Abstract
A new automated 96 channel microdispenser is described for precise, high speed dispensing of microliter volumes of reagents. The Hydra 96™ is a programmable instrument composed of 96 glass syringes arrayed in microplate format that fill and dispense in unison under computer control. Studies show the instrument has less than a 2% coefficient of variance (C.V.) across the syringe array when dispensing between 0.5–20.0 μl of reagent. Blot hybridization studies demonstrate a simple rinsing protocol using 2% bleach efficiently cleans the system of DNA preventing downstream sample contamination without affecting subsequent PCR reactions. Current uses of the instrument in assembling microassays used in large scale genetic mapping and sequencing projects and compound library screening are discussed.
INTRODUCTION
Sample amplification techniques such as PCR coupled with improved signal enhancement and detection methods have led to a new generation of microassays. In addition, screening of large libraries of compounds to identify drug candidates has led to new requirements for rapid, parallel preparation of various ELISA and bioassays. In laboratory situations where a multitude of small volume reactions are prepared in parallel, robotic multichannel dispensers are normally used. Many commercial systems have limited precision when dispensing below 10μl and have high fixed operating expenses from the use of disposable tips. Also, robotic systems tend to be inefficient when used for assembling identical reaction mixtures used in many screening type applications.
The Hydra-96 Channel Microdispenser was designed for special applications where multiple, identical small volume reaction mixtures are prepared in microplate format. It provides high speed, precise delivery of reagents down to 0.5μl using an array of 96 glass syringes that fill and dispense in unison. All instrument operations such as fill and dispense volumes are controlled via an integral microprocessor with desired parameters stored in non-volatile memory. The instrument can also be controlled through an external computer when used in conjunction with other automated laboratory equipment.
In this report, results are provided from studies designed to measure the dispensing precision or uniformity of the syringe array. Also, since the system does not use disposable tips and removal of residual reagents between uses is of paramount importance, data is presented from a study using DNA samples of the effectiveness of simple rinsing to clean the syringes that does not effect subsequent PCR reactions.
Current applications of the instrument in the human genome project and for combinatorial library screening will be discussed.
MATERIALS AND METHODS
Instrument Construction and Operation
The Hydra-96 (Figure 1) is constructed of 96 glass syringes vertically mounted in a bracket that spaces them precisely 9 mm center to center (two syringes sizes are available, 290 and 580μl). Up and down motion of the syringe plungers for filling and emptying the syringes is achieved by the action of a precision screw and stepper motor that moves a plate to which the plungers are attached.

The Hydra-96 Microdispenser. The unit pictured is being used to dispense microliter volumes of reagents from its array of 96 syringes into a microplate.
The motion of this system is accurate to 2.5 microns. Syringe needles two inches in length are Teflon ™ coated on the outside at their tips to ensure uniform drop sizes are formed during dispensing. Prior to each fill or dispense operation the microplate, reservoir, etc. to be used is positioned on a platform that raises it below the ends of the needles to the height needed for the vessel selected with a positional accuracy of 6.5 microns. The precision of the platform positioning system is required for delicate operations including dispensing onto membranes and agar plates and for “touch off” of drops into dry plates.
To operate the system, a single “practice” run is performed to program the unit. The dispense volume, fill volume, wash volume and cycles and platform vertical motion are selected by push buttons and stored in one of 16 separate files in non-volatile memory. An LED display on the front of the unit allows the operator to scroll through the files to confirm or edit parameters prior to operation. Dispense, fill, wash and empty functions are activated through push buttons on the front of the unit. An RS232 port on the side of unit is used to control the instrument by a remote computer using a simple ASCII communication protocol.
Measurement of Dispenser Uniformity
Syringes were filled with a concentrated solution of Napthol Blue Black and volumes from 0.5 to 20.0μl dispensed into a series of 96 well microplates containing 100μ1 of distilled water per well. After filling the syringes and prior to dispensing into the microplates, a few dispenses were normally performed back into the dye reservoir. The microplates containing the dye/water solution were mixed by hand using an 8-channel pipettor and then read on a Molecular Devices Thermomax plate reader (Molecular Devices, Sunnyvale, CA). Data is presented after subtraction of values from a blank plate containing water only.
Effectiveness of Bleach Treatment to Remove Residual DNA
Preparation of DNA
Human genomic DNA was prepared from nuclei from 1 ml of whole blood from a DRB1–0301 homozygote using a modification of an ammonium acetate salting out procedure (1).
Final DNA preparation had an O.D. 260/280 of >1.8. A segment of the DRB1 locus was then amplified by PCR for 30 cycles under standard conditions using DRB1 specific primers (2) using a Perkin Elmer Model 9600 thermal cycler. (Perkin Elmer, Foster City, CA) and thin wall Strip-Ease ™ reaction tubes (Robbins Scientific, Sunnyvale, CA). The amplified product was then denatured in 1/5 volume 2N NaOH then neutralized with 4/5 volume of 3M sodium acetate. The final solution of denatured DNA used for blotting was 0.22μg/μl.
Dot Blot Preparation
Dot blots were prepared with the Hydra-96 Microdispenser using the DNA preparation described above. Syringes were filled with 50μl of the DNA solution except syringes A1, B1, and C1 which contained a dilution series of the DNA and syringe D7 which contained water only. A series of four identical blots were prepared by dispensing 2μl of the solution per syringe on to HyBond-N membranes (Amersham Corporation, Arlington Heights IL).
The syringes were emptied and then rinsed with three up down cycles with 2% bleach in water followed by three cycles with distilled water. The syringes were then refilled with distilled water and a dot blot prepared as described above except positions A1, B1 and C1 contained the dilution series of DNA used in the first series of blots as hybridization controls. The syringes were then emptied again, rinsed with distilled water only for three cycles and another dot blot prepared. This process was repeated twice more generating a total of four blots which would contain residual DNA left in the syringes.
Dot Blot Hybridization
Membranes were baked at 80° C for two hours then prehybridized for 2 hours at 57° C in a solution containing 6X SSPE, 5X Denhardt's, 0.5% SDS and l00μg/μ1 denatured herring sperm DNA (3). Blots were then hybridized in the same solution for an additional 4 hours at 57° C using 32 Pend labeled DRB 7004 (3) as probe. Blots were then washed twice in 2XSSPE/0.1% SDS then 3–5 times in 6X SSPE/1.0% SDS at 60°C. After air drying, the blots were exposed to X-film overnight at −70° C with intensifying screens.
Effect of Bleach Treatment on Subsequent PCRs
The three distilled water solutions used to rinse the syringes described in the previous section was in the preparation of a series of PCR reactions. Reactions conditions were as follows: 13μl of H2 O containing 0.3μg human DNA, 15.2 ul of amplification mix containing, 2 units of Taq polymerase (Perkin Elmer), nucleotides, MgCl2, and DRB1 primers and 31.8 ul of the distilled water rinse solutions. Positive control reactions contained pure distilled water in place of the rinse solutions. PCR was performed for 30 cycles and the amplified products separated on a 1.5% agarose gel and stained with ethidium bromide.
RESULTS
Dispensing Uniformity
Figure 2 shows the results of studies to measure the dispensing uniformity of the syringe array when delivering from 0.5 to 20.0μl. Data presented is from analysis of data points from the individual wells across each microplate used for each volume evaluated. Results show that the volume delivered as measured by the dye concentration in the wells is a linear function of the volume selected on the instrument. Statistical analysis of the O.D. readings from the wells of each plate identical reactions prepared from distilled water rinse #1, lanes 5–8 from rinse #2 and lanes 9–12 from rinse #3. Positive control is shown in lane C. Molecular weight markers (lanes marked M) is an Eco Rl digest of OX174.
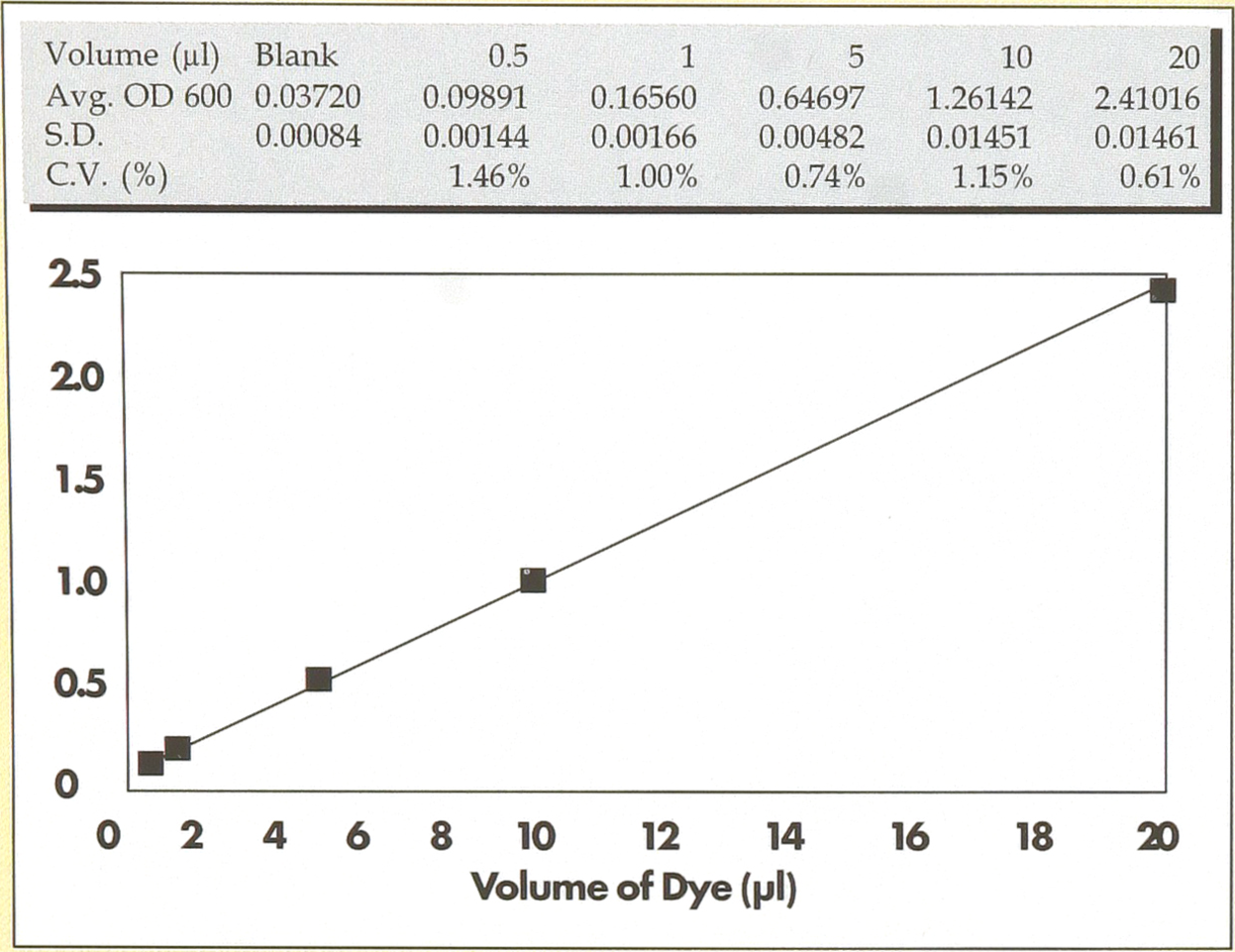
Dispensing uniformity of the Hydra. Using a concentrated dye solution as a detection reagent, the dispensing uniformity of the instrument was measured when delivering between 0.5 to 20μl.

Uniformity of dot blots prepared with the Hydra. Four identical dot blots were prepared with the instrument, probed and auto radiographed overnight. Positions A1, B1 and C1 were serial dilutions of the DNA sample containing 220, 44 and 9 ng of DNA respectively. Position D7 is a negative hybridization control.

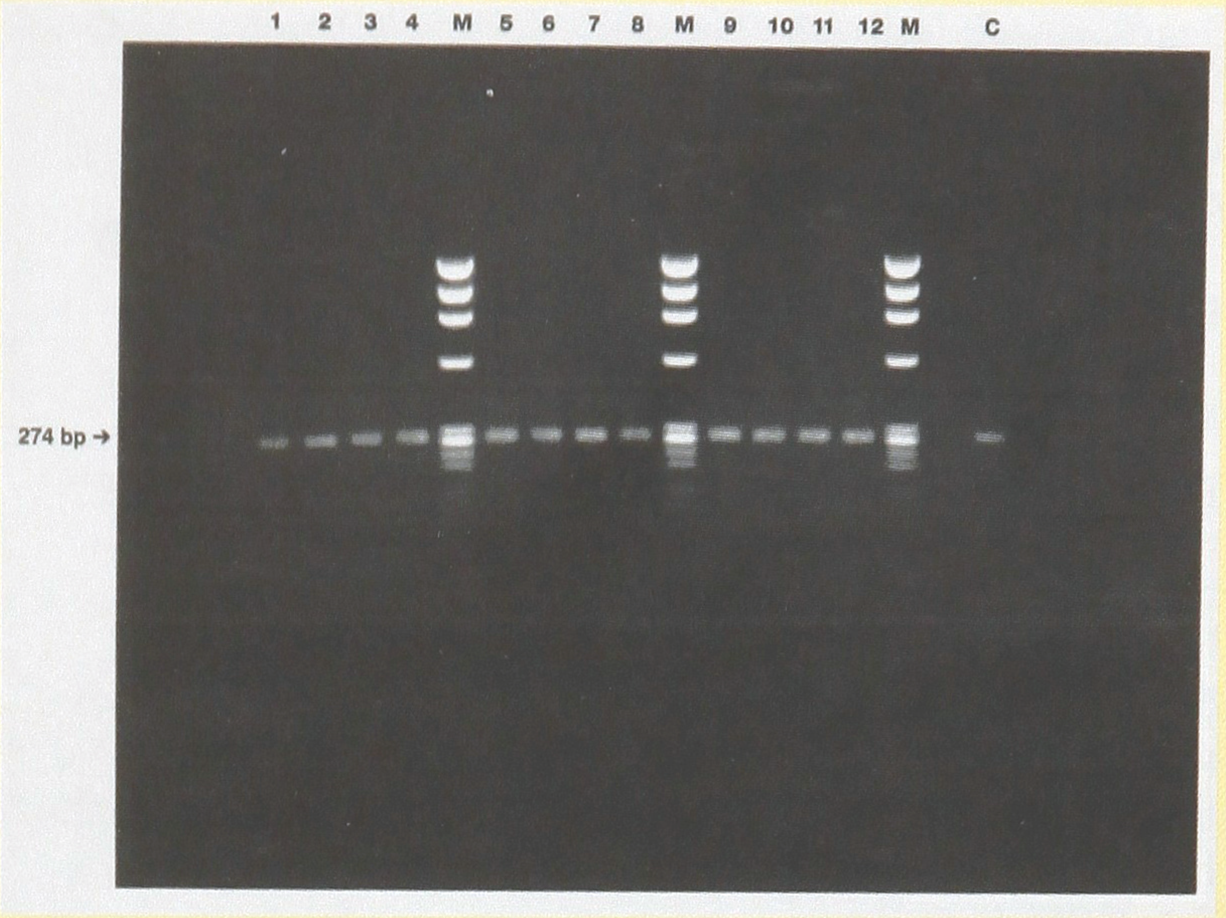
Effect of bleach treatment on subsequent PCRs Reaction conditions are described in the text. Lanes 1–4 are identical reactions prepared from distilled water rinse #1, lanes 5–8 from rinse #2 and lanes 9–12 from rinse #3. Positive control is shown in lane C. Molecular weight markers (lanes marked M) is an EcoR1 digest of 0X174.
DISCUSSION
The Hydra-96 Microdispenser was developed to meet the specialized needs of laboratories that require high speed, precise, repetitive dispensing of microliter volumes of reagents into microplates, PCR plates and filter membranes. Studies in this initial report have focused on examining the instrument's ability to uniformly deliver volumes down to 0.5 ul and the effectiveness of rinsing the syringes to remove any residual sample.
Results show that the instrument has less than 2% variance in dispensing volume, performance that meets the requirements of the most demanding applications. Also shown is that simple rinsing of the syringes with dilute bleach effectively removes DNA that might contaminate downstream reactions and that rinsing with distilled water removes residual bleach that could interfere with subsequent PCR reactions prepared with the instrument. These results show that the instrument can be a useful tool in a variety of PCR based applications. It further suggests that disposable tips are not necessary to prevent sample carry-over, a feature that provides an important cost-saving feature relative to all known multichannel automated liquid handling devices.
Because of its unique characteristics, the Hydra has found uses in a variety of applications in diverse laboratory environments. Drug development laboratories involved in screening large combinatorial libraries use the instrument for dispensing small volumes of candidate compounds, detection reagents and other precious reagents. Human genome analysis laboratories use the instrument for preparing PCR and cycle sequencing reactions and for preparation of DNA sequencing templates. The Hydra is also being used under sterile conditions for dispensing reagents into cell cultures used in various bioassays and for removing and adding media in micro-cell cultures used in single cell cloning techniques.
