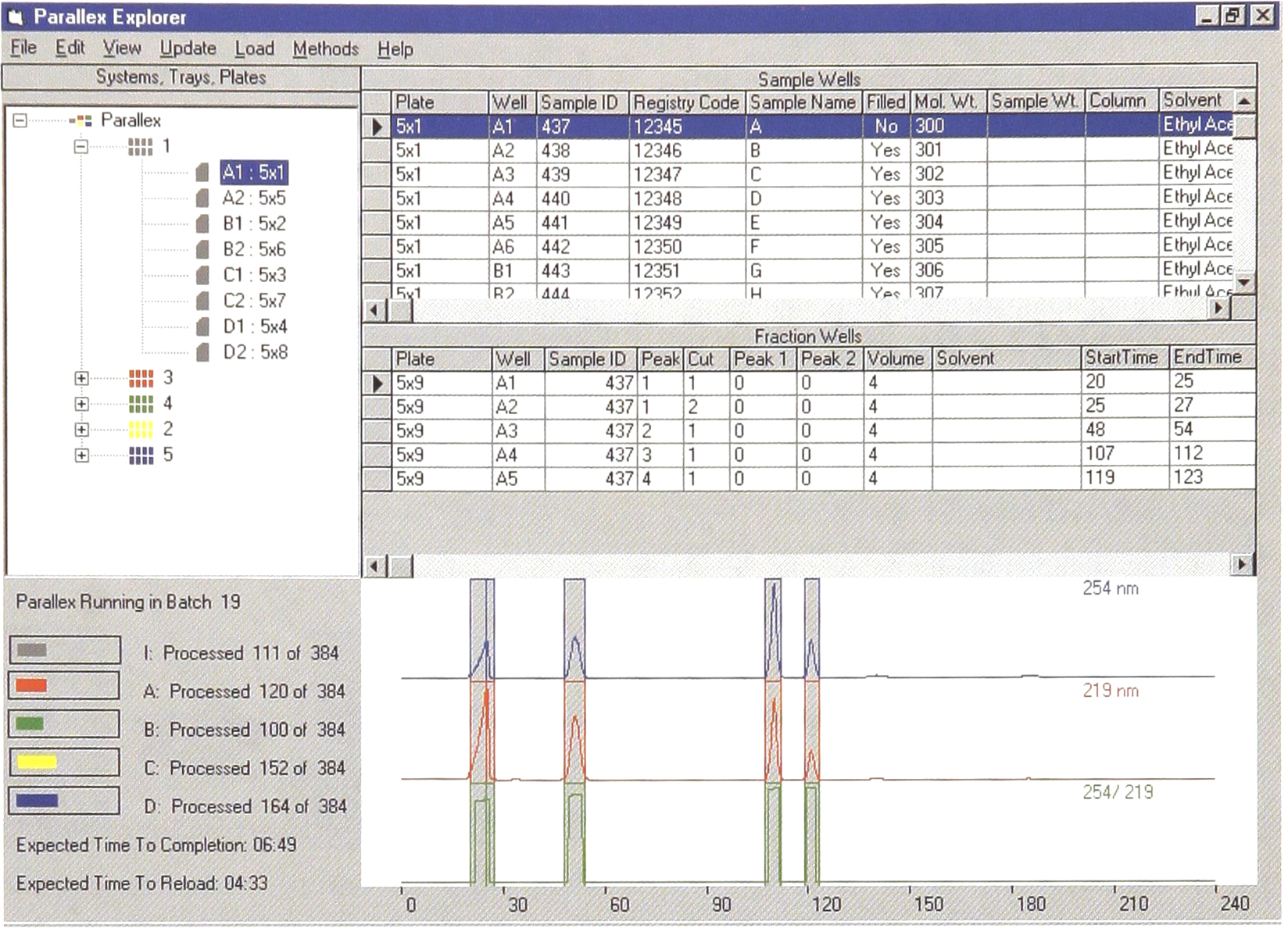
The Parallex Explorer integrates information on systems, trays, microtiter plates, chromatograms, system operating status, and textual data on samples and fractions in a simple browser screen.
Chromatography
The Parallex HPLC is fully programmable in terms of flow rates, gradient ramp rate, injection volumes, etc. While the Parallex HPLC could be used in normal phase or ion exchange chromatography, we believe it will find its greatest application in rapid reverse-phase chromatography using a “universal gradient”.
Routine reverse-phase purification in this area has been extensively developed by Dr. Harold Weller of Bristol-Myers Squibb [2], where rapid reverse-phase gradients are routinely used for both the results of parallel synthesis and for compounds synthesized traditionally. Typically, a short 5μm C18-packed column is used at high flow rates.
Figure 5 shows two chromatograms of a solution-phase synthesis of urea. The “universal” gradient (10% — 90% acetonitrile/water) gives a separation in six (6) minutes (right) that is similar to a 20 minute separation (left). The sample was provided by PanLabs, Inc.
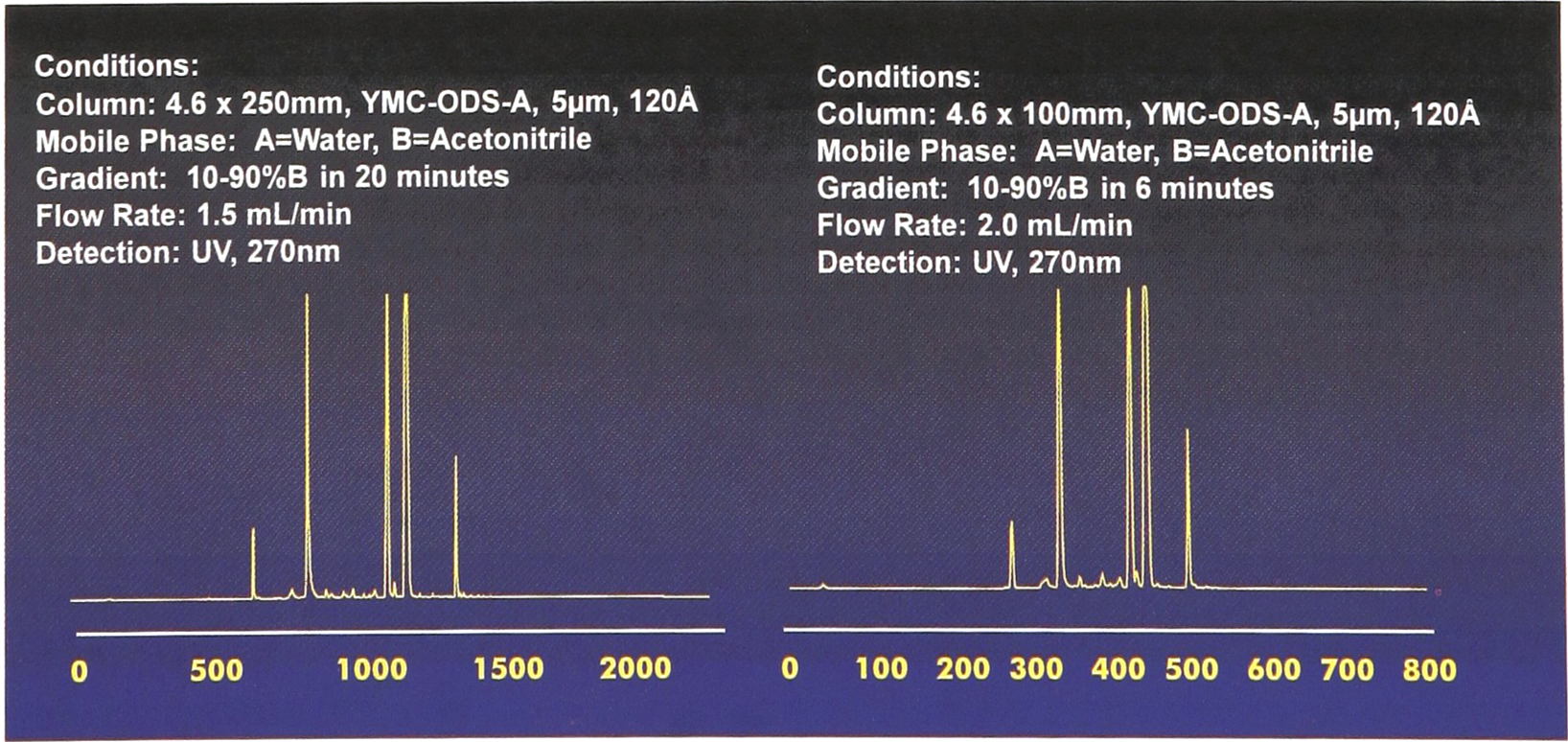
In this example, the Parallex HPLC universal gradient separated a sample in six minutes with nearly equivalent resolution to that shown in 20 minutes. (sample supplied by PanLabs).
Since the Parallex HPLC is fully programmable, resolution may be further improved by extending the time for the gradient with concomitant increase in solvent use. These tradeoffs between throughput, solvent use, and resolution are under the control of the operator.
Communication with Synthesis and Characterization
The Parallex HPLC is only one element in the overall production of compound libraries. Figure 6 shows one way that the transition from synthesis to purification to characterization might be organized.
The operator of the Parallex HPLC reads the barcode label for each plate, automatically importing sample information prepared by the liquid handler. At a minimum, this information should include a unique identifier for each well and the molecular weight of the compound that was (supposedly) synthesized.
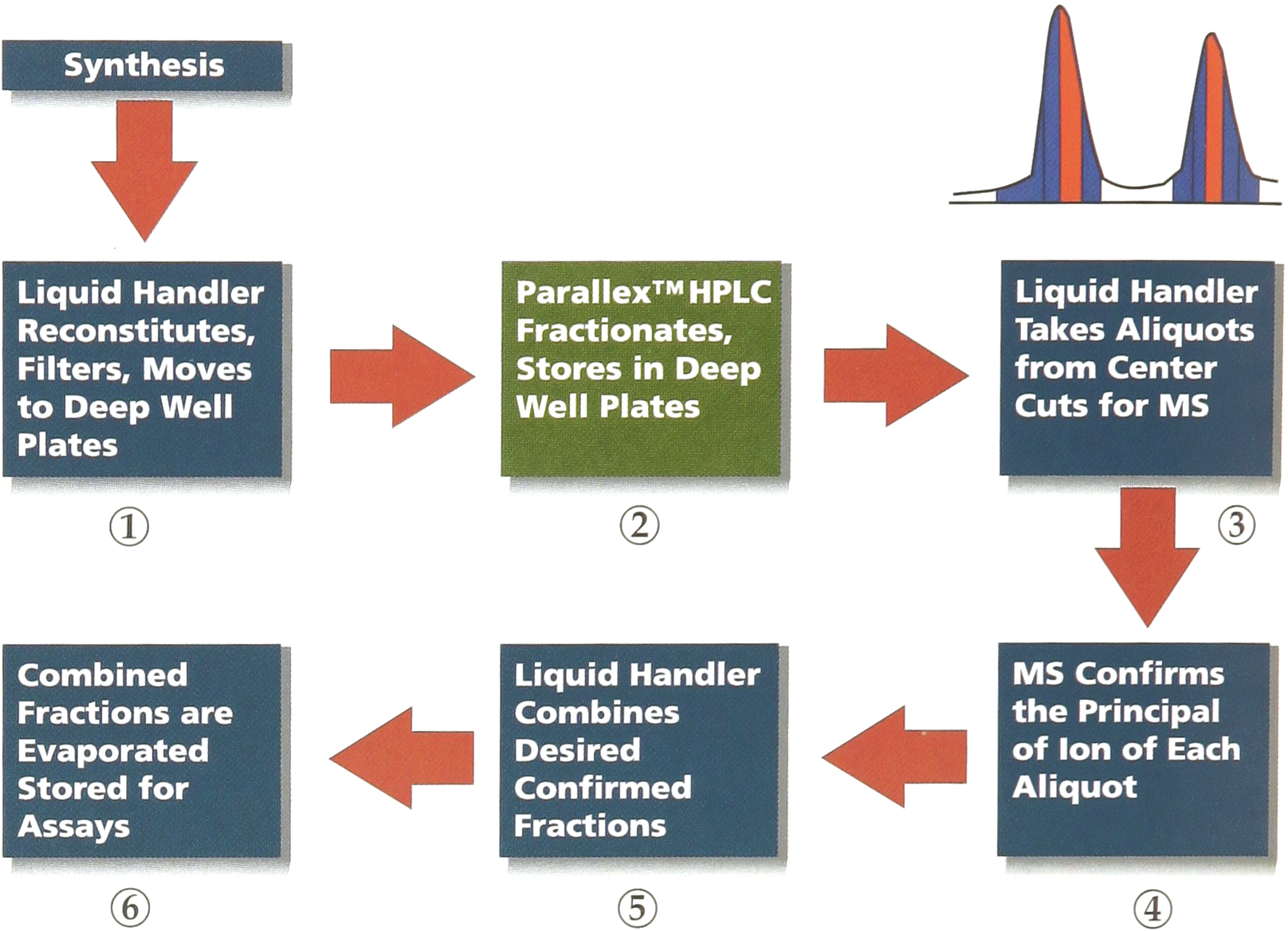
Typical overall organization of a production process for organic libraries using the Parallex HPLC for the purification stage. The Parallex HPLC connects to the synthesis and characterization stages through liquid handlers. Data is transmitted either by import/export of Excel™ spreadsheets or ASCII tables or through direct connection of databases. By sending only the center cut of each peak (see upper right of figure) to the mass spectrometer for analysis by loop injection, the number of wells that must be tested is minimized. By using the mass spectrometer to identify the desired peak before evaporation, the amount of solvent that must be evaporated can be greatly reduced.
Cost per sample
In designing the Parallex HPLC, we have been especially concerned about the running cost per sample. In traditional preparative chromatography, where only a few samples are purified at a time, this has not typically been an area of concern. However in purifying organic libraries that may contain thousands of compounds, the cost per sample becomes very important. We have identified five principal areas of cost:
Areas of Further Work
We are planning on actively investigating the use of additional on-line detectors (MS, ELS) and on further automating the transfer of both information and material between the Parallex HPLC and liquid handlers.
For further information on the Parallex HPLC and its high-throughput parallel purification of combinatorial libraries, contact the Biotage, a Division of Dyax Corp., 1500 Avon Street Extended, Charlottesville, VA 22902. Tel: 804-979-2319, Fax: 804-979-4743.
Parellex and Parallel Purification are trademarks of Dyax Corp.
