Abstract
Reprinted with permission of author and publisher from
Over the last few years there has been a growing demand for assay miniaturization in high throughput screening. The successful adaptation of the 96-well microplate serves as a practical example of the power of miniaturization. Assays that once required 2 to 3 mL volumes are now done in 1/10 this volume. However, before the 96-well plate became a standard in HTS there were many questions about the suitability of this small format to screening assays and the availability of all the necessary peripheral equipment to support it. These problems were quickly solved. Today we are faced with the same questions about the 384-well format.
Because the acceptance of the 384-well microplate was unproven, instrument companies were unwilling to commit R&D resources to develop such equipment. Scientists could not develop 384-well assays without analytical instruments and instrument companies would not develop equipment without some indication that there would be assays to measure.
This unproductive cycle has been broken with the development and introduction of the TopCount™ HTS. The HTS has the capability to measure radioisotopes or luminescence in both 96 and 384-well microplates. This development will allow laboratories to continue using the 96-well format while scaling down assays to the 384-well format. The 40 plate external stacker allows the unattended processing of over 15,000 samples in 384 plates or almost 4,000 samples in 96-well plates. Twelve wells are measured simultaneously and 96- and 384-well plates can be mixed in the stacker. This will add significantly to the efficiency, flexibility and future adaptability of the screening laboratory.
Photo courtesy of Nunc, Inc.

There are two questions most often asked regarding the use of the 384-well microplate. First, can assays be scaled down to 100 μL, 50 μL or even less and still have a strong signal and a good signal to noise ratio? Second, will equipment be available for operations such as pipetting and plate washing? The answer to both of these questions is yes.
In this brief article, examples will be given of scaling down the scintillation proximity assay (Amersham International) and the luminescent luciferase enzyme assay (LucLite™, Packard Instrument Company).
A major advantage of the 384-well microplate is that much of the current equipment in use today can be adapted to this format. An example illustrated here is the use of the MultiPROBE™ liquid handling robot (Packard) to prepare a SPA assay in the 384-well plate.
The 384-well plate has exactly one-half the spacing between wells compared to a 96-well plate. This allows the use of the MultiPROBE, as well as hand-held, multichannel pipettes. Samples are pipetted into every other well, the pipette is then indexed and the alternate wells are processed. With regard to other 384-well plate equipment, microplate readers for fluorescence and absorbance have been introduced and the adaptation of plate washers to the 384-well format is also inevitable. Because the 384-well plate has the same outside dimensions as the 96-well plate, much of the current plate handling equipment should be readily adaptable.
Microplate centrifuges, incubators, plate carousels, shakers and roboticarms should all fit this new format, thus eliminating the need to replace new and expensive equipment.
Results and Discussion
To illustrate the capabilities of the 384-well microplate, two common assays were selected for initial testing. The first was the simple, mix and measure SPA assay and the second was the homogeneous LucLite luciferase assay. The microplates used for these experiments are OptiPlate™-384 (Packard Instrument Company) These white, opaque polystyrene plates have square wells and hold a total volume of 125 μL. The maximum usable volume is approximately 100 μL, All samples, either 96 or 384-well, were analyzed on the TopCount HTS.
Figure 1 shows the results of a SPA assay (1251 cyclic-AMP) where the assay components, including SPA beads, were pipetted with the MultiPROBE or a hand held pipette. The total assay volume was 100 μL. Both samples were prepared in OptiPlate-384 and counted on the TopCount HTS for 3 min. As would be expected, the results of either method gave the same displacement curve. Another important assay parameter is the signal to noise (Bo to NSB) ratio which is a measure of the assay window. A comparison of this same SPA assay showed that the use of the MultiPROBE with the 384-well plate gave a Bo/NSB of greater than 15 compared to a value of less than 13 for a 96-well plate prepared manually. The ability to use automated equipment becomes particularly important with the 384-well plate. While scientists have become very skilled at identifying any given position in a 96-well plate, doing the same with 384-wells may not be possible. This, and the obvious fatigue of pipetting, could lead to unacceptably high error rates in manual assay preparation.
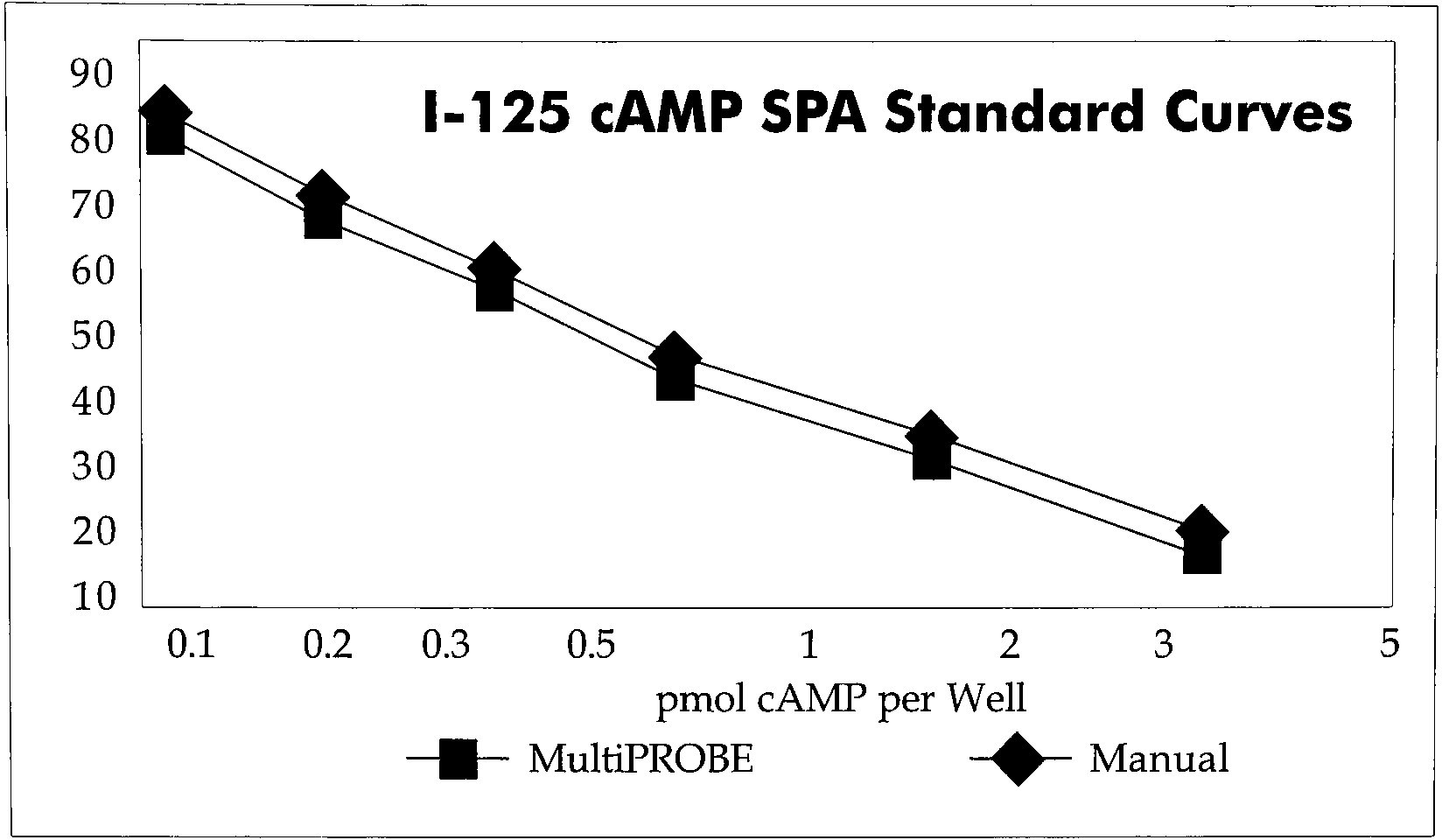
Optiplate-384, Manual versus MultiPROBE Pipetting
A further test of the 384-well plate for SPA was carried out at Zeneca Pharmaceuticals and is shown in Figure 2. In this example, an 1251 SPA dose response curve was run in a 50 μL total volume and counted for 1 min. The data points gave a good fit with the same midpoint as measured in the 96-well assay. The error bars around the midpoint (indicated by the lines around the closed circle) show the excellent fit of the data.
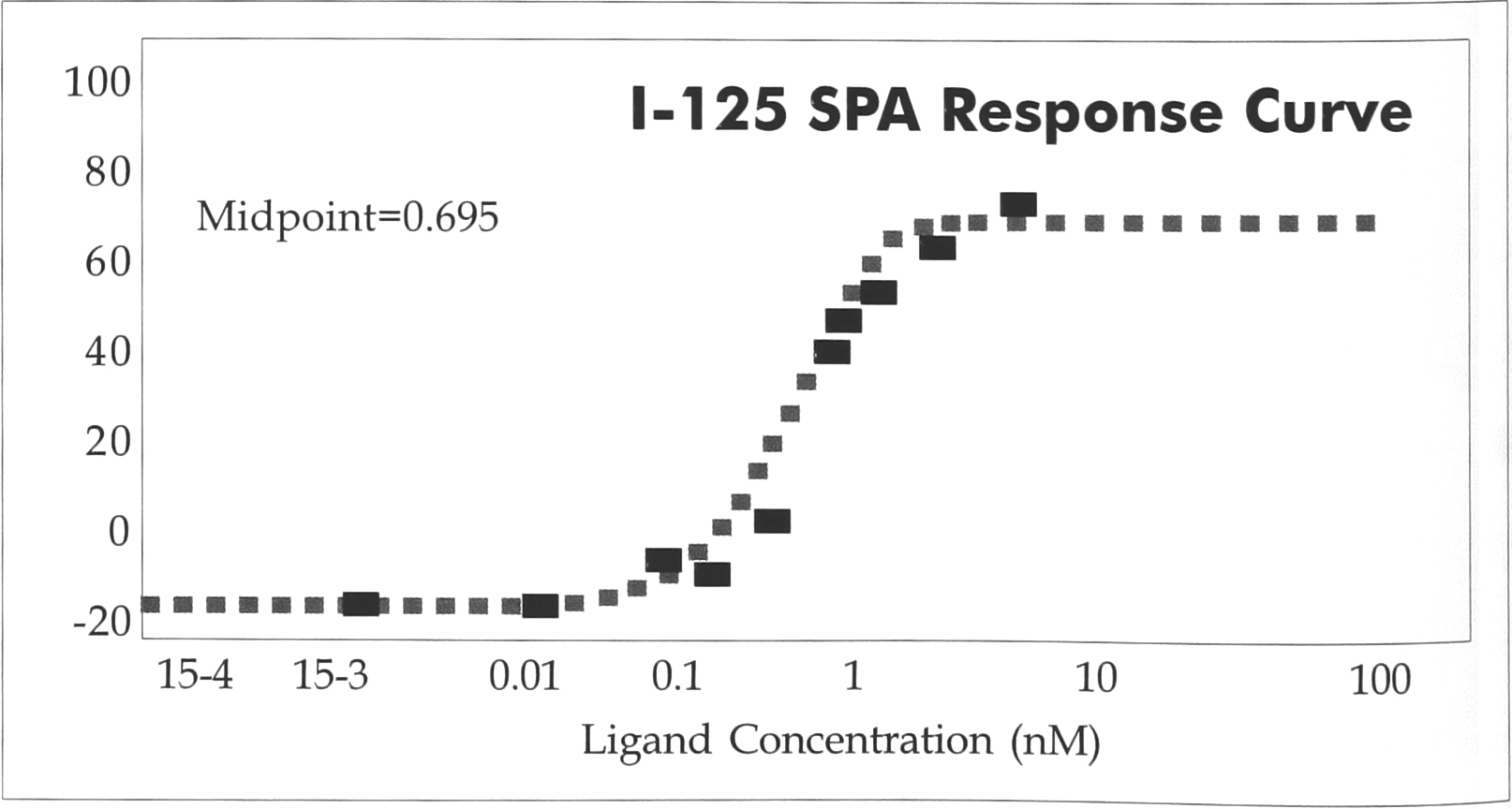
Optiplate-384, 50μl.
The assay window for the 50 μL and 25 μL volumes was well above the minimum window required for acceptance as a screening assay. The dose response curve run at 25 μL and also counted for 1 min exhibited more scatter (data not shown). This illustrates a limit in miniaturization for some assays because of diminishing signal. Low activity samples could be counted longer, but this would limit throughput. There are ongoing developments in 384-well microplate design that promise to significantly increase the assay signal and thus make lower volumes practical.
The luciferase reporter gene is a popular luminescence assay for transcriptional regulation. The standard flash chemistry has recently been improved upon with the introduction of LucLite. This assay kit extends the half-life of the luciferase luminescent signal to over five hours. This eliminates critical timing steps and allows the batch processing of many thousands of samples. This assay was measured in the TopCount HTS in the single photon (luminescence) counting mode. Known amounts of luciferase enzyme were used to compare the signal in the 96-well plate (100μL) and 384-well plate (50μL). These results are shown in Figure 3. The two curves are parallel and signals from several hundred cps to many hundred thousand cps can be measured. The 384-well plate offers several cost advantages with the use of fewer cells per assay, lower culture reagent costs, less labor and more efficient use of cell culture facilities.
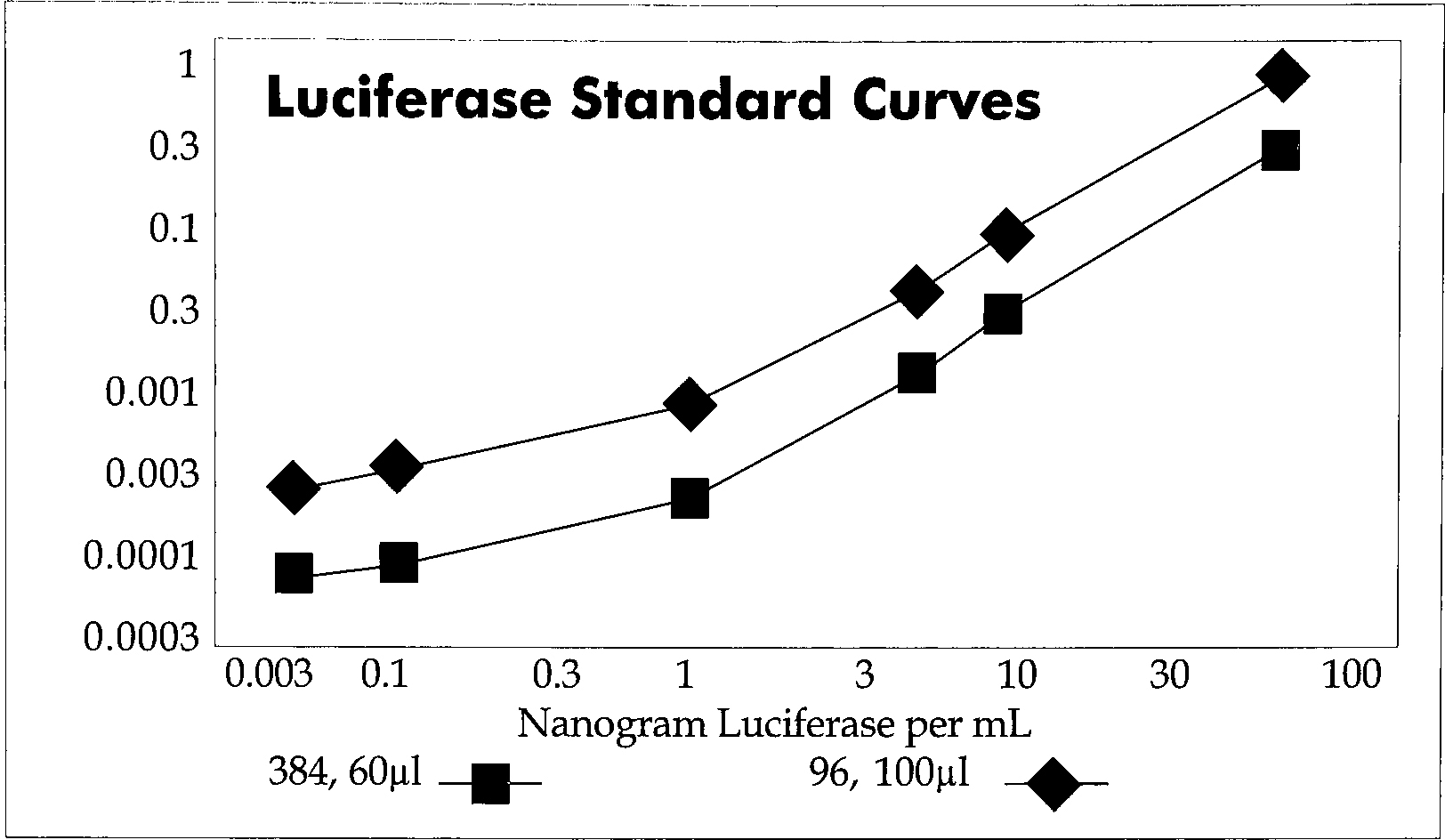
Lucite Optiplate-384 and Optiplate-96.
Conclusion
The 384-well microplate is a very promising format for HTS. It will allow significant reductions in reagent and disposal costs and can use currently available assay methods and equipment. This saves the considerable expense to completely re-equip a laboratory with new automated pipettes, robots and peripheral devices. It could cost several hundred thousand dollars to enlarge a robot with more plate carousels, larger incubators, a longer track or a larger room. Instead, the same equipment and the same robot real estate could be used to accommodate four times the samples by simply converting to 384-well plates.
Screening technology and assay miniaturization is a very dynamic field. While this makes HTS an exciting discipline to work in, it also creates anxiety when it comes to selecting between various methods. A question that is commonly asked is whether it is worth converting to 384-well plates or would it be better to wait for the next step in assay miniaturization? Since the next step in miniaturization is not clearly defined, the answer to this question is also not clear. The danger in waiting for the next step could mean that two or more years of enhanced productivity would be lost by not incorporating the 384-well format. Advances in nanoliter dispensing technology, single photon imaging and highly sensitive fluorescent assays are all under development and many of these instruments and assay methods will still be suitable for use in the 96 and 384-well formats. With these prospects for the future, it would seem that there is greater risk in ignoring the 384-well format than there is in incorporating it as a part of an ongoing HTS program.
